Chimeric Antigen Receptor T Cell Manufacturing on an Automated Cell Processor
Summary
This article details the manufacturing process for chimeric antigen receptor T cells for clinical use, specifically using an automated cell processor capable of performing viral transduction and cultivation of T cells. We provide recommendations and describe pitfalls that should be considered during the process development and implementation of an early-phase clinical trial.
Abstract
Chimeric antigen receptor (CAR)-T cells represent a promising immunotherapeutic approach for the treatment of various malignant and non-malignant diseases. CAR-T cells are genetically modified T cells that express a chimeric protein that recognizes and binds to a cell surface target, resulting in the killing of the target cell. Traditional CAR-T cell manufacturing methods are labor-intensive, expensive, and may carry the risk of contamination. The CliniMACS Prodigy, an automated cell processor, allows for manufacturing cell therapy products at a clinical scale in a closed system, minimizing the risk of contamination. Processing occurs semi-automatically under the control of a computer and thus minimizes human involvement in the process, which saves time and reduces variability and errors.
This manuscript and video describes the T cell transduction (TCT) process for manufacturing CAR-T cells using this processor. The TCT process involves CD4+/CD8+ T cell enrichment, activation, transduction with a viral vector, expansion, and harvest. Using the Activity Matrix, a functionality that allows ordering and timing of these steps, the TCT process can be customized extensively. We provide a walk-through of CAR-T cell manufacturing in compliance with current Good Manufacturing Practice (cGMP) and discuss required release testing and preclinical experiments that will support an Investigational New Drug (IND) application. We demonstrate the feasibility and discuss the advantages and disadvantages of using a semi-automatic process for clinical CAR-T cell manufacturing. Finally, we describe an ongoing investigator-initiated clinical trial that targets pediatric B-cell malignancies [NCT05480449] as an example of how this manufacturing process can be applied in a clinical setting.
Introduction
Adoptive transfer of T cells engineered to express a chimeric antigen receptor (CAR) has shown remarkable efficacy in treating patients with refractory B-cell malignancies1,2,3,4,5. However, the traditional manufacturing methods for CAR-T cells are labor-intensive, time-consuming, and require highly trained technicians to carry out highly specialized steps. For example, the traditional manufacturing process of an autologous CAR-T cell product involves density gradient centrifugation, elutriation or magnetic separation to enrich T cells, activation, and transduction with a viral vector in a sterile flask, and expansion in a bioreactor prior to harvest and formulation. Various systems have emerged recently that aim to partially automate this process. For example, the Miltenyi CliniMACS Prodigy (hereafter referred to as the "processor") is an automated cell processing device that can perform many of these steps in an automated fashion6,7,8,9. An in-depth discussion of traditional and automated CAR-T manufacturing methods is presented in a recent review article10.
The processor builds upon the functionality of the CliniMACS Plus, a U.S. Food and Drug Administration (FDA)-approved medical device for the processing of hematopoietic progenitor cells. The processor includes a cell cultivation unit that allows for automated washing, fractionation, and cultivation of cells (Figure 1). The T cell transduction (TCT) process is a preset program within the processor device that largely replicates manual CAR-T cell manufacture. TCT allows for customizable cell processing using a graphical user interface (the "Activity Matrix," Figure 2). Because the processor automates many steps and consolidates the functionality of multiple devices into one machine, it requires less training and specialized troubleshooting skills from technologists. Because all steps are performed within a closed, single-use tubing set, the processor may be operated in facilities with less stringent air-handling infrastructure than would be considered acceptable for an open manufacturing process. For example, we are operating the processor in a facility certified as ISO class 8 (comparable to EU grade C).
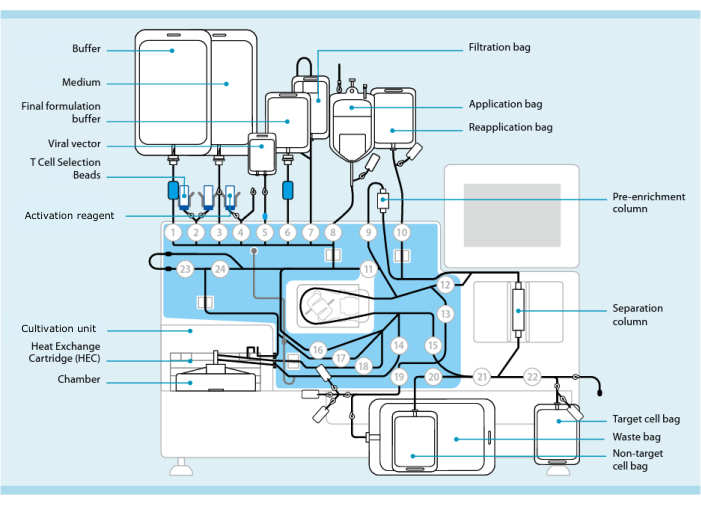
Figure 1: CAR-T cell manufacturing using the T cell transduction system. Shown is the processor with the tubing set installed. The tubing set allows for connecting other components such as bags containing processing buffer, culture medium, and lentiviral vector via sterile welding. Once the leukapheresis product is added to the Application bag, it can be labeled with T Cell Selection Beads, passed through the Separation column, and then transferred into the Reapplication bag. Selected cells are then directed to the Cultivation unit of the instrument for culture and activated with the Activation reagent (see Table of Materials). The final product is collected in the Target cell bag. Throughout the process, it is possible to remove samples for quality control aseptically. Grey numbers inside of circles represent the numbered valves on the processor that direct the liquid path through the tubing set. Reproduced with permission from 11. Please click here to view a larger version of this figure.
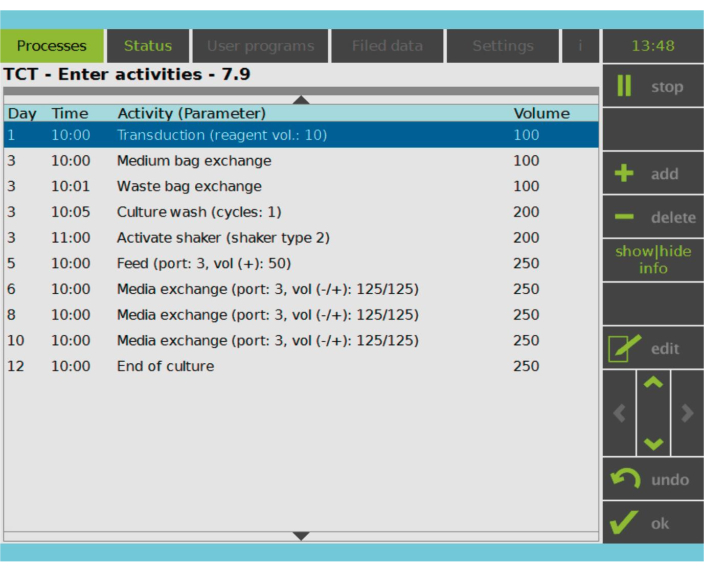
Figure 2: Activity Matrix. After T cell selection and activation, the remainder of the CAR-T cell manufacturing process is fully customizable. Activities can be added or deleted and scheduled for the appropriate day and time, and the culture volume after the activity can be specified (Volume). For example, the Transduction activity was configured to begin at 10:00 AM on Day 1, and the culture volume at the end of the activity was set as 100 mL. The Activity Matrix can be edited throughout the cultivation period. The status of the process can be monitored on the integrated screen of the processing device. Please click here to view a larger version of this figure.
The aim of this manuscript is to provide a detailed walk-through of manufacturing CAR-T cells using the processor and additionally provide guidance on the in-process and product release testing that will likely be required by regulators to approve an investigational new drug (IND) application. The presented protocol stays close to the vendor's recommended approach and is the underlying protocol for IND 28617, which is currently being evaluated in a single-center investigator-initiated phase I/II clinical trial. This trial aims to determine the safety and efficacy of using this processor to manufacture humanized CD19-directed autologous CAR-T cells for patients with B cell acute lymphoblastic leukemia (B-ALL) or B-lineage lymphoblastic lymphoma (B-Lly) [NCT05480449]. The trial started in September 2022 and is planned to enroll up to 89 patients ages 0-29 years with B-ALL or B-Lly. We report some manufacturing results from the trial in the manuscript.
We would like to point out that although the manuscript is presented as a protocol with steps to follow, it should be considered a starting point for others to begin optimizing their own CAR-T cell manufacturing process. A non-comprehensive list of possible variations to the presented protocol includes: using fresh instead of cryopreserved T cells as starting material; using a different method of T cell enrichment or omitting it altogether; using different media and cytokine cocktails such as IL7/IL15 instead of IL2; varying the concentration of human AB serum or omitting it altogether; timing of transduction; using "multi-hit" transductions; varying agitation, culture volumes, and feeding schedule; using different methods of genetic transfer including electroporation of nucleic acids or non-lentiviral vectors; using a different final formulation buffer and/or cryoprotectant; and infusing CAR-T cells fresh instead of cryopreserving for infusion at a later time. These variations may have a significant impact on the cellular composition and potency of the therapeutic product.
| Overall Process Step | Process Day | Technical Details | |||
| Cell Enrichment | Day 0 | Selection of CD4+/CD8+ T cells | |||
| Cell Activation | T cell culture seeding and activation | ||||
| Cell Transduction | Day 1 | Lentiviral transduction (100 mL culture volume) | |||
| Cell Expansion (followed by cell formulation) | Day 2 | — | |||
| Day 3 | Culture Wash (1 cycle); Shaker activated; Culture volume increases to 200 mL | ||||
| Day 4 | — | ||||
| Day 5 | Feed (50 mL); Culture volume reaches final volume of 250 mL | ||||
| Day 6 | In-process sample; Media exchange (-125 mL / +125 mL) | ||||
| Day 7 | Media exchange (-150 mL / +150 mL) or Harvest | ||||
| Day 8 | In-process sample; Media exchange (-150 mL / +150 mL) or Harvest | ||||
| Day 9 | Media exchange (-180 mL / +180 mL) or Harvest | ||||
| Day 10 | In-process sample; Media exchange (-180 mL / +180 mL) or Harvest | ||||
| Day 11 | Media exchange (-180 mL / +180 mL) or Harvest | ||||
| Day 12 | Media exchange (-180 mL / +180 mL) or Harvest | ||||
| Day 13 | Harvest | ||||
Table 1: Process timeline and overview. This table summarizes the TCT process steps employed in a current clinical trial [NCT05480449]. The process starts with T cell enrichment by CD4+/CD8+ selection, culture seeding, and activation on Day 0, followed by transduction on Day 1. Cells rest for 48 h, followed by a culture wash, an increase of the culture volume to 200 mL, and agitation using a shaking mechanism. On Day 6, the first in-process sample is taken. Cells are harvested once sufficient cells are available for at least three full doses of CAR-T cells (5 × 106 CAR-T cells/kg if the patient is <50 kg, otherwise 2.5 × 108 CAR-T cells) and quality control testing (~2 × 106 CAR-T cells); or once the culture reaches a total of 4-5 x 109 cells. Abbreviations: TCT = T cell transduction; CAR-T = chimeric antigen receptor T cells; MACS = magnetic-activated cell sorting.
Protocol
All research was performed in compliance with institutional guidelines with approval by the hospital's Institutional Review Board (IRB), and all subjects have provided informed consent for publication of the data collected within the context of the trial.
NOTE: The first section of the Protocol provides a high-level overview of the CAR-T manufacturing process. The remaining sections provide the step-by-step instructions. The protocol describes the workflow using TCT software version 1.4, which is the current version as of this writing. The user interface of other versions of the TCT software may vary.
1. Process timeline and overview (Table 1)
- Prepare for the procedure on a Monday (Day -1) with preflight checks. Ensure that the processor and other equipment are running as expected and that all reagents and consumables are available and in-date for the entire manufacturing run.
- On day 0, install the tubing set on the machine. Thaw previously cryopreserved T cells in a dry bath and connect them by sterile welding to the tubing set installed on the instrument.
NOTE: This protocol assumes that autologous T cells were collected via apheresis, cryopreserved, and stored until the start of manufacturing. While it is possible to use freshly collected T cells, this increases the logistical burden as it requires coordinating apheresis collection with CAR-T cell manufacturing. We strongly recommend using a dry-thaw process instead of a water bath to minimize the risk of bacterial contamination. - Label T cells with CD4 and CD8 reagents (see Table of Materials) and enrich by magnetic selection.
NOTE: It is possible to omit T cell enrichment or perform it prior to loading cells onto the processor. See section 5 of the Protocol. - After enrichment of CD4+/CD8+ cells, take a sample for a cell count. Seed the culture with 1-2 × 108 cells in an initial volume of 70 mL of medium (see Table of Materials) supplemented with 5% human AB serum and recombinant human IL2 (25 ng/mL).
- Add a vial of activation reagent (a colloidal polymeric nanomatrix conjugated to anti-CD3 and anti-CD28 antibodies; see Table of Materials) to activate T cells and incubate the culture without agitation for 24 h at 37 ºC in a 5% CO2 atmosphere. Cryopreserve any leftover CD4+/CD8+ cells as backup, if desired.
NOTE: In case of manufacturing failure, leftover CD4+/CD8+ selected cells may be used as starting material. If the failure is purely technical, such as contamination or operator error, and sufficient cells remain, then using leftover cells to perform an additional manufacturing run can be considered. If the quality of starting material is of concern, then a new apheresis procedure could be warranted; however, this is ultimately a clinical decision. In either case, a manufacturing failure is a significant event that should be investigated, and the sponsor and possibly regulators should be informed. - After 24 h of activation, transduce T cells with lentiviral vector at an appropriate multiplicity of infection (MOI).
NOTE: It is critical to determine the lentiviral vector titer and establish an appropriate MOI before starting to manufacture clinical products. The vector titer should be determined by performing a small-scale experiment in which human primary T cells are transduced at various concentrations of the vector. Considerations for the appropriate MOI include the cost of the vector, the desired transduction efficiency, and an acceptable vector copy number. This protocol uses an MOI of 30-50% to minimize the cost of the vector and keep the average vector copy number below 8 copies per transduced cell. - On Day 3, trigger the Culture Wash activity, and start low-level agitation. Increase the culture volume to 200 mL.
- On Day 5, add 50 mL of medium to the culture, increasing the culture volume to 250 mL.
- On Day 6 of cultivation, take a sample from the culture, and enumerate CAR-T cells by flow cytometry. Use this measurement to estimate the rate at which the culture is growing and identify the optimal time of harvest.
NOTE: Each in-process sampling yields 3 mL for testing and removes 7 mL total of culture volume. - From Day 7 to 13, if the culture has not been terminated, take up to two additional in-process samples on alternate days, and perform daily media exchanges to feed the growing culture. Harvest the product when the total nucleated cell (TNC) count reaches 5 × 109 and/or when sufficient cells are available for the required number of doses and release testing.
- On the day of harvest, take an in-process sample from the actively growing culture. Use this sample for mycoplasma, endotoxin, replication-competent lentivirus (RCL) testing, vector copy number (VCN) testing, cell count, cell size analysis, flow cytometry, and gram stain.
- Initiate the final harvest program, which triggers the removal of the culture medium and a cell wash with the final formulation buffer (a sterile isotonic crystalloid solution supplemented with 4% human serum albumin; see Table of Materials). After harvest is completed, the target cell bag will hold 100 mL of cell product in final formulation buffer. Add dimethyl sulfoxide (DMSO) to a concentration of 10% (v/v), aliquot the product into individual doses, and cryopreserve using a controlled-rate freezer.
2. Day -1: Preparation and preflight checks
- Verify that CO2 gas levels and compressed air are sufficient to produce an inlet pressure of at least 20 psi on each line.
- Turn on the processor and confirm that there are no errors on startup. If needed, set the clock to the correct time. Shut down the processor.
- Ensure cell count, volume, and total CD4+ and total CD8+ count of the T cell starting material are known.
- Ensure there are sufficient quantities of the viral vector, reagents, and consumables for the entire manufacturing run.
- Place a bottle of human AB serum in the refrigerator to thaw overnight.
3. Day 0: Tubing set installation
- Prepare 3 L of processing buffer (0.5 % (w/v) human serum albumin (HSA) in phosphate-buffered saline / ethylenediaminetetraacetic acid (PBS/EDTA) buffer).
- Prepare 2 L of culture medium (2 L of medium supplemented with 100 mL of human AB serum to a final concentration of 5% and two vials of recombinant human IL2 at 25 µg/vial; see Table of Materials for more information on these reagents). Transfer 10 mL culture medium into a 20 mL reagent bag and store at 4 ºC overnight.
- Turn on the instrument and select the T Cell Transduction Process (TCT) from the touch screen interface. Click Run to start the TCT process; let the instrument guide the user through the procedure using on-screen instructions and prompts.
- On the Parameter Input screen, enter the operator's initials, the tubing set lot number, and its expiration date when prompted.
- The Process Setup screen presents four different processes. Choose Full process (1).
NOTE: Other available processes include starting from CD4+/CD8+ selected T cells (see section 5 below) and restarting a previously aborted manufacturing run. - When prompted, choose two vials of selection reagent to reflect the CD4+/CD8+ selection method.
- Install the tubing set per the on-screen instructions. Ensure all Luer connections are tight and that there are no defects in the tubing set.
- Follow the on-screen instructions to initiate the automated upper and lower integrity tests.
NOTE: An integrity test may fail due to a faulty tubing set, an incorrectly installed tubing set, or a defect in the machine's peristaltic pump. We strongly recommend having at least one additional tubing set on hand for production runs. We also recommend that a second technologist verify the correct installation of the tubing set. - Follow the on-screen instructions for attaching the medium and processing buffer bags.
- Begin the automatic priming of the tubing set.
NOTE: The TCT process must continue within 3 h after priming. Ensure T cell starting material will be ready.
4. T cell enrichment
- When the Transfer cell product screen appears, begin thawing the cryopreserved T cell product.
NOTE: The acceptable volume of T cells that can be added to the tubing set ranges from 50 to 280 mL. The maximum number of target (sum of CD4+ and CD8+) cells is 3 × 109, and the maximum TNC count is 2 × 1010. - Transfer the thawed cells to a 150 mL transfer bag. Sterile weld the transfer bag to the Application bag of the tubing set.
- Remove a sample from the Application bag using the QC pouch and perform a cell count.
- Connect the CD4 and CD8 reagent vials.
- Start the selection (T cell enrichment) process.
- After enrichment, remove a sample of the CD4+/CD8+ selected cells from the Reapplication bag's QC pouch for cell count, flow cytometry, and cell size analysis.
NOTE: The cell count result is needed to proceed to the next step.
5. Alternative: Starting with CD4+/CD8+ selected cells
- Prepare the Medium as described in step 3.2. No processing buffer is needed.
- Turn on the processor and select T cell cultivation with TS installation (3) on the Process Setup screen. Follow the on-screen instructions and prompts.
- Following the on-screen instructions, prime the tubing set with medium instead of processing buffer.
- When the "Prepare cultivation-Connect cell product" screen appears, begin thawing the CD4+/CD8+ selected T cells.
NOTE: The minimum number of T cells (sum of CD4+ and CD8+ T cells) for the process is 1.0 × 108. - Transfer cells to a 150 mL transfer bag and dilute with medium to a final volume of 50 mL.
- Sterile weld the cell suspension to the instrument's Reapplication bag.
- Remove a sample of the CD4+/CD8+ selected cells from the Reapplication bag's QC pouch for cell count and cell size analysis.
6. Culture setup and programming of the Activity Matrix
- Enter the cell concentration and desired starting number (1-2 × 108 T cells).
NOTE: The instrument will automatically pump the appropriate volume from the Reapplication bag into the culture chamber and adjust the final volume to 70 mL. - Attach a vial of the activation reagent per the on-screen instructions.
- Enter 5% for CO2 concentration and 39 °C for culture chamber temperature.
NOTE: The manufacturer recommends entering 39 °C or a value specifically calibrated for the instrument. - Set up the Activity Matrix. Use the Enhanced feeding protocol as a starting point and modify individual steps within the protocol to user-defined specifications (Figure 2).
- Ensure that the time of Transduction activity is 24 h after seeding (Day 1).
- Ensure that the time of Culture Wash activity is 48 h after Transduction (Day 3).
- Set the time of Activate Shaker (shaker type 2) to 30 min after the start of the Culture Wash.
- Delete any Medium Bag Exchange and Waste Bag Exchange activities.
- Touch ok on the screen to begin cultivation.
- Cryopreserve any leftover CD4+/CD8+ selected cells in the Application bag for future use, if needed.
7. Day 1: T cell transduction
- Calculate the volume of the lentiviral vector to use based on the desired MOI.
- Retrieve the 20 mL reagent bag containing 10 mL culture medium that was stored at 4 ºC overnight (step 3.2). Thaw the vector vial and transfer the volume calculated in 7.1 into the 20 mL reagent bag.
NOTE: We recommend freezing any leftover vector for retention or for research and development. - Modify the Activity matrix to set the time of Transduction to 2 min into the future. When prompted, touch ok to start the Transduction activity.
- Sterile weld the vector bag to the tubing set following the on-screen instructions.
- Modify the Activity matrix based on the actual time that the Transduction activity was started.
NOTE: We suggest starting transduction within 20-24 h after seeding to ensure hands-on activities are performed during normal working hours.- Ensure that the time of Culture Wash is 48 h after Transduction (Day 3).
- Set the time of Activate Shaker (shaker type 2) to 30 min after Culture Wash (Day 3).
- Ensure that the time of Media Exchange is in the afternoon (1 PM) of Day 6.
8. Day 6: First in-process sample
- Touch the Sample button and follow the on-screen instructions to obtain a QC sample from the active culture.
- Perform cell count, flow cytometry, and cell size analyses. Send 1 mL of the sample for a Gram stain.
NOTE: Cell counts should be repeated approximately every other day to monitor culture growth. - Prepare another 2 L of culture medium (see step 3.2).
- Add a Medium Bag Exchange activity to the Activity Matrix, timed to start 2 min into the future. Adjust the Media Exchange activity to start 20 min into the future. Follow on-screen instructions.
NOTE: Media Exchange is the replacement of the culture medium in the cultivation chamber. Medium Bag Exchange is the replacement of the culture medium bag hung on the instrument.
9. Day of harvest (anywhere from day 7 to 13): Harvest and cryopreservation
- Touch the Sample button and follow the on-screen instructions to obtain a QC sample from the active culture.
- Separate the QC sample into three aliquots of 1 mL each. Use 1 mL for flow cytometry, cell count, and cell size analyses. Use 1 mL for mycoplasma, vector copy number, replication-competent lentivirus, and endotoxin testing. Send the final 1 mL for Gram stain.
- Prepare the final formulation buffer (a sterile isotonic crystalloid solution supplemented with 4 % HSA in a 2 L bag, see Table of Materials). Save 100 mL of this buffer to prepare the cryoprotectant solution at a later step.
- Modify the Activity Matrix to set the time of End of Culture to 2 min into the future and delete all other remaining activities. Follow the on-screen instructions to attach the final formulation buffer to the tubing set and begin harvest.
NOTE: The processor will automatically transfer the cells into the Target cell bag. The volume will be 100 mL. - Take a 0.5 mL sample from the Target cell bag QC pouch and perform a cell count.
- Seal off the Target cell bag and remove it from the tubing set. Divide the CAR-T product into appropriate doses and cryopreserve them in a controlled-rate freezer. Store cells in the vapor phase of liquid nitrogen storage tank at ≤ -150 ºC.
NOTE: Final formulation and aliquoting of doses are specific to the protocol. We provide here an example in which three equal doses of CAR-T cells and QC vials are cryopreserved. See section 10 for details. - Download the process data from the instrument, remove and dispose of the tubing set, and perform a shutdown.
10. Cryopreservation of CAR-T cells
NOTE: This protocol assumes CAR-T cells are cryopreserved after manufacture and stored until the patient is ready for infusion. While it is possible to infuse freshly manufactured CAR-T cells, this increases the logistical burden as it requires coordinating CAR-T cell manufacturing with CAR-T cell infusion. This may be problematic in case of a manufacturing failure. Especially if the clinical protocol requires lymphodepleting chemotherapy prior to CAR-T infusion, we strongly suggest cryopreserving, because a manufacturing failure may expose the patient to the risk of unnecessary chemotherapy. Regulators may require investigators to demonstrate the product passes all release testing prior to infusion, which may be difficult to achieve without cryopreservation.
- From the final 100 mL of harvested product, determine and remove the volume required to meet the desired CAR-T doses and quality control (QC) vials for performing additional release testing (e.g., viability), retention, and a sterility sample.
NOTE: It is critical to develop a fully defined strategy for aliquoting the final product. We suggest manufacturing multiple doses of the product to allow for reinfusions, given that sufficient material is available. A product volume of 10-20 mL in a bag allows for rapid thawing and easy infusion by syringe push. It is recommended to fill each product to the desired CAR-T cell dose (e.g., 5 × 106 CAR-T cells per kg or 2.5 × 108 CAR-T cells) as this will avoid the need for dose calculations on the day of infusion. Also recommended are cryopreserving at least four QC vials and accounting for the possibility that there might be leftover material; we suggest saving this material as it can be useful for research and development purposes. - Centrifuge the cells to reduce the volume.
- Bring up the product(s) to half of the desired final volume using the portion of final formulation buffer put aside in step 9.3.
- Prepare 2x cryoprotectant by creating a 20% DMSO solution in final formulation buffer.
NOTE: Cryoprotectant formulation and DMSO concentration can be modified. - Add 2x cryoprotectant to cells at equal volume for a final DMSO concentration of 10%.
NOTE: Duration of exposure of the product to cryoprotectant should be minimized. - Fill the dose bags and QC vials. Send 1 mL for sterility.
NOTE: Regulators commonly require a 14-day sterility assay performed on the final product. However, rapid sterility assays are emerging; at this time, the compendial 14-day method is the most common. - Cryopreserve products using a controlled-rate freezer.
NOTE: The controlled-rate freezer program should be validated to produce a cooling rate of ~ -1 ºC/min to ~ -45 ºC, with compensation for the eutectic point. - Store the product in the vapor phase of a liquid nitrogen ultralow freezer at ≤ -150 °C.
11. Examination of procedure performance
NOTE: Throughout the TCT process, several QC samples are drawn from the active culture. Table 2 provides a grid that can help the reader organize the results for reference and calculate procedure performance metrics. The terms below consisting of a letter and a number (e.g., "B4") refer to cells in the grid of this table. The following values are used in the performance calculations: B3 = total nucleated cells (TNC) pre-enrichment; B4 = TNC post-enrichment; E2 = The sum of CD4+ and CD8+ T cells as a percentage of the total cells of the initial apheresis product; E4 = The sum of the CD4+ and CD8+ T cells as a percentage of the total cells post-enrichment; G2 = CD19+ cells as a percentage of the total cells of the initial product; G4 = CD19+ cells as a percentage of the total cells after CD4+/CD8+ enrichment; B10 = TNC of the Active Culture on day of harvest.
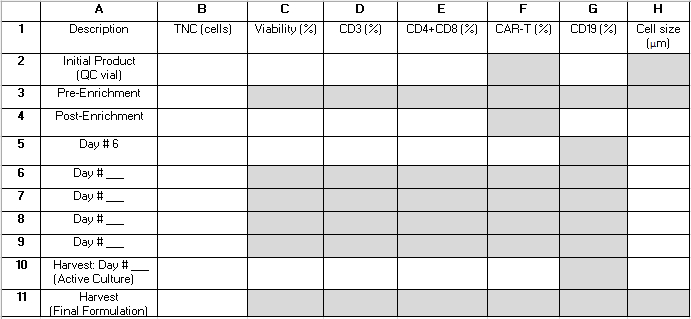
Table 2: Procedure performance grid. We provide this grid to help organize in-process test reults needed to calculate procedure performance statistics. Rows represent samples analyzed at various time points during the procedure and are labeled with numbers 1-11. Rows 6-9 can be used to capture results from samples taken after day 6 of cultivation but prior to the day of harvest. Columns represent measured parameters and are labeled with letters A-H. Fields that are shaded gray do not apply. Some additional fields may not apply depending on whether CD4+/CD8+ enrichment is performed as part of the procedure and the length of the cultivation. We suggest writing "N/A" into those fields. Abbreviations: TNC = total nucleated cell count; QC = quality control. Please click here to download this Table.
- Calculate recovery of CD4+/CD8+ cells after selection by dividing the total number of CD4+ plus CD8+ T cells post-enrichment by the total number of CD4+ plus CD8+ T cells prior to enrichment using equation (1).
CD4+/CD8+ T cell recovery =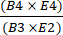 (1)
(1) - Calculate depletion of CD19+ cells after enrichment by dividing the total number of CD19+ cells post CD4+/CD8+ enrichment by the total number of CD19+ cells prior to enrichment. Since this number is expected to be very small, report the logarithm of this fraction as shown in equation (2):
Log CD19 cell depletion = log10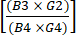 (2)
(2) - Calculate the total fold growth by dividing the total number of cells in active culture at harvest by the number of cells seeded using equation (3):
Total fold growth =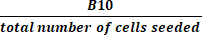 (3)
(3) - Calculate the average daily growth by taking the root of the total fold growth with respect to the day of harvest using equation (4):
Average daily growth = day of harvest √ (total fold growth) (4)
Representative Results
Results from the initial three CAR-T manufacturing runs of the NCT05480449 trial are presented below in Table 3. The starting material, vector, culture cytokines, and AB serum concentrations were kept consistent for each run. Products were harvested on day 7 or 8. The average daily cell growth was 46% (increase in total cell count), indicating that the TCT process was effective in promoting cell expansion. These results suggest that the processor can produce consistent and reproducible CAR-T cell products.
The final product test results, summarized in Table 4, demonstrate that the CAR-T cells passed quality control standards. Cell viabilities were between 88% and 94%, and the CD3 T cell purity was 89-93%. Importantly, endotoxin, mycoplasma, sterility, replication-competent lentivirus, and residual leukemic cells were not detectable. These results confirm that the TCT process produces high-quality CAR-T cells that meet regulatory standards for clinical use.
Three flow cytometry panels were developed for this procedure (Figure 3). They include the CD4/CD8 panel to test for the percentage of CD4+ and CD8+ T cells pre and post enrichment, the CD19 CAR-T panel to test for transduction efficiency, and the CD3/CD19 panel to determine viability and the content of residual leukemic (CD19+) cells in the final product. The results of these panels confirm the successful manufacturing of CAR-T cells and the absence of residual leukemic cells in the final product.
Overall, the results suggest that the TCT process can produce consistent and high-quality CAR-T cell products suitable for clinical use.
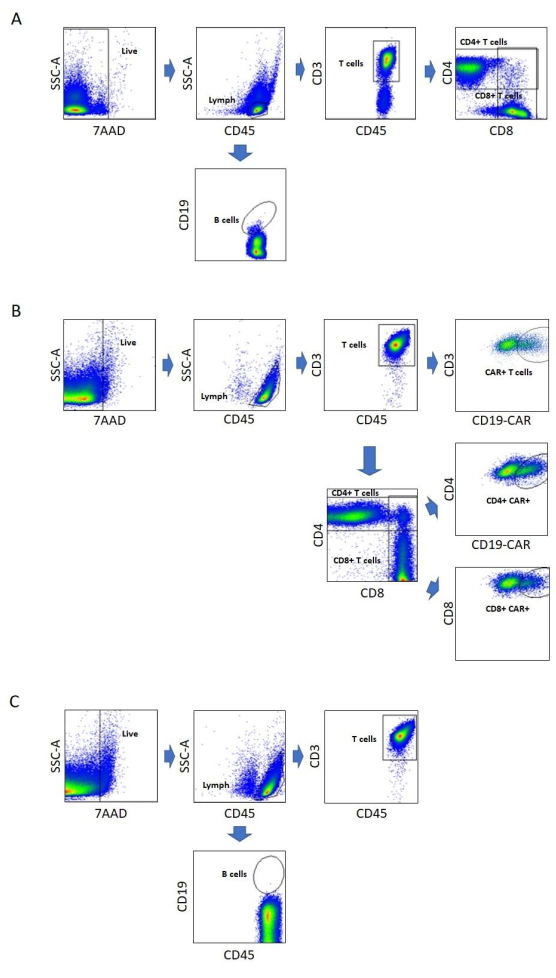
Figure 3: Flow cytometry assays. The gating strategy used for flow cytometry analysis is depicted for each panel. Gates are drawn as black boxes or ellipses and labeled, and blue arrows indicate the direction of the gating. (A) CD4/CD8 panel. B cells are defined as the CD19+ subset and T cells as the CD3+ subset of the Lymph gate. CD4+ and CD8+ T cells are subsets of the T cell gate. (B) CD19 CAR-T panel. CAR+ T cells are defined as the CD19-CAR+ subset of CD3+ T cells. In addition, CD4+ and CD8+ subsets that are CD19 CAR+ are enumerated. (C) CD3/CD19 panel. This panel is identical to the CD4/CD8 panel, except it omits the CD4 and CD8 markers. Abbreviations: SSC-A = side scatter area; lymph = lymphocyte; CD19-CAR = CD19-specific chimeric antigen receptor; 7AAD = 7-aminoactinomycin D. Please click here to view a larger version of this figure.
| Run # | Starting Material | Vector | Culture Cytokines | AB Serum % | Final TNC | Total CAR-T Cells Harvested | Total Fold Growth | Average Daily Growth | Harvest Day | ||
| 1 | Patient T Cells, Apheresis, Cryopreserved |
huCART19 | IL2 | 5% | 2.75×109 | 2.24×108 | 14 | 45% | 7 | ||
| 2 | Patient T Cells, Apheresis, Cryopreserved |
huCART19 | IL2 | 5% | 4.15×109 | 1.14×108 | 21 | 46% | 8 | ||
| 3 | Patient T Cells, Apheresis, Cryopreserved |
huCART19 | IL2 | 5% | 4.20×109 | 8.40×107 | 21 | 46% | 8 | ||
Table 3: Parameters and growth characteristics of three clinical-scale CAR-T manufacturing runs. Details about culture conditions, growth, and harvest day for the first three patient manufacturing runs using the processor for the NCT05480449 clinical trial are shown. huCART19, humanized CD19-directed CAR lentiviral vector. Note that transduction efficiency and viability for these products are shown in Table 4. Abbreviations: CAR-T cells = chimeric antigen receptor T cells; TNC = total nucleated cells.
| Release Testing | ||||||||||
| Assay | Cell Viability | CD3 % | Endotoxin | Myco-plasma | Transduction Efficiency | Residual Leukemic Cells | Sterility Day 14 | Vector DNA Sequence per Cell | Vector DNA Sequence per Transduced Cell | Replication Competent Lentivirus |
| Specification | ≥ 70% | ≥ 80% | ≤ 3.5 EU/mL |
Negative | ≥ 2 % | < 1 % | No Growth | Report Result | 0.04-8 copies/ transduced cell |
< 50 copies/µg DNA |
| Results | ||||||||||
| Run 1 | 90 | 89 | <0.4 | Negative | 36 % | Not Detected |
No Growth |
1.04 | 2.89 | Not Detected |
| Run 2 | 88 | 93 | <0.4 | Negative | 39 % | Not Detected |
No Growth |
1.16 | 2.97 | Not Detected |
| Run 3 | 94 | 91 | <0.4 | Negative | 18 % | Not Detected |
No Growth |
0.43 | 2.39 | Not Detected |
Table 4: Final product tests and release specifications. Specifications shown in this table are for the NCT05480449 clinical trial (IND 28617). Post-thaw viability, CD3%, transduction efficiency, and residual leukemic cell content were measured by flow cytometry. Endotoxin was measured using an FDA-licensed limulus amebocyte lysate-based chromogenic test. Sterility testing was performed using a USP<71> compliant method. Mycoplasma, vector DNA sequence number per cell, and replication-competent lentivirus were measured using quantitative polymerase chain reaction (qPCR). Vector DNA sequence number per transduced cell was calculated by dividing the Vector DNA sequence number by the transduction efficiency. Abbreviations: EU = endotoxin unit; IND = investigational new drug; FDA = Food and Drug Administration.
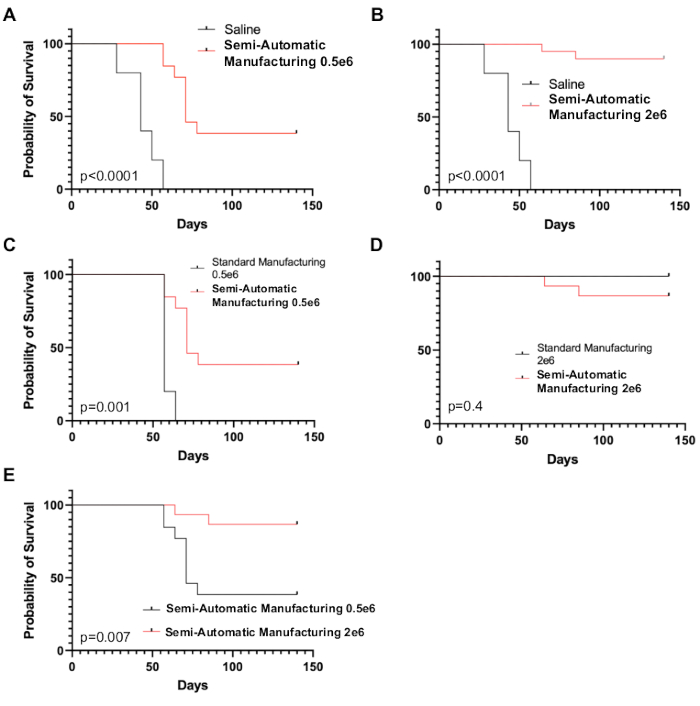
Supplemental Figure S1: Preclinical testing of CAR-T cells manufactured using the TCT process. Immunodeficient (NSG) mice were injected with patient-derived xenograft leukemia cells on day 0 and subsequently treated with CD19-directed CAR-T cells manufactured on the processor (“Semi-Automatic Manufacturing”), traditionally manufactured CD19-directed CAR-T cells (“Standard Manufacturing”), or saline on day 7. (A, B) Survival of mice treated with semi-automatic manufacturing CAR-T cells at two dose levels (0.5 × 106 or 2 × 106 CAR-T cells) compared to saline (p < 0.0001 for both dose levels). (C) Comparison of semi-automatic with standard manufacturing CAR-T cells at a dose level known to be non-curative for standard cells (0.5 × 106 CAR-T cells). Survival was significantly better in mice treated with CAR-T cells from the processor (p = 0.001). (D) Comparison of semi-automatic with standard manufacturing CAR-T cells at a dose level known to be curative for standard CD19 CAR-T cells. Survival was not significantly different (p = 0.4). (E) Comparison of CAR-T cells from the processor at the two dose levels. A significant dose effect was observed with better survival in mice treated with 2 × 106 huCART19 cells (p = 0.007). Each group contained 5 mice. Abbreviations: NSG = non-obese diabetic severe combined immunodeficiency gamma; TCT = T cell transduction; CAR-T = chimeric antigen receptor T cells. Please click here to download this File.
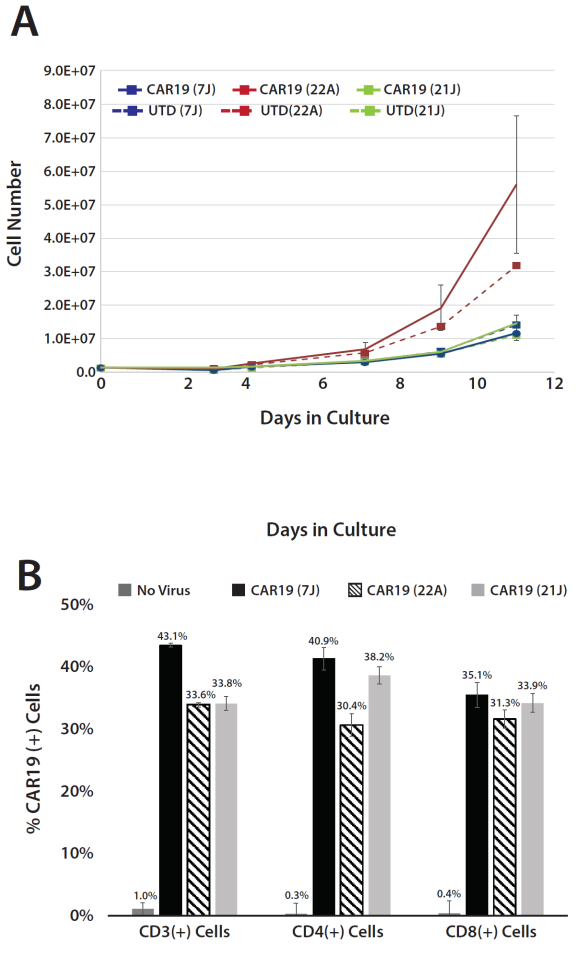
Supplemental Figure S2: Comparison of growth and lentiviral transduction of human T cells cultured in medium supplemented with different lots of human AB serum. (A) Expansion of human T cells untransduced (UTD) or transduced with CD19 CAR-T lentiviral vector (CAR19) in cultures supplemented with three different lots of human AB serum (7J, 22A, or 21J). Growth rates for one of the lots (22A) were substantially greater than that of the two others. (B) Mean percentage (3 replicates) of CAR19+ cells in the CD3+ and CD3+/CD4+ or CD3+/CD8+ subpopulations of ex vivo-expanded T cells derived from healthy donors. Note that differences in growth rates did not appear to influence the ability of the cells to take up lentivirus as determined by flow cytometric analysis of the cells from the expanded cultures. Transduction efficiency of T cells and CD4+ and CD8+ subpopulations was consistent from culture to culture. Error bars represent standard deviations of 3 repeat cultures. Please click here to download this File.
Discussion
CAR-T cell therapy has emerged as a promising treatment approach for B-cell and other malignancies. However, traditional CAR-T cell manufacturing methods have several limitations, such as high cost, labor-intensive production, and open steps that increase the risk of contamination. Recently, several semi-automated platforms, including the Miltenyi CliniMACS Prodigy (the "processor"), have emerged to address these limitations. The T cell transduction (TCT) process, integrated into the processor described in this manuscript, encompasses T cell enrichment, activation, viral transduction, culture expansion, and harvest in a closed system. Competing automated cell processor platforms are less widely used at this time but may have better in-process monitoring of culture parameters, favorable equipment or material cost, or a smaller footprint, which may allow use in a biosafety cabinet instead of a dedicated cleanroom space. It is important to choose a platform that is optimally suited for a given application and environment.
A significant limitation of cell and gene therapies such as CAR-T cell therapy is the high cost of commercial products, which limits access to life-saving therapy. This is especially the case in resource-poor settings and non-first-world countries. We have found that it is possible to manufacture CAR-T cells at a dramatically lower cost than a comparable commercial product when deploying a semi-automated process into the setting of an academic hospital that has existing current Good Manufacturing Practice (cGMP)-compliant cleanroom facilities in the context of a stem cell laboratory and the support of a clinical laboratory that can perform some of the in-process and release tests. Under these circumstances, we find that the marginal cost to perform a manufacturing run, including materials and labor for manufacturing and all in-process and release testing (but not including equipment and facilities) is approximately $30,000. It should be noted that the equipment capital cost of the processor is less than the cost of a single commercial CAR-T cell product.
One of the potential concerns with switching from a manual to an automated method is decreased flexibility. The TCT process, while programmed with strong guardrails, is still extensively customizable. The process offers multiple starting points, including a full process with T cell enrichment, a full process without enrichment (e.g., for use with CD4+/CD8+ pre-enriched cells, see section 5 of the Protocol above), and a resumption of an aborted process. Additionally, the Activity Matrix feature allows for the customization of the entire cultivation process. With the growing interest in rapid CAR-T manufacturing, it should be noted that is possible to configure the Activity Matrix to begin harvest as early as a few hours after transduction, if desired. The rapid procedure has the potential double benefit of decreased manufacturing time and a more potent product by avoiding differentiation and loss of anti-leukemic activity12. Furthermore, a variant of the TCT has been designed for non-viral vectors by using electroporation, which further increases the flexibility of the system.
The optimal harvest time for CAR-T cells depends on various factors, including the desired target dose, the number of doses required, and the maximum cell density that the culture vessel can support without compromising cell viability. The manufacturer recommends not exceeding 5 billion cells in a volume of 250 mL. Determining the desired target dose should be based on preclinical data in an animal model. For example, in this study, we used NOD scid gamma knockout (NSG) mice injected with patient-derived leukemic xenografts and treated with CAR-T cells to estimate that an effective dose would be approximately between 2 and 5 million CAR-T cells per kilogram (Supplemental Figure S1). Monitoring cell size can also be useful in determining the optimal time for harvesting CAR-T cells. By carefully considering these factors, researchers can optimize the harvest time of CAR-T cells to ensure that the final product is of high quality and efficacy.
Developing an appropriate release testing plan is a critical component of obtaining approval for an IND application. We present a set of release tests including specifications for our IND 28617, and we believe this will be a good starting point for similar products. However, the regulators' opinions are constantly evolving, and variations of the presented protocol may lead to regulators requiring different tests or different specification thresholds. We strongly recommend closely reading current regulatory guidance documents such as the FDA's Chemistry, Manufacturing, and Control (CMC) Information for Human Gene Therapy Investigational New Drug Applications (INDs)13. A general principle is that tests demonstrating the safety of a product are evaluated more stringently than tests demonstrating potency. Generally, tests such as sterility, endotoxin, mycoplasma, and replication-competent lentivirus should be validated, and their performance criteria should be known. In contrast, potency tests are relatively de-emphasized in early-phase trials and may not need to be fully validated. A recent FDA draft guideline states that "transgene expression alone as a measure of potency may be sufficient to support early-phase IND studies"14. In addition, tests that are used for dose determination should be validated. This may be problematic for new CAR targets for which there may not be any standardized flow assays or reference materials. For this case, the FDA suggests that "the reference material for an assay may be a well-characterized lot of the gene therapy product itself."13 For example, we established a positive control for the CD19 CAR flow assay using CAR-T cells from early engineering runs.
Developing and optimizing the manufacturing process for CAR-T cell therapy can be challenging and time-consuming, as we experienced in our work. We encountered several setbacks, including issues with bacterial contamination. We strongly advocate using a dry thaw method for thawing cells, as water baths can increase the risk of contamination. Because of cultivation, even a small inoculum can lead to frank contamination, resulting in an unusuable product. Additionally, we recommend planning logistics for release testing, which may involve additional validations and contracting with outside parties to ensure timely and accurate testing. Another important element when developing a CAR-T manufacturing process is optimizing transduction efficiency. An IND application should include data to demonstrate the potency of the viral vector. To meet this requirement and at the same time obtain information to optimize the MOI, we strongly recommend performing a small-scale titration of the clinical grade viral vector on human T cells. It is also important to consider the use of traditionally manufactured CAR-T cells as control for mouse experiments (as shown in Supplemental Figure S1). Obtaining these cells may be difficult and require careful planning. Another factor to consider is the use of human AB serum, which is a poorly defined reagent that can have a significant effect on cell growth and other characteristics of the product. To address this, we suggest testing multiple lots of AB serum and conducting small-scale experiments to assess lot-specific effects on cell growth (Supplemental Figure S2). Once a suitable lot is identified, a sufficiently large volume should be purchased to ensure consistency across the trial cohort and minimize the risk of batch effects in patient outcomes that are due to changes in serum lot.
In summary, the processor and the included TCT process offer a manufacturing method that can address several limitations of current CAR-T manufacturing procedures, including high costs, labor-intensive production, and open manipulation steps. By providing a closed and semi-automated system, this processor has the potential to improve the availability and affordability of CAR-T cell therapy. It can accommodate rapid manufacturing and non-viral vectors, making it a flexible alternative to traditional manufacturing methods. By carefully considering factors such as the desired target dose, maximum cell density, and the effects of human AB serum on cell growth, researchers can optimize the harvest time of CAR-T cells to ensure the quality and efficacy of the final product. Although the process can be complex and laborious, planning logistics for release testing and eliminating sources of contamination can help to mitigate risks and ensure successful CAR-T cell manufacture. Overall, the TCT process presents a promising avenue for CAR-T cell therapy, with the potential to overcome current limitations and improve patient accessibility, ultimately benefiting patients with cancer.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge the contributions of several individuals and organizations to this work. The Cell and Gene Therapy Laboratory and the Penn Translational and Correlative Studies Laboratory provided valuable assistance with process development and preparation for IND submissions. Melissa Varghese and Amanda DiNofia contributed to the process development and preparation for IND submissions that underly this manuscript. This work was supported by an Acceleration Grant of the Cell and Gene Therapy Collaborative of the Children's Hospital of Philadelphia. The authors would also like to thank Miltenyi Biotec for their technical and research support. Figure 1 is covered by copyright © 2023 Miltenyi Biotec B.V. & Co. KG; all rights reserved.
Materials
| 12 x 75 borosilicate tubes | Charles River | TL1000 | |
| 20 mL Reagent Bag | Miltenyi Biotec | 170-076-631 | |
| 50 mL Conical Tube | Fisher | 05-539-10 | |
| 150 mL Transfer Set | Fenwal | 4R2001 | |
| 2,000 mL Transfer Set | Fenwal | 4R2041 | |
| 7AAD | Fisher Scientific | BDB559925 | |
| Alcohol Prep | Tyco/Healthcare | ||
| Bag Access | Medline | 2300E-0500 | |
| CD19 APC-Vio770 REAfinity | Miltenyi Biotec | 130-113-643 | |
| CD19 CAR Detection Reagent Biotin | Miltenyi Biotec | 130-129-550 | |
| CD19 PE | BD | 555413 | |
| CD3 APC | BD | 340440 | |
| CD4 VioBright FITC REAfinity | Miltenyi Biotec | 130-113-229 | |
| CD45 VioBlue REAfinity | Miltenyi Biotec | 130-110-637 | |
| CD8 APC-Vio770 REAfinity | Miltenyi Biotec | 130-110-681 | |
| Cellometer Reference Beads 10um | Nexcelom | B10-02-020 | |
| Cellometer Reference Beads 15um | Nexcelom | B15-02-010 | |
| Cellometer Reference Beads 5um | Nexcelom | B05-02-050 | |
| Cellometer Slides | Nexcelom | CHT4-SD100-002 | |
| CliniMACS CD4 GMP MicroBeads | Miltenyi Biotec | 276-01 | The CD4 reagent |
| CliniMACS CD8 GMP MicroBeads | Miltenyi Biotec | 275-01 | The CD8 reagent |
| CliniMACS PBS/EDTA Buffer | Miltenyi Biotec | 130-021-201 | The buffer |
| DMSO | Origen | CP-10 | |
| Freezing Bag 50 mL | Miltenyi Biotec | 200-074-400 | |
| Freezing Vial, 1.8 mL | Nunc | 12565171N | |
| Freezing Vial, 4.5 mL | Nunc | 12565161N | |
| Human AB serum | Valley Biomedical | Sterile filtered, heat inactivated | |
| Human Serum Albumin 25% | Grifols | 68516-5216-1 | |
| Human Serum Albumin 5% | Grifols | 68516-5214-1 | |
| MACS GMP Recombinant Human IL-2 | Miltenyi Biotec | 170-076-148 | The cytokines |
| MACS GMP T Cell TransAct | Miltenyi Biotec | 200-076-202 | The activation reagent |
| MycoSeq Mycoplasma Detection Kit | Life Technologies | 4460623 | |
| Needles, Hypodermic 14G | Medline | SWD200573 | |
| Needles, SlideSafe 18G | BD | B-D305918 | |
| Pipet tips, 2-200 μL, individually wrapped | Eppendorf | 022492209 | |
| Pipet tips, 50-1000 μL, individually wrapped | Eppendorf | 022492225 | |
| Pipets 10 mL | Fisher | 13-678-27F | |
| Pipets 25 mL | Fisher | 13-675-30 | |
| Pipets 5 mL | Fisher | 13-678-27E | |
| Plasmalyte-A | Baxter | 2B2544X | The electrolyte solution |
| Prodigy TS520 Tubing Set | Miltenyi Biotec | 170-076- 600 | The tubing set |
| Sterile Field | Medline | NON21001 | |
| Streptavidin PE-Vio770 | Miltenyi Biotec | 130-106-793 | |
| Syringe 1 mL | BD | 309628 | |
| Syringe 10 mL | BD | 302995 | |
| Syringe 3 mL | BD | 309657 | |
| Syringe 30 mL | BD | 302832 | |
| Syringe 50 mL | BD | 309653 | |
| TexMACS GMP Medium | Miltenyi Biotec | 170-076-306 | The medium |
| Triple Sampling Adapter | Miltenyi Biotec | 170-076-609 | |
| Viral Vector | CHOP Clinical Vector Core | huCART19 | |
| Equipment | |||
| Biological Safety Cabinet | The Baker Co | ||
| Cellometer Auto 2000 | Nexcelom | ||
| CliniMACS Prodigy | Miltenyi Biotec | 200-075-301 | The processor |
| Controlled Rate Freezer | Planer/Kryosave | ||
| Endosafe nexgen-PTS150K | Charles River | ||
| Mettler Balance | Mettler | ||
| Refrigerated Centrifuge | Thermo Fisher | ||
| Refrigerated Centrifuge | Fisher Sci | ||
| SCD Sterile Tubing Welder | Terumo | ||
| Sebra Tube Sealer | Sebra | ||
| Varitherm | Barkey | The dry thaw device | |
| XN-330 Hematology Analyzer | Sysmex |
References
- Maude, S. L., et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. New England Journal of Medicine. 378 (5), 439-448 (2018).
- Shah, N. N., et al. Bispecific anti-CD20, anti-CD19 CAR T cells for relapsed B cell malignancies: A phase 1 dose escalation and expansion trial. Nature Medicine. 26 (10), 1569-1575 (2020).
- Maude, S. L., et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. New England Journal of Medicine. 371 (16), 1507-1517 (2014).
- Grupp, S. A., et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. New England Journal of Medicine. 368 (16), 1509-1518 (2013).
- Maude, S. L., et al. Efficacy of humanized CD19-targeted chimeric antigen receptor (CAR)-modified T cells in children and young adults with relapsed/refractory acute lymphoblastic leukemia. Blood. 128 (22), 217 (2016).
- Mock, U., et al. Automated manufacturing of CAR-T cells for adoptive immunotherapy using CliniMACS Prodigy. Cytotherapy. 18 (8), 1002-1011 (2016).
- Fernández, L., et al. GMP-compliant manufacturing of NKG2D CAR memory T cells using CliniMACS Prodigy. Frontiers in Immunology. 10 (10), 2361 (2019).
- Zhu, F., et al. Closed-system manufacturing of CD19 and dual-targeted CD20/19 chimeric antigen receptor T Cells using CliniMACS Prodigy device at an academic medical center. Cytotherapy. 20 (3), 394-406 (2018).
- Zhang, W., Jordan, K. R., Schulte, B., Purev, E. Characterization of clinical grade CD19 chimeric antigen receptor T cells produced using automated CliniMACS prodigy system. Drug Design, Development and Therapy. 12 (12), 3343-3356 (2018).
- Abou-El-Enein, M., et al. Scalable manufacturing of CAR T cells for cancer immunotherapy. Blood Cancer Discovery. 2 (5), 408-422 (2021).
- Miltenyi Biotec. . CliniMACS Prodigy User Manual. , (2021).
- Ghassemi, S., et al. Rapid manufacturing of non-activated potent CAR T cells. Nature Biomedical Engineering. 6 (2), 118-128 (2022).
- U.S. Department of Health and Human Services, Food and Drug Administration. . Chemistry, manufacturing, and control (CMC) information for human gene therapy investigational new drug applications (INDs) guidance for industry. , (2020).
- U.S. Department of Health and Human Services, Food and Drug Administration. . Considerations for the development of chimeric antigen receptor (CAR) T cell products draft guidance for industry. , (2022).

