Non-invasive Assay for Chlorophyll Biosynthesis Kinetics Determination during Early Stages of Arabidopsis De-etiolation
Summary
Here, we describe an advanced tool designed for chlorophyll biosynthesis monitoring during the early stages of Arabidopsis seedling de-etiolation. The novel methodology provides non-invasive real-time chlorophyll fluorescence imaging at high spatial and temporal resolution.
Abstract
Chlorophyll biosynthesis is a hallmark of de-etiolation, one of the most dramatic stages in the plant life cycle. The tightly controlled and highly dynamic process of chlorophyll biosynthesis is triggered during the shift from the dark to the light in flowering plants. At the moment when etiolated seedlings are exposed to the first traces of sunlight, rapid (in order of seconds) conversion of protochlorophyllide into chlorophyllide is mediated by unique light-accepting protein complexes, leading via subsequent metabolic steps to the production of fully functional chlorophyll. Standard techniques for chlorophyll content analysis include pigment extraction from detached plant tissues, which does not apply to studying such fast processes. To investigate chlorophyll kinetics in vivo with high accuracy and spatiotemporal resolution in the first hours after light-induced de-etiolation, an instrument and protocol were developed. Here, we present a detailed procedure designed for statistically robust quantification of chlorophyll in the early stages of Arabidopsis de-etiolation.
Introduction
De-etiolation represents the most dramatic phase in the plant life cycle, characterized by a number of morphological changes and complete rearrangement of plant metabolism (from hetero- to auto-tropic)1. Chlorophyll biosynthesis is a hallmark of light-induced de-etiolation in plants and a very dynamic process. Formation of chlorophyll from dark-produced precursor protochlorophyllide must be tightly coordinated to avoid damage due to reactive byproducts2. The protochlorophyllide reduction to chlorophyllide is catalyzed by light-dependent protochlorophyllide oxidoreductases (PORs), unique enzymes activated directly by light. The reaction is very fast, taking place in the order of ms to s3, leading to recognizable chlorophyll accumulation within minutes after etiolated seedling irradiation4,5,6. More time (from hours to days) is required for chloroplast biogenesis to establish a fully functional photosynthetic apparatus3.
Various methods exist to analyze chlorophyll content, including high-performance liquid chromatography (HPLC) or spectrophotometry. Usually, these techniques demand the destruction of plant tissue4,5,6, restricting the determination of changes in chlorophyll levels over time. Methods allowing non-invasive chlorophyll kinetics establishment may open a whole new perspective to study plants in diverse aspects ranging from fundamental research questions, such as analyzing the process of chlorophyll synthesis in time and space, to more practical applications, such as assessment of stress tolerance or effect of biostimulants on the chlorophyll kinetics. Considering this, we introduced a system for monitoring chlorophyll formation, iReenCAM7. It incorporates a CCD camera, emission filters, light sources, and a pipeline for automated fluorescence analysis (Figure 1). The main feature of the developed device is high spatial and temporal resolution, outperforming in the parameters used in current approaches, and sufficient sensitivity and specificity when compared with standard analytical methods7.
The non-invasive procedure described here requires minimum reagents and comprises simple steps, allowing to obtain a chlorophyll kinetics profile in living Arabidopsis seedlings during very early stages of de-etiolation. The protocol can be useful for the study of highly dynamic process of chlorophyl synthesis influenced by number of factors, both exogenous (salt, drought, biostimulants, heavy metals, etc.) and endogenous (typically associated with changes in the gene activity) in origin without a need to detach any plant tissue, thus avoiding additional stress.
Protocol
1. Medium preparation
- Prepare the cultivation medium by mixing 0.75 g of gelling agent with 50 mL of sterile deionized water in a glass bottle to achieve a 1.5% (w/v) concentration for one Petri plate (120 x 120 x 17 mm). Gently shake the mixture and then heat it in a microwave until boiling to dissolve the gelling agent (the solution becomes clear).
- Allow the medium to cool down to 58-60 °C before proceeding to the next steps. All subsequent steps must be performed under sterile conditions within a laminar-flow hood to prevent contamination.
- Use Petri plates with light-tight edges to avoid excessive actinic light reflection and high background autofluorescence during the measurements. For this, apply black adhesive tape (or other means available) to cover all the sides of the empty Petri plate (Figure 2).
- Perform sterilization of the plate(s) after tape application by irradiation with germicide UV lamp for 20-30 min.
- If the experiment involves chemical treatment (i.e., abiotic/biotic stressors, plant hormones and/or growth regulators, etc.), add the appropriate amount of corresponding chemical directly to the medium. Be aware of a chosen chemical stability being added to the media (for example, if the chemical is not thermostable, add to the medium when it is cooled down right before pouring it into plate). Mix the medium thoroughly by shaking to ensure an even distribution of the chemical.
- Avoid illumination of plates to UV light after the media is poured (would lead to oxygen radical production that might interfere with the experiment).
- Pour the prepared medium into the square Petri plate(s) and allow the medium to solidify at room temperature.
2. Seed surface sterilization and plant growth conditions
- Get the required amount of Arabidopsis thaliana Col-0 seeds from the stock balk (10-20 mg) and add it to a 2 mL microcentrifuge tube.
NOTE: No specific modifications are necessary while working with different Arabidopsis lines (ecotype/mutant lines). Revision of the sawing grid and measuring steps should be performed for other plant species taking into account difference in seed size, germination rate and seedling size. - Surface-sterilize Arabidopsis seeds by adding 70% ethanol to the tube for 2 min. Gently shake the tubes during sterilization.
- Remove ethanol by pipetting carefully, taking care not to lose any seeds. Wash the seeds by adding sterile water to the tube for 5 min to remove any residual ethanol (gently shake the tubes during the washing period).
- Let the seeds sediment to the bottom of the tube by gravity, remove the remaining water.
- Rinse the seeds again 2x with sterile water as described in step 2.3 to ensure they are free from any ethanol traits. Let the seeds sediment to the bottom of the tube by gravity, remove the remaining water.
- Add an equal volume of sterile water to the tube containing the seeds to create a seed-water suspension.
- Use a sowing grid to evenly distribute seed-water suspension of the given genotype on the selected areas of the medium plate (Figure 2 and Supplementary Figure 1). Distribute the seeds (approximately 30-40) in a row in each area using a wide pipette tip.
- Let the water dry in the seed areas for about 30 min, keeping the plate(s) open in a laminar flow hood to prevent contamination. Seal the plate with micropore tape and wrap it with aluminum foil.
- Stratify the seeds for 3 days at 4 °C in darkness to (depending on the ecotype used) overcome the seed dormancy and/or to promote uniform germination.
- Transfer the plate with stratified seeds to white light (150 µmol/m2/s) for 1 h to induce germination (unwrap the foil only for the light treatment).
- After the light treatment, wrap the plate with aluminum foil to protect the seeds from light and place it in a vertical position in a growth chamber and cultivate for 4 days in the dark at 21 ˚C.
3. Chlorophyll fluorescence measurement and analysis
- Turn on the iReenCAM system and ensure that the system is ready and properly configured in the automatically initiated PS server software (e.g., if there is enough storage space for the experiment data, if the fluorescence camera is connected to PC, etc.).
- Activate the Scheduler software to create the experimental plan for the measurement by clicking Experiments > New Experiment. Provide a descriptive name for the experiment and fill in the details (description).
- Set the required actions for the experiment by clicking Add Action which will lead to the schedule of experimental actions.
NOTE: The word action here means performing a complete experiment (i.e., including all the steps necessary to perform one plate measurement). - Specify the conditions for a single round measurement (i.e., the length of light/darkness period).
- By clicking Generate List define the time intervals between measuring rounds. Choose the time when the round will start and finish (4 h in total) and the intervals between the rounds (in the current setup one round every 2 min).
- Click Generate and ensure that the time frame and intervals between light impulses are correct by checking the list generated on the left side of the screen.
- Choose the measuring protocol (Supplementary Figure 2). Save all modifications to the database for future reference.
- Just before the measurement starts, use green light of low intensity (see Table of Materials) inside the dark room and adjust the level of the shelf inside the measuring chamber or perform other preparational steps before removing the foil from the Petri plate. Then switch off the light and transfer the plates into the measuring chamber in the complete darkness.
- Carefully remove the aluminum foil covering the Petri plate containing the 4-day-old seedlings. Place the Petri plate horizontally inside the device measuring chamber. Inside the chamber, induce actinic light pulses, and perform imaging according to the experiment plan (actions) set in steps 3.1-3.7.
NOTE: It is critical to avoid any illumination of the plates with etiolated seedlings before placing them into the measuring chamber. The manipulation with the plate with etiolated seedlings must be performed in the dark room/chamber (for a possible experimental setup see Supplementary Figure 3).
4. Data extraction and analysis
- After completing the measurement, open the corresponding experiment in the analyzer software.
- To analyze the fluorescence of the seedlings, generate two types of masks- a rough (tray) mask that covers the area where the seedlings are located, and a precise (plant) mask that covers only the tissue of interest (typically cotyledons).
- Generate a tray mask by clicking the Create New Tray Type option. Assign the appropriate genotype names to the respective areas on the plate image.
- For assigning genotype, choose an image number at Round and then click Load Image in the upper part of the screen. The image of the plate will be shown on the screen. By clicking on any of the set of buttons that represent different shapes drawing tools (Supplementary Figure 4), enter New Shape Mode that allows to draw the area of interest on the plate image. Choose the necessary areas (e.g., different genotypes on the plate) and provide their appropriate naming.
- Click Esc to exit the shape mode. Save the generated tray mask (after providing a name for it) by clicking Store Tray Type.
- Go back a step and apply the mask that was generated in previous steps by selecting its name from the Change by Tray Type option.
- To generate plant mask with high accuracy, use the image acquired after 180 min of measurement (round 91) to set the minimum threshold value for fluorescence signal intensity, enabling background noise subtraction. For this, remove the tick from Auto Threshold and set a Manual Threshold at 0 (Supplementary Figure 4).
- Click Preview to ensure that the tray mask covers all necessary areas (genotypes) on the plate image. For this choose round 91 and click Refresh Preview.
- Enter the Run menu by clicking Run. Run the analysis exclusively for round 91 by putting a tick only on round 91. Then choose the output path and click Start Analysis.
- After the analysis is finished the Finish menu will open automatically. Pick the executed round (it will be the only one) from experiments and click Switch Analyzer to Analyzed Data to export the data for this specific time point (round 91).
- Extract the .xsel file from the exported archive, as this file contains the essential plant mask information by clicking Open Export Part icon.
- Re-open the experiment by clicking on Open Local Analysis Part icon. Enter the Mask Builder menu again, click Load Image, choose round 1 and then Load from File in the upper right corner of the screen and load the previously extracted .xsel file. The image of the plate will be shown with the tray mask applied.
- Save the mask by clicking Store Tray Type and apply it by selecting its name in Change by Tray Type option.
- Generate the plant mask by adjusting the fluorescence signal intensity threshold. Increase the value of Manual Threshold till the mask generated in the Preview menu fits perfectly to the ROI (e.g., cotyledons) in each of the genotypes (Supplementary Figure 4). Check if the mask fits to all rounds of the measurements by scrolling throughout the rounds (checking the proper mask positioning at round 1, 61 and 121 should be enough) in the Preview menu.
- Perform the analysis for all measuring rounds and export data.
NOTE: The output file includes chlorophyll fluorescence values of a given genotype for each time point, enabling the construction of charts of choice and facilitating further statistical evaluation.
Representative Results
The typical output obtained using the newly developed procedure in the 4-day-old de-etiolated Arabidopsis seedlings of wild-type (WT), ecotype Columbia-0 (Col-0) is shown in Figure 3. Under control conditions (DMSO-supplemented MS media), the chlorophyll biosynthetic curve starts with an initial burst of the chlorophyll synthesis, in which the protochlorphyllide pool synthesized during the scotomorphogenic phase of the growth, is quickly converted to chlorophyll owing to the light-induced PORs7,8,9. The initial phase of fast chlorophyll accumulation takes approximately 10 min and is followed by a lag phase, during which the minima of the dark-synthesized protochlorophyllide are reached (approximately 30 min after irradiation; for the HPLC-measured protochlorophyllide curve, see7). During the lag phase, the chlorophyll biosynthetic genes are upregulated10, leading to light-induced production of protochlorophyllide. The newly synthesized protochlorophyllide is promptly converted to chlorophyll, detectable as exponential phase (in case of WT Col-0 starting at approximately 120 min after irradiation), finishing with another lag phase at approximately 4 h after the de-etiolated seedling irradiation (Figure 3). In the presence of 6-benzylaminopurine (BAP), the first significant differences are detectable during the exponential phase7, suggesting negative effect of BAP on the chlorophyll kinetics in later stages of chlorophyll biosynthesis (at about 2 h after the start of illumination with actinic light; Figure 3).
For comparison of different conditions and/or genotypes, normalization of the raw data is necessary. As no chlorophyll was detectable using HPLC under various conditions and/or different genotypes in etiolated seedlings, we performed the normalization (F/F0) to the T0 fluorescence levels (F0) measured for the corresponding treatment and/or genotype7. To demonstrate the importance of normalization, we present both raw data and the data normalized to the mean chlorophyll fluorescence value of control measured at T0 (F0; Figure 3A and Figure 3B, respectively).
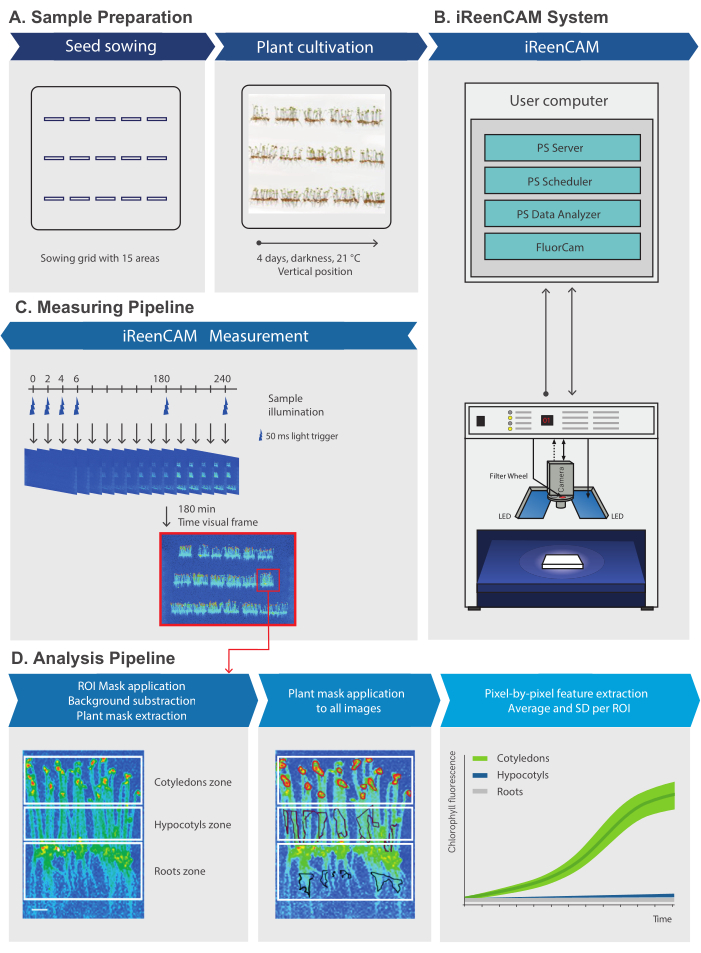
Figure 1: Measuring device protocol overview. The scheme of iReenCAM measurement and analysis pipeline. (A) Sample preparation by grid-defined seed sowing, stratification, light induction of germination and vertically oriented Petri plate cultivation in darkness. (B) Control module for automatic and programmable image acquisition and data management based on PlantScreen phenotyping SW toolbox organizes the operation of the entire system by controlling and synchronizing the HW operation with user defined measurement and analysis protocol. (C) The measuring protocol is designed for dynamic measurements of the fluorescent sample images in 2 min intervals for 4 h in total, i.e., 120 measuring rounds. Time visual frame of representative false-color image of vertically oriented 4-day-old Arabidopsis seedlings acquired at time 180 min is used for ROI mask generation. (D) Mask defining ROI for a tissue of interest (e.g., cotyledon, hypocotyl, or root zone) is applied on the time visual frame, background subtraction is performed and pixel by pixel fluorescence values for each ROI (defined by the plant mask) from all measurement rounds is extracted. Finally, the raw data (fluorescence F) is normalized to the mean fluorescence value at T0 (F0). Scale bars = 1 cm (A) and 0.25 cm (C). The figure was modified from7. Please click here to view a larger version of this figure.
Figure 2: Seed placement. The figure shows placing Arabidopsis seeds on the Petri plate with light-tight edges using the sawing grid. Please click here to view a larger version of this figure.
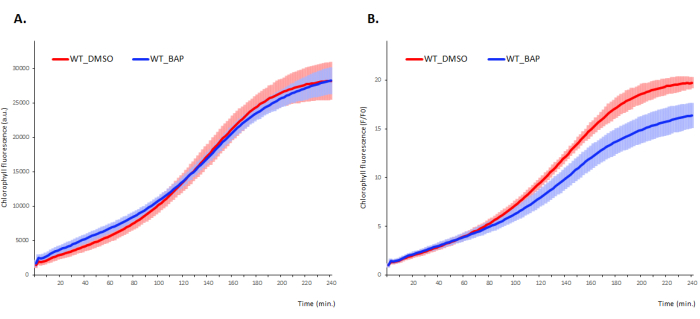
Figure 3: Chlorophyll accumulation kinetics in the early stages of Arabidopsis de-etiolation. Etiolated WT Col-0 seedlings were grown on media supplemented with BAP or DMSO (mock). (A) The mean value ± SD (shaded area), n=9 of raw data and (B) data normalized to the mean fluorescence value at T0 (F0). Please click here to view a larger version of this figure.
Supplementary Figure 1: Sowing grid. Left: Sowing grid with outlined rectangular boxes for placing the seeds of each genotype into a measuring spot located in the area of actinic light homogeneity (light intensity ≥ 0.7 of the maximum light intensity). Right: Schematic representation of 4 days old, etiolated Arabidopsis seedlings grown for the analysis. The sowing grid provides a possible scheme for Arabidopsis seeds positioning ensuring light homogeneity and proper seed density (each slot can be used for seed placement as the size of a slot, distance between the slots and the light homogeneity in the area of the grid are unified). Please click here to download this File.
Supplementary Figure 2: Fluorescence measuring protocol. Please click here to download this File.
Supplementary Figure 3: Experimental configuration allowing to avoid unwanted light exposure.(A) The measuring device is placed in the walk-in phytotron, (B) to the chamber separated by light-tight door. (C) The in vitro cultivation boxes (red arrowhead) dedicated for the etiolated seedlings growth under defined conditions (temperature and relative humidity) are placed just beneath the device (yellow arrowhead), ensuring the minimal risk of light exposure. The source of green dim light (blue arrowhead) is mounted on the wall next to the control PC (orange arrowhead). Please click here to download this File.
Supplementary Figure 4: Mask generation procedure in the workflow using PS data analyzer software. Print screenshots of individual steps to be performed for the tray mask (steps 4.3-4.6) and plant mask (step 4.12) generation and data analysis (steps 4.7 and 4.13). Please click here to download this File.
Supplementary Figure 5: Sowing density affecting measurement variability. Chlorophyll accumulation kinetics in 4-days-old, etiolated Arabidopsis WT Col-0 seedlings grown (A) separately (as individual seedlings, here n=5) or in a group of (B) high (HD, n=30-40) or (C) low density (LD, n=10-15). The n corresponds to the number of seedlings per slot of the sowing grid, data represent the mean values ± SD (shaded region). The high or low density corresponds to the number of seedlings per slot of the sowing grid as mentioned. Please click here to download this File.
Supplementary Figure 6: Cultivation interval and space requires species-specific optimization. Growth of various plant species using the Arabidopsis-optimized protocol. (A) Seeds placement on the sowing grid. (B) 4-day-old, etiolated seedlings of (from the left to the right) Arabidopsis thaliana, Brassica napus, and Crambe abyssinica. Please click here to download this File.
Discussion
Critical steps of the protocol and troubleshooting – no light and take care of the mask
As highlighted directly in the protocol description above, avoiding even the trace amounts of light both during cultivation of etiolated plants seedlings or just before starting the protocol is of critical importance11. In our setup, we use a dedicated dark chamber located in the walk-in phytotron and separated from the rest of the phytotron with light-tight rotating door (Supplementary Figure 3). The chamber is equipped with plant cultivation space and workbench allowing to accommodate the device together with control PC and fume hood. This allows us both to grow the plants in darkness and start the measurement without the need for plate transportation.
The way of plant cultivation and mask generation is critical for subsequent quantification of chlorophyll fluorescence signal via the assay proposed. One should avoid getting clumps of gelling agent or small visual trash/dust in the media as it might cause light reflection. As the recognition of chlorophyll fluorescence by the software is limited by the mask, a plant area that will not be covered by the mask will simply not be analyzed by the software. During the 4 h measurement, seedlings grow/move a bit, therefore designated mask could require adjustments as it may not fit through all the measurement rounds for the accurate signal intensity quantification. According to our experience, the imperfect mask definition seems to be the main source of variability. During the optimization experiments, we monitored the changes in the variability of chlorophyll fluorescence values taken for analysis in i) individual seedlings and a group of seedlings with ii) higher (30-40 seeds) and iii) lower (10-15 seeds) density sowing (Supplementary Figure 5). We used a higher density of seed sowing since it showed the lowest variability. The higher variability seen in case of individual seedlings/lower density sowing originates mostly from the higher proportion of pixels located at the edge between the signal and the background and the slight movement of the seedlings during the 4 h measurement interval.
To ensure that all measurements are accurate, check if the plant mask fits the first round, then to the 15th, 30th, 45th, 60th 75th and so on till the last one (by picking the number of the round and clicking Refresh Preview). This will take only a couple of minutes but will ensure that the whole cotyledon area is being covered and evaluated. If at any round the plant mask does not fit (the required area is not fully covered), divide the experiment into several parts. Then perform the protocol starting from Step 4 (4.1-4.13) to create a specific mask separately for each part of the experiment. For example, if you notice the displacement of the plant mask at the round 60-61 (or slightly earlier), divide the experiment into two parts – 1st part (1-60 rounds) and 2nd part (61-121 rounds). For the 1st part use the image of round 41 to generate a plant mask and for the 2nd part use round 91. At Step 4.13, when analyzing the data, be careful to choose the rounds according to the corresponding part of the experiment (e.g., rounds 1-60 for the first part and 61-121 for the second one, as in the aforementioned example) before clicking Analyze. When working with Arabidopsis, the movement of plants is negligible as they grow rather slowly but if applying the protocol for different species (see below), the growth pace should be taken into account.
Modifications and limitations
The number of parameters can be modified, including the intensity and wavelength of the actinic light and/or the light being applied between the intervals of actinic light application. The measuring device includes the integrated, fully motorized, and software-controlled filter wheel. Thus, the measuring algorithm suitable for the quantification of other pigments, typically also products of the tetrapyrolle biosynthetic pathway10,12, might be included in the protocol in case of adding the appropriate filters.
Also, as it was mentioned earlier, the protocol can be used not only for Arabidopsis plants. However, when working with other plant species, each step of the protocol should be revised accordingly taking into consideration the species-specific features including the germination and growth rate and/or the size (Supplementary Figure 6).
One of the important limitations of the protocol is the timespan, during which the chlorophyll quantification analysis can be performed. After chlorophyll becomes integrated into the photosystem complexes, the fluorescence signal becomes biased by the photosynthesis energy consumption (what is called the variable chlorophyll florescence is present) as has been observed during later stages of de-etiolation13. As assayed using OJIP transients assay14,15, no signs of photosynthetic activity was detectable using this experimental setup during the first 4 h of de-etiolation7. However, if the extended time period of photomorphogenesis is supposed/necessary to be assayed, the level of photosystems assembly and possible effect of photosynthesis on the overall fluorescence levels should be tested.
Finally, it should be mentioned that our protocol based on the fluorescence measurements, allows relative, not absolute chlorophyll quantification. If absolute quantification is needed, corresponding calibration must be performed using an alternative, e.g. HPLC approach.
Significance with respect to existing methods – simple, fast, and statistically robust chlorophyll quantification with high time and spatial resolution
The procedure described here allows the real-time detection and quantification of chlorophyll in living Arabidopsis seedlings during early stages of de-etiolation. Compared to other approaches mostly relying on chlorophyll extraction from detached plant material16,17 or recently developed optical methods18,19 this approach is purely non-invasive, allowing chlorophyll quantification by in vivo measurement of fluorescence intensity. Also, there is no need for additional reagents necessary for sample preparation as with other existing alternative methods including the aforementioned HPLC- or spectrophotometry-based approaches. The newly introduced protocol is simple, fast, and accurate, as previously verified using HPLC5. Using the standard protocol settings, the final curve of a single biological repeat is made out of 120 measuring points (fluorescence intensity means) taken during 4 h of the measurement, each consisting of up to 15 measuring spots. Typically, the final curve includes the data of three biological replicas (e.g., three independently prepared plates), and three measuring spots (three technical replicas), each consisting of 30-40 seedlings. Thus, there are around 300 seedlings assayed in each time interval, providing statistically robust dataset, allowing to reliably detect even small differences as demonstrated on mutants affected in various steps of chlorophyll biosynthesis7. Here we encourage the user to employ the recently developed statistical approach based on generalized linear mixed models combined with classical time series models as a suitable tool for the chlorophyll kinetics data analysis20.
Future applications of the technique – fast and cheap screening
The aforementioned features make this approach a useful tool suitable for fast and cheap screening and highly precise quantification of traits associated (directly or indirectly) with chlorophyll biosynthesis. This might include studies employing the forward genetic screening to better characterize the complex and multi-level regulations of chlorophyll biosynthesis10,12,21. Considering the possibility of various compound treatment, the protocol is also highly valuable to study, for example, the importance of light-mediated hormonal regulations22,23 or screening for low-molecular compounds with possible impact on chlorophyll accumulation kinetics.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported from the European Regional Development Fund-Project SINGING PLANT (No. CZ.02.1.01/0.0/0.0/16_026/0008446). This project has received funding through the MSCA4Ukraine project (ID 1233580), which is funded by the European Union. We are grateful to Lenka Sochurkova for the graphical design of Figure 1.
Materials
| 6-benzylaminopurine | Duchefa Biochemie | B0904.0001 | |
| Aluminum foil | Merck | Z691577 | |
| Arabidopsis thaliana Col-0 seeds | NASC collection | N1092 | |
| Cultivation chamber | PSI | custom made | |
| Dimethilsulfoxid | Thermo Fisher Scientific | 042780.AK | |
| Eppendorf single-channeled, variable (100-1000 μL) | Merck | EP3123000063 | |
| Gelrite | Duchefa Biochemie | G1101 | |
| iReenCAM device | PSI | custom made/prototype | |
| Laboratory bottles, with caps (Duran), 100mL | Merck | Z305170-10EA | |
| Laminar-flow box | UniGreenScheme | ITEM-31156 | |
| Linerless Rubber Splicing Tape, 19 mm width, black, Scotch | 3M Science. Applied to Life | 7000006085 | |
| Microcentrifuge tube, 2 mL with lid, PPT, BRAND | Merck | BR780546-500EA | |
| Micropore tape | 3M Science. Applied to Life | 7100225115 | |
| Osram lumilux green l18w/66 | Ovalamp | 200008833 | |
| Petri plates – Greiner dishes, square, 120 x 120 x17mm, vented | Merck | Z617679-240EA | |
| Pipet tips, 1000 μL, Axygen | Merck | AXYT1000B | |
| The Plant Screen Data Analyzer software | PSI | delivered as a part of the iReenCAM |
References
- Arsovski, A. A., Galstyan, A., Guseman, J. M., Nemhauser, J. L. Photomorphogenesis. Arabidopsis Book. 10, e0147 (2012).
- Reinbothe, S., Reinbothe, C., Apel, K., Lebedev, N. Evolution of chlorophyll biosynthesis–the challenge to survive photooxidation. Cell. 86 (5), 703-705 (1996).
- Heyes, D. J., et al. Photocatalysis as the ‘master switch’ of photomorphogenesis in early plant development. Nat Plants. 7 (3), 268-276 (2021).
- Hu, X. Y., Tanaka, A., Tanaka, R. Simple extraction methods that prevent the artifactual conversion of chlorophyll to chlorophyllide during pigment isolation from leaf samples. Plant Methods. 9 (1), 19 (2013).
- Chazaux, M., Schiphorst, C., Lazzari, G., Caffarri, S. Precise estimation of chlorophyll a, b and carotenoid content by deconvolution of the absorption spectrum and new simultaneous equations for chl determination. Plant J. 109 (6), 1630-1648 (2022).
- Marr, I. L., Suryana, N., Lukulay, P., Marr, M. I. Determination of chlorophyll-a and chlorophyll-b by simultaneous multicomponent spectrophotometry. Fresenius J Anal Chem. 352 (5), 456-460 (1995).
- Balakhonova, V., et al. Ireencam: Automated imaging system for kinetic analysis of photosynthetic pigment biosynthesis at high spatiotemporal resolution during early deetiolation. Front Plant Sci. 14, 1093292 (2023).
- Virgin, H. I., Kahn, A., Vonwettstein, D. The physiology of chlorophyll formation in relation to structural changes in chloroplasts. Photochem Photobiol. 2 (2), 83-91 (1963).
- Reinbothe, C., et al. Chlorophyll biosynthesis: Spotlight on protochlorophyllide reduction. Trends Plant Sci. 15 (11), 614-624 (2010).
- Kobayashi, K., Masuda, T. Transcriptional regulation of tetrapyrrole biosynthesis in arabidopsis thaliana. Front Plant Sci. 7, 1811 (2016).
- Wang, Y., Folta, K. M. Contributions of green light to plant growth and development. Am J Bot. 100 (1), 70-78 (2013).
- Brzezowski, P., Richter, A. S., Grimm, B. Regulation and function of tetrapyrrole biosynthesis in plants and algae. Biochim Biophys Acta. 1847 (9), 968-985 (2015).
- Pipitone, R., et al. A multifaceted analysis reveals two distinct phases of chloroplast biogenesis during de-etiolation in arabidopsis. eLife. 10, e62709 (2021).
- Stirbet, A. G. On the relation between the kautsky effect (chlorophyll a fluorescence induction) and photosystem ii: Basics and applications of the ojip fluorescence transient. J Photochem Photobiol B-Biol. 104 (1-2), 236-257 (2011).
- Kupper, H., et al. Analysis of ojip chlorophyll fluorescence kinetics and q(a) reoxidation kinetics by direct fast imaging. Plant Physiol. 179 (2), 369-381 (2019).
- Avin-Wittenberg, T., et al. Global analysis of the role of autophagy in cellular metabolism and energy homeostasis in arabidopsis seedlings under carbon starvation. Plant Cell. 27 (2), 306-322 (2015).
- Kósa, A., Preininger, &. #. 2. 0. 1. ;., Böddi, B. Nitrogen deficiency hinders etioplast development in stems of dark-grown pea (pisum sativum) shoot cultures. Physiol Plant. 155 (3), 330-337 (2015).
- Liang, Y., et al. A nondestructive method to estimate the chlorophyll content of arabidopsis seedlings. Plant Met. 13 (1), 26 (2017).
- Pérez-Patricio, M., et al. Optical method for estimating the chlorophyll contents in plant leaves. Sensors. 18 (2), 650 (2018).
- Spyroglou, I., et al. Mixed models as a tool for comparing groups of time series in plant sciences. Plants (Basel). 10 (2), (2021).
- Tanaka, R., Kobayashi, K., Masuda, T. Tetrapyrrole metabolism in arabidopsis thaliana. The Arabidopsis book / Am Soc Plant Biol. 9. 9, e0145 (2011).
- De Wit, M., Galvao, V. C., Fankhauser, C. Light-mediated hormonal regulation of plant growth and development. Annu Rev Plant Biol. 67, 513-537 (2016).
- Liu, X., Li, Y., Zhong, S. Interplay between light and plant hormones in the control of arabidopsis seedling chlorophyll biosynthesis. Front Plant Sci. 8, 1433 (2017).

