Drosophila Late Pupa Indirect Flight Muscle (IFM) Dissection: A Method for High-Throughput Tissue Collection
Abstract
Source: Kao, S. Y., et al. Dissection of Drosophila melanogaster Flight Muscles for Omics Approaches. J. Vis. Exp. (2019).
The Drosophila flight muscles are used as a model system for muscle development and physiology. This video describes the flight musculature of adults and highlights a protocol to dissect developing indirect flight muscles that are suitable for RNA sequencing from pupae.
Protocol
This protocol is an excerpt from Kao et al., Dissection of Drosophila melanogaster Flight Muscles for Omics Approaches, J. Vis. Exp. (2019).
1. IFM Dissection After 48 h APF
- Assemble necessary equipment including two #5 biology grade forceps, fine scissors, standard glass microscope slides, double-stick tape, pipette, pipette tips, dry ice, and (for RNA applications) isolation reagent (see Table of Materials). Chill the 1x PBS and microcentrifuge tubes on ice.
- Using a lightly wetted paintbrush, transfer the staged pupae to a strip of double-sided sticky tape mounted on a microscope slide (Figure 1A). Place the pupae in a line, oriented in the same orientation (ventral down and anterior towards the bottom of the slide).
NOTE: Be careful not to use too much water on the paintbrush or filter, or the pupae will not stick well. If pupae do not stick, dry them by first transferring to a dry filter or tissue paper. Mount as many pupae as can be dissected within a 30 min time window, ideally ~10 pupae. - Remove the pupa from the pupal case. Use forceps to tease apart and open the pupal case above the anterior spiracles (Figure 1B).
- Gently slide a pair of forceps dorsally towards the posterior, cutting the pupal case as the forceps move (Figure 1B'). Be careful not to rupture the underlying pupa. Liberate the pupa from the opened case and immediately transfer it to a drop of 1x PBS on a second microscope slide (Figure 1B",C).
- Repeat steps 1.3 and 1.4 for all pupae in the line, then set the double-stick tape slide aside.
- Using the fine scissors, cut the abdomen of the pupa away from the thorax and push it into a separate pile (Figure 1D,D'). Repeat for the remaining pupae.
NOTE: Begin timing the length of dissection with step 1.6, as soon as pupal integrity is disrupted. Dissect as many flies as possible in 20–30 min to prevent cell death and associated transcriptomic and proteomic changes. When dissecting 1 d adults or >90 h pupae, it is often convenient for later steps to additionally remove the head with the fine scissors. - Using a tissue paper, remove the majority of the 1x PBS (generally cloudy with suspended fat) as well as the pile of abdomens (Figure 1E). Add a drop of fresh, chilled 1x PBS to the remaining thoraxes.
- Use the scissors to cut the thorax in half (Figure 1F,F') by cutting from the head down the longitudinal body axis in a single motion. Alternately, if the head has been removed, first insert the scissors where the head was attached and cut the top half of the thorax longitudinally between the IFMs. Then, cut the ventral side of the thorax with a second cut in the same orientation.
- Repeat steps 1.7 and 1.8 for all pupae to be dissected, generating a pile of thorax hemisections near the center of the slide. Ensure there is enough chilled 1x PBS on the slide so that the hemisections do not dry out.
NOTE: After 48 h APF, IFMs are large enough to be visible under a standard dissecting microscope to the trained eye. At this point in the protocol, muscles with a fluorescent label can be moved to a fluorescent dissecting scope to aid in IFM identification or for training purposes, but this is not necessary. - Dissect the IFMs out of the thorax. Isolate one of the hemisections using the #5 forceps (Figure 1G,H). Gently insert the tips of one forceps above and below the middle of the IFMs (Figure 1G',H'). While holding the first forceps still, use fine scissors to cut one end of the IFM away from the cuticle and tendons. Then, cut the other end of the IFM free from the cuticle (Figure 1G'',H'').
NOTE: Depending on the orientation of the thorax after the first IFM cut, it is useful to rotate the thorax 180° so that the second IFM cut is easier to perform. - Remove the IFM bundle from the thorax with forceps (Figure 1G''',H'''), transferring it to the edge of the PBS bubble to use water tension to hold it in place (Figure 1I). Push the carcass to the opposite side of the slide. Repeat for the remaining thorax hemisections, generating a collection of dissected IFMs.
NOTE: If the IFMs do not stay in a neat pile, remove some of the 1x PBS with a tissue. Be careful not to let all of the PBS evaporate, and ensure that the dissected IFMs and hemithoraxes remain covered by buffer. - After dissecting all IFMs, quickly perform a quality control on the dissected muscle. Using #5 forceps, remove any jump muscle or cuticle fragments that may have found their way into the sample (Figure 1J-K'').
NOTE: Jump muscle appears different from IFM. If dissecting Mef2-Gal4 labeled muscle under fluorescence, jump muscle has a weaker fluorescence and a different shape and texture. Under normal light, it appears nearly translucent while the IFMs are an opaque, milky yellow (Figure 1J-J'',K). - Using water tension, capture (but do not squish) the dissected IFMs between a pair of forceps (Figure 1L). Transfer the IFMs to a 1.5 mL microcentrifuge tube pre-filled with 250 μL of chilled 1x PBS (Figure 1M). Proceed immediately with section 2.
NOTE: When forceps tips are brought into proximity of each other and lifted out of a buffer solution, water tension causes a bubble of buffer to be captured between the forceps tips. If IFMs are also present in this bubble, they can be lifted out of the solution and easily transferred to another buffer-filled receptacle. It is important to squeeze the forceps to bring the tips near one another without touching each other, to avoid macerating the tissue captured in the buffer bubble.
2. Pellet and Preserve the IFM Sample
- Pellet the IFMs by centrifuging the 1.5 mL microcentrifuge tube for 3–5 min at 2,000 x g in a table-top centrifuge (Figure 2A,B).
- Remove the buffer using a pipette tip (Figure 2C).
- For RNA applications, resuspend the IFM pellet in 50–100 μL of the desired RNA isolation buffer (see Table of Materials, Figure 2D). Otherwise, proceed to step 2.4.
NOTE: IFMs can be dry-frozen after step 2.2 for mass spectrometry preparations or isolation of RNA with commercial kits (see representative results). For RNA applications, better results are obtained by immediately resuspending and freezing the IFM pellet in isolation buffer. - Freeze sample on dry ice or snap freeze in liquid nitrogen (Figure 2E). Store at -80 °C until ready for subsequent steps in sample preparation for downstream analysis.
NOTE: After cryopreservation, samples can be stored for several months before processing for downstream investigation.
Representative Results
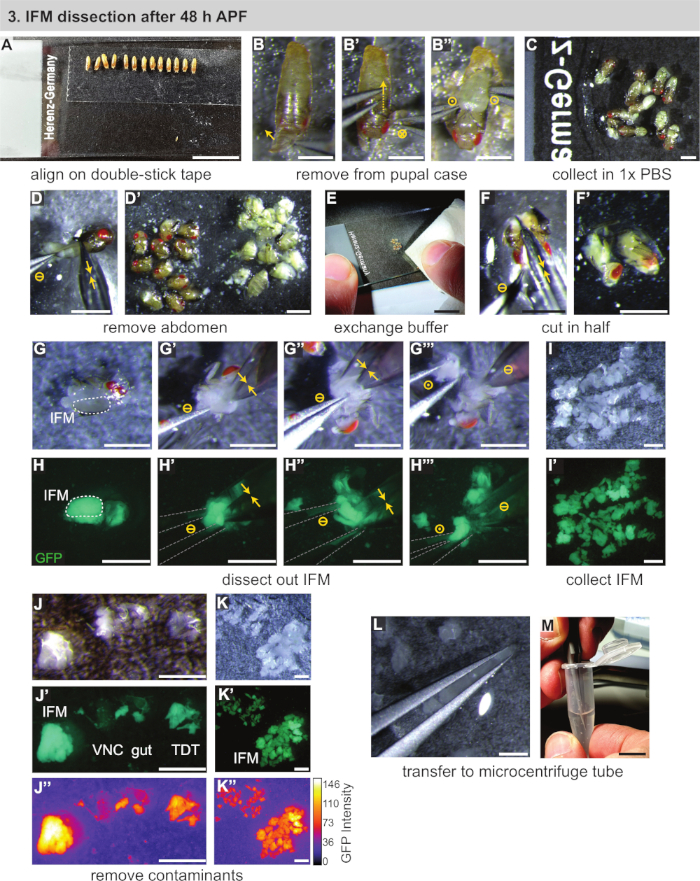
Figure 1: Dissection of IFMs after 48 h APF. (A) Aligning of pupae on double-stick tape. (B) Removal of pupae from the pupal case by opening anteriorly, cutting the case dorsally (B'), and lifting out the pupa (B''). Circle symbols represented the same as Figure 2. (C) Transfer of pupae to buffer. (D) Removal of the abdomen by cutting with scissors (yellow double arrows) and separation from thoraxes (D'). (E,F) Addition of clean buffer (E), then cutting of thoraxes in half longitudinally (F,F'). (G,H) Dissections can be performed under white light (G) or fluorescence to visualize the GFP (H); cutting of the IFMs on one side (G'), then the other side (G''); lifting out of the thorax with forceps (outlined in grey) (G'''). (I,J,K) Collection of IFMs in buffer (I) and removal of contaminating ventral nerve cord (VNC), gut, and jump muscle (TDT) (J) to generate a clean IFM sample (K). TDT has lower GFP expression and a different shape than IFM fibers (J'', K'). (L,M) Use of forceps to transfer IFMs (L) to a microcentrifuge tube (M). Scale bars = 1 cm (A,E,M), 1 mm (B-D',F-L). Please click here to view a larger version of this figure.
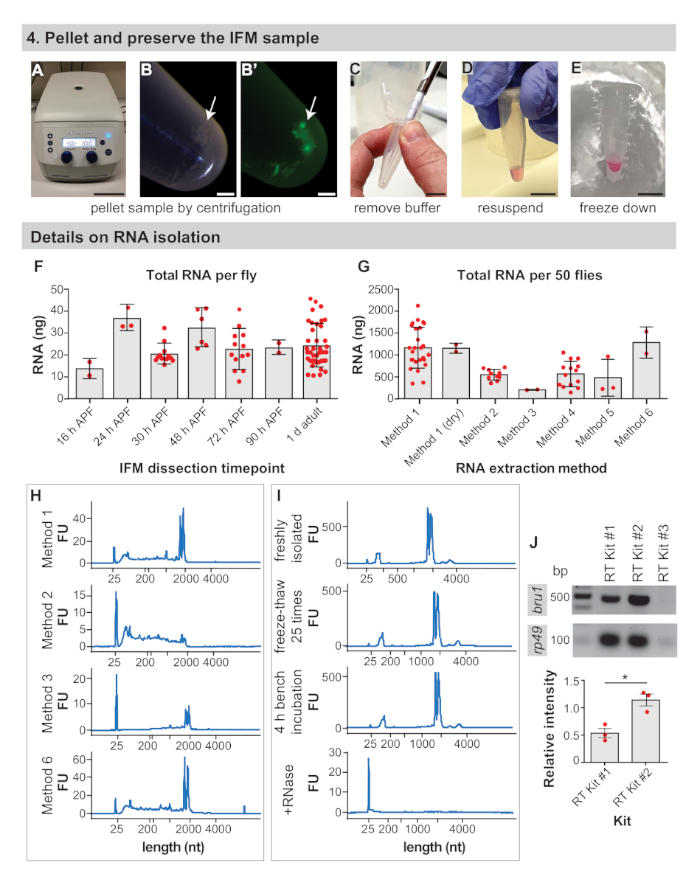
Figure 2: IFM preservation and RNA isolation details. (A) IFMs are pelleted by centrifugation for 5 min at 2000 x g. (B) IFM pellet (arrow) and pellet under fluorescence (B'). (C) Removal of all buffer with a pipette tip. (D) For RNA extraction, resuspension of pellet in isolation buffer. This step can be skipped to dry-freeze dissected IFMs. (E) Freezing of sample in liquid nitrogen or on dry ice and storage at -80 °C. Scale bars = 10 cm (A), 1 mm (B,B'), 1 cm (C,D,E). (F) Nanograms (ng) of total RNA from dissected IFM obtained per fly at 16 h APF, 24 h APF, 30 h APF, 48 h APF, 72 h APF, 90 h APF, and 1 d adult. Error bars = SD. (G) Total RNA isolated from IFM dissected from 50 1 d adult flies using different extraction methods. Error bars = SD. (H) Representative traces to assay RNA integrity after different extraction methods. The ribosomal bands run just below 2000 nucleotides (nt) and the marker band at 25 nt. Additional traces available in Supplemental Figure 1. (I) Representative traces of a freshly isolated RNA sample (top), a sample freeze-thawed 25x on dry ice (second plot), a sample left for 4 h on the bench (third plot), and a sample treated with RNase A (bottom plot). Note complete degradation of RNA upon addition of RNase A. (J) RT-PCR gel from kits as labeled for bru1 and rp49. The relative intensity of the bru1 band normalized against rp49 is plotted below. Error bars = SEM (unpaired t-test, p = 0.0119). Please click here to view a larger version of this figure.
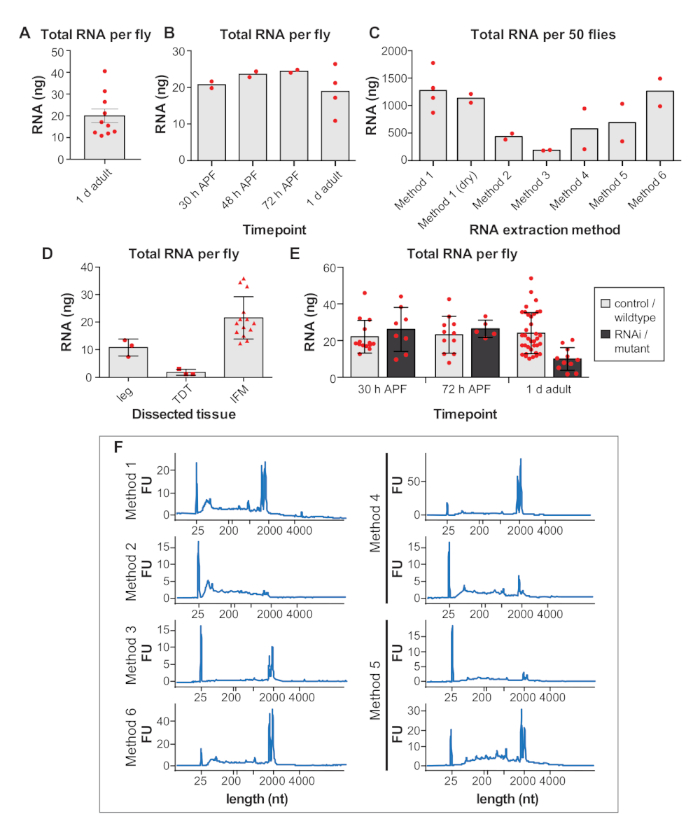
Supplemental Figure 1. (A,B,C) RNA yields from samples of the same genotype dissected by the same researcher in the same week. After all samples were dissected, RNA was isolated and measured the same day. (A) Nanograms (ng) of total RNA obtained from IFM dissections per 1 d adult fly. Error bars = SEM. (B) Total RNA obtained from dissected IFM per fly at 30 h APF, 48 h APF, 72 h APF and 1 d adult. (C) Total RNA isolated from IFM dissected from 50 1 d adult flies using different extraction methods. (D) Total RNA concentrations per fly from dissected legs, jump muscle (TDT) and IFM. More RNA is obtained from the larger IFMs. Error bars = SD. (E) Total RNA concentrations per fly of IFM dissected from controls compared to RNAi or mutant samples at 30 h APF, 72 h APF and 1 d adult. For mutants, w1118 was used as wildtype control. Mutant data are compiled from bru1-IR, salm-/- and another RNA-binding protein mutant. Note that for these manipulations, RNA yields are decreased in 1 d adult due to muscle atrophy and loss, so more flies need to be dissected to obtain sufficient quantities for omics approaches. Errors bars = SD. (F) Additional traces showing RNA integrity for the RNA isolation methods shown in Figure 2G and in C. Please click here to view a larger version of this figure.
Materials
| 60 mm culture dishes | Sigma-Aldrich | Z643084-600EA | Greiner dishes, 60 mm x 15 mM, vented |
| Cell phone camera, Samsung Galaxy S9 | Samsung | SM-G960F/DS | used for photos not taken under a microscope |
| Double stick tape | Scotch/3M | 3M ID 70005108587 | Double-sided tape, available at most office supply handlers |
| Dumont #5 Forceps | Fine Science Tools | 11252-20 | Inox straight tip 11 cm forceps, Biology grade with 0.05 mm x 0.02 mm tip |
| fluorescent dissecting microscope camera, Leica DFC310 FX camera | Leica | www.leica-microsystems.com | |
| Fluorescent dissecting microscope software, Leica Application Suite (LAS) version 4.0.0 | Leica | www.leica-microsystems.com | |
| Fluorescent dissecting microscope, Leica M165 FC | Leica | www.leica-microsystems.com | |
| Fly: Bru1[M2] | Fly stock; This paper | ||
| Fly: Bru1[M3] | Fly stock; This paper | ||
| Fly: Mef2-GAL4 | Bloomington Stock Center | BDSC:27390 | Fly stock |
| Fly: salm[1] | Bloomington Stock Center | 3274 | Fly stock |
| Fly: salm[FRT] | Fly stock; see Spletter et al., Elife, 2018 | ||
| Fly: UAS-Bru1IR | Vienna Drosophila Research Center | GD41568 | Fly stock, RNAi hairpin |
| Fly: UAS-GFP::Gma | Bloomington Stock Center | BDSC:31776 | Fly stock |
| Fly: UAS-mCD8a::GFP | Bloomington Stock Center | BDSC:5130 | Fly stock |
| Fly: w[1118] | Bloomington Stock Center | 3605 | Fly stock |
| Fly: weeP26 | Fly stock; see Clyne et al., Genetics, 2003 | ||
| GFP detection reagent, GFP-Booster | ChromoTek | gba488-100 | |
| Glycogen | Invitrogen | 10814-010 | |
| Image processing software, Photoshop CS6 | Adobe | www.adobe.com | |
| Isopropanol | Sigma-Aldrich | I9516-25ML | 2-propanol |
| Method 1 (RNA isolation): TRIzol | Life Technologies | 15596018 | Guanidinium isothiocyanate and phenol monophasic solution |
| Method 2 (RNA isolation): Method 1 + TURBO DNA-free Kit | Invitrogen | AM1907 | TRIzol isolation followed by treatment with a kit to remove DNA |
| Method 3 (RNA isolation): Direct-zol RNA Miniprep Plus Kit | Zymo Research | R2070S | RNA isolation in TRIzol, but over commercial columns instead of using phase separation. Recommended DNase treatment performed with Monarch Dnase I in Monarch DNase I Reaction buffer. |
| Method 4 (RNA isolation): RNeasy Plus Mini Kit | Qiagen | 74134 | We used the provided DNase treatment. IFM pellets were homogenized in RTL buffer as suggested for animal tissues. |
| Method 5 (RNA isolation): ReliaPrep RNA Tissue Miniprep System | Promega | Z6110 | We applied the protocol for 'Purification of RNA from Fibrous Tissues'. |
| Method 6 (RNA isolation): Monarch Total RNA Miniprep Kit | New England Biolabs | T2010G | We applied the protocol for tissues/leukocytes and lysed in 300 µL of RNA Protection Reagent. |
| Microcentrifuge tubes | Thermo Fisher | AM12400 | RNase-free Microfuge Tubes, 1.5 mL |
| Microscope slides | Thermo Fisher | 12342108 | Standard slides, uncharged, 1 mm |
| Paintbrush | Marabu | 1910000000 | Marabu Fino Round No. 0, or similar brush from any art supply |
| Paraformaldehyde | Sigma-Aldrich | 158127 | |
| PBS buffer (1x) | Sigma-Aldrich | P4417 | Phosphate buffered saline tablets for 1 L solutions, pH 7.4 |
| PFA PureTip Pipette Tips | Elemental Scientific | ES-7000-0101 | Optional substitute for standard pipette tips to reduce sample loss; 100 mL, 0.8 mm orifice |
| Pipette tips | Sigma-Aldrich | P5161 | Universal 200 mL pipette tips |
| RNA concentration Approach 1 & RNA integrity traces, Bioanalyzer | Agilent Technologies | G2939BA | |
| RNA concentration Approach 2, Nanodrop | Thermo Fisher | ND-2000 | |
| RNA concentration Approach 3, Qubit 4 Fluorometer | Invitrogen | Q33238 | |
| RNase A | Promega | A7937 | |
| RNase-free water, Diethyl pyrocarbonate (DEPC) | Sigma-Aldrich | D5758 | DEPC treat water overnight and then autoclave, to remove all RNase. |
| RT Kit #1: Super Script III Reverse Transcriptase Kit | Invitrogen | 18080-044 | reverse transcription kit |
| RT Kit #2: LunaScript | New England Biolabs | E3010S | reverse transcription kit |
| RT Kit #3: QuantiNova Reverse Transcription Kit | Qiagen | 205410 | reverse transcription kit |
| Statistical software: GraphPad Prism | GraphPad Prism | www.graphpad.com | |
| Statistical software: Microscoft Excel | Microsoft | Purchased as part of the bundle: Office Home & Student 2019 | |
| Table-top centrifuge | Eppendorf | 5405000760 | Eppendorf Centrifuge 5425 or equivalent |
| Tissue/ Kimwipes | Sigma-Aldrich | Z188956 | Standard tissue wipes |
| Transfer pipette | Sigma-Aldrich | Z350796 | Plastic pipette |
| Vannas spring scissors | Fine Science Tools | 15000-00 | 3 mm cutting edge, tip diameter 0.05 mm, length 8 cm |
| Whatman paper | Sigma-Aldrich | 1004-070 | Filter paper circles, Grade 4, 70 mm |

