Co-culture of Living Microbiome with Microengineered Human Intestinal Villi in a Gut-on-a-Chip Microfluidic Device
Summary
We describe an in vitro protocol to co-culture gut microbiome and intestinal villi for an extended period using a human gut-on-a-chip microphysiological system.
Abstract
Here, we describe a protocol to perform long-term co-culture of multi-species human gut microbiome with microengineered intestinal villi in a human gut-on-a-chip microphysiological device. We recapitulate the intestinal lumen-capillary tissue interface in a microfluidic device, where physiological mechanical deformations and fluid shear flow are constantly applied to mimic peristalsis. In the lumen microchannel, human intestinal epithelial Caco-2 cells are cultured to form a 'germ-free' villus epithelium and regenerate small intestinal villi. Pre-cultured microbial cells are inoculated into the lumen side to establish a host-microbe ecosystem. After microbial cells adhere to the apical surface of the villi, fluid flow and mechanical deformations are resumed to produce a steady-state microenvironment in which fresh culture medium is constantly supplied and unbound bacteria (as well as bacterial wastes) are continuously removed. After extended co-culture from days to weeks, multiple microcolonies are found to be randomly located between the villi, and both microbial and epithelial cells remain viable and functional for at least one week in culture. Our co-culture protocol can be adapted to provide a versatile platform for other host-microbiome ecosystems that can be found in various human organs, which may facilitate in vitro study of the role of human microbiome in orchestrating health and disease.
Introduction
The human intestine harbors a stunningly diverse array of microbial species (<1,000 species) and a tremendous number of microbial cells (10 times more than the human host cells) and genes (100 times more than the human genome)1. These human microbiomes play a key role in metabolizing nutrients and xenobiotics, regulating immune responses, and maintaining intestinal homeostasis2. Not surprisingly, given these diverse functions, the commensal gut microbiome extensively modulates health and disease3. Thus, understanding the role of gut microbiome and host-microbe interactions are of great importance to promote gastrointestinal (GI) health and explore new therapeutics for intestinal disorders4. However, existing in vitro intestine models (e.g., static cultures) restrict host-microbiome co-culture to a short period of time (<1 day) because microbial cells overgrow and compromise intestinal barrier function5. Surrogate animal models (e.g., germ-free6 or genetically engineered mice7) are also not commonly used to study host-gut microbiome crosstalk because the colonization and stable maintenance of human gut microbiome are difficult.
To overcome these challenges, we recently developed a biomimetic human "Gut-on-a-Chip" microphysiological system (Figure 1A, left) to emulate the host-gut microbiome interactions that occur in the living human intestine5,8. The gut-on-a-chip microdevice contains two parallel microfluidic channels separated by a flexible, porous, extracellular matrix (ECM)-coated membrane lined by human intestinal epithelial Caco-2 cells, mimicking the intestinal lumen-capillary tissue interface (Figure 1A, right)9. Vacuum-driven cyclic rhythmical deformations induce physiological mechanical deformations that mimic changes normally induced by peristalsis (Figure 1A, right). Interestingly, when Caco-2 cells are grown in the gut-on-a-chip for more than 100 hr, they spontaneously form three-dimensional (3D) intestinal villi with tight junctions, apical brush borders, proliferative cells limited to basal crypts, mucus production, increased drug metabolizing activity (e.g., cytochrome P450 3A4, CYP3A4), and enhanced glucose reuptake8. In this 'germ-free' microenvironment, it was possible to co-culture the probiotic Lactobacillus rhamnosus GG or a therapeutic formation of a probiotic bacterial mixture with host epithelial cells for up to two weeks5,10.
In this study, we describe the detailed protocol to perform host-gut microbiome co-culture in the gut-on-a-chip device for an extended period. In addition, we discuss critical issues and potential challenges to be considered for a broad application of this host-microbiome co-culture protocol.
Protocol
1. Microfabrication of a Gut-on-a-chip Device
Note: The gut-on-a-chip is a microfluidic device made by transparent, gas-permeable silicone polymer (polydimethylsiloxane, PDMS), containing two parallel microchannels (1 mm width x 150 µm height x 1 cm length) separated by a flexible porous (10 µm in pore diameter, 25 µm in spacing pore to pore) PDMS membrane5,9. Fabricate the gut-on-a-chip (Figure 1A, left) following the steps provided.
- Microfabrication Procedure of the Gut-on-a-chip5,9.
- Prepare uncured, degassed PDMS by mixing the PDMS prepolymer and the curing agent (15:1 w/w), and place it in the vacuum desiccator for 30 min.
- Pour 30 g and 3 g of uncured, degassed PDMS onto each silicon mold that has SU-8 based micropatterns of the upper and the lower microchannels of the gut-on-a-chip, respectively. Then cure at 60 °C in a dry oven for at least 4 hr.
- Demold the upper and lower PDMS layers from each silicon mold by cutting around the edge using a surgical scalpel. Punch 6 holes (two inlets, two outlets, and two vacuum chambers) using a biopsy puncher (2.0 mm in hole diameter).
- Prepare the porous membrane by pouring 10 g of uncured and degassed PDMS on a silanized silicon wafer containing post arrays with circular pillars (10 µm in diameter, 25 µm in height) overlaying with a flat, silanized PDMS support layer (15:1, w/w; 1 cm thick). Place a 3 kg weight presser on the setup and cure the polymer at 60 °C for 12 hr or longer.
- Demold the setup of a porous PDMS membrane adhered to the PDMS support slab from the silicon wafer by carefully lifting up the corner of the slab from the wafer, and peeling off until the porous PDMS membrane is completely detached from the wafer.
- Expose the channel side of an upper layer (PDMS, 15:1, w/w) and the porous membrane side to the plasma generated by a handheld corona treater for 1 min, and 3 sec, respectively.
- Place the plasma-treated surfaces of the porous PDMS membrane and upper microchannel layer in a conformal contact by placing each piece in parallel without any air bubbles.
- Incubate the whole setup at 80 °C O/N for irreversible bonding of the two PDMS layers.
- Peel off the PDMS support layer from the assembly of an upper layer with a porous membrane by carefully lifting up the corner of the support layer, and peeling off until the support layer is completely detached from the upper layer with the membrane.
- Tear off the portions of this membrane located over the vacuum chambers (the two chambers located on either side of the main channel) using a fine-tip tweezers to make hollow vacuum chambers.
- Expose the side with the membrane attached on the top piece and the side with the channels engraved on the lower piece to plasma in a same way as described in step 1.1.6 for 1 min.
- Align the upper microchannel layer with a porous membrane and the lower layer with a conformal contact by placing each piece in parallel without any air bubbles under a stereoscope.
- Cure the whole setup in a dry oven at 80 °C O/N to produce a complete scale gut-on-a-chip microfluidic device containing two hollow vacuum chambers next to the microfluidic cell channel.
- Connect tubing to the inlet and outlet of each port linked to the upper or the lower microchannels in a gut-on-a-chip via a 90° bent stainless steel blunt-end needle.
- Connect the tubing to the holes linked to the vacuum chambers using a 90° bent stainless steel blunt-end needle.
- Sterilize the microchannels and tubing by flowing 70% (v/v) ethanol using a 1 ml sterilized disposable syringe.
- Dry out the microchannels and tubing in a 60 °C dry oven O/N.
2. Growth of Microengineered Intestinal Villi in the Gut-on-a-chip Device
- Seed Intestinal Epithelial Cells (e.g. Caco-2BBE) on a Gut-on-a-chip Microdevice5,9.
- Sterilize the device with 70% (v/v) ethanol (see step 1.4) using a 1 ml sterilized disposable syringe followed by drying in a 60 °C dry oven O/N (see step 1.5) prior to the surface activation.
- Expose the complete setup of a gut-on-a-chip microsystem (i.e., a body of the gut-on-a-chip connected to tubing) to UV light (253.7 nm) and ozone simultaneously for 40 min.
- Cool down the device at RT for 15 min under UV light in a biosafety cabinet (BSC).
- Introduce 100 µl of the ECM coating solution- a mixture of 50 µg/ml type I collagen and 300 µg/ml extracellular matrix mixture diluted in the serum-free Dulbecco's Modified Eagle Medium (DMEM) – into both upper and lower microchannels using a disposable, sterile 1 ml syringe.
- Incubate the whole setup in a 37 °C humidified 5% CO2 incubator for 1 hr.
- Degas 50 ml of pre-warmed (37 °C) complete culture medium (DMEM, 20%, v/v, fetal bovine serum (FBS), 100 units/ml penicillin, and 100 µg/ml streptomycin; aliquoted in a 3 ml disposable syringe) using a 50 ml filtration system (0.45 µm in pore size) in the BSC for 1 min.
- Pass the pre-warmed medium through the filtration system and tap gently on the filtered medium for 1 min to remove any bubbles or dissolved gas in the medium.
- Place the aliquot of degassed complete medium in a 3 ml disposable syringe and incubate in a 37 °C humidified 5% CO2 incubator for 1 hr.
- Connect two 3 ml disposable syringes containing degassed, pre-warmed complete culture medium to the upper and lower microchannels.
- Flow the degassed complete cell culture medium into the upper microchannel using a syringe pump at the volumetric flow rate of 30 µl/hr (equivalent to the shear stress at 0.02 dyne/cm2) in a 37 °C humidified 5% CO2 incubator O/N.
- Formation of Microengineered Caco-2 Villi in the Gut-on-a-chip.
- Use Caco-2BBE cells with the passage number between 50 and 65.
- Grow Caco-2 cells in a T75 flask containing complete culture medium in a 37 °C humidified 5% CO2 incubator for 4-5 days to obtain fully confluent cells.
- Add 10 ml of Ca2+ and Mg2+-free PBS to fully confluent Caco-2 cells grown in a T75 flask, wash cells, then aspirate out PBS. Repeat this step twice.
- Add 1 ml of 0.05% trypsin/EDTA solution and incubate in a 37 °C humidified 5% CO2 incubator for 5 min.
- Resuspend the dissociated cells with 10 ml pre-warmed complete cell culture medium (with FBS) by pipetting up and down three to five times.
- Spin down the cell suspension by centrifugation at 500 x g for 5 min, then remove the supernatant.
- Resuspend again with 1 ml pre-warmed complete medium to adjust cell density at ~1.5 x 105 cells/cm2 in the device. Use hemocytometer to estimate the cell density.
- Infuse 100 µl of the resuspended Caco-2 cells into the upper microchannel (lumen side) by placing a 1 ml disposable syringe attached to a 25 G5/8 needle on the outlet of tubing connected to the upper microchannel.
- Clamp all inlets and outlets of tubing connected to the upper and lower microchannels using binder clips.
- Incubate the whole setup in a 37 °C, humidified 5% CO2 incubator for 1 hr to allow dissociated Caco-2 cells to adhere to the surface of the porous membrane.
- Remove the clamps and resume the flow of culture medium only to the upper microchannel at 30 µl/hr using a syringe pump until cells form an intact monolayer for 24-36 hr. Use the phase contrast or the differential interference contrast (DIC) microscopy to confirm cell-cell junctions in a cell monolayer.
- When cells form a monolayer, perfuse the culture medium into both upper (lumen) and lower (capillary) microchannels at the same flow rate of 30 µl/hr.
- Application of mechanical deformations mimicking peristalsis-like motions5,9.
- Turn on the vacuum pump equipped with the tension equipment.
- Connect the vacuum chambers to the vacuum controller via tubing with a stainless steel connector.
- Set the stretching motion of 10% mean cell strain at a frequency of 0.15 Hz with the cyclic sine function mode on the vacuum controlling software, then click "Start".
- Maintain the constant flow of culture medium (30 µl/hr) in both upper and lower microchannel to the confluent Caco-2 monolayer under mechanical deformations (10%, 0.15 Hz) for ~100 hr.
Note: Caco-2 cells spontaneously undergo villus morphogenesis with undulated 3D projections extended towards the lumen of the epithelial microchannel5,8-10.
- To emulate the organ-level functions of the living human intestine with lumen-capillary tissue interface in the gut-on-a-chip10, follow the steps as described.
- Grow microengineered Caco-2 villi by repeating steps from 2.1 to 2.2.
- Flow the pre-warmed co-culture medium (a mixture of the complete Caco-2 cell culture medium and the complete HMVECs culture medium, 1:1 v/v) to the upper and lower microchannels at 30 µl/hr.
- Introduce 100 µl of dissociated normal human capillary microvascular endothelial cells (HMVECs; final cell density, ~5.0 x 105 cells/cm2) into the lower microchannel by the identical method described in steps from 2.2.3 to 2.2.9.
- Place a cured rectangular PDMS piece (0.5 cm x 1 cm x 1 cm; width x length x height; 15:1, w/w elastomer:curing agent) on top of the gut-on-a-chip device, then flip the whole device setup upside down (i.e., the upper microchannel faces to the downside, and the lower microchannel faces to the upside).
- Incubate the setup in a 37 °C, humidified 5% CO2 incubator for 1 hr to allow dissociated HMVECs to adhere to the surface of the porous membrane in the lower microchannel.
- Take out a whole setup from the CO2 incubator, and flip the setup over again.
- Flow pre-warmed co-culture medium (a mixture of the complete Caco-2 cell culture medium and the complete HMVECs culture medium, 1:1 v/v) into both upper and lower microchannels at 30 µl/hr with mechanical stretching motions (10%, 0.15 Hz) for at least three days to form cell-cell junctions of an endothelial monolayer.
3. Host-gut Microbiome Co-culture in a Gut-on-a-chip Microdevice
- Perform pre-culture of the bacterial cells as follows to co-culture commensal gut microbiome on the microengineered intestinal villi.
- Resuspend the freeze-dried probiotic bacterial powdery mixture in 10 ml of the mixture (1:1, v/v) of autoclaved Lactobacilli MRS Broth and Reinforced Clostridial Medium.
- Adjust the final cell density to approximately 0.2 optical density units (at 600 nm) by adding appropriate amount of medium, then aliquot 3 ml of microbial cell suspension in a 10 ml disposable sterile tube.
- Place tubes containing resuspended microbial cells in the anaerobic container. Add two packs of anaerobic gas generating sachet in the container. Close the container lid tightly and incubate the container setup without shaking in a 37 °C humidified 5% CO2 incubator O/N.
- Inoculation of Microbiome and Co-culture with Intestinal Epithelial Cells5,10.
- Prepare 3 ml disposable syringes containing degassed antibiotic-free cell culture medium (i.e., DMEM with 20% FBS). See 2.1.6 for the degassing procedure of the culture medium.
- Take out a whole setup of the gut-on-a-chip device containing microengineered villi from the CO2 incubator, then move to the biosafety cabinet.
- Remove syringes connected to tubing linked to the upper and lower microchannels. Connect syringes prepared in 3.2.1 to the device, bring back to the CO2 incubator, then flow this antibiotic-free culture medium for 12 hr prior to seeding of microbiome.
- Spin down the pre-cultured probiotic bacterial mixture cells (see steps in 3.1) at 10,000 x g for 5 min. Aspirate out the supernatant using vacuum, then resuspend in antibiotic-free DMEM medium (final cell density, ~1.0 x 107 CFU/ml).
- Infuse the cell suspension into the lumen side of the microchannel containing the "germ-free" intestinal villi using a 1 ml disposable syringe attached to a 25 G5/8 needle. Allow the adherence of microbial cells to the apical surface of the intestinal villi for ~1.5 hr without flow by clamping to all the tubing end.
- Perfuse the pre-warmed antibiotic-free culture medium into both upper and lower microchannels at 40 µl/hr with cyclic rhythmical deformations (10%, 0.15 Hz).
- For the co-culture of green fluorescence protein (GFP)-labeled E. coli (GFP E. coli; Non-pathogenic DH5-Alpha E. coli host)10,11 cells with microengineered villi, pre-cultivate GFP E. coli cells in autoclaved LB medium at 37 °C under shaking condition (200 rpm) for 12 hr. Repeat the procedure from 3.2.5 to 3.2.6 to carry out co-culture of GFP E. coli cells with microengineered villi.
- Image Cells5,8-10
- Perform DIC, epifluorescence, or laser scanning confocal microscopy for recording microbial and epithelial morphology5,8,10.
- For the DIC imaging, take out a setup of gut-on-a-chip microsystem from the CO2 incubator, place the device setup on the stage of a microscope, then record the cell morphology.
- For fluorescence imaging of villus epithelium, flow PBS for washing the cells at 100 µl/hr for 10 min.
- Fix villi with 4% (w/v) paraformaldehyde for 15 min. Then flow PBS for washing the cells at 100 µl/hr for 10 min.
- Permeabilize the villi with 0.3% (v/v) Triton X-100 diluted in PBS containing 2% (w/v) bovine serum albumin (BSA) for 10 min. Then flow PBS for washing the cells at 100 µl/hr for 10 min.
- Block cells with 2% (w/v) BSA solution in PBS for 1 hr. Then flow PBS for washing the cells at 100 µl/hr for 10 min.
- Add 300 nM of 4',6-diamidino-2-phenylindole dihydrochloride (DAPI) solution diluted in PBS for the nuclear staining under light protection.
- Add 25 units/ml of fluorescent phalloidin (Phalloidin-CF647 conjugate) dissolved in PBS for F-actin staining under light protection.
- Record images of the fluorescently stained cells using a laser scanning confocal microscope.
Note: A 25X objective was applied with appropriate optical zoom during the confocal microscopy. In Figure 3B, approximately 525X magnification was used.
- Perform DIC, epifluorescence, or laser scanning confocal microscopy for recording microbial and epithelial morphology5,8,10.
Representative Results
To emulate the human intestinal host-microbiome ecosystem in vitro, it is necessary to develop an experimental protocol to reconstitute the stable long-term co-culture of gut bacteria and human intestinal epithelial cells under physiological conditions such as peristalsis-like mechanics and fluid flow. Here, we utilize a biomimetic gut-on-a-chip microdevice (Figure 1A) to co-culture living microbial cells in direct contact with living human villi for periods of a week or more in vitro. The intestinal epithelial cells spontaneously form well differentiated villi when cultured on one side of a porous ECM-coated membrane in one channel of a 2-channel microfluidic device in the presence of physiologically relevant fluid flow and cyclic mechanical deformations5. The microengineered villi replicate structures and functions of human small intestinal villi, including formation of columnar epithelial cells lined by an apical brush border, basal proliferative crypts with migration and differentiation of daughter cells progressing from the crypt to villus tip, high levels of mucus production, enhanced drug metabolizing activity, and increased glucose reuptake8. Living endothelial cells can be cultured on the opposite side of the same membrane to recreate the tissue-tissue interface of the intestinal wall, and immune cells can be cultured in the system as well10. To validate the protective function of intestinal epithelial cells, tight junction barrier of intestinal villi formed in a gut-on-a-chip is intermittently quantitated by measuring the transepithelial electrical resistance (TEER)5,10,12. Routine monitoring of TEER value is required to estimate the integrity of a cell monolayer or intestinal villi at every juncture (e.g., prior to adding bacteria into the lumen).
To reestablish the host-microbe ecosystem, living microbial cells resuspended in the antibiotic-free cell culture medium (cell density, ~1.0 x 107 CFU/ml) are inoculated into the lumen of the epithelial microchannel (Figure 1B, "Inoculation"). After microbial cells adhere on the apical surface of villi in the absence of flow for ~1.5 hr (Figure 1B, "Attachment"), physiological flow (40 µl/hr) is resumed through both channels with cyclic mechanical deformations (10% strain at 0.15 Hz) to remove unbound gut bacteria and supply nutrients to both bacterial and villus epithelial cells (Figure 1B, "Co-culture"). This co-culture protocol allows the stable colonization of multiple species of probiotic bacteria in the intervillus space, with viable bacterial microcolonies being maintained for up to two weeks in co-culture (Figures 2A, 2B). In this study, a commercial probiotic formulation that contains a mixed population of 8 different facultative or obligate anaerobic, probiotic strains of Bifidobacterium breve, B. longum, B. infantis, Lactobacillus acidophilus, L. plantarum, L. paracasei, L. bulgaricus, and Streptococcus thermophiles is used.
This co-culture method can be broadly applied to emulate the human intestinal host-microbe ecosystem. For instance, non-pathogenic GFP E. coli can be co-cultured on the surface of intestinal villi grown in a gut-on-a-chip device. Based on the same protocol described above, adhered GFP E. coli cells on the villi grow from single cells at 1 hr to multiple microcolonies with 3 days (Figure 3A, 1 hr vs. 72 hr) that locate in the intervillus spaces (Figure 3B, 1 hr vs. 72 hr) when cultured under trickling flow (40 µl/hr) in the presence of peristalsis-like deformations (10%, 0.15 Hz).
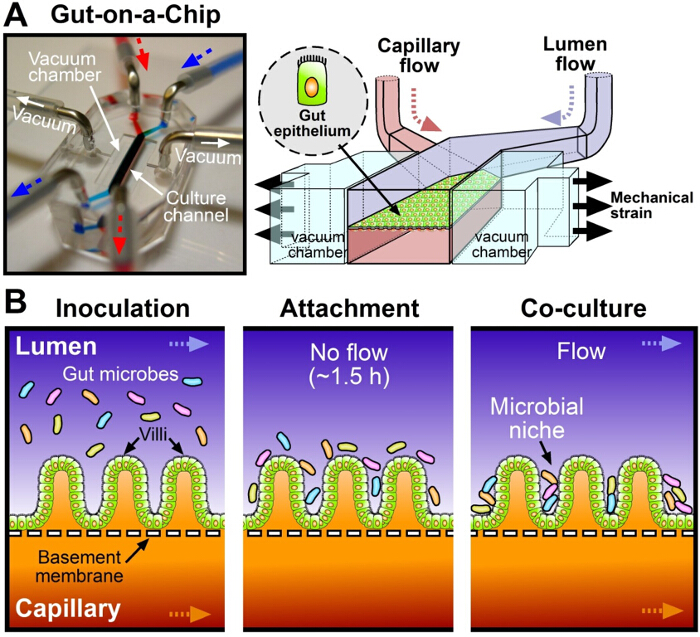
Figure 1. The human Gut-on-a-Chip microphysiological system for host-gut microbiome co-culture. (A) A photograph (left) and a schematic (right) of a 2-channel, gut-on-a-chip microfluidic device. Arrows indicate the direction of culture medium flow (blue, top microchannel; red, bottom microchannel). (B) Schematic diagrams of the procedure of host-gut microbiome co-culture in the gut-on-a-chip. After intestinal epithelial cells form villi (~100 hr), microbial cells resuspended in the cell culture medium are introduced to the lumen microchannel (Inoculation), then the flow of culture medium is suspended for ~1.5 hr (Attachment). After microbial cells adhere to the apical surface of the intestinal villi, flow is resumed (Co-culture). Please click here to view a larger version of this figure.
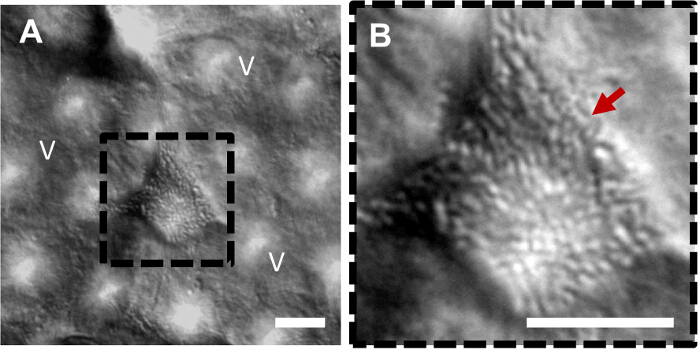
Figure 2. Co-culture of multiple probiotic bacteria with intestinal villi in the gut-on-a-chip. (A) A differential interference contrast micrograph shows a microcolony of probiotic bacterial cells growing between the villi in the gut chip. (B) A higher power magnification view of A (a black dotted square) showing the microcolony of probiotic bacterial cells (a red arrow). V, villi; Scale bar = 20 µm. Please click here to view a larger version of this figure.
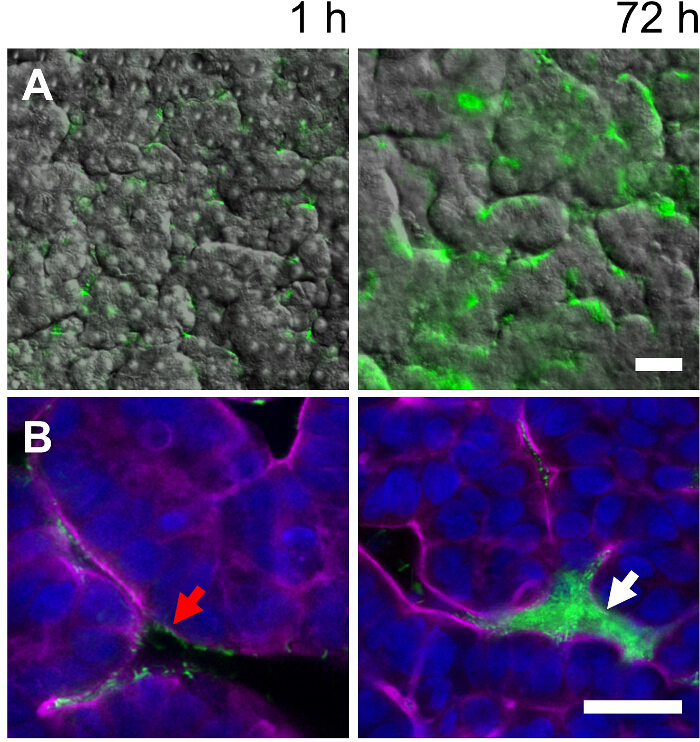
Figure 3. Co-culture of non-pathogenic, GFP-labeled E. coli with intestinal villi in the gut-on-a-chip. (A) Overlaid fluorescence and DIC microscopic images taken at 1 and 72 hr, displaying the colonization of GFP E. coli on the intestinal villi grown in the gut-on-a-chip. (B) High power magnification views of overlaid fluorescence confocal micrographs taken at 1 and 72 hr showing the growth of GFP-labeled E. coli from a single cell (a red arrow) to a microcolony (a white arrow). Blue, nuclei; magenta, F-actin; Scale bar = 30 µm. Please click here to view a larger version of this figure.
Discussion
Understanding host-microbiome interactions is critical for advancing medicine; however, traditional cell culture models performed in a plastic dish or a static well plate do not support the stable co-culture of human intestinal cells with living gut microbes for more than 1-2 days because microbial cells mostly overgrow the mammalian cells in vitro. The overgrowing microbial population rapidly consumes oxygen and nutrients, subsequently producing excessive amount of metabolic wastes (e.g., organic acids), which seriously compromise intestinal barrier functions and cause intestinal epithelial cell death. Hence, preventing the microbial overgrowth that causes the depletion of nutrients and the accumulation of microbial wastes is crucial for sustaining viable host-microbiome coexistence for an extended period (from days to weeks) in the co-culture microenvironment.
To overcome these challenges, a microphysiological gut-on-a-chip device5,8 has been developed to form fully differentiated human intestinal villi and to maintain the steady-state microenvironment of the intestinal lumen in vitro. The design and functional units (e.g., microfluidic flow cell chambers, vacuum-driven cyclic deformations) of a gut-on-a-chip microfluidic device are modified to precisely emulate the physiological, physical and chemical microenvironment for the microbial co-culture5,10. The shear stress applied in the gut-on-a-chip microfluidic culture was determined by the physiological range of luminal flow in the human intestine5,13. For the host-microbe co-culture, maintaining the "steady-state" of microbial population in the luminal microchannel of the gut-on-a-chip is critical in supporting chemically and nutritionally feasible conditions. In the gut-on-a-chip microsystem, it is possible to estimate the intra-luminal steady-state in the gut-on-a-chip by measuring the pH of the culture medium and the microbial cell density. Because fresh culture medium continuously flows through the microchannels, both microbial and epithelial cells do not undergo nutrient depletion when the initial seeding density of microbial cells and the volumetric flow rate of the culture medium are optimized. The conditioned medium passing through the microchannel flows out at a defined volumetric flow rate (here, 40 µl/hr), so metabolic wastes (e.g., organic acids) as well as cellular secretomes (e.g., cytokines) are continuously removed from the co-culture microenvironment. Thus, the gut-on-a-chip device can be considered as a "continuous bioreactor" with a steady-state microenvironment necessary and sufficient to effectively wash out unbound or overgrown microbial cells from the lumen of the intestinal microchannel, which facilitates generation of a stable microbial niche within 2-3 days.
There are crucial factors that must be considered as troubleshooting for the successful co-culture of microbial cells on the intestinal villi. First, it is necessary to determine the optimized incubation time required for the microbial cells to attach to the surface of villi because microbial adhesion can vary between microbial species. For instance, probiotic bacteria, such as Lactobacillus rhamnosus GG5, generally require ~1.0-1.5 hr to attach, but some other microbial species may require shorter (e.g., pathogenic microbes) or longer times, depending on their adhesion kinetics14. Second, the initial seeding density of the microbial cells should be identified because excessive microbial cell numbers may lead to the outgrowth of bacteria in the early stage of co-culture, which can interfere with the achievement of a stable steady-state. To optimize this seeding density, it is recommended to characterize the growth profile of the target microbial strain in the antibiotic-free cell culture medium at various seeding densities. Finally, adjustment of the volumetric flow rate may be required depending on the microbial species. For example, increased flow rates will be required if the microbial cells proliferate very rapidly. It was experimentally confirmed that flow rates up to 300 µl/hr (0.2 dyne/cm2) do not compromise the barrier function and cell morphology of the microengineered villi (data not shown). Although Lactobacillus rhamnosus GG5, over-the-counter probiotic mixture, VSL#3, non-pathogenic lab strain E. coli as well as the pathogenic enteroinvasive E. coli strain10 were successfully applied for the long-term co-culture in the gut-on-a-chip, it is still required to test a variety of microbial species from commensal gut microbiota to pathogenic infectious microorganisms. The cultivability of microbial cells in vitro should be contemplated in the presence or the absence of host cells in the context of symbiosis and evolution, where the test of cultivating the unculturable gut microbiome is an intriguing challenge. Finally, a robust protocol for the co-culture of both aerobic and anaerobic microbes in the gut-on-a-chip is a critical unmet need to be discovered in the future.
There are steps that can be taken to further improve the model, such as the use of primary intestinal epithelial or undifferentiated stem cells from individual human subjects who have specific gastrointestinal diseases such as Crohn's disease or colorectal cancer; or induced pluripotent stem cells (iPSCs). However, with the burgeoning interest in the role of human microbiome in orchestrating human health and disease, our host-microbiome co-culture method has an enormous significance and potential to be applied to mimic other host-microbiome ecosystems found in the human body (e.g., oral cavity15, skin16, or urogenital tract17). Finally, this co-culture method may innovate the conventional drug development process by improving the predictability in terms of how the gut microbiome influences on the bioavailability, efficacy, and toxicity of new drug compounds. Taken together, the gut-on-a-chip microphysiological system can provide a robust platform to establish a stable steady-state intestinal microenvironment that can be used to reconstitute host-microbiome ecosystem.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Sri Kosuri (Wyss Institute at Harvard University) for providing the GFP-labeled E. coli strain. This work was supported by the Defense Advanced Research Projects Agency under Cooperative Agreement Number W911NF-12-2-0036, Food and Drug Administration under contract #HHSF223201310079C, and the Wyss Institute for Biologically Inspired Engineering at Harvard University. The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the Army Research Office, Army Research Laboratory, Food and Drug Administration, or the U.S. Government. The U.S. Government is authorized to reproduce and distribute reprints for Government purposes notwithstanding any copyright notation hereon.
Materials
| Dulbecco's Modified Eagle Medium (DMEM) containing 25 mM glucose and 25 mM HEPES | Gibco | 10564-011 | Warm it up at 37°C in a water bath. |
| Difco Lactobacilli MRS broth | BD | 288120 | Run autoclave at 121°C for 15 min. |
| Poly(dimethylsiloxane) | Dow Corning | 3097358-1004 | 15:1 (w/w), PDMS : cureing agent |
| Caco-2BBE human colorectal carcinoma line | Harvard Digestive Disease Center | Human colorectal adenocarcinoma | |
| Heat-inactivated FBS | Gibco | 10082-147 | 20% (v/v) in DMEM |
| Trypsin/EDTA solution (0.05%) | Gibco | 25300-054 | Warm it up at 37℃ in a water bath. |
| Penicillin-streptomycin-glutamine | Gibco | 10378-016 | 1/100 dilution in DMEM |
| 4′,6-Diamidino-2-phenylindole dihydrochloride | Molecular Probes | D1306 | Nuclei staining |
| Phalloidin-CF647 conjugate (25 units/mL) | Biotium | 00041 | F-actin staining |
| Flexcell FX-5000 tension system | Flexcell International Corporation | FX5K | Peristalsis-like stretcing motion (10% cell strain, 0.15 Hz frequency) |
| Inverted epifluorescence microscope | Zeiss | Axio Observer Z1 | Imaging, DIC |
| Scanning confocal microscope | Leica | DMI6000 | Imaging, Fluorescence |
| UVO Cleaner | Jelight Company Inc | 342 | Surface activation of the gut-chip |
| Type I collagen | Gibco | A10483-01 | Extracellular matrix component for cell culture into the chip |
| Matrigel | BD | 354234 | Extracellular matrix component for cell culture into the chip |
| 1 mL disposable syringe | BD | 309628 | Cell and media injection stuff |
| 25G5/8 needle | BD | 329651 | Cell and media injection stuff |
| Syringe pump | Braintree Scientific Inc. | BS-8000 | Injection equipment into the chip |
| VSL#3 | Sigma-Tau Pharmaceuticals | 7-45749-01782-6 | A formulation of 8 different commensal gut microbes |
| Reinforced Clostridial Medium | BD | 218081 | Anaerobic bacteria culture medium |
| GasPak EZ Anaerobe Container System with Indicator | BD | 260001 | Anaerobic gas generating sachet |
| 4% paraformaldehyde | Electron Microscopy Science | 157-4-100 | Fixing the cells for staining |
| Triton X-100 | Sigma-Aldrich | T8787 | Permeabilizing the cells |
| Bovine serum albumin | Sigma-Aldrich | A7030 | Blocking agent for staining of the cells |
| Corona treater | Electro-Technic Products | BD-20AC | Plasma generator for fabrication of the chip |
| Steriflip | Millipore | SE1M003M00 | Degasing the complete culture medium |
| Disposable hemocytometer | iNCYTO | DHC-N01 | For manual cell counting |
References
- Turnbaugh, P. J., et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 444, 1027-1031 (2006).
- Tremaroli, V., Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature. 489, 242-249 (2012).
- Sekirov, I., Russell, S. L., Antunes, L. C. M., Brett Finlay, B. Gut Microbiota in Health and Disease. Physiol. Rev. 90, 859-904 (2010).
- Turnbaugh, P. J., et al. The human microbiome project. Nature. 449, 804-810 (2007).
- Kim, H. J., Huh, D., Hamilton, G., Ingber, D. E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip. 12, 2165-2174 (2012).
- Round, J. L., Mazmanian, S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 9, 313-323 (2009).
- Garrett, W. S., et al. Communicable ulcerative colitis induced by T-bet deficiency in the innate immune system. Cell. 131, 33-45 (2007).
- Kim, H. J., Ingber, D. E. Gut-on-a-Chip microenvironment induces human intestinal cells to undergo villus differentiation. Integr Biol. 5, 1130-1140 (2013).
- Huh, D., Kim, H. J., et al. Microfabrication of human organs-on-chips. Nat Protoc. 8, 2135-2157 (2013).
- Kim, H. J., Li, H., Collin, J. J., Ingber, D. E. Contributions of microbiome and mechanical deformation to intestinal bacterial overgrowth and inflammation in a human gut-on-a-chip. Proc. Natl. Acad. Sci. 113, E7-E15 (2016).
- Miller, W. G., Lindow, S. E. An improved GFP cloning cassette designed for prokaryotic transcriptional fusions. Gene. 191, 149-153 (1997).
- Odijk, M., et al. Measuring direct current trans-epithelial electrical resistance in organ-on-a-chip microsystems. Lab Chip. 15, 745-752 (2015).
- Lentle, R. G., Janssen, P. W. Physical characteristics of digesta and their influence on flow and mixing in the mammalian intestine: a review. J Comp Physiol B. 178, 673-690 (2008).
- Granato, D., et al. Cell surface-associated lipoteichoic acid acts as an adhesion factor for attachment of Lactobacillus johnsonii La1 to human enterocyte-like Caco-2 cells. Appl Environ Microbiol. 65, 1071-1077 (1999).
- Dewhirst, F. E., et al. The human oral microbiome. J Bacteriol. 192, 5002-5017 (2010).
- Grice, E. A., Segre, J. A. The skin microbiome. Nat Rev Microbiol. 9, 244-253 (2011).
- Hay, P. E., et al. Abnormal bacterial colonisation of the genital tract and subsequent preterm delivery and late miscarriage. Br Med J. 308, 295-298 (1994).

