Processing Embryo, Eggshell, and Fungal Culture for Scanning Electron Microscopy
Summary
Here, we present detailed processing protocols for imaging delicate tissue samples using scanning electron microscopy (SEM). Three different processing methods, namely, hexamethyl disilazana (HMDS) chemical drying, simple air drying, and critical point drying are described for preparing rigid eggshells, embryos at early developmental stages, and fungal cultures respectively.
Abstract
Although scanning electron microscopy (SEM) is being widely used for the ultra-structural analysis of various biological and non-biological samples, methods involved in processing different biological samples involve unique practices. All conventional practices described in the literature for processing samples still find useful applications, but subtle changes in the sample preparation can alter image quality, as well as, introduce artifacts. Hence, using a unique sample preparation technique specific to the type of tissue analyzed is required to obtain a good quality image with ultrastructural resolution. The focus of this study is to provide the optimal sample preparation protocols for imaging embryos, rigid eggshells, and fungal cultures using SEM. The following optimizations were recommended to yield good results for the three different delicate biological samples studied. Use of milder fixatives like 4% paraformaldehyde or 3% glutaraldehyde followed by dehydration with ethanol series is mandatory. Fungal mycelium on agar blocks obtained by slide cultures yields a better ultrastructural integrity compared to cultures taken directly from agar plates. Chemical drying of embryos with HMDS provides drying without introducing surface tension artifacts compared to critical point drying. HMDS prevents cracking caused by shrinkage as samples are less brittle during drying. However, for fungal culture, critical point drying provides acceptable image quality compared to chemical drying. Eggshells can be imaged with no special preparation steps except for thorough washing and air drying prior to mounting. Preparation methodologies were standardized based on acceptable image quality obtained with each trial.
Introduction
Scanning electron microscope (SEM) ultrastructural analysis and intracellular imaging supplement light microscopy for three-dimensional profiling of prokaryotes, plants, and animals. The high spatial resolution of an SEM makes it one of the most versatile and powerful techniques available for the examination of microstructural characteristics of specimens at the nanometer to micrometer scale. Desiccated specimens are resolved to compositional and topographical structures with intense detail, which provides the foundation for developing valid conclusions about functional relationships1,2,3,4,5,6,7,8,9. When interpreting SEM images of biological specimens, it is a great challenge to distinguish between native structures and the artifacts that are created during processing. SEM is generally operated at very high vacuums to avoid any interference from gas molecules affecting the primary, secondary or backscattered electron beams emitted from the sample10,11. Also, biological materials are susceptible to radiation damage due to their poor or non-conducting properties. It is essential for the specimens loaded into the SEM to be completely dry and free of any organic contaminants to eliminate any possible outgassing in a high vacuum environment10,11. As biological specimens are mostly composed of water, additional preparative techniques are required to ensure that the native structures are retained.
The resolution obtained is based on optimizing preparation methods specific to specimen types and instrumental parameters utilized. Thus, it is necessary to avoid using generalized processing steps for all tissue types. Some biological specimens will require less stringent processing to preserve their structure while more time and care might be needed for delicate types of samples to avoid the introduction of drying artifacts, such as shrinkage and collapse. Sample preparation is a critical step in SEM imaging; the findings of morphometric studies are remarkably influenced by specimen preparation procedures12,13. Common preparation steps for many biological samples are fixation, dehydration and coating with a metal such as gold, platinum or palladium to convert their surfaces to be conductive for SEM analysis. The nature and combination of steps used will vary depending on the type of the tissue, and the specific goals of the study. Charge build-up, sensitivity to vacuum and electron beam damage pose problems when processing soft delicate biological samples, necessitating additional processing steps to retain the native structure of the object. Using conventional methods such as osmium tetroxide fixing, and dehydration cause shrinkage and the collapse of delicate tissues14,15,16,17. The aim of the study is to establish elegant methodologies derived by combining ideas from earlier studies with modifications to prepare and image soft delicate tissues (e.g., reptile embryos, eggshell of painted turtles, and fungal cultures).
Selection of a suitable fixing method is the first most important step for microscopic analysis of biological specimens. Fixing the tissues immediately after isolating from an organism is essential to prevent alteration in their morphology due to decomposition. An effective fixative should terminate cellular processes by permeating the cells quickly and maintaining the effect irreversibly to stabilize the structure of the sample to withstand both subsequent processing steps and examination under the SEM17,18. Although several chemical and physical fixation methods are known, chemical fixation is more commonly used for biological specimens to avoid any cellular changes due to autolysis, putrefaction, and drying effects. There are numerous fixative chemical formulations discussed in literature17,19,20,21,22,23, fixatives that work by denaturing and coagulating biological macromolecules, and those that fix by covalently cross-linking macromolecules. Alcohols are used as denaturing fixatives that preserve ultrastructure very poorly and are used mostly for light microscopy and not recommended for electron microscopic analysis. Cross-linking fixatives like formaldehyde, glutaraldehyde, and osmium tetroxide create intermolecular and intramolecular crosslinking between macromolecules within the tissues, providing excellent preservation of ultra-structures11,24,25,26. Biological samples are sensitive to temperature. The temperature at the beginning of fixation is recommended to be 4 °C to reduce the lateral mobility of membrane proteins, to slow the diffusion of intercellular molecules, and to slow the rate of fixation11. The time required for fixing tissues largely depends on the size of the sample and the speed at which the fixative diffuses and reacts with the components of the specimen. An overnight fixation in 4% paraformaldehyde or 3% glutaraldehyde in PBS at 4 °C is the preferred method for SEM analysis of specimens used in this study for their sequential penetrative properties, which allow smaller delicate samples to be processed17,18,19,20,27. A post-fixation step with osmium tetroxide is eliminated not only due to its toxic nature but also found to implement no added advantage to improve image quality for the samples analyzed in this study.
Biological samples contain fluids that interfere with the SEM operation; hence, the samples need to be dried before inserting it in the SEM sample chamber. Once dehydration is ensured, the solvent must be removed from the tissue without creating artifacts into the specimens due to the surface tension/drying. Three different drying methods were commonly used during processing tissues for SEM imaging: air drying, critical point drying, and freeze drying samples28,29,30,31. Few studies report all three drying methods producing identical results with animal tissue samples28,29,30,31. A general practice used for smaller specimens are chemical dehydration by ascending concentration series of alcohol and hexamethyldisilazane (HMDS), but larger specimens are dried using a critical point drying (CPD) instrument32. During the drying process, considerable forces formed in small cavities that are passed through the specimen by a liquid/gas interface; this can even lead to a complete collapse of the hollow structures33. Any deformation occurring due to the treatment could then be mistaken as a native structural feature of the specimen. Thus, the generalized phenomenon for processing should be eliminated and a unique drying process should be standardized for each type of tissue especially when delicate tissue specimens are analyzed.
In several trials conducted using various combination of all the above-mentioned processes, we standardized the methods that can be used for SEM analysis of three delicate tissues: reptile embryos, eggshells of painted turtles, and fungal cultures. Developmental biologists and morphologists describe normal and abnormal morphogenesis during embryo development in representative vertebrate animals. Investigations on gene signaling pathways depend on the morphological description of novel structures. To avoid any abrupt change in the vertebrate embryo structure during SEM analysis, we recommend chemical drying following dehydration. Chemical drying using HMDS is the relatively newest drying method and the advantages include relative quickness, ease of use, lost cost, and the limited expertise and equipment needed9. CPD is a commonly used drying technique using passaging CO2 across the specimens at a specific temperature and pressure. We identified that HMDS is suitable for drying soft delicate tissues and allows larger samples to be processed compared to critical point drying, which caused extensive deformation to embryonic tissues. Several methods have been used to prepare samples for SEM imaging to study the morphological characteristics of fungi34. Fungal specimens are commonly fixed in osmium tetroxide followed by ethanol dehydration and critical point drying, which may provide satisfactory results, although the toxic effects of osmium tetroxide6,7,35 and losing fungal materials while changing solutions during processing are pronounced disadvantages. The sample preparation technique using air-drying without fixation has also been practiced36 but results in shrunken and collapsed structures, and observation of such specimens can easily be misinterpreted while characterizing the species. Fungal hypha loses its integrity in contact with liquids and an even drying may not be achieved to restore the structure. Due to this effect, freeze-drying is commonly used for drying soft tissues like fungal mycelium. Freeze drying works well for clean materials but the presence of any salts or secretion will obscure surface detail that will be identified only at the SEM viewing stage. We coupled the slide culture method with glutaraldehyde fixing and critical point drying to yield structural details of intact fungal hyphae and spores. Although CPD drying caused shrinkage in embryos, it resulted in well preserved mycelial structures when coupled with glutaraldehyde fixation. The eggshell is of primary importance to the embryo of oviparous animals by not only acting as a protective covering but also providing mechanical stability, permeability to gas and water, and a calcium reserve for the developing embryo. Freshwater turtle eggshells are classified as "rigid" based on their structure, and due to their availability have received significant attention from biologists1,2,3,4,5,6,7,37,38.
We detail simple methods for easy examination of eggshell and shell membranes of painted turtle that can be applied to any rigid eggshell species. Preparation methodologies were evaluated based on resulting image quality and reduced potential artifacts.
Protocol
NOTE: Painted turtle (Chrysemys picta) eggs used in this study were collected during the nesting season of May through June 2015-16 from Rice Creek Field Station, Oswego New York with permission obtained from the New York State Department of Environmental Conservation (DEC).
1. Chemical drying method to process embryos for SEM
- Collect turtle eggs from field sites during the nesting season. Prepare the incubation chambers in advance, made of plastic boxes with lids (L x W x H) 6.7 cm x 25.4 cm x 10.2 cm filled with bedding medium prepared with a moist mixture of vermiculite and peat moss (1:1 ratio). Make 4-6 holes of approximately 0.25 cm along the sides of the boxes and on the lid to allow aeration.
- Gently remove the soil from the nest to uncover the eggs. Wipe the surface of turtle eggs with diluted iodine tincture (1:25,000) to control microbial contamination during incubation. Place the clutches separate from each other, clutch size of painted turtle may vary from 5-9 eggs and place a maximum of 8-9 eggs per box.
NOTE: Carefully handle the eggs during collection, wiping, labeling and placing inside the boxes. Position and alignment of eggs need to be the same as they were laid, any movement will inhibit embryo development. - Manually bury the eggs half in the bedding, cover and place the box inside the incubator set at 30 °C. Incubate the eggs for 10-17 days to obtain the embryonic stages 12, 13, and 18 respectively used in this study. Add distilled water to partially wet the bedding medium every other day to avoid dehydration and to maintain the moisture level for normal development of the embryos.
NOTE: Incubation and staging embryos are according to a complete developmental table published earlier39. - Fix the embryos by making the first cut on one side of the dorsal eggshell and yolk membrane together, vertical to the long axis using pointed scissors. Insert the scissors into the yolk carefully to avoid cutting the embryonic disc. Now cut the lateral side of the egg along the long axis and then cut the other side of the egg along the short axis.
- Peel open the excised piece with the embryo side up using forceps. Cut the other lateral side of the eggshell and place the excised piece into phosphate buffered saline (PBS at a pH of 7.4).
NOTE: The turtle egg is filled with highly viscous egg yolk and the dorsal side of the embryo adheres to the eggshell membrane. The eggshell membrane near the embryo changes with the incubation from translucent white to an opaque, chalky white. This allows one to locate the embryo in the center of the long axis and be seen as a dark calcified spot from the exterior40,41. - Use a stereomicroscope to isolate the embryos along with the yolk membrane by peeling them from the eggshell using forceps. Remove extra-embryonic membranes using forceps and micro-scissors. Transfer the embryo using an embryo spoon to fresh PBS in a Petri dish to wash any blood or yolk.
- Use clear 12-well plates to fix the embryos overnight in 4% paraformaldehyde in PBS at 4 °C or in 2-3% glutaraldehyde in PBS. Place one to three embryos in each well depending on the size of the embryos. Ensure complete infiltration (samples will appear white) for older embryos by extending fixation time for 2-3 days. Rinse embryos 3x with fresh PBS for 5 min each rinse.
NOTE: Avoid damaging the surface of the embryo by using polystyrene inserts with polyester mesh bottoms for 12-well plates to transfer specimens from one solvent to another. - Dehydrate samples using a series of ethanol concentration in distilled water: 30%, 50%, 70%, 80%, 95%, and 100% and treat samples for 1 h in each dehydration solution. Repeat the step with 100% ethanol twice to ensure complete dehydration. If not used immediately, store samples in 70% ethanol at -20 °C for a longer period.
- Dry embryos using a series of hexamethyldisilazana (HMDS) to 100% ethanol concentration: 1:2, 2:1 and 100%. Leave the samples in each solution for 20 min and keep the Petri dish partially covered during the process.
NOTE: Carry out all steps involving HMDS in fume hood with necessary personal protection gear as HMDS is highly toxic. - Leave the embryos in the final 100% HMDS solution covered completely or partially in a fume hood overnight aiding in evaporation of HMDS, leaving samples ready for mounting and sputter coating. Cover the dish to eliminate dust settling over the samples.
NOTE: The tissue will appear white after complete drying and partially dried samples will look yellow in color, leave these tissues in the fume hood for a longer time. - Choose the size of aluminum stubs and carbon adhesive tape per size of the sample analyzed. Mount the dried samples carefully on a standard aluminum pin stub (12.7 mm x 8 mm) using double stick carbon conductive tape (12 mm).
- Introduce mounted samples into the chamber of the sputter coater to coat the specimen with a very thin film of gold to eliminate the charge effect. Gold plate the specimens for 60-120 s at a 35 mA sputter.
- Mount the stubs to corresponding pore-plates by securely fastening the setscrews. Transfer the sample holder into or out of the sample chamber of SEM using the sample exchange tool. Image the samples at high vacuum mode with an accelerating beam voltage of 10 kV and emission current 10 µA.
NOTE: Always wear gloves while handling samples, sample holders, mounting stubs, and transfer tools to avoid grease contamination from hands to the SEM system. - Test alternative techniques listed below to compare the resolution of specimens obtained from above procedure.
- Include a post-fixation with 1% osmium tetroxide for 1 h at room temperature after step 1.7.
- Partially or completely remove the HMDS at step 1.10 for fast rapid evaporation of HMDS.
- Process embryos after step 1.8 using critical point drying (CPD) by following steps 3.3-3.4.
2. Preparing the eggshell for SEM using an air-drying method
- Collect and incubate the painted turtle (Chrysemys picta) eggs to fix the embryos as specified in steps 1.1-1.7.
- Save eggshells in distilled water following embryo fixation in step 2.1. Clean the eggshells thoroughly by soaking in distilled water for at least 1 h to eliminate yolk and albumin contamination.
- Air-dry eggshells after washing on delicate antistatic wipes in the fume hood overnight. Store dried eggshells in clean specimen bottles labeled by number and stage.
- Mount, sputter coat and image the specimens by following steps specified in steps 1.11-1.13.
3. Critical point drying method for preparing fungal cultures for SEM
- Establish slide cultures
- Prepare potato dextrose agar (PDA) media for fungal cultures: Add 39 g of PDA power in 1 L of distilled water in an Erlenmeyer flask. Mix well by swirling the flask and autoclave the media at 121 °C for 30 min. Allow the media solution to cool and then add the antibiotic chloramphenicol (25 µg/mL) using a sterile micropipette.
NOTE: After sterilization, the agar solution should be hot and it will not solidify soon. Cool it enough so that it will not inactivate the antibiotics. - Mix it by swirling and pour plates (approximately 10-12 mL of media for each 10 cm Petri dish), carefully stack up and let solidify.
- Use a sterile scalpel blade to cut out small blocks of agar about ½ to ¾ of an inch. Remove and place an agar block onto a clean glass microscope slide.
- Place the slide in a clean Petri dish to prevent contamination and preserve moisture during incubation.
- Raise the slide off the bottom of the Petri dish using a sterile toothpick to create surface tension between the plate and the slide to remove the glass slide without disrupting the delicate growth following incubation.
- Use a sterile loop or a needle to transfer some of the fungi from the specimen inoculum to each of the four sides of the agar block on the slide.
- Place a clean coverslip on the surface of the agar block following inoculation. Add a few drops of sterile distilled water to the Petri dish around the slide to ensure moisture for the growing fungi.
- Seal the plate partially using paraffin film and incubate the plate at 30 ˚C for an appropriate length of time (for Fusarium species incubate for 36 to 48 h).
- Remove the slide from the Petri dish and separate the tightly adhered coverslip from the agar block using sterile forceps. Fix the agar blocks in 3% glutaraldehyde in PBS overnight at 4 ˚C.
- Prepare potato dextrose agar (PDA) media for fungal cultures: Add 39 g of PDA power in 1 L of distilled water in an Erlenmeyer flask. Mix well by swirling the flask and autoclave the media at 121 °C for 30 min. Allow the media solution to cool and then add the antibiotic chloramphenicol (25 µg/mL) using a sterile micropipette.
- Dehydrate the samples by passing through an ethanol series: 10%, 25%, 50%, 75% and 90% with 15 min per change. Process the samples for final dehydration with two changes in 100% ethanol lasting 30 minutes each to ensure complete saturation.
- Critical point drying: Place dehydrated samples in the chamber of the CPD apparatus. Seal and cool the chamber by opening the valves to allow liquid CO2 in and vent ethanol out, until liquid CO2 completely fills the chamber.
- Seal and heat the chamber slowly to achieve a critical point when the chamber pressure exceeds 1000 psi and the temperature exceeds 31 °C, the liquid and gas phase of CO2 is in equilibrium. Slowly drain the CO2 from the chamber and the sample as gas to avoid effects of surface tension.
- Perform mounting, gold plating and imaging the specimens by following steps specified in steps 1.11-1.13.
- Perform comparative analysis by chemically drying the specimens from step 3.2 using HMDS by following steps 1.9-1.13.
Representative Results
Figure 1 show scanning electron micrographic analysis of painted turtle (Chrysemys picta) embryos. Painted turtle eggs collected and incubated on a bedding medium, mounted on aluminum stubs following chemical drying were used for SEM imaging (Figure 1A-E). A lateral view of a stage 12 embryo shows the craniofacial structures; maxillary prominence extends beyond the mandibular and limits a well-marked nasal pit medially; five pharyngeal arches were also observed (Figure 1F). Newly formed somites at the posterior tail region of the embryo are easily countable compared to the segmented body somites. Forelimb buds appear longer compared to hind limb buds and points more caudally than ventrally. A well-defined outgrowth of a carapace ridge is seen along the entire inter-limb flank region of the stage 15 embryo (Figure 1G. At stage 18, painted turtles possess a short, feebly projecting snout. The lower beak rests within the upper jaw and is slightly upturned with a terminal hook that fits into the central notch of the upper beak (Figure 1H). The upper jaw of the painted turtles shows a notched appearance forming a medial U-V shaped distal tip with a small cusp on each side, a tiny egg tooth begins to form at the tip of the upper jaw. Limb buds that appeared longer during earlier stages form a paddle-like structure during stages 13-14 with digital plate vaguely indicated. Limb mesenchyme demarcated by the apical ectodermal ridge is seen along the anterior-posterior margins (Figure 1I-J).
Figure 2 shows ultra-structural analysis of Chrysemys picta eggshell and shell membrane using SEM imaging. Painted turtle eggshells were obtained as described above and were subjected to air-drying following washing with double distilled water (Figure 2A,B). Eggshells mounted on aluminum stubs with double-sided carbon tape (Figure 2C) were imaged using SEM. A lateral view of the shell showed outer calcareous eggshell layer firmly attaching to the inner filamentous shell membrane (Figure 2D). The outer surface of the eggshell consists of well-distinguished mineralized shell units, made of globular/spherical nodules arranged in groups and in between adjacent shell units a concentration of small rounded depressions or pores of various sizes were observed (Figure 2E). Nodules from each group of shell units meet at a connection junction, the center point (Figure 2F). The outer surface was manually peeled using forceps to observe the surface of the shell membrane. Rows of central plaques were seen which provides the attachment point between the shell units and underlying multilayered fibrous membrane (Figure 2G).
Figure 3 shows the morphological characterization of ascomycete fungal isolate from slide cultures observed using SEM imaging. Scheme of slide culture setup established (Figure 3A) showing fungal growth on the agar block within two days of inoculation. Colonies growing rapidly with white to cream-colored aerial mycelium (Figure 3B). SEM imaging following critical point drying and sputter coating of agar slices revealed fungal hyphae, curved conidia, and rice like spores. Conidiophores were seen arising laterally from the septate aerial hyphae (Figure 3C). Macroconidia produced on shorter, branched conidiophores are moderately curved, with short, blunt apical and indistinctly pedicellate basal cells mostly septate (Figure 3D).
Figure 4 shows the comparative results for processing similar specimens using alternative techniques. Immersing heads of stage 15 embryos in HMDS and drying by gradual evaporation show well preserved craniofacial structures, almost no shrinkage or distortion is seen with extended times of slow evaporation from HMDS (Figure 4A). Embryos post-fixed with osmium tetroxide showed no distinguishable difference in image quality except for slight shrinkage of tissue compared to HMDS slow processing (Figure 4B). Drying embryos by removing HMDS partially or completely allowing fast evaporation results in structural distortion and shrinkage (Figure 4C), whereas slow evaporation resolved excellent surface structures of a similarly staged embryo (Figure 4A). The drying artifacts were seen in the embryos treated with the CPD technique causing extensive shrinkage and destruction of tissue (Figure 4D,E). Agar slices from slide cultures show incomplete drying of fungal isolate with HMDS treatment compared to CPD (Figure F). Complete drying is achieved with CPD treatment of agar slices with a well dried, white specimen showing intact high-resolution structural features of fungal hyphae and spores (Figure 4G-G'). Agar blocks with fungal cultures appear shrunken and yellow in color making them not suitable for SEM imaging (Figure 4H-H').

Figure 1: Scanning electron micrographs of Chrysemys picta embryos prepared by chemical drying. (A) Chrysemys picta, nesting during the breeding season at Rice Creek Field Station, Oswego, NY. (B) Well-constructed nest from outside. (C) Surface soil removed with care to exposure the eggs laid. (D) Eggs placed into the incubation chamber with bedding medium. (E) Mounting embryos onto aluminum stubs following chemical drying, E4 showing the sputter coated embryo. (F) Lateral view of stage 12 embryo showing facial structures, limb buds and somites. (G) Well defined carapace ridge and paddle-shaped limb buds are seen at stage 15. (H) Craniofacial structures of stage 18 embryo showing upper and lower jaw, note a small egg tooth formed at the tip of the upper beak. (I) Dorsal view of right forelimb at stage 13-14 and a close-up view of AER (J) at the dorsal-ventral boundary. Scale bars: F-H, 500 µm; I, 100 µm, and J, 10 µm. Keys: mb, midbrain; np, nasal pit; mxp, maxillary prominence; pa, pharyngeal arches; h, heart; fl, forelimb; s, somite; hl, hind limb; t, tail tip; cr, carapacial ridge; et, egg tooth; oc, oral cavity; e, eye; uj, upper jaw; lj, lower jaw; m, mesenchyme, AER, apical ectodermal ridge. Please click here to view a larger version of this figure.
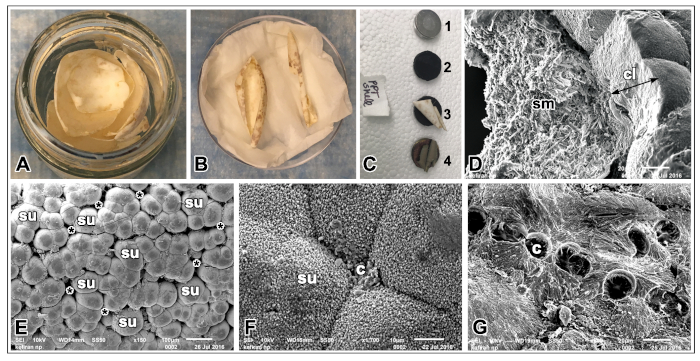
Figure 2: Ultrastructural analyses of the air-dried eggshell and the shell membrane of painted turtle eggs. (A) After fixing the embryos, eggshells were washed thoroughly in distilled water to remove yolk and albumin. (B) Clean eggshells were air dried at least overnight. (C) Mounting of an eggshell onto aluminum stubs. (D) Radial view of shell fragment showing outer calcareous layer and inner shell membrane. (E) Outer calcareous layer shows globular shell units (su) arranged in groups and pores seen in between these units (asterisks). (F) Magnified view of a nodule showing the center point (c) between the shell units. (G) Removal of calcareous layer shows the outer surface of the shell membrane with depressions left broken from shell units. Scale bars: E, 100 µm; F, 10 µm, and G, 20 µm. Please click here to view a larger version of this figure.
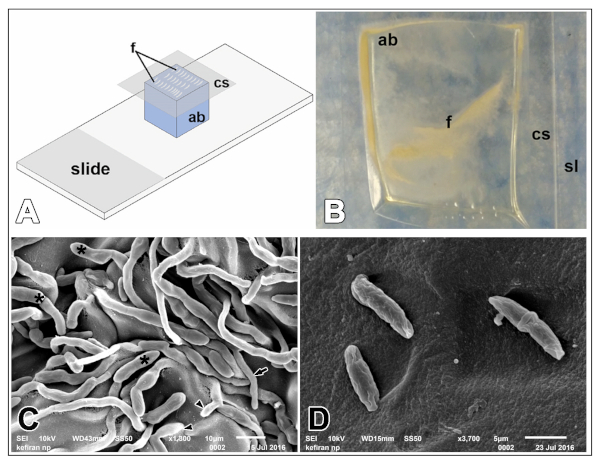
Figure 3: Preparing fungal culture for SEM imaging. (A) Schematic diagram to show the setup of slide culture. (B) White colored fungal colonies seen on the agar block after two days of incubation at 30°C. (C) SEM image showing a section of mycelium of Fusarium solani, black asterisks marking the macroconidia in sporodochia, an arrow pointing to phialide, and arrowheads directed towards microconidia, scale bar, 10 µm. (D) Magnified image of septate chlamydospores, scale bar 5 µm. Keys: ab, agar block; cs, coverslip; f, fungal culture. Please click here to view a larger version of this figure.
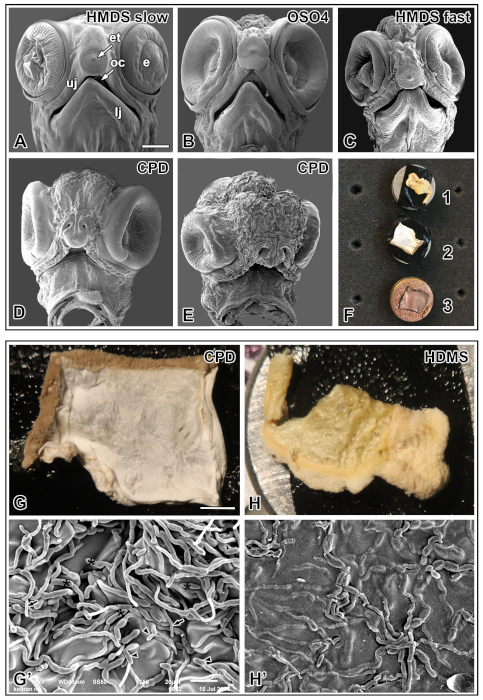
Figure 4: Comparative analysis of alternative techniques to process similar specimens. (A) Frontal view of painted turtle head dried with slow evaporation of HMDS (A), after post-fixing with osmium tetroxide (B), rapid faster evaporation of HMDS (C), and CPD (D-E). Including a post-fixation step using osmium tetroxide do not show any visible difference in image quality compared to HMDS treatment without post-fixation. Deformations are seen after fast evaporation of HMDS, while gradual drying with slow evaporation provides a better surface structure. Different degrees of shrinkage and collapsed structures are seen in the regions of brain, eyes and facial prominences in embryos treated with CPD. (F) Mounting of slide cultures processed with HMDS (1), CPD (2) and sputter coated CPD treated specimen (3) on aluminum stubs. (G-G') Complete drying seen as intact white colored agar slice with fungal inoculate achieved by CPD preserving structures of fungal mycelium and spores of Fusarium solani. (H-H') Inadequate drying of slide culture, shrinkage and damaged structural integrity seen with HMDS preservation technique. Scale bars: A-E, 500 µm; G-H, 2 mm, and G'-H', 20 µm. Keys: et, egg tooth; oc, oral cavity; e, eye; uj, upper jaw; lj, lower jaw; black asterisks, macroconidia in sporodochia; arrow, phialide; and arrowheads, microconidia. Please click here to view a larger version of this figure.
Discussion
In our study, different fixation agents, dehydration and drying methods were tested to prepare three different delicate biological samples for SEM imaging: embryos, eggshells, and fungal cultures. SEM is commonly used for surface analysis, so fixative penetration is less concerning, but it must be understood that poorly fixed internal structures will cause inward shrinking or/and collapsed surface structures. Extended fixation time should also be considered for larger tissue samples, replacing the fixative solution a few times depending on the fixation duration. Due to active interaction between the fixative and the tissue, the osmotic properties of cells would change considerably. Tissue fluids would dilute the fixative and thus an aldehyde effect contributing to the overall osmolality can be neglected as far as effects on cell volume are concerned. Also, formaldehyde would penetrate faster into tissues and is more effective as the cross-linker compared to glutaraldehyde. A buffered formaldehyde-glutaraldehyde mixture was also reported for fixing a wide variety of tissues, although for monolayers and cell suspensions diluted solutions were more suitable for fixing15,16. However, the diluted solutions are also hypertonic, thus the aldehydes are not themselves osmotically active. For delicate samples used in this study, PBS at pH 7.4 was used as a vehicle for the fixing agents, as phosphate buffers are thought to be more similar to the cytoplasmic environments of most biological samples. Further, the osmolarity is identified to be within the physiological range required for the sample while PBS is acting as a vehicle for the fixing agent. Post-fixation with osmium tetroxide is often recommended for various biological samples16,22,42 but has been eliminated for all the biological samples used in this study. Samples without osmium tetroxide yielded crisp images and specimens fixed with osmium tetroxide yielded indistinguishable results (Figure 4A). Osmium tetroxide causes distortion of leaf tissue structures and yields poorer specimen preservation compared to glutaraldehyde and formaldehyde mixture43, and it has been suggested that a build-up of osmium molecules could inhibit infiltration of dehydrating agents and transitional agents used in CPD, and the solvents like HMDS used for chemical drying. The effect is tissue-specific and excluding osmium tetroxide will not have any effect on the image quality for the type of delicate specimens analyzed in this study. For other types of tissues use of osmium tetroxide may be eliminated with extended primary fixation.
HMDS dried turtle embryo specimens showed well-preserved surfaces, and less distortion or shrinkage compared to CPD (Figure 4). CPD minimizes the chance for distortion in cell morphology and artifacts production due to the zero or minimal surface tension created during the process. However, CPD can cause a potential physical hazard for delicate fragile samples. In line with our observation, HMDS yielded similar or higher quality imaging by minimizing the surface tension caused due to drying based on previous studies published44,45,46,47. Compared to CPD treatment, HMDS preserved structural details of the organic meshwork excellently in etched bivalves and barnacle shells48 and provided high resolution of the structural features of the complex internal organization of mermithid nematodes49 and insect internal tissues28. The drying plate artifacts were seen in high quantities on the specimens prepared by the CPD technique as compared to the HMDS technique. The membrane blebs and pellet artifacts were increased in cervical cells prepared by using CPD technique50. HMDS reacts with water to produce hexamethyldisiloxane and ammonia, both of which evaporate from the object. HMDS is commonly used in gas chromatography to create silyl ethers of compounds such as sugars, amino acids, alcohols, and numerous other compounds. It is not known if HMDS reacts with some of these compounds in tissues. HMDS might crosslink proteins and stiffen the tissue during the drying process28, although the exact reason that HMDS provides better preservation of embryos could not be explained except for tissue specificity. Based on the results of our investigation, CPD caused extensive shrinkage compared to HMDS, and is much more suitable for processing embryonic tissues, especially to analyze surface structure for early staged embryos. The mechanism by which HMDS works on tissue drying has not been elucidated, although slow drying with gradual evaporation in absolute anhydrous surroundings could be a reason for obtaining an excellent surface structure of the animals.
Small pieces of agar blocks with fungal colonies from previously established slide cultures were used in this study to avoid issues with maintaining the original fluffiness of the mycelial mat. CPD drying of agar pieces with fungal colonies resulted in a washing effect, while changes in the fixatives, buffers, and ethanol was found to be a better choice overcoming the difficulties with freeze drying. HMDS that works well for embryos of all stages was unable to resolve fungal cultures, while air drying with HMDS posed a significant problem: curling up and losing the structural rigidity. While CPD works well for fungal cultures, it induced instant damage to embryos especially at early stages in development, causing shrinkage on the surface. No charging or drying artifacts were observed when HMDS and CPD were used for embryos and fungal cultures, respectively. For rigid tissue like eggshell of turtles, no special processing is required except for washing and air drying at room temperature prior to mounting for imaging.
Regardless of the preparation method, gradual ethanol gradient steps should be used to reduce the potential for drying artifacts. Initially, both HMDS and CPD samples were found to have surface artifacts with shrinkage following the dehydration and drying protocols available in the literature. After a series of standardizations, we determined that the artifacts were the result of the alcohol dehydration. It is critical to use a slow dehydration process using more gradual ethanol increases during dehydration. Also, the duration of each iteration should be longer for embryos compared to fungal cultures. Further, an additional step at 100% ethanol ensured complete uniform saturation. HMDS can be removed completely or partially to allow air drying of samples, but extended exposure to HMDS and gradual air drying by evaporation were found to be effective for embryos to avoid disruption (Figure 4A,C) caused by large surface tension as observed for microbial cell attachment51. Immersing specimens in HMDS and slow drying overnight revealed an excellent surface structure in Daphnia species52 suggesting that soft delicate tissues could benefit from the slow drying process. Acceptable image quality for embryos can be obtained with slow chemical drying by gradually increasing the concentration of HMDS to ethanol following dehydration.
All samples were mounted onto aluminum stubs using a double-sided carbon tape to provide a conductive surface, a thin coat of colorless nail polish could also be used if the same sample needs to be imaged in a different orientation. Samples can be easily removed from the stubs when nail polish is used on the stub surface and positioning the samples in alternative planes is possible compared to using double-sided carbon tape. As the samples are nonconductive, it is necessary to sputter a thin layer of metal on the samples to increase conductance. A gold target was used in this study during sputter coating, and gold-palladium also yields similar results avoiding charging effects. The correct beam voltage is sample dependent, for lightly coated specimens, imaging with a field emission SEM at 2-5 kV will provide a good definition of surface structures. For tissues with adequate coating, 5 kV generally provides better signal without more beam penetration. For specimens that lack depth and are coated with gold alone, imaging at a higher voltage (10 kV) allowed better visualization of surface topography.
Preparation of delicate tissues for SEM poses distinctive challenges, and a range of sample preparation techniques can be used to overcome and minimize imaging artifacts. We provide comprehensive methods to process three different delicate biological tissue samples, for SEM imaging: embryos, rigid eggshells, and fungal cultures. In our investigation, subtle alterations to popular methods available from previously published studies were made, and we identified the most appropriate dehydration and drying methods specific to each delicate tissue type. These protocols could be adapted to obtain acceptable SEM image quality from similar biological samples.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Dr. Daniel Baldassarre, SUNY Oswego for helpful discussions and comments on the manuscript. This study was supported by Rice Creek Associate Grants, Oswego; Challenge Grants SUNY Oswego and National Science Foundation (NSF) Small Grants to PGL and JG.
Materials
| Agar | Fischer Scientific | S25127A | for slide cultures |
| Aluminum pin stub | Tedpella | 16111 | 12.7 mm x 8 mm |
| BD Difco Dehydrated Culture Media: Potato Dextrose Agar | BD 213400 | DF0013-17-6 | Media for isolation and cultivation of Fungi, yeast and molds |
| Chloramphenicol | Fischer BioReagents | BP904-100 | Antibiotic for media |
| Coarse Vermiculite | Greenhouse Megastore | SO-VER-12 | bedding medium |
| Clear 12- well plate | Corning | 07-201-589 | for fixing embryo |
| Coverslips | Fischer Scientific | S17525B | for slide culture |
| Critical Point Dryer | Quorum CPD | EMS850 | critical point drying |
| Culture dishes | Fischer Scientific | 08 747B | DISH PETRI 100X10MM 12/PK |
| Ethanol | Fischer Scientific | A406P 4 | dehydration agent |
| Forceps- Aquarius Tweezers | Tedpella | 5804 | style 4, length 108mm, widh x thickness 0.017 x 0.17 mm |
| Glutaraldehyde | Fischer Scientific | G151-1 | fixative |
| Gold target for sputter coater | DENTON VACUUM | TAR001-0158 | Gold Target, 2.375″ D X .002″ |
| Hexamethyldisilazana | Fischer Scientific | C19479-5000 | chemical drying agent |
| Kim wipes | Kimtech | S-8115 | cleaning |
| Microscope slides | Thermo Scientific | 67-762-16 | for slide culture |
| Microscopy Scissors | Tedpella | 1327 | Double pointed, stainless steel, 100 mm L (3-5/8"). |
| Micro-scissors | Tedpella | 1346 | Vannas-type, straight, 80mm L |
| Moria Perforated Embryo Spoon | Fine Science Tools | 10370-17 | Length 14.5 cm, tip diameter 20 mm, spoon depth 5 mm |
| Netwell Inserts | Corning | 0330B09 | 15 mm Inserts with 74 µm Mesh Size Polyester Membrane act as handy carriers during specimen processing into different solvents |
| Paraformaldehyde | Fischer Scientific | T353 500 | fixative |
| Peat moss | Walmart- Miracle Gro | 551705263 | bedding medium |
| PELCO tabs double stick carbon conductive tape | Tedpella | 5000 | 12 mm OD |
| Sputter coater | DENTON VACUUM | DESK V | thin metal coating |
| SEM | JEOL USA | JEOL JSM 6610LV scanning electron scope | electron microscopy |
References
- Packard, M. J. Ultrastructural Morphology of the Shell and Shell Membrane of Eggs of Common Snapping Turtles (Chelydra serpentina). Journal of Morphology. 165 (2), 187-204 (1980).
- Solomon, S. E., Watt, J. M. The structure of the eggshell of the leatherback turtle (Dermochelys coriacea). Animal Technology. 36 (1), 19-27 (1985).
- Sahoo, G., Mohapatra, B. K., Sahoo, R. K., Mohanty-Hejmadi, P. Ultrastructure and Characteristics of Eggshells of the Olive Ridley Turtle (Lepidochelys olivacea) from Gahirmatha, India. Acta Anatomica. 156, 261-267 (1996).
- Mitrus, S. The calcareous layer eggshell of the turtle Emys Orbicularis: Ultrastructure and composition. Italian Journal of Zoology. 70 (1), 13-16 (2003).
- Chang, Y., Chen, P. Y. Hierarchical structure and mechanical properties of snake (Naja atra) and turtle (Ocadia sinensis) eggshells. Acta Biomaterialia. 31, 33-49 (2016).
- Samson, R. A., Stalpers, J. A., Verkerke, W. A simplified technique to prepare fungal specimens for scanning electron microscopy. Cytobios. 24, 7-11 (1979).
- Kaminskyj, S. G. W., Dahms, T. E. S. High spatial resolution surface imaging and analysis of fungal cells using SEM and AFM. Micron. 39, 349-361 (2008).
- Wisse, E., et al. Fixation methods for electron microscopy of human and other liver. World Journal of Gastroenterology. 16 (23), 2851-2866 (2010).
- Braet, F., De Zanger, R., Wisse, E. Drying cells for SEM, AFM and TEM by hexamethyldisilazane: a study on hepatic endothelial cells. Journal of Microscopy. 186 (1), 84-87 (1997).
- Echlin, P. . Handbook of Sample Preparation for Scanning Electron Microscopy and X-Ray Microanalysis. , (2009).
- Bell, P. B., Safiejko-Mroczka, B. Preparing whole mounts of biological specimens for imaging macromolecular structures by light and electron microscopy. International Journal of Imaging System Technology. 8 (3), 225-239 (1997).
- Bahr, G. F., Bloom, G., Friberg, U. Volume changes of tissues in physiological fluids during fixation in osmium tetroxide or formaldehyde and during subsequent treatment. Experimental Cell Research. 12 (2), 342-355 (1957).
- Baker, J. R. . Principles of biological microtechnique; a study of fixation and dyeing. , (1958).
- Glauert, A. M. Practical methods in electron microscopy. Journal of Microscopy. 3, 5-65 (1974).
- Hayst, M. A. Biological Applications. Principles and techniques of electron microscopy, 4th ed. , 13-105 (1970).
- Pathan, A. K., Bond, J., Gaskin, R. E. Sample preparation for SEM of plant surfaces. Materials Today. 12 (1), 32-43 (2010).
- Hayat, M. A. . Electron microscopy: Biological applications. 4th ed. , (2000).
- Hayat, M. A. . Fixation for electron microscopy. , (1981).
- Karnovsky, M. J. A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. Journal of Cell Biology. 27, 137-138 (1965).
- McDowell, E. M., Trump, B. F. Histologic fixatives suitable for diagnostic light and electron microscopy. Archives Pathology and Laboratory Medicine. 100, 405 (1976).
- Wisse, E., et al. Fixation methods for electron microscopy of human and other liver. World Journal of Gastroenterology. 16 (23), 2851-2866 (2010).
- Fischer, E. R., Hnasen, B. T., Nair, V., Hoyt, F. H., Dorward, D. W. Scanning Electron Microscopy. Current Protocols in Microbiology. , (2012).
- Magalhaes, M., et al. Embryonic development of the Giant South American River Turtle, Podocnemis expansa (Testudines: Podocnemididae). Zoomorphology. 136 (4), 523-537 (2017).
- Hopwood, D. Fixatives and fixation: a review. The Histochemistry Journal. 1 (4), 323-360 (1969).
- Fox, C. H., Johnson, F. B., Whiting, J., Roller, P. P. Formaldehyde fixation. Journal of Histochemistry and Cytochemistry. 33 (8), 845-853 (1985).
- Bell, P. B., Rundquist, I., Svensson, I., Collins, V. P. Formaldehyde sensitivity of a GFAP epitope, removed by extraction of the cytoskeleton with high salt. Journal of Histochemistry and Cytochemistry. 35 (12), 1375-1380 (1987).
- Hayat, M. A. Glutaraldehyde: Role in electron microscopy. Micron and Microscopica Acta. 17 (2), 115-135 (1986).
- Nation, J. L. A new method using hexamethyldisilazane for preparation of soft insect tissues for scanning electron microscope. Stain Technology. 58 (6), 347-351 (1983).
- Bray, D. F., Bagu, J., Koegler, P. Comparison of hexamethyldisilazane (HMDS), Peldri II, and critical point drying methods for scanning electron microscopy of biological specimens. Microscopy Research Technique. 26 (6), 489-495 (1993).
- Barre, C., O’Neil, D., Bricelj, V. M. Preparation of large bivalve specimens for scanning electron microscopy using Hexamethyldisilazane (HMDS). Journal of Shellfish Research. 25 (2), 639-641 (2006).
- Lee, J. T. Y., Chow, K. L. SEM sample preparation for cells on 3D scaffolds by freeze-drying and HMDS. SCANNING. 33, 1-14 (2011).
- Araujo, J. C., et al. Comparison of hexamethyldisilazane and critical point drying treatments for SEM analysis of anaerobic biofilms and granular sludge. Journal of Electron Microscopy. 52, 429-433 (2003).
- Boyde, A., Bailey, E., Jones, S. J., Tamarin, A. Dimensional changes during specimen preparation for scanning electron microscopy. SEM/IITRI. 10, 507-518 (1977).
- Read, N. D., Porter, R., Beckett, A. A comparison of preparative techniques for the examination of the external morphology of fungal material with the scanning electron microscope. Canadian Journal of Botany. 61 (8), 2059-2078 (1983).
- Melo, I. S., Faull, J. L. Scanning electron microscopy of conidia of Thichoderma stromaticum, a biocontrol agent of witches broom disease of cocoa. Brazilian Journal of Microbiology. 35, 330-332 (2004).
- Pathan, A. K., Bond, J., Gaskin, R. E. Sample preparation for scanning electron microscope of plant surfaces- Horses for courses. Micron. 39, 1049-1061 (2008).
- Packard, G. C., Taigen, T. L., Packard, M. J., Boardman, T. J. Water relations of pliable-shelled eggs of common snapping turtle (Chelydra serpentine). Canadian Journal of Zoology. 58, 1404-1411 (1979).
- Packard, M. J., Packard, G. C., Boardman, T. J. Structure of eggshells and water relations of reptilian eggs. Herpetologica. 38, 136-155 (1982).
- Cordero, G. A., Janzen, F. J. An Enhanced Developmental Staging Table for the Pianted Turtle, Chrysemys picta (Testudines: Emydidae). Journal of Morphology. 275, 442-455 (2014).
- Yntema, C. L. Procurement and use of turtle embryos for experimental procedures. The Anatomical Record. 149, 577-586 (1964).
- Matsubara, Y., Kuroiwa, A., Suzuki, T. Efficient harvesting methods for early-stage snake and turtle embryos. Development Growth Differentiation. 58, 241-249 (2016).
- Moran, P., Coats, B. Biological Sample Preparation for SEM Imaging of Porcine Retina. Microscopy Today. , 10-12 (2012).
- Bray, D. F., Bagu, J., Koegler, P. Comparison of hexamethyldisilazane (HMDS), Peldri II, and critical point drying methods for scanning electron microscopy of biological specimens. Microscopy Research Technique. 26, 489-495 (1993).
- Botes, L., Price, B., Waldron, M., Pitcher, G. C. A simple and rapid Scanning Electron Microscope preparative technique for delicate “Gymnodinioid” Dinoflagellates. Microscopy Research Technique. 59, 128-130 (2002).
- Dekker, N. P., Lammel, C. J., Brooks, G. F. Scanning Electron Microscopy of piliated Neisseria gonorrhoeae processed with hexamethyldisilazane. Journal of Electron Microscopy. 19, 461-467 (1991).
- Fratesi, S., Lynch, F. L., Kirkland, B. L., Brown, L. R. Effects of SEM preparation techniques on the appearance of bacteria and biofilms in the Carter Sandstone. Journal of Sedimentary Research. 74, 858-867 (2004).
- Jung, S. W., Joo, H. M., Park, J. S., Lee, J. H. Development of a rapid and effective method for preparing delicate dinoflagellates for scanning electron microscopy. Journal of Applied Phycology. 22, 313-317 (2010).
- Bernd, S., Bentley, D. Use of HMDS (hexamethyldisilazane) to dry organic microstructures in etched bivalve mollusk and barnacle shells. Nautilus -Greenville then Sanibel. 116, 25-31 (2002).
- Bowen, W. R., Hemann, C. C., Johnson, A. A., Good, B. H. Mermithid Nematodes: SEM Observations Comparing Hexamethyldisilazane and Critical Point Drying Methods. Journal of the Arkansas Academy of Science. 44, (1990).
- Jusman, J., Ng, S. C., Osman, N. A. A. Investigation of CPD and HMDS Sample Preparation Techniques for Cervical Cells in Devloping Computer-Aided Screening System Based on FE-SEM/EDX. The Scientific World Journal. 289817, 1-11 (2014).
- Hazrin-Chong, N. H., Manefield, M. An alternative SEM drying method using hexamethyldisilazane (HMDS) for microbial cell attachment studies on sub-bituminous coal. Journal of Microbiological Methods. 90, 96-99 (2012).
- Laforsch, C., Tollrian, R. A new preparation technique of daphnids for Scanning Electron Microscopy using hexamethyldisilazane. Arch Hydrobiology. 149 (4), 587-596 (2000).

