Murine Orchiectomy and Ovariectomy to Reduce Sex Hormone Production
Summary
This manuscript describes a consistent way to quickly perform survival rodent orchiectomies and ovariectomies.
Abstract
Sex hormone signaling plays a critical role in multiple organ systems as well as in the progression of various diseases, including neurodegenerative disease. The manipulation of sex hormone levels in the murine model system allows for the study of their impact on organs/tissues and within disease progression. Orchiectomy – the surgical removal of the testes – and ovariectomy – the surgical removal of the ovaries – provide a method to deplete the endogenous sex hormones so that the precise hormone levels can be provided through drug or other delivery methods. Here, we provide rapid and minimally invasive methods for both orchiectomy and ovariectomy in the murine model system for the reduction of sex hormones. This protocol details the surgical preparation and excision of the testes through the scrotal sac, and excision of the ovaries via two incisions in the right and left lateral dorsum.
Introduction
The testes and ovaries are the primary organs responsible for sex hormone production. The cascade of hormonal communication leading to the production of testosterone and estrogen is a well-characterized process that begins in the hypothalamus with the release of gonadotropin-releasing hormone (GnRH)1. The release of GnRH causes the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the pituitary gland. As these hormones enter the bloodstream, they then affect other tissues in the body. The primary target of LH is the testes (in males) and the ovaries (in females)2. In response to LH, the testes produce and release testosterone3. Similarly, the ovaries produce estrogen4. While the intended effects of these hormones are to prepare the cells and body for fertilization and ensure a functioning reproductive system, many other bodily systems can be affected.
Sex hormones have been linked to several physiological functions. For instance, estrogen helps maintain bone homeostasis by preventing the resorption of bone by osteoclasts. For this reason, ovariectomized mouse models can be used to study the physiology of bone diseases such as osteoporosis5,6,7. Testosterone and estrogen are also research targets for many cardiovascular and neurodegenerative diseases. Recently, elevated testosterone production coupled with high-fat diet has been linked to vascular oxidative stress8. In the brain, changes in LH after ovariectomy have caused alterations in spatial memory9. Reduction of estrogens following ovariectomy has also become a model system for studying cell death in the hippocampus, as this can induce apoptosis, resulting in memory deficits10. Testosterone has also shown a role in the growth of kidneys both in mouse models and humans following kidney transplantation11.
The creation of a hormone-deprived murine model allows for the study of sex hormones and their hormone cascades on various diseases or tissues. This can be accomplished by surgical removal of the testes (orchiectomy) or ovaries (ovariectomy). This procedure can be performed in mice of any strain when they are as young as weaning age (twenty-one days) or any adult age. Ovariectomy is performed in female mice, while orchiectomy is performed in male mice. By removing these organs, the levels of estrogen and testosterone, and many of their derivatives, such as progesterone, can be greatly reduced12,13. The process of performing orchiectomies or ovariectomies in mice can be rapid and minimally invasive with the proper technique. Rapid excision of these organs in a safe and efficient manner can allow for quick surgical processing while keeping mouse numbers minimal by having a 100% survival rate when performed correctly. Here, we detail a protocol for the rapid excision of the testes and ovaries and demonstrate the proper post-surgical monitoring to enable researchers to perform this surgery quickly and safely. We also include visual examples of the sex organs and surrounding tissues to provide the surgeon with anatomical landmarks when performing this procedure.
Protocol
All animal experiments were approved by the Institutional Animal Care and Use Committee at UTSW (APN#2019-102840).
1. Murine orchiectomy
- Prepare an aseptic working field and ensure that necessary surgical instruments have been sterilized and are readily available. Follow aseptic practices for rodent survival surgery.
- Record the weight of the male mouse and administer the preferred method of anesthesia according to the institutional guidelines. In this protocol, 2% isoflurane was delivered via a precision vaporizer to maintain anesthesia. Before the procedure, administer pain medicine according to the institutional guidelines. In this protocol, 1.0 mg/kg Buprenorphine SR and 5 mg/kg Meloxicam were delivered subcutaneously.
- Once the mouse is under anesthesia, cover the animal's eyes with lubricating eye gel to avoid the formation of eye injury, corneal desiccation, or ulcer.
- Before proceeding, ensure the proper plane of anesthesia by performing a toe pinch response test.
- Prepare the animal's surgical area before placing it on the aseptic, draped surgical field.
- Next, shave the inguinal and scrotal areas of the mouse using a pair of clippers.
NOTE: Shaving the lower abdomen superior to the penis will allow for better visualization during the surgery. - Using a cotton-taped applicator, apply a thin layer of depilatory cream (see Table of Materials) to the animal's skin, covering the scrotum and surrounding area that was just shaved. Wait for 30 s and remove the cream with a piece of clean gauze.
- Use 70% ethanol-soaked gauze sponges to wipe any remaining cream and hair off the area.
NOTE: 70% ethanol in this step is to aid in the complete removal of depilatory cream. Antiseptic preparation of the surgical site is carried out in step 1.8.
- Next, shave the inguinal and scrotal areas of the mouse using a pair of clippers.
- Transfer the mouse to the surgical area and make sure a heat source is available to maintain the animal's body temperature during the operation. Place the animal in a supine position on a surgical drape and use soft adhesive tape to adhere the mouse's feet to the surgical mat.
- If the testes ascend into the abdomen, carefully palpate the abdomen to have the testes descend. Use a gloved hand to apply gentle rolling pressure downward on the abdomen.
NOTE: It is easier to perform this surgery under a dissection scope or using a pair of loupes for enhanced magnification. If performing this under a dissection scope, take a moment to position the mouse's scrotum under the visual field of the scope. A soft tape can be used to adhere the mouse's legs or abdomen to the surgical field to ensure it stays in the correct placement. - Scrub the skin with Betadine surgical scrub (or a similar antiseptic skin preparation scrub), followed by 70% alcohol scrub at least three times to ensure proper skin disinfection. For each scrub, scrub in a radial motion outward, so the middle of the incision site is scrubbed first and ending with the outer border of the shaved surgical area being scrubbed last.
- At this time, put on sterile surgical gloves. For the next steps of the procedure, use aseptic techniques. Cover the mouse with a sterile surgical drape with a small incision (cut to approximately 0.5-1 inch square to fit over the surgical site) over the animal to cover the body in the drape material.
- Create a ventral midline incision in the scrotum approximately 1 cm -1.5 cm in length using a surgical scalpel blade or similar instruments following your approved institutional animal welfare guidelines.
- Grasping the cut border of the skin, use a blunt tipped instrument to separate the skin from the underneath tissue. Take time to do this on the lateral, superior, and inferior borders of the incision.
- Isolate a testicle by using the spatula and forceps to move the incision in the skin to be centered atop one of the testicles. If this is difficult, go back to 1.11 and further separate the skin from the underneath layers.
- Take a pair of curved forceps and place them on either side of the testicle. Apply a gentle downward pressure to exteriorize the testicle. Grab the thin, transparent layer of muscle that is on top of the testicle using forceps. This is the cremaster muscle. Verification of the correct muscle layer is made by observing this layer and its circulation, moving independently from the testicle beneath it.
NOTE: Identification of correct tissue is confirmed by its transparency and ability to move independently from the underlying tissues.- Manipulate the muscle tissue using forceps until the apex of the muscle is grasped. This should be the lowest portion, with the testes and cauda epididymis located underneath.
- Grasp the cremaster muscle with forceps and move gently away from the testes and epididymis. Create a 0.5 cm incision through the cremaster muscle of the first testicle at its apex using a small pair of Vannas spring scissors.
- Place a locking clamp on the posterior edge of the cut cremaster muscle. Use a narrow pair of hemostats or a pair of locking micro needle holders. Once the tissue is clamped, gently lay the instrument on the lateral side of the mouse to keep the tissue clamped down during the following procedural steps.
- Use forceps to grab the superior border of the incision made in step 1.14. While the two ends of the cremaster muscle are being retracted, use another pair of forceps to reach inside the cavity and gently grasp the testicle.
- Pull it outward through the hole in the muscle. Be careful to watch for any signs of nicked blood vessels and blood in the surgical field.
- The testicle, epididymis, attached spermatic cord, and blood vessel will be apparent when the tissues are properly externalized.
- Near the caudal-dorsal end of the epididymis and testicle, there is a fibrous point of attachment of the testicle to the cremaster muscle. Roll the exteriorized tissue laterally to locate this insertion point. Sever this to avoid damage to the muscle and allow for further isolation and visualization of tissues. Use two pairs of forceps to perform a clamp and tear technique.
- Look for fatty tissue around the testicle from the inguinal fat pad. Use a pair of forceps to grab onto the fat pad and gently pull to exteriorize it.
- Do not grab the blood vessel that travels along the fat pad, as pulling on this can lead to hemorrhage.
- Locate the spermatic cord, blood vessels, and remaining fat pad that is proximal to the exteriorized tissues and use a pair of hemostats to clamp it. While holding slight tension on the distal testicle, cauterize the spermatic cord and blood vessels distal to the hemostat clamp.
- Once complete, slowly release the clamp proximal to the cauterized end and check for signs of bleeding. If present, repeat this step. It is helpful to use a pair of forceps to hold gentle tension on the distal end of the tissue being cauterized.
- Allow the severed stump of the spermatic cord to retract back into the body. Grab the two cut ends of the cremaster muscle and bring them together. Assess the size of the cut in the tissue, and close the tissue using 4-0 or 5-0 absorbable sutures. Depending on the incision size, 1 or 2 sutures will be needed. Trim the suture ends to 0.5 cm using suture scissors.
- Repeat steps 1.13-1.20 on the other side of the scrotum to remove the second testicle.
- Prepare to close the skin incision.
- Ensure good hemostasis of both tissue stumps. Poor hemostasis will result in residual bleeding in the surgical field. If this occurs, locate the bleeding and cauterize. This is avoided by slow and careful manipulation of the tissues with blunt instruments.
- If blood is seen in the surgical field, use a sterilized cotton-tipped applicator to dry the area for better visualization. Use a syringe to drip saline onto the area to irrigate any blood or fluid away from the surgical field.
- Pull the two sides of the skin incision together to prepare it for closure. Ensure the sutured ends of the cremaster incisions are not protruding into the skin incision. If needed, further trim the ends of the suture.
- Close the skin.
- If using wound clips, use forceps to evert the skin and pull it away from the underlying tissue. Place one wound clip centered over the incision. Check that the skin is well clipped. If the incision is slightly too large for one wound clip to sufficiently close it, surgical skin glue can be applied to the superior and inferior portions of the incision.
- If using sutures, place the needed number of sutures through the skin, without grabbing the underlying tissue. This type of incision will require 2-3 single interrupted sutures using 4-0 non-absorbable suture material.
- Clean skin around the surgical site gently with saline and a sterilized cotton-tipped applicator to remove any dried blood or residual antiseptic scrub.
2. Murine ovariectomy
- Prepare an aseptic working field and ensure that necessary surgical instruments have been sterilized and are readily available. Follow aseptic practices for rodent survival surgery.
- Record the weight of the female mouse and administer the preferred method of anesthesia according to your institutional guidelines. In this protocol, 2% isoflurane was delivered via a precision vaporizer to maintain anesthesia. Before the procedure, administer pain medicine according to your institutional guidelines. In this protocol, 1.0 mg/kg Buprenorphine SR and 5 mg/kg Meloxicam were delivered subcutaneously.
- Administer lubricating eye gel on the animal's eyes to avoid the formation of eye injury, corneal desiccation, or ulcer. Make sure a heat source is available to maintain the animal's body temperature during the operation. Ensure the proper plane of anesthesia by checking for a toe pinch response.
- Prepare the animal's surgical area before placing it in the surgical field.
- Gently shave the hair on the dorsolateral region of the animal's back using a pair of clippers. Remove hair from the region above the superior border of the hips and below the inferior border of the rib cage, as well as the areas between these landmarks.
- Using a cotton-tipped applicator, apply a layer of depilatory cream to the animal's skin, covering the area that was just shaved. Wait for 30 s and remove the cream with a piece of gauze, removing the fine hairs that remain.
- Use a 70% ethanol-soaked gauze sponge to wipe any remaining cream and hair from the surgical area.
NOTE: 70% ethanol in this step is to aid in completely removing depilatory cream. Antiseptic preparation of the surgical site is performed in step 2.5.
- Place the animal in the lateral position (for incision to remove one ovary at a time) in the surgical field and make sure that a source of heat is available. Scrub the operative area with Betadine surgical scrub (or a similar antiseptic skin preparation scrub), followed by 70% alcohol at least three times to ensure proper skin disinfection. For each scrub, scrub in a radial motion outward, so the middle of the incision is scrubbed first and the outer border of the shaved surgical area is scrubbed last.
- At this time, put on sterile surgical gloves. For the next steps of the procedure, use aseptic techniques. Place a surgical drape with a small incision in it (cut an approximately 0.5-1 inch square in the drape to fit the surgical site) over the animal so that the body is covered in drape material.
- Locate the ideal location for the surgical incision by finding the halfway point between the hips and ribs of the mouse (see Figure 2). Grasp the skin with forceps and make a 1.0 cm incision into the skin at this location.
- Use a blunt tipped instrument (probe, surgical spatula, or blunt tipped hemostats) to separate the skin from the underneath muscle tissue. Take time to do this on the lateral, superior, and inferior borders of the incision.
- Locate the dorsolateral muscle wall of the abdomen. If needed, remove fat tissue that lies between the skin and muscle layers. In mature mice, this fat tissue is more prominent. Move this fat tissue toward the caudal end of the mouse, exposing the muscle wall.
- Fat tissue is differentiated from the abdominal wall by color: fatty tissue appears pale white and lies more superficial, while the abdominal wall appears pink and lies deeper than the fatty tissue.
- Grasp the abdominal wall with rat tooth forceps and make a 0.5 cm incision into it.
- Grab one border of this incision with one pair of forceps, use a second pair to reach inside the body cavity, and find the ovary, uterine horn, and fat pad. Gently pull this through the incision in the muscle wall to separate the muscle from the underlying tissue and exteriorize it. Locate the transition between the uterine horn and the ovary.
- Sever the ovary by performing a crush and tear technique.
- Place one clamp on the distal end of the uterine horn and place the second clamp just distal to the first. While applying firm pressure with both clamps, move the more distal one away from the body, tearing the connection between them. To minimize bleeding and tissue damage, keep the proximal clamp stationary during this process and be careful not to also pull it away from the body.
- Alternatively, use a cautery. Use a pair of forceps or hemostats to clamp the uterine horn just proximal to the desired cautery point. Use a cautery tool to sever the tissues.
- Gently and slowly release the proximal clamp and check for signs of bleeding.
- Allow the tissue stump to retreat into the body cavity. Grab the abdominal wall and gently pull upwards. Do not force the tissue stump inwards, as this increases the likelihood of bleeding.
- Locate the borders of the incision into the muscle wall and bring them together to prepare to suture it.
- Place 1-2 single interrupted sutures into the abdominal muscle wall using 4-0 absorbable suture material. Trim the suture ends to 0.5 cm using suture scissors.
- Close the incision by allowing the skin to return to its natural resting state and then evert it upwards, being careful not to grab underlying tissue or the ends of the suture material. Place 1-2 wound clips on the everted skin. Based on your institutional guidelines, the skin can also be closed using 4-0 non-absorbable suture material.
- To remove the other ovary, switch the mouse's position to have the other lateral side facing up. Be gentle when switching the mouse's position so as not to put too much pressure on the wound clipped skin from step 2.17.
- On this side, repeat steps 2.5 – 2.17.
- Clean skin around the surgical site gently with saline and a sterilized cotton-tipped applicator to remove any dried blood or residual antiseptic scrub.
3. Post-operative care
- Per your institutional guidelines, administer pain medicine for up to 72 h post-operation. Document the animals' surgical procedure, noting the date, time, anesthetic, and analgesic used. Here, Buprenorphine slow-release and meloxicam is administered pre-operatively.
- After the completion of the animals' surgery, transfer them to a clean cage lined with a dry paper towel. Place this cage 2/3 on a heat source, leaving the other 1/3 of the cage off heat. Do not place mice together in a cage until they have recovered from anesthesia.
- Allow the animal to regain the ability to freely walk around the cage before placing them back into a clean cage with normal animal bedding. At this time, mice can be housed together if all of them have recovered from anesthesia.
- For the first several days following surgery, ensure moist food and water is easily accessible. Place some moistened food on the bottom of the cage. Frequently check on the mouse surgical site and look for any signs of infection or bleeding.
- Following surgery, ensure mice are either housed alone or with other animals that have undergone the same procedure simultaneously. A mouse recovering from surgery located in the same cage as one without surgery can introduce a hazard and should be avoided.
- For mice who have undergone ovariectomy and have wound clips on the dorsolateral sides of the back, remove any rodent igloo habitats from the cage to reduce the chance of the mouse's wound clip getting caught on the igloo during the first week of healing.
- Keep monitoring the animal for signs of infection, pain, or surgical complications every 12 h through the first 72 h of the healing time.
- Remove wound clips using a wound clip remover tool 10-14 days after surgery.
Representative Results
The procedure presented here is performed in one- to three-month-old mice in the C57BL/6J background. Male mice weighed 16-28 g, and female mice weighed 14-24 g at the time of the procedure. This procedure has been optimized to be applicable for mice of many ages, from weaning through adulthood.
Surgical orchiectomy involves a single skin incision in the ventral scrotal sac, as depicted in Figure 1A. Both testes are removed one at a time and are severed through the vas deferens and spermatic blood vessels, resulting in the removal of the testis and attached epididymis (Figure 1B). The removed contents include the testis, epididymis, and inguinal fat pad, shown in Figure 1C. Successful removal of the testicles can be confirmed by visual observation of removed tissues and ensuring good hemostasis of the tissue stump prior to closing the body cavity. The success of orchiectomy can be measured by observation of well-recovered mice that demonstrate healed incision sites (Figure 1D). Mice that have undergone orchiectomy will also demonstrate a reduction in the level of testosterone measured in mouse serum as early as a week after the procedure (Figure 1E).
Surgical bilateral ovariectomy is done by incisions into the left and right dorsum of female mice (Figure 2A). The lateral incision approach can be used if only one ovary is to be removed. In this approach, the abdominal wall is incised, and the ovarian fat pad is located. The fat pad, ovary, ovarian duct, and distal uterine horn, as depicted in Figure 2B, is exteriorized, followed by severing the distal uterine horn. This technique results in the removal of the ovary and oviduct. The ovary and ovarian duct can be removed without including the distal uterine horn, but there is a risk that any remaining ovarian cells will continue to keep the mouse in estrous cycles. Including the distal uterine horn ensures that the full ovary and duct are removed, there will be a depletion of the sex hormones, and the mice will no longer cycle. Figure 2C depicts an example of the murine female reproductive organs dissected out of the body. It can be observed that the ovaries themselves are much smaller than the surrounding tissues. A dissection scope or loupes aids in the identification of these tissues. Figure 2D demonstrates the ovarian anatomy under a dissecting scope with tissue from the uterine fat pad removed, allowing for better visualization. The success of this procedure can be measured by the observation of well-recovered mice that demonstrate healed incision sites (Figure 2E).
Ovariectomy surgery is safe and efficient when performed rapidly and with minimal disturbance to surrounding tissues. Following the procedure outlined here accomplishes this by ensuring that the skin and abdominal wall incision are positioned correctly, allowing for quick location of the uterine tissues. Identifying the inferior border of the rib cage and the superior border of the leg bones ensures the incision is made near the tissue of interest. Making an incision that is about halfway between the inferior border of the ribs and superior border of the leg bones and is located about 1.5 cm lateral to the spine allows for good positioning of the surgical site when performing a lateral incision approach (Figure 2A).
Confirmation of surgical ovariectomy is shown by observation of uterine atrophy. Figure 2F shows a uterine horn dissected from a 6-month-old mouse, while Figure 2G shows a uterine horn from a 6-month-old mouse that underwent surgical ovariectomy at three months of age. The ovariectomized uterine horn appears thinner and lacks the attachment to the ovary on the distal end. This method of confirmation of the surgical technique effectively shows that the removal of the ovary resulted in atrophy of the uterine tissue.
Once tissues have been successfully identified and excised, the surgery can be completed by closure of the wounds and careful post-operative monitoring. Proper aseptic technique14 is important in ensuring good outcomes when performing survival surgery. As the mouse begins to heal from the incisions, monitoring is performed to look for signs of pain and surgical complications. A possible surgical complication is internal bleeding because of poking or nicking blood vessels while trying to incise or manipulate the tissue. This is denoted by a persistent red, flushed appearance underneath the skin around the surgical site. Avoiding complications such as these is achieved by proper localization of incision sites and aiding the surgeon's vision by use of a surgical microscope or loupes. Visual examination is recommended every 12 hours for the first three days post-surgery. Successful surgery will result in clean, healing wounds (Figure 1D, 2E).
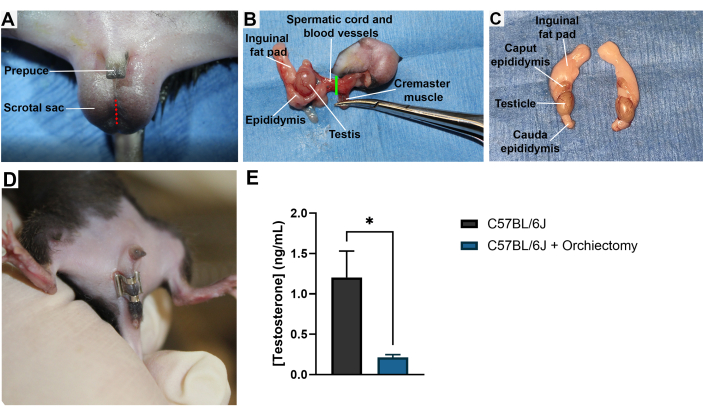
Figure 1: Male murine orchiectomy. (A) A male mouse is shown in the supine position with the location of a surgical incision into the scrotal sac for the removal of the testis, which is shown in red. (B) Image taken during the orchiectomy surgical procedure depicting the point of detachment in green. (C) Dissected-out testis, fat pads, and epididymis from a male mouse. (D) A male mouse with a well-healed wound clip over the scrotal incision following orchiectomy. (E) Mouse serum testosterone concentration in ng/mL as determined by ELISA interpolation. Both groups represent 9-week-old mice. Surgical orchiectomies were performed at 8 weeks of age. N = 3 mice per group. Samples were run in triplicate. Serum samples were collected by cardiac puncture and frozen until the time of analysis. *, p < 0.05. Error bars = SEM. Please click here to view a larger version of this figure.
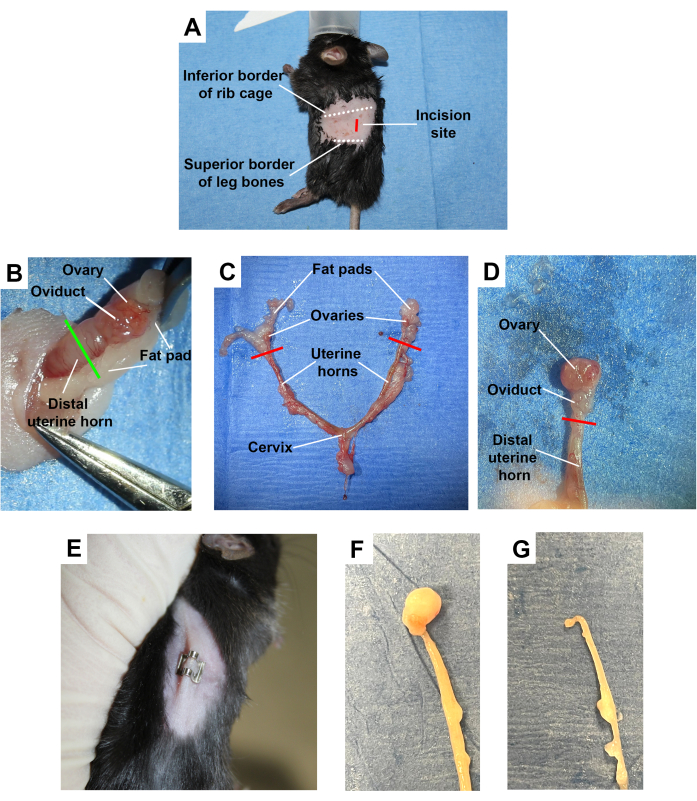
Figure 2: Female murine ovariectomy. (A) A female mouse shown in the lateral position with the surgical incision site for the removal of a single ovary from the dorsal lateral side, shown in red. Other important anatomical landmarks are highlighted. (B) Image taken during surgery depicting the exteriorized ovary, oviduct, and distal uterine horn with the point of detachment shown in green. (C) Dissected uterine horns, ovaries, and fat pads from a female mouse. (D) A close-up view of the dissected ovary, ovarian duct, and distal portion of the uterine horn with fat from the fat pad removed, allowing for better visualization of the ovary. (E) A female mouse with a well-healed wound clip over the incision following ovariectomy. (F) Dissected mouse uterine horn. (G) Dissected mouse uterine horn 3 months post-ovariectomy procedure, showing uterine atrophy. Please click here to view a larger version of this figure.
Figure 3: Graphical abstract for murine ovariectomy and orchiectomy. A pictorial representation of the orchiectomy procedure for male mice involving the removal of the testis and ovariectomy procedure involving the removal of the ovaries, both of which result in a sex hormone depleted mouse model. Please click here to view a larger version of this figure.
Discussion
Surgical removal of testes and ovaries allows for studying murine physiology under controlled hormone deprivation. This technique is important for many fields of science, including neurodegeneration, mineral metabolism, cardiovascular, and reproductive health15,16,17,18,19,20,21. Here, we detail a protocol for the fast, safe, and effective removal of murine ovaries and testis using survival surgery to deplete the sex hormones. When performed by an experienced surgeon, these procedures can take as little as 5 minutes, resulting in a very high survival rate in mice.
Some considerations when planning for a mouse surgery include the best incision site and the physiology of the animal. In the case of orchiectomy, the mouse testis can also be removed through the abdomen by a midline ventral incision13. This technique not only opens the peritoneal cavity, but also relies on the quick identification of the inguinal fat pads. For new surgeons, it is difficult to rapidly perform this technique. When mice are very young, the fat pads are also less developed and, therefore, more difficult to locate. The scrotal approach outlined in this protocol is a suitable option for researchers who are new to mouse surgery, operating on small, young mice, or who wish to minimize the chance of disturbing other organs in the peritoneal cavity by an abdominal incision.
Surgical ovariectomy involves the removal of one or both ovaries. As for most surgical procedures, safe and fast methods usually involve making the incision into the skin and underlying fascia as small as possible. This allows for reduced sutures and wound clips and faster healing. While being able to make the incisions smaller is a skill that comes with practice, being confident about the location of the incision is beneficial. Figure 2A demonstrates an example of how to locate the best incision points. Due to the small mouse anatomy, the ovary will lie near the liver, and on the left side of the mouse, near to the spleen. Accidentally probing and pulling on these organs can result in undesired bleeding or harm. We provide a detailed guide to the correct incision location and identification of the surrounding anatomical landmarks to aid in this technique and reduce the likelihood of adverse complications.
Here, we provide an example of how this procedure can be used to effectively knock down the circulating hormone levels in mice less than a week after the surgical procedure, as well as how the remaining gonadal tissue atrophies over time after the procedure. Previous methods and research detail how these can be done to generate menopause or andropause mouse models, but many of these are performed in rat models rather than mice12,22. While rats and mice have similar anatomy, the murine system has smaller anatomical structures. Here, we provide a method that reliably works in mouse models as young as 3 weeks of age. Overall, these two protocols represent a consistent way to quickly perform survival rodent orchiectomies and ovariectomies, recognize the mouse anatomy, and minimize surgical complications, creating a basis for consistent and effective results.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank the University of Texas Southwestern Medical Center Animal Resource Center for their help in surgical training and protocol review. We thank the Wert Lab support team for their invaluable assistance. This work has been supported by funds from the National Institute of Health (NIH P30EY030413). Biorender.com was used for the creation of cartoon schematics.
Materials
| 1mL Syringe | BD | 309659 | |
| 30G 1/2" Needle | BD | 305106 | |
| AutoClip System | Fine Science Tools | 12020-00 | |
| Betadine Solution | Fisher Scientific | NC0158124 | |
| Cotton-Tipped Applicators | Fisher Scientific | 10-000-692 | |
| Double -ended Micro Spatula | Fine Science Tools | 10091-12 | |
| Galilean Loupes | Fine Science Tools | 28050-30 | Optional, can provide better clarity during procedure |
| Gauze Sponges, 4"x4" | Fisher Scientific | 13-761-52 | |
| Graefe Forceps | Fine Science Tools | 11150-10 | |
| High Temp Cautery Kit | Fine Science Tools | 18010-00 | Using the fine tip attachment |
| Needle Holders | Fine Science Tools | 12001-13 | |
| PGA Absorbable Suture:4-0 / NFS-2 Reverse Cutting 19MM / 30 IN | Covetrus | 29242 | 4-0 or 5-0 Absorbable sutures are best |
| Rodent Warming pad | Kent Scientific | RT-0515 | |
| Sterile Alcohol Prep Pads | Fisher Scientific | 22-363-750 | |
| Straight Locking Micro Needle Holders | Fine Science Tools | 12060-01 | |
| Surgical Scissors | Fine Science Tools | 140-60-09 | |
| Vannas Spring Scissors – 2.5mm Cutting Edge | Fine Science Tools | 15000-08 | |
| Veet Sensitive Hair Remover Gel Cream | Amazon | N/A | |
| Wahl Professional Animal Compact Trimmer and Grooming Kit, Blue | Amazon | #9861-900 |
References
- Kaprara, A., Huhtaniemi, I. T. The hypothalamus-pituitary-gonad axis: Tales of mice and men. Metabolism. 86, 3-17 (2018).
- Stamatiades, G. A., Kaiser, U. B. Gonadotropin regulation by pulsatile GnRH: Signaling and gene expression. Molecular and Cellular Endocrinology. 463, 131-141 (2018).
- Plant, T. M., Marshall, G. R. The functional significance of FSH in spermatogenesis and the control of its secretion in male primates. Endocrine Reviews. 22 (6), 764-786 (2001).
- Fuentes, N., Silveyra, P. Estrogen receptor signaling mechanisms. Advances in Protein Chemistry and Structural Biology. 116, 135-170 (2019).
- Guo, X., Yu, X., Yao, Q., Qin, J. Early effects of ovariectomy on bone microstructure, bone turnover markers and mechanical properties in rats. BMC Musculoskeletal Disorder. 23 (1), 316 (2022).
- Yu, H., et al. High-mobility group box chromosomal protein-1 deletion alleviates osteoporosis in OVX rat model via suppressing the osteoclastogenesis and inflammation. Journal of Orthopedic Surgery and Research. 17 (1), 232 (2022).
- Sun, J., et al. Quercetin attenuates osteoporosis in orchiectomy mice by regulating glucose and lipid metabolism. Frontiers in Endocrinology (Lausanne). 13, 849544 (2022).
- Costa, R. M., et al. Testosterone contributes to vascular dysfunction in young mice fed a high fat diet by promoting nuclear factor E2-related factor 2 downregulation and oxidative stress. Frontiers in Physiology. 13, 837603 (2022).
- Bohm-Levine, N., Goldberg, A. R., Mariani, M., Frankfurt, M., Thornton, J. Reducing luteinizing hormone levels after ovariectomy improves spatial memory: Possible role of brain-derived neurotrophic factor. Hormones and Behavior. 118, 104590 (2020).
- Pandey, R., et al. Estrogen deficiency induces memory loss via altered hippocampal HB-EGF and autophagy. Journal of Endocrinology. 244 (1), 53-70 (2020).
- Laouari, D., et al. The sexual dimorphism of kidney growth in mice and humans. Kidney International. 102 (1), 78-95 (2022).
- Ström, J. O., Theodorsson, A., Ingberg, E., Isaksson, I. M., Theodorsson, E. Ovariectomy and 17β-estradiol replacement in rats and mice: a visual demonstration. Journal of Visualized Experiments. (64), e4013 (2012).
- Valkenburg, K. C., Amend, S. R., Pienta, K. J. Murine prostate micro-dissection and surgical castration. Journal of Visualized Experiments. (111), e53984 (2016).
- Pritchett-Corning, K. R., Luo, Y., Mulder, G. B., White, W. J. Principles of rodent surgery for the new surgeon. Journal of Visualized Experiments. (47), e2586 (2011).
- Haider, A., et al. Role of sex hormones in modulating myocardial perfusion and coronary flow reserve. European Journal of Nuclear Medicine and Molecular Imaging. 49 (7), 2209-2218 (2022).
- Joll, J. E., Bersi, M. R., Nyman, J. S., Merryman, W. D. Evaluation of early bilateral ovariectomy in mice as a model of left heart disease. American Journal of Physiology Heart-Circulartory Physiology. 322 (6), H1080-H1085 (2022).
- Lu, H., Ma, K., Jin, L., Zhu, H., Cao, R. 17β-estradiol rescues damages following traumatic brain injury from molecule to behavior in mice. Journal of Cell Physiology. 233 (2), 1712-1722 (2018).
- Meydan, S., et al. Effects of testosterone on orchiectomy-induced oxidative damage in the rat hippocampus. Journal of Chemical Neuroanatomy. 40 (4), 281-285 (2010).
- Ohlson, N., Bergh, A., Persson, M. L., Wikström, P. Castration rapidly decreases local insulin-like growth factor-1 levels and inhibits its effects in the ventral prostate in mice. Prostate. 66 (16), 1687-1697 (2006).
- Tehranipour, M., Moghimi, A. Neuroprotective effects of testosterone on regenerating spinal cord motoneurons in rats. Journal of Motor Behavior. 42 (3), 151-155 (2010).
- Yamada, K., et al. The impact of ovariectomy on olfactory neuron regeneration in mice. Chemical Senses. 45 (3), 203-209 (2020).
- Koebele, S. V., Bimonte-Nelson, H. A. Modeling menopause: The utility of rodents in translational behavioral endocrinology research. Maturitas. 87, 5-17 (2016).

