Meta-Analysis of the Effectiveness and Safety of Shugan Jieyu Capsules for the Treatment of Insomnia
Summary
The presented protocol describes an investigation strategy and systematic review to evaluate the clinical effectiveness and safety of Shugan Jieyu capsules for treating insomnia.
Abstract
A systematic review and meta-analysis were conducted to evaluate the clinical effectiveness and safety of Shugan Jieyu capsules for treating insomnia by searching seven databases up to February 21, 2022. The study was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The quality of the studies was assessed using the risk of bias assessment tool. This article describes in detail how to retrieve and screen the literature. The detailed steps for conducting the meta-analysis are also included in the protocol. Fourteen studies were found to be eligible, including 1,283 insomnia patients (644 with and 639 without Shugan Jieyu capsules at baseline). The meta-analysis showed a better total clinical effectiveness (odds ratio [OR]: 5.71, 95% confidence interval [CI]: 3.56 to 9.15) and a lower Pittsburgh Sleep Quality Index (PSQI) score (mean difference [MD]: −2.95, 95% CI: −4.97 to −0.93) with combined Shugan Jieyu capsules and Western medicine compared to Western medicine alone. The secondary outcomes showed that the Shugan Jieyu capsule group had significantly reduced adverse reactions and improvements in sleep duration, night awakening, nightmares with excessive dreaming, daytime sleepiness, and low energy. Further multicenter randomized trials must be encouraged to provide more concrete evidence on whether Shugan Jieyu capsules are beneficial in routine clinical practice.
Introduction
Insomnia is a sleep disorder characterized by difficulty in falling asleep, difficulty in maintaining sleep, and premature or intermittent waking1, which can cause discomfort and disrupt daily functioning. According to the China Sleep Study Report (2022), over 300 million Chinese people suffered from sleep disorders in 2021, with a prevalence of insomnia among adults as high as 38.2%2.
Western medicine for insomnia treatment typically involves psychological therapies or medication3. Psychotherapies, such as simple insomnia behavior therapy and cognitive insomnia behavior therapy, aim to modify the behaviors that contribute to sleep difficulties. Sedative-hypnotic drugs, which act quickly to induce sleep, are also commonly used to treat insomnia4. However, these drugs can have toxic side effects, including respiratory depression, hypotension, and excessive sedation. The prolonged usage of these drugs can lead to dependence, resulting in significant harm and burden to the patients and their families5,6,7. In traditional Chinese medicine (TCM), insomnia is referred to as "no sleep" or "sleeplessness" and is believed to result from deficiencies in organs such as the liver, kidneys, spleen, and heart, as well as dysfunction of the heart and mind8. In the history of traditional Chinese medicine, several experts have described the mechanisms underlying insomnia; specifically, insomnia is thought to occur due to disorder of the function of the internal organs, the loss of harmony between the qi and blood, and the disorder of yin and yang, which lead to a loss of nourishment or the disturbance of the heart and mind. The basic pathological alternation of insomnia is the loss of yang and yin9. Utilizing the active ingredients of TCM and following the principles of TCM compounding for complex conditions, research10,11,12,13 has shown that TCM plays a critical role in preventing and treating insomnia. One example of a TCM treatment for insomnia is the Shugan Jieyu capsule14, which comprises Hypericumperforatum L15,16 and Acanthopanax senticosus17.
Acanthopanax senticosus18,19, a traditional Chinese herb, has been used for tonifying the middle qi and kidney and calming the mind. It has been found to effectively improve symptoms such as insomnia, excessive dreaming, and neurasthenia. Hypericum perforatum20, another traditional Chinese herb, has demonstrated antidepressant properties, with its active components potentially possessing similar antidepressant effects to tricyclic antidepressants (TCAS) and selective serotonin reuptake inhibitors (SSRIs), but with better tolerability. The combination of Hypericum perforatum and Acanthopanax senticosus has been found to regulate neurotransmitters, improve neuronal synaptic plasticity, and clinically relieve insomnia caused by mental illness; indeed, it improves the sleep quality and cognitive function of patients with liver-depression type insomnia21,22. Syndrome differentiation is a unique characteristic of TCM. The Shugan Jieyu capsule containing Hypericum perforatum and Acanthopanax senticosus is formulated to treat insomnia caused by stagnation of liver qi. It has been reported to be effective in improving the condition and safety of patients21,22. Combining these two herbs has been proven to relieve depression, calm the mind, soothe the liver, and strengthen the spleen23,24,25. However, there currently needs to be more conclusive evidence regarding the clinical effectiveness and safety of Shugan Jieyu capsules in treating insomnia.
There are various forms of Chinese herbal medicine for treating insomnia, including decoction, patent medicine, plaster, and auricular point sticking26. Previous research has examined the effect of traditional Chinese herbs on insomnia using meta-analyses. It has been found that traditional Chinese herbs significantly improve the symptoms of patients with primary insomnia, are superior to placebo, and have promising potential for research and development27,28. This study aims to evaluate the clinical effectiveness and safety of the Shugan Jieyu capsule in treating insomnia through a systematic evaluation and meta-analysis in order to provide clinical evidence and guidance for using these capsules.
The inclusion criteria for this study were defined using the population, intervention or exposure, comparison, outcome(s) (PICO) framework29. The inclusion criteria for this study were as follows: (1) randomized controlled trials (RCTs) or semi-RCTs that enrolled patients diagnosed with insomnia30; (2) studies in which the experimental group received Western medicine in combination with Shugan Jieyu capsules, while the control group received Western medicine alone (Western medicines include alprazolam tablets31, risperidone32, estazolam tablets33, aripiprazole orally disintegrating tablets34, mirtazapine35, escitalopram oxalate tablets36, eszopiclone tablets37, zopiclone tablets38, and fluoxetine dispersible tablets39); (3) studies published in Chinese or English; (4) studies in which the primary outcomes were the overall clinical effectiveness and the Pittsburgh Sleep Quality Index (PSQI)40. According to the "Diagnostic Effectiveness Criteria for Chinese Medicine Evidence"41 and the "Guidelines for Clinical Research on New Chinese Medicines"42, effective clinical outcomes are defined as patients being clinically documented as cured or showing efficacious results. The secondary outcomes identified include sleep latency, sleep duration, nighttime awakening, nightmares with excessive dreaming, daytime sleepiness, lack of energy, and sleep quality, all evaluated by the PSQI scale and the frequency of adverse events. Among these outcome measures, sleep latency, sleep duration, nighttime awakening, nightmares, daytime sleepiness, lack of energy, and sleep quality are all evaluated using the PSQI scale43.
The exclusion criteria were defined according to the PICO framework. The exclusion criteria for this study were as follows: (1) studies unrelated to insomnia; (2) duplicate published research; (3) non-randomized controlled trials; (4) non-clinical patient studies, such as preclinical and animal model studies; and (5) case reports, comments, conferences, reviews, and dissertations.
Protocol
1. Software installation
- RevMan software installation
- Go to RevMan's official website (see Table of Materials), and according to the configuration of the computer system, select the compatible software version, and download it in the form of an exe file.
- Double-click to run the exe file: Next > Select the appropriate installation path > Next > Finish.
- EndNote software installation
- Go to EndNote's official website (see Table of Materials) to download the installation package compatible with the computer's version as a ZIP file.
- Decompress the ZIP file within a local folder. Double-click to start the installer.
- Configure the installation options: Next > Welcome to Endnote X9 > I would like a 30-day free trail > Next > I accept the license agreement > Next > Typical > Next > Finish.
2. Literature search and screening
- Create the retrieval strategy. Determine the search terms according to the inclusion and exclusion criteria. The details of the retrieval strategy for this study are provided in Supplementary File 1.
- Perform a literature search through various databases (common databases: Pubmed, CBM, Web of Science, CNKI, Cochrane Library, VIP, Embase, and Wan fang; see Table of Materials). Take PubMed as an example:
- Go to the PubMed database website.
- Enter the retrieval strategy based on the Pubmed search rules.
NOTE: In this study, the query might be ("insomnia" OR "restlessness" OR "sleeplessness" OR "wakefulness" OR "inability to sleep") AND ("Medicine, Chinese Traditional" OR "Traditional Chinese Medicine" OR "Zhong Yi Xue" OR "Chinese Traditional Medicine" OR "Chinese patent medicine" OR "Shugan Jieyu Capsules"). In this query, the operator "AND" indicates that the studies must have all the keywords, the operator "OR" indicates that the studies must have at least one of the keywords, and the parentheses indicate the order of these logical operations. The literature search time for this study was up to February 21, 2022 (Supplementary Figure 1A). - Download all the retrieved literature: Save > PubMed > Create file (Supplementary Figure 1B).
- Perform the literature screening.
NOTE: Take Pubmed as an example using Endnote.- Import the retrieved literature: File > Import > Options > Import Options > PubMed (NLM) > Import (Supplementary Figure 2A).
- Create inclusion and exclusion groups: My Groups > Create group set or Create group. For example, the exclusion groups contain inappropriate interventions, inappropriate patients, duplication of literature, etc. (Supplementary Figure 2B).
- Remove the duplicates: All References > References > Find Duplicates > Cancel. Then, all the duplicate literature is selected. Drag all the selected documents to the left side of the "Trash" to exclude them. After automatically eliminating the duplicate literature, manually check again to eliminate any duplicate literature (Supplementary Figure 2C).
- For literature screening, firstly, read the titles and abstracts of the studies to delete any that do not meet the inclusion criteria, and then further read the full texts to determine whether each study is included.
- Consider performing a manual search of the referenced works in the selected studies to increase the search's exhaustiveness.
- Consider reaching out to the corresponding authors of the literature to request any missing or unclear data to maximize study inclusion and avoid uncertainty in the data collection.
NOTE: The literature search and screening must be performed by two or more independent researchers. Final decisions should be made after discussion, or a third researcher must judge in the case of disagreement. - Use Microsoft Word to create a PRISMA flowchart44 documenting the number of studies retrieved or excluded for each reason (Supplementary File 2).
3. Data extraction
- Create an Excel extraction spreadsheet containing a list of specific information to be extracted for each literature. For example, this information could include the first author, year of publication, sample size (experimental group/control group), gender, patient age, intervention, duration of treatment, and medication use in the experimental and control groups.
- Ensure that two reviewers independently conduct the data extraction and make the final decision after discussion or after a judgment by a third researcher when there is disagreement.
- Summarize the outcome indicators that reflect the effectiveness and safety of the treatment of insomnia with Shugan Jieyu capsules in the two groups. Extract the sample size and composition ratio for the outcome indicators of dichotomous variables; however, extract the mean and standard deviation for continuous variables.
NOTE: When two or more included studies mention the same outcome index, extract the specific data of that outcome index. For example, this could include the total clinical effectiveness rate, PSQI score, adverse effects, etc.
4. Risk of bias assessment and quality of evidence
- Use the Cochrane Risk Assessment Manual45 to evaluate the risk of bias (RoB) of the included literature.
- Divide the evaluation grades into unclear, low, and high risk based on seven aspects: random sequence generation, blinding of participants and personnel, allocation concealment, incomplete outcome data, blinding of outcome assessment, selective reporting45, and other biases (other biases refer to the potential risk of bias in the included studies, such as in relation to the age of the participants, co-interventions, etc.).
- Produce quality evaluation charts.
- Create a new file. Open the RevMan software: File > New > Next, and then select an appropriate type of systematic evaluation: systematic evaluation > Intervention review > fill in the title > Next > Full review > Finish (Supplementary Figure 3A).
- Add the included literature: Studies and references > References to studies > Included studies > Add Study (Supplementary Figure 3B). Enter the study ID (e.g., Xiao Juan 2017)24.
- Enter the quality evaluation results: Tables > Characteristics of studies > Characteristics of included studies. Select the literature to be evaluated, and enter the evaluation result and basis for each item (Supplementary Figure 3C).
NOTE: If the RoB is "Unclear risk", enter "Unclear risk" in the text box after it; otherwise, it cannot be displayed in the quality evaluation chart. - Create a graph of the RoB: Figures > Add Figure > Risk of bias graph or Risk of bias summary > Save (Supplementary Figure 4A).
- Use the GRADE guidelines46 to assess the quality of evidence, and generate a summary sheet in which the results of each study included in the meta-analysis are classified based on evidence quality.
NOTE: The overall quality is downgraded according to five factors (risk of bias, indirectness, imprecision, inconsistency, and publication bias) and upgraded according to three factors (large effect size, negative bias, and dose-effect relationships). The GRADE guidelines classify the quality of evidence as high, medium, low, and very low, reflecting the degree of confidence that the effect estimates are correct. - Ensure that two reviewers independently assess the risk of bias and quality of the evidence and that disagreements are resolved by discussion or by an adjudicator.
5. Meta-analysis
- Start the meta-analysis by using RevMan software: Data and analyses > Add Comparison, then add a comparison and enter the name of the control group > Finish.
- Add the outcome indicators: Add Outcome > select the variable type ("Dichotomous" for dichotomous variables; "Continuous" for continuous variables) > Next, and then enter the outcome indicators (e.g., "Total clinical effectiveness", "PSQI overall score") > Finish (Supplementary Figure 4B).
- Add the study data: Add Study Data, then select the literature > Finish (Supplementary Figure 5A).
- Create forest plots47: Enter the extracted data > FE or RE > Forest Plot (Supplementary Figure 5B).
NOTE: Due to the non-homogeneity of the population and intervention, random effect models were used to combine all the effect sizes in this study.
Representative Results
Search results
A total of 1,117 studies were identified from databases, including 204 in Chinese and 913 in English. After eliminating 392 duplicates, 725 studies remained. After reading the titles and abstracts, 31 studies remained. Eventually, 14 studies were included through reading the full texts. The search process and selection procedure are shown in Figure 1.
Study characteristics
The characteristics of the included studies are provided in Table 1. The 14 studies involved 1,283 patients with insomnia (644 treated with and 639 treated without Shugan Jieyu capsules), including 445 females (35%), 528 males (41%), and 310 patients of unknown gender. The patients ranged in age from 18 to 74 years, with an average of 46 years. There were no statistically significant baseline differences in patient gender, age, or basic disease between the experimental and control groups in the 14 studies. All the randomized controlled trials (RCTs) were conducted in China.
Methodological quality
The methodological quality assessment results of the 14 studies are shown in Figure 1 and Figure 2. Five trials reported specific randomization methods (random number table)48,49,50,51,52 and, thus, were assessed as low risk. One trial53 was judged as high risk, using the order of consultation, and other trials mentioned randomization but no specific methods. None of the trials reported minute details of allocation concealment and blinding of the outcome assessment. None of the studies used placebo, and blinding appeared unlikely to be implemented, so the risk of participant bias and lack of personnel blinding was considered high. In all the studies, the risk of incomplete outcome reporting was lower. For five25,50,53,54,55 studies, it was difficult to determine whether there was a risk of selective reporting. Two studies47,48 had unclear risks due to incomplete baseline measurements.
Primary outcomes
Nine studies24,49,50,51,52,54,56,57,58, including 866 patients, reported the total number of clinically effective cases. The total clinical effectiveness rate was 94% (406/433) in the experimental group compared to 73% (317/433) in the control group. Compared with the control group, the total clinical effectiveness was significantly higher in the experimental group (OR: 5.71, 95% CI: 3.56 to 9.15, I2 = 0%) (Figure 3). Eight studies stated that the experimental group showed reduced overall PSQI scores compared to the control group, and one study reported opposite results25. After pooling the data, the mean difference (MD) was −2.95 (95% CI: −4.97 to −0.93, I2= 97%) (Figure 4).
Secondary outcomes
Eight studies24,48,49,52,53,56,57,58, including 717 patients, reported a number of adverse events. The adverse reaction rate was 13% (45/358) in the experimental group compared to 21% (74/359) in the control group. The experimental group had significantly fewer adverse reactions (OR: 0.56, 95% CI: 0.32 to 0.97, I2 = 35%) (Figure 5). The experimental group did not show a significant reduction in sleep latency (MD: −0.26, 95% CI: −1.32 to 0.80, I2 = 96%) (Figure 6). Four studies49,50,52,55, including 392 patients, reported the sleep duration. Three studies showed that the experimental group showed statistically significant improvements in sleep duration, and one study reported that the experimental group did not differ significantly from the control group in terms of sleep duration52. After pooling the data, the MD was 1.06 (95% CI: 0.11 to 2.01, I2 = 91%) (Figure 7).
In comparison with the control group, the experimental group was associated with a significant decrease of −0.46 in night awakening (95% CI: −0.57 to −0.35, I2 = 0%), of −0.48 in nightmares with excessive dreaming (95% CI: −0.56 to −0.39, I2 = 0%), of −0.40 in daytime sleepiness (95% CI: −0.69 to −0.10, I2 = 87%), and of −0.41 in low energy (95% CI: −0.54 to −0.29, I2 = 0%) (Figure 8, Figure 9, Figure 10, Figure 11). Additionally, the experimental group showed significantly improved sleep quality (MD: −0.78, 95% CI: −1.54 to −0.02, I2 = 90%) (Figure 12).
Quality of evidence
The results of the GRADE46 evaluation on the studies examining the effectiveness and safety of Chinese medicine Shugan Jieyu capsules for insomnia treatment showed that the evidence qualities were medium, low, or very low. The details can be found in Supplementary Table 1.
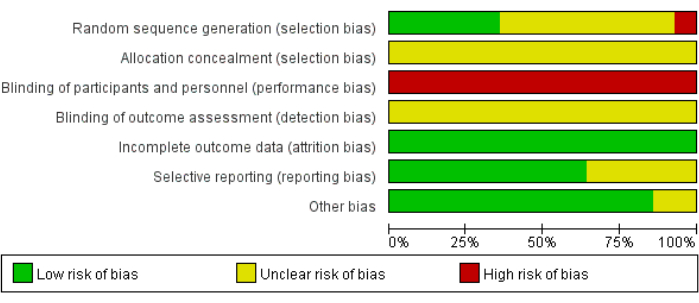
Figure 1: Assessment of the risk of bias according to the authors' judgment. This graph represents the percentages of respective bias across all the included literature. Please click here to view a larger version of this figure.

Figure 2: Risk of bias summary. The specific biases identified for each study included in the analysis. The red, yellow, and green circles represent high, unclear, and low risk of bias, respectively. For further details, please see Table 1. Please click here to view a larger version of this figure.
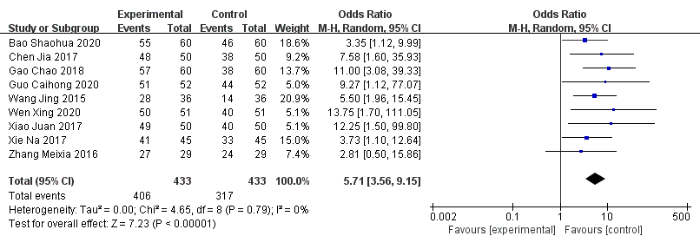
Figure 3: Forest plot for the pooled analysis of the total clinical effectiveness. Please click here to view a larger version of this figure.
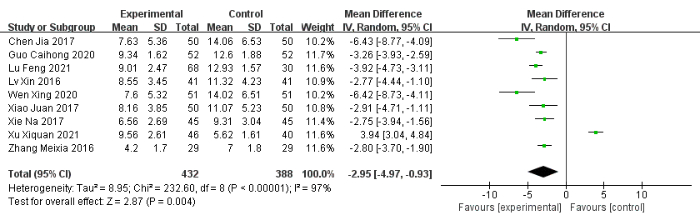
Figure 4: Forest plot for the pooled analysis of the PSQI total score. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
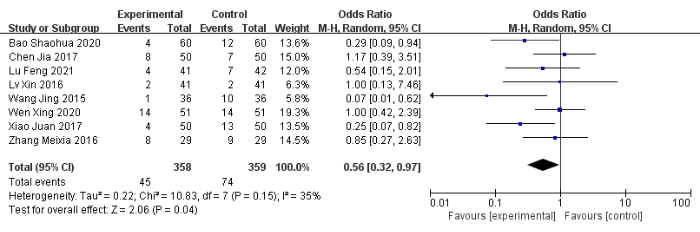
Figure 5: Forest plot for the pooled analysis of the improvement of adverse reactions. Please click here to view a larger version of this figure.

Figure 6: Forest plot for the pooled analysis of sleep latency improvement. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
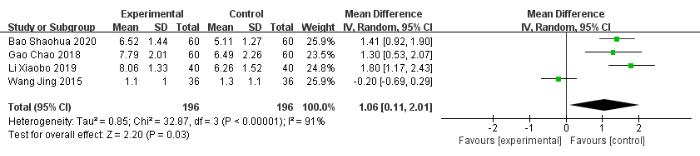
Figure 7: Forest plot for the pooled analysis of sleep duration improvement. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
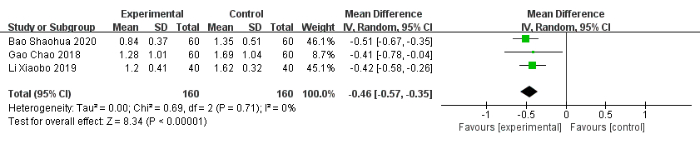
Figure 8: Forest plot for the pooled analysis of night awakening. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
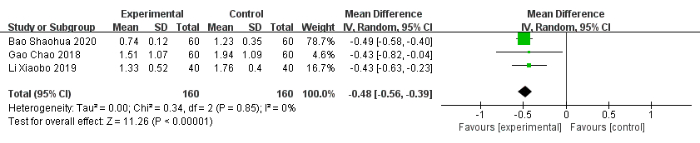
Figure 9: Forest plot for the pooled analysis of nightmares with excessive dreaming. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
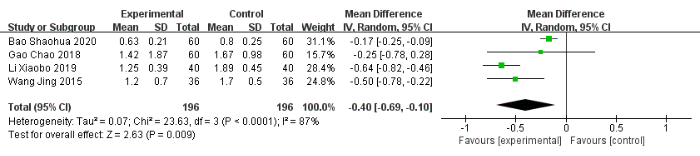
Figure 10: Forest plot for the pooled analysis of daytime sleepiness. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
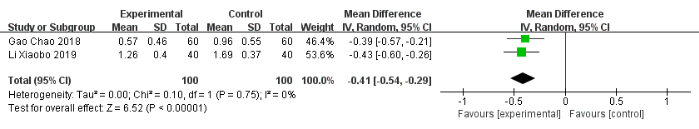
Figure 11: Forest plot for the pooled analysis of low energy. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.

Figure 12: Forest plot for the pooled analysis of sleep quality. Abbreviation: SD = standard deviation. Please click here to view a larger version of this figure.
Table 1: Basic information of the included studies. *: (1) Total clinical efficiency; (2) PSQI overall score; (3) Adverse reactions; (4) Sleep latency; (5) Sleep duration; (6) Night awakening; (7) Nightmares with excessive dreaming; (8) Daytime sleepiness; (9) Low energy; (10) Sleep quality. Abbreviations: T = experimental group; C = control group; "-" = not mentioned. Please click here to download this Table.
Supplementary Figure 1: Handling the PubMed database. (A) The main window of the literature search in Pubmed. (B) The main window for downloading all the retrieved literature in Pubmed. Please click here to download this File.
Supplementary Figure 2: Working in Endnote. (A) The main window for importing the retrieved literature in Endnote. (B) The main window for creating the inclusion and exclusion groups in Endnote. (C) The main window for removing duplicates in Endnote. Please click here to download this File.
Supplementary Figure 3: Adding research to RevMan. (A) The main window for creating a new file in RevMan. (B) The main window for adding the included literature in RevMan. (C) The main window for entering the quality evaluation results in RevMan. Please click here to download this File.
Supplementary Figure 4: Literature quality evaluation in RevMan. (A) The main window for creating a graph of RoB in RevMan. (B) The main window for adding the outcome indicators in RevMan. Please click here to download this File.
Supplementary Figure 5: Meta-analysis. (A) The main window for adding the study data in RevMan. (B) The main window for creating forest plots in RevMan. Please click here to download this File.
Supplementary Table 1: GRADE evidence profile. 1, downgrade one level: the quality of included literature is low; 2, downgrade one level: heterogeneity in the data synthesis results, I2 > 50%; 3, downgrade one level: the confidence interval is too wide; 4, downgrade one level: the sample size is too small. Abbreviations: MD = mean difference; OR = odds ratio; CI = confidence interval. *: the overall quality of the evidence. Please click here to download this File.
Supplementary File 1: The details of the retrieval strategy for this study. Please click here to download this File.
Supplementary File 2: PRISMA checklist. Overall, 1,117 articles were identified initially through the literature search, and 14 full-text articles were reviewed and entered into the final analysis. Please click here to download this File.
Discussion
In the present study, a comprehensive evaluation of the effectiveness and safety of Shugan Jieyu capsules for treating insomnia was conducted, and the following results were observed. In the experimental group, using Shugan Jieyu capsules in combination with conventional Western medicine improved the overall clinical effectiveness for patients with insomnia and resulted in higher PSQI scores. Additionally, the Shugan Jieyu capsules group showed improvements in clinical outcomes, such as sleep latency, night awakening, nightmares, daytime sleepiness, lack of energy, and sleep quality compared to the control group.
Previous research has demonstrated that Hypericumperforatum L can inhibit the reuptake of central neurotransmitters such as 5-hydroxytryptamine, dopamine, and norepinephrine, thereby increasing the concentration of neurotransmitters in the synapse and producing antidepressant and anxiolytic effects59. Acanthopanax senticosus, on the other hand, has been shown to improve the blood flow to the heart and brain, regulate the function of the central nervous and endocrine systems, and increase the viability of neurons60, thus producing calming, fatigue-reducing, and immune-boosting effects. Since Shugan Jieyu capsules are mainly composed of these two ingredients, they may be effective in treating insomnia61.
In terms of safety, the use of Shugan Jieyu capsules resulted in significantly fewer clinical adverse reactions in the treatment of insomnia compared to the use of Western medicine alone (including alprazolam tablets, risperidone, dexzopiclone tablets, aripiprazole orally disintegrating tablets, mirtazapine, escitalopram oxalate tablets, eszopiclone tablets, zopiclone tablets, and fluoxetine dispersible tablets) in the control group. A previous study by Shi et al.62 found that the combination of Shugan Jieyu capsules and non-benzodiazepines (NBZDs) was associated with improved clinical effectiveness and PSQI scores compared to NBZDs alone for the adjunctive treatment of insomnia, but their study did not report any findings on safety. A study by Wang et al.63 on using Shumian capsules for insomnia found that while the clinical effectiveness was not significantly improved compared to Western medicine, the capsules demonstrated better safety compared to Western medicine. The results of the present meta-analysis suggest that using Shugan Jieyu capsules has higher effectiveness than Western medicine alone. Still, due to the low quality of the literature, high-quality clinical studies are needed to fully explore and compare the clinical effectiveness and safety of these two treatments.
Meta-analysis is a statistical method used to compare and synthesize the results of studies on a particular clinical scientific problem, and meta-analyses are often utilized in systematic reviews to perform a quantitative pooled analysis. The reliability of the conclusions drawn is dependent on the quality of the included studies, making the formulation of appropriate inclusion and exclusion criteria and retrieval strategies according to the PICO framework crucial. Any retrieval bias or omission in the literature can lead to significant information bias in the meta-analysis results.
Before combining the effect sizes, a meta-analysis should test for heterogeneity. When the heterogeneity is considerable, the results of the meta-analysis may be less reliable, and techniques such as random-effects models, subgroup analysis, and meta-regression should be used to correct the results, compare the combined effects before and after the correction, or remove the low-quality literature before re-running the meta-analysis64. However, these correction methods do not guarantee the elimination of heterogeneity, as this study still showed high levels of heterogeneity after both using random-effects models and removing low-quality literature. Further investigation into better methods for addressing heterogeneity is necessary.
The combination of Shugan Jieyu capsules with Western medicine appears to improve clinical symptoms and the overall effectiveness rate for insomnia patients to a certain extent while reducing the side effects. Further multicenter randomized trials examining clinical endpoints are needed to provide evidence on the usefulness of Shugan Jieyu capsules in routine clinical practice.
Limitations
Several limitations exist in this meta-analysis, which must be considered. Above all, the methodological quality of the studies was poor. None of the trials reported specific details of allocation concealment, blinding of the outcome assessment, and blinding of the participants and personnel. Secondly, some of the outcome measures were evaluated using the PSQI scale, and the data in the evaluation form were from patients' subjective self-assessment, so there may have been cases of patients filling the forms incorrectly or deviation between subjective evaluations and the actual situation, which would affect the quality of the evaluation. In addition, selection bias may exist because no trials showed negative results in the included studies. Thus, the effectiveness of Shugan Jieyu capsules for promoting insomnia might be overestimated. Meanwhile, insomnia patients often also experience depression, schizophrenia, perimenopausal syndrome, etc. The heterogeneity of the patients' accompanying diseases may affect the accuracy of the results. Moreover, subgroup analysis65 could not be performed, as it was not possible to collect the clinical outcomes of patients with different syndromes separately from the included literature. Finally, most studies had small sample sizes and short-term follow-ups, so further trials with high-quality, large sample sizes and long-term follow-ups are recommended to generate high-quality clinical evidence.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Nature Foundation of China (grant number 82004504) and the Chinese Medicine Administration of Sichuan Province (grant number 2021MS199).
Materials
| Endnote | Clarivate | Free trial version | EndNote is a widely used reference management tool to help you collect, organize, and share your references. Official website: https://endnote.com/downloads |
| Microsoft Excel | Microsoft | Official Version | Microsoft Excel, a spreadsheet software with an intuitive interface, excellent computing capabilities and charting tools, is one of the most popular data processing software for personal computers. |
| Microsoft Word | Microsoft | Official Version | Microsoft Word offers a number of easy-to-use document creation tools, as well as a rich set of features for creating complex documents, as well as text formatting or image manipulation. |
| Revman | Cochrane collaboration | Official Version | Review Manager (RevMan) is the international Cochrane Collaboration's proprietary software for systematic reviewers to produce and maintain Cochrane systematic reviews. Official website: https://training.cochrane.org/online-learning/core-software-cochrane-reviews/revman/revman-5-download/download-and-installation |
| Database websites | |||
| CBM | http://www.sinomed.ac.cn/ | ||
| CNKI | https://www.cnki.net/ | ||
| Cochrane Library | https://www.cochranelibrary.com/ | ||
| Embase | https://www.embase.com/ | ||
| Pubmed | https://pubmed.ncbi.nlm.nih.gov/ | ||
| VIP | http://www.cqvip.com/ | ||
| Wan fang | https://www.wanfangdata.com.cn/ | ||
| Web of Science | http://www.webofscience.com/ |
References
- Burman, D. Sleep disorders: Insomnia. Fp Essentials. 460, 22-28 (2017).
- Guo, L. Improving the quality of people's sleep should become a "livelihood project". Xiaokang. 488 (5), 28-31 (2022).
- Ebben, M. R., Narizhnaya. M. Cognitive and behavioral treatment options for insomnia. Mount Sinai Journal of Medicine. 79 (4), 512-523 (2012).
- Sun, Y. K., et al. Effects of sedative-hypnotic pharmacotherapy on cognitive function in patients with insomnia. Chinese Journal of Neuropsychiatric Disorders. 43 (11), 701-704 (2017).
- Chapoutot, M. et al. Cognitive behavioral therapy and acceptance and commitment therapy for the discontinuation of long-term benzodiazepine use in insomnia and anxiety disorders. International Journal of Environmental Research and Public Health. 18 (19), 10222 (2017).
- Atkin, T., Comai., S., Gobbi. G. Drugs for insomnia beyond benzodiazepines: Pharmacology, clinical applications, and discovery. Pharmacological Reviews. 70 (2), 197-245 (2018).
- Singh, A., Zhao. K. Treatment of insomnia with traditional Chinese herbal medicine. International Review of Neurobiology. 135, 97-115 (2017).
- Weidong, W., Tao, L., Xue, Z., Xueyu, L. V. Clininal practice guidelines of insomnia disorder. World Journal of Sleep Medicine. 3 (2), 65-79 (2016).
- Xu, X. W., Lv, W., Xiao, P. Q., Sun, Q., Liu, F. L. Overview of theoretical and clinical research on insomnia with Chinese medicine. World Journal of Sleep Medicine. 6 (7), 1001-1008 (2019).
- Hou, J. J., Lu, Y., Lv, Y., Yang, Q. Y., Xue, R. W. Clinical research progress of Chinese medicine for insomnia. Shaanxi Journal of Traditional Chinese Medicine. 40 (2), 270-272 (2019).
- Liang, J. H., Guan, B. H., Wu, L. M., Wang, B., Gu, W. X. Clinical research progress of traditional Chinese medicine in treating insomnia. Chinese Medicine Modern Distance Education of China. 20 (16), 198-201 (2022).
- Yu, Q. M., Chen, W. Y., Wang, Q., Li, M., Research progress of Chinese herbal medicines commonly used in the treatment of insomnia in Chinese medicine. Acta Chinese Medicine and Pharmacology. 50 (3), 102-107 (2022).
- Zhao, Q., Li, J. C., Huang, T., He, Q., Li, Y. P. Research progress of the classic prescription Tianwang Buxin Dan in the treatment of insomnia. China Pharmacy. 33 (18), 2295-2298, 2304 (2022).
- Expert panel of the Chinese Academy of Traditional Chinese Medicine, Psychosomatic Medicine Branch. Expert consensus on the clinical application of Shu liver relief capsules for mild to moderate depressive disorders. Journal of Beijing University of Traditional Chinese Medicine. 44 (11), 969-977 (2021).
- Sun, S. B. et al. A review of studies on the improvement of sleep function by Schisandra chinensis, Acanthopanax spp. and Ziziphi jujubae. Medical Food Therapy and Health. 19 (05), 196-198 (2021).
- Liu, H., Dai, Q. M., Zhong, L. L., Li, J. Advances in the mechanism of antidepressant effects of Guan Ye Lian Qiao. Journal of Liaoning University of Traditional Chinese Medicine. 23 (07), 40-44 (2021).
- Lv, Y., et al. Overview of clinical research on the treatment of depression with Shuhelian Jieyu capsule. Journal of Shanxi College of Traditional Chinese Medicin.e. 19 (5), 74-77 (2018).
- Liu, H. D. et al. Exploring potential mechanism of ciwujia tablets for insomnia by UPLC-Q-TOF-MS/MS, network pharmacology, and experimental validation. Frontiers in Pharmacology. 13, 990996 (2022).
- Zhou L., Wang, M. N., Zhu, X., Cheng, H. M., Wang, Y. X. Active ingredients, pharmacological actions, and clinical applications of Acanthopanax senticosus in the central nervous system. Journal of Hunan University of Chinese Medicine. 38 (8), 961-964 (2018).
- Kumar, V. et al. Hypericum perforatum: Nature's mood stabilizer. Indian Journal of Experimental Biology. 38 (11), 1077-1085 (2000).
- Dang, J. J. et al. Study on the evidence of chronic insomnia. Chinese Journal of Basic Chinese Medicine. 28 (9), 1547-1550 (2022).
- Zhou, J. B., Yu, Z. J., Mai, S. Z., Lu, X. X., Liang, M. M. A randomized controlled trial of ShuMian capsule and ShuLiver relief capsule in the treatment of primary insomnia with liver qi stagnation evidence. China Medical Innovation. 16 (19), 120-123 (2019).
- Li, L., Zhao, Q. L. Clinical observation on the treatment of post-stroke depression with Shuhelian Xieyu capsule. Chinese Journal of Practical Neurological Disorders. 14 (9), 18-20 (2011).
- Xiao, J., Wang, C. J., Kong, X. F., Wang, L., Ping, H. K. Observation on the efficacy of combining liver detoxification capsule with western medicine in the treatment of perimenopausal insomnia. Journal of Modern Traditional Chinese and Western Medicine. 26 (23), 2584-2586 (2017).
- Xu, X. Q., Zheng, Y. Q., Xu, Q. W., Cai, Q. J. Efficacy of Shuhelian Xieyu capsule combined with risperidone in the treatment of schizophrenia with insomnia in the elderly and the effect on patients' sleep quality and cognitive ability. World Journal of Sleep Medicine. 8 (6), 997-998 (2021).
- Zhang, Z. C., Li, S., Chen, Y., Liu, H., Zhang, Z. Progresson traditional Chinese medicine in the treatment of insomnia. Guangming Journal of Chinese Medicine. 37 (15), 2740-2743 (2022).
- Ni, X. et al. Traditional use of Chinese herbal medicine for insomnia and priorities setting of future clinical research. Journal of Alternative and Complementary Medicine. 25 (1), 8-15 (2019).
- Zhang, H., Liu, P., Wu, X., Zhang, Y., Cong, D. Effectiveness of Chinese herbal medicine for patients with primary insomnia: A PRISMA-compliant meta-analysis. Medicine. 98 (24), e15967 (2019).
- Patterson, P. D., et al. Evidence-based guidelines for fatigue risk management in EMS: Formulating research questions and selecting outcomes. Prehospital Emergency Care. 21 (2), 149-156 (2017).
- Gao, H. Diagnostic criteria for chronic insomnia disorder in the International Classification of Sleep Disorders (3rd edition). World Journal of Sleep Medicine. 5 (5), 555-557 (2018).
- Alprazolam is initially effective in the treatment of insomnia. InPharma. 610 (1), 9 (1987).
- Yin, Y., et al. Low-dose atypical antipsychotic risperidone improves the 5-year outcome in Alzheimer's disease patients with sleep disturbances. Pharmacology. 96 (3-4) 155-162 (2015).
- Post, G. L. et al. Estazolam treatment of insomnia in generalized anxiety disorder: A placebo-controlled study. Journal of Clinical Psychopharmacology. 11 (4), 249-253 (1991).
- Takaki, M. Aripiprazole as monotherapy at bedtime was effective for treatment of two cases of obsessive-compulsive disorder and insomnia. American Psychiatric Publishing. 26 (3), E64 (2014).
- Dolev, Z. Case series of perimenopausal women with insomnia treated with mirtazapine followed by prolonged-release melatonin add-on and monotherapy. Archives of Women's Mental Health. 14 (3), 269-273 (2011).
- Guthrie, K. A. et al. Effects of pharmacologic and nonpharmacologic interventions on insomnia symptoms and self-reported sleep quality in women with hot flashes: A pooled analysis of individual participant data from four MsFLASH trials. Sleep. 41 (1), zsx190 (2018).
- Huo, S. et al. Effects of eszopiclone on sleep quality and cognitive function in elderly patients with Alzheimer's disease and sleep disorder: A randomized controlled trial. Brain and Behavior. 12 (2), e2488 (2022).
- Teaima, M. et al. Construction of sublingual trilaminated Eszopiclone fast dissolving film for the treatment of Insomnia: Formulation, characterization and in vivo clinical comparative pharmacokinetic study in healthy human subjects. PLoS One. 17 (6), e0266019 (2022).
- Satterlee, W. G., Faries, D. The effects of fluoxetine on symptoms of insomnia in depressed patients. Psychopharmacology Bulletin. 31 (2), 227-237 (1995).
- Lu, T. Y., Li, Y., Xia, P., Zhang, G. Q., Wu, D. R. Reliability and validity analysis of the Pittsburgh Sleep Quality Index. Chongqing Medicine. (3), 260-263 (2014).
- Diagnostic efficacy criteria for Chinese medicine illnesses by the State Administration of Traditional Chinese Medicine.31.-32. Nanjing University Press. Nanjing (1994).
- State Administration of Traditional Chinese Medicine. Guiding principles for clinical research of new Chinese medicines. Beijing: China Pharmaceutical Science and Technology Press. 135-139 (2002).
- Buysse, D. J., Reynolds, C. F. 3rd, Monk, T. H., Berman, S. R., Kupfer, D. J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research. 28 (2), 193-213 (1989).
- Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Medicine. 6 (7), e1000097 (2010).
- Higgins, J. P. T., Alterman, D. G., Sterne J. A. C. Chapter 8: Assessing risk of bias in included studies. In Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0., edited by Higgins, J. P. T., Green, S. The Cochrane Collaboration. London, UK (2011).
- Guyatt, G. H. et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. The British Medical Journal. 336 (7650), 924-926 (2008).
- Lewis, S., Clarke, M. Forest plots: Trying to see the wood and the trees. The British Medical Journal. 322 (7300), 1479-1480 (2001).
- Lu, F., Lan, Y., Wang, Y., Zhou, Y. Observation of the efficacy of combining dexzopiclone with Shuhebeijiao capsule on patients with post-stroke depression with insomnia. World Journal of Sleep Medicine. 8 (09), 1495-1497 (2021).
- Bao, S. H., Guo, T., Zhang, K., Yuan, Z. L., Tang, L. Efficacy and mechanism of action of Shuhelian Xiezhu capsule combined with escitalopram in the treatment of depression with insomnia in the elderly. Journal of Modern Traditional Chinese and Western Medicine. 29 (30), 3385-3389 (2020).
- Gao, C., Zhang, J. H. Effects of combining liver detoxification capsule with western medicine on sleep and cognition in patients with liver depression and qi stagnation type insomnia. World Traditional Chinese Medicine. 13 (05), 1127-1130 (2018).
- Xie, N., Zhang, J. C., Wang, C. J., Liu, Y. C., Wang, L. Effect of liver detoxification treatment on the psychological and BDNF effects of perimenopausal sleep disorder (anxiety-depression type). Journal of Liaoning University of Traditional Chinese Medicine. 19 (10), 135-138 (2017).
- Wang, J., Hu, Y. L. Efficacy of combined Chinese and Western medicine in the treatment of insomnia in depressive disorders. Liaoning Journal of Traditional Chinese Medicine. 42 (06), 1275-1277 (2015).
- Lv, X., Guo, S. S. Clinical study on the treatment of post-stroke sleep disorder with Shuhelian Xieyu capsule combined with dexzopiclone. Journal of Integrated Cardiovascular and Cerebrovascular Diseases of Chinese and Western Medicine. 14 (13), 1543-1544+1568 (2016).
- Guo, C. H., He, X. Q., He, X. Y. Clinical observation on the treatment of insomnia in depressive disorder by combining Shuhelian Xiezhu capsule with mirtazapine. Ningxia Medical Journal. 42 (2), 179-181 (2020).
- Li, X. B., Wang, A. H. Analysis of the effects of combining liver detoxification capsule with western medicine on patients' sleep and cognition in treating liver-depression and qi-stagnation type insomnia. Medical Theory and Practice. 32(11), 1675-1676 (2019).
- Chen, J., Huang, H. Y., Zhou, W. D., Liu, Q. J. Effects of liver detoxification capsule on sleep index and mental status of patients with schizophrenia combined with chronic insomnia. International Journal of Psychiatry. 44 (06), 1038-1040+1044 (2017).
- Zhang, M. X., Wang, L. N., Wang, X. J., Sun, Q. Q., Wang, C. H. Efficacy and safety observation of fluoxetine combined with Shuhelian Xiezhu capsule in the treatment of depression with insomnia. Journal of Xinxiang Medical College. 33 (06), 489-491, 496 (2016).
- Wen, X. Clinical efficacy and safety of liver detoxification capsule for schizophrenia with chronic insomnia. Journal of Clinical Rational Drug Use. 13 (12), 67-68 (2020).
- Chen, J. B., Jia, X. N., Quan, W., Zhang, X. H. A clinical study on the efficacy of Shuhelian Xiezhu capsule combined with escitalopram oxalate on anxiety and depression status in depressed patients. International Journal of Psychiatry., 44 (4), 635-637 (2017).
- Jia, J. M., Wang, H. T., Wang, Z. Q., Song, J., Zhao, S. H. Advances in the pharmacological activity of Acanthopanax spinosus. China Modern Traditional Chinese Medicine. 12 (2), 7-10+18 (2010).
- Huang, W., Teng, H. Y., Mao, Y. Y. Efficacy of combining liver detoxification capsule with eszopiclone in the treatment of post-stroke sleep disorder. Journal of Integrative Cardiovascular and Cerebrovascular Diseases. 16 (8), 1105-1107 (2018).
- Shi, M. L. et al. Meta-analysis of the effectiveness and safety of proprietary Chinese medicines in the treatment of insomnia. Chinese Journal of Traditional Chinese Medicine. 46 (20), 5403-5417 (2021).
- Wang, C. et al. Efficacy and safety of Shumian capsules in treating insomnia: A systematic review and meta-analysis. Medicine. 100 (50), e28194 (2021).
- Wang, D. et al. Discussing on the research of heterogeneity in meta-analysis.Chinese Journal of Evidence-Based Medicine. 9 (10), 1115-1118 (2009).
- Wang, Y. Z., Wang, J., Huang, Q. The use of subgroup analysis in drug clinical trials. Chinese Journal of Clinical Pharmacology. 28 (6), 477-480 (2012).

