Urethral Stricture Induction Followed by Buccal Mucosa Graft Urethroplasty in a Rat Model
Summary
In the present protocol, a urethral stricture induction was developed in Wistar rats, followed by urethral reconstruction with a buccal mucosa graft. A retrograde urethrogram and laser Doppler assessment were performed, validating urethral reconstruction (after stricture formation) and graft placement.
Abstract
Urethral reconstruction is an important area of expertise for urologists. The buccal mucosa is considered the best option when urethral grafting is necessary, although in some cases, it is inappropriate or needs to be optimized to repair a given stricture. Therefore, developing innovative procedures and evaluating their putative success in experimental models is crucial to fit the clinical need. With this goal, this study describes a protocol in which urethral stricture was induced by electrocautery in Wistar rats. Urethral reconstruction was performed 1 week later with a buccal mucosa graft, harvested from the lower lip, and placed in a ventral onlay fashion. A retrograde urethrogram showed a significant improvement in urethral diameter after urethroplasty compared to the respective value after stricture induction. Additionally, the graft placement was assessed by blood perfusion analysis using laser Doppler. As expected, a dark blue area corresponded to the non-vascularized buccal mucosa graft. This procedure can successfully simulate the normal pathophysiological process of urethral injury and tissue modulation, as well as urethral reconstruction using a buccal mucosa graft in a reproducible manner, and serve as the basis for future research based on tissue engineering or urethral grafts.
Introduction
Urethral reconstruction is a major challenge for urological surgeons in the management of urethral injury in the setting of strictures, trauma, or congenital defects1. With curative intent, urethroplasty is the treatment of choice for most patients, with long (>2 cm) and anterior urethral defects requiring some form of substitution urethroplasty2. Many tissues have been used as urethral substitutes, including full or split-thickness skin grafts of genital or extragenital areas, bladder wall mucosa, or the widespread buccal mucosa2. Buccal mucosa grafts have several advantages, such as coming from a wet and hairless environment, being easy to harvest, being resistant to infection, a thick epithelium, a reduced likelihood of pseudo-diverticulum formation, and a thin lamina, allowing early imbibition and inosculation3. In contrast with flaps, grafts have no blood supply, depending on the recipient’s vascular bed to survive4.
Animal models of grafts or flaps have been used extensively to develop or refine surgical techniques, study and understand tissue physiology, underlying mechanisms, and causes of failure, and evaluate innovative treatment strategies5,6. Although bigger animals facilitate technical execution, rodents, namely rats and mice, are easier to handle and maintain, resistant to diseases, more cost-effective, and, importantly, with the tools to investigate molecular mechanisms, crucial to test innovative therapies5,6. Several models of flaps and grafts have been described in rats using different tissues, namely skin, bone, muscle6, vessels5, and even solid organs7. However, there is scarce investigation in murine models into grafts for urethral reconstruction or tissue engineering.
Nevertheless, advances in translational science depend on animal models that mimic disease. So far, the local pathophysiology environment has not been addressed, since urethral reconstruction is performed immediately after its stricture. Herein, this study aims to perform a urethral reconstruction using a buccal mucosa graft in a local pathophysiology environment. With this objective, the urethral stricture was induced 1 week before its reconstruction. This experimental model, performed in rats, allows to test innovative therapies and investigate their molecular mechanisms and clinical advantages in the future.
Protocol
All animal procedures were performed according to Directive 2010/63/EU. The procedures were approved by the institutional Animal Welfare Body, licensed by DGAV, the Portuguese competent authority for animal protection (license number 0421/000/000/2021). Male Wistar Han IGS (Crl:WI(Han) rats (400-500 g) at 12-14 weeks of age were used for the present study. The animals were obtained from a commercial source (see Table of Materials).
1. Preparation of solutions
- Anesthetic solution
- Fill a 3 mL syringe with 2.3 mL of medetomidine (1 mg/mL; 0.715 mg/kg body weight) and transfer to a 15 mL centrifuge tube.
- Fill a 3 mL syringe with 1.55 mL of fentanyl (0.05 mg/mL; 0.02 mg/kg body weight) and transfer to the same 15 mL centrifuge tube.
- Fill a 10 mL syringe with 6.15 mL of midazolam (5 mg/mL; 9.5 mg/kg body weight) and transfer to the same 15 mL centrifuge tube, thus making a 10 mL anesthetic solution.
- Label the tube and store it at 4 °C in the dark.
NOTE: This anesthetic combination (medetomidine, fentanyl, and midazolam) provides an up to 3 h anesthetic surgical window. The administration of atipamezole and flumazenil may revert the effect.
- Anti-sedative solution
- Fill a 1 mL syringe with 0.7 mL of atipamezole (5 mg/mL; 3.72 mg/kg body weight) and transfer to a 15 mL centrifuge tube.
- Fill a 10 mL syringe with 9.3 mL of flumazenil (0.1 mg/mL; 1.56 mg/kg body weight) and transfer to the same 15 mL centrifuge tube, thus making a 10 mL anti-sedative solution.
- Label the tube and store it at 4 °C in the dark.
- Postoperative analgesia solutions
- Fill a 1 mL syringe with 0.5 mL of carprofen (50 mg/mL; 5 mg/kg body weight) and transfer to a 15 mL centrifuge tube.
- Add 9.5 mL of NaCl solution (0.9%), obtaining a 10 mL carprofen solution.
- Fill a 1 mL syringe with 1 mL of buprenorphine (0.3 mg/mL; 0.01-0.05 mg/kg body weight) and transfer to a 15 mL centrifuge tube.
- Add 9 mL of NaCl solution (0.9%), obtaining a 10 mL buprenorphine solution.
- Label the tubes and store them at 4 °C in the dark.
- Perioperative antibiotic
- Fill a 2.5 mL syringe with 2.5 mL of enrofloxacin (25 mg/mL; 10 mg/kg body weight) and transfer to a 15 mL centrifuge tube.
- Add 7.5 mL of NaCl solution (0.9%), obtaining a 10 mL enrofloxacin solution.
- Label the tube and store it at 4 °C in the dark.
NOTE: The details of all the reagents to prepare the above solutions are listed in the Table of Materials.
2. Surgical induction of urethral stricture
NOTE: The surgical procedures were performed using a stereo microscope (10x) (see Table of Materials).
- Sterilize all surgical tools before use: scalpel blade (number 11), pointed forceps, spring scissors, surgical forceps, ophthalmic needle holder, surgical scissors, and a needle holder. Use sterile cotton balls to clean the surgical field.
- Load the syringe with the anesthetic solution before holding the rat.
- Restrain the animal using a tube or a towel and elevate the tail to expose the abdomen.
- Keep the animal restrained and perform an intraperitoneal injection of the anesthetic solution (prepared in step 1.1).
- Test the rat's pedal withdrawal reflexes to assess anesthesia.
- Apply protective eye gel in both eyes of the animal. Perform a subcutaneous injection of the antibiotic solution (prepared in step 1.1), at 10 mg/kg of body weight.
- Place the rat in the dorsal decubitus position on a heated pad and use a dissection microscope at 10x or 20x magnification to perform the surgical procedure.
- Clean the penis and surrounding abdominal skin with povidone-iodine (100 mg/mL).
- Manually retract the foreskin and place a superficial stay suture (7.0 suture; see Table of Materials) in the dorsal aspect of the penile glans to apply traction to the penis, leaving the needle holder in place to keep the penis retracted.
- Place a 22 G venous catheter into the urethra for catheterization using a lubricant gel.
- Using a surgical scalpel blade (number 11), perform a 1 cm longitudinal ventral incision in the penile skin.
- Using the pointed forceps and the spring scissors, dissect the penile tissue layers until exposing the urethra at the mid-shaft level.
- With an electrocautery device (see Table of Materials), apply a current with 10 W, for 1 s, in the lateral aspects of the urethra (in one location on each side), ventrally at the level of the midshaft of the penis.
- Close the incision with a 6.0 absorbable running suture (see Table of Materials).
- Remove the urethral catheter. Remove the penile traction suture.
- Perform the subcutaneous injection of analgesia: Carprofen at 5 mg/kg of body weight, and buprenorphine at 0.03 mg/kg of body weight.
- Load the syringe with the anti-sedative solution (prepared in step 1.2) and, with the animal in ventral decubitus, tent the loose skin to administer a subcutaneous injection of the anti-sedative solution.
3. Surgical procedure of urethroplasty with a buccal mucosa graft
NOTE: The surgical procedures were performed using a stereo microscope (10x) (see Table of Materials).
- Sterilize all surgical tools needed for this intervention: pointed forceps, spring scissors, ophthalmic needle holder, surgical scissors, needle holder, three mosquito forceps, and a scalpel blade (number 11). Use small sponges to clean the surgical field.
- Administer the anesthetic, the antibiotic, the restrain and place the animal as described previously (steps 2.2-2.8).
- Clean the buccal mucosa of the lower lip, the penis, and the surrounding abdominal skin with povidone-iodine (100 mg/mL).
- Place three stay sutures (7.0 sutures) in the lower lip, at both sides and in the middle, and leave a mosquito in each one to retract the lower lip and expose the inner mucosa.
- Using spring scissors and pointed forceps, harvest a 4 mm diameter graft of the lower lip inner buccal mucosa, and place it in a small recipient with sterile saline (0.9% NaCl).
- Apply compression in the donor area with a sponge for hemostasis.
- Remove the previously placed stay sutures of the lower lip.
- Expose the penis as described previously (step 2.9).
- Place a 22 G venous catheter into the urethra for catheterization using a lubricant gel.
- Using spring scissors, perform a circumferential, sub-coronal incision and deglove the penis to the base.
- Using the pointed forceps and the spring scissors, dissect the remaining layers and expose the urethra.
- Using a surgical scalpel blade (number 11) and spring scissors, perform a longitudinal ventral incision, starting 3 mm distal to the coronal sulcus in an extension of 4 mm, spatulating the urethra at the level of the previously inducted stricture (step 2).
- Place two stay sutures of 7.0 material, one at each side of the spatulation, and leave a mosquito in each one to retract the urethra.
- Place two 7.0 non-absorbable sutures, one at each end of the spatulation.
- Place the buccal mucosa graft in a ventral onlay fashion with the mucosa side facing the urethral lumen.
- Pass one of the sutures through the graft end and perform a half ellipse with a running suture.
- Repeat step 3.15 and step 3.16 with the other suture on the other side of the graft.
- Remove the urethral catheter. Reposition the penile skin.
- Close the circumferential, sub-coronal incision with a 6.0 absorbable interrupted suture.
- Remove the penile traction suture.
- Administer the analgesia followed by the anti-sedative solution, as described in steps 2.16-2.17.
4. Postoperative monitoring
- Observe the rats three to four times every hour, confirming their recovery from anesthesia. Monitor respiration and assess pedal and eye reflexes.
- Subcutaneously inject analgesia every 12 h for 48 h.
NOTE: Carprofen was administered at 1 mL/500 mg of body weight, and buprenorphine was administered at 0.5 mL/500 mg of body weight (see Table of Materials). - Provide soft food for 48 h after each procedure and water ad libitum.
- Monitor the rats daily following surgery and register their health status and surgery site appearance. Evaluated signs include facial expression, vocalization, activity state, any sign of pain, food, and drink ingestion, voiding, and bleeding.
5. Blood perfusion evaluation
NOTE: Blood flow is measured immediately before stricture induction, immediately before urethroplasty, and immediately after urethroplasty.
- Perform laser Doppler perfusion imaging.
- Anesthetize the rats using the anesthetic solution (step 1.1).
- Put the rat supine on a 37 °C heating pad with penile traction, as described in step 2.9.
- Start the laser Doppler perfusion imager (see Table of Materials) to obtain data. Pre-set the area of interest to be read by the laser beam.
- Apply the anti-sedative solution (step 1.2) to revert the anesthesia.
- Using the image analysis software, draw the region of interest (ROI) around the area of the penis and register the flux values over time.
6. Radiographic evaluation
NOTE: Stricture induction confirmation and stricture resolution after urethroplasty are confirmed with a retrograde urethrogram. This evaluation is performed 1 week after stricture induction (before urethroplasty) and 2 weeks after urethroplasty.
- Perform a retrograde urethrogram using a monoplan angiography system (see Table of Materials).
- Anesthetize the rats using the anesthetic solution.
- Place the animal on the angiography mattress in a right oblique decubitus, with penile traction, as described in step 2.9.
- Focus the cone beam on the animal's pelvic area, including the penis.
- Place a 22 G venous catheter advanced 2 mm into the distal urethra.
- Start instillating 1 mL of iodine radiographic contrast material (1:1 ratio of 623 mg/mL iopromide solution and 0.9% NaCl) into the urethra.
- Simultaneously, perform plain radiography to identify the radiographic contrast fulfilling the urethral lumen and assess the urethral diameter.
- On completion of imaging, revert the anesthesia with the anti-sedative solution.
7. Euthanasia
NOTE: Euthanasia is performed 3 weeks after urethroplasty (4 weeks after stricture induction), immediately after the last perfusion evaluation.
- Fill a 2.5 mL syringe with 2 mL/kg pentobarbital sodium (400 mg/mL).
- Perform an intraperitoneal injection of the solution to euthanize the animal.
Representative Results
A total of 12 male Wistar rats, weighing 400-500 g and 12-14 weeks old, were used for urethral stricture induction. A retrograde urethrogram (RUG1) was performed 1 week later8, confirming the success of the technique. The urethral diameter was measured in millimeters at the level of the stricture induction. After this, a urethroplasty with a buccal mucosa graft was performed in the ventral face of the rat urethra. The same rats were submitted to a second retrograde urethrogram (RUG2) 14 days after urethroplasty, and the urethral diameter was measured in millimeters at the level of the graft placement. The mean RUG1 and RUG2 diameters were 1.04 mm and 1.52 mm, respectively, showing a significant improvement (p < 0,0001) in urethral permeability (Figure 1), thereby confirming the success of the surgical intervention and good consistency between the measurements.
Local perfusion was also evaluated by laser Doppler immediately before and after urethroplasty as a noninvasive method to monitor the tissue microcirculatory environment. Tissue perfusion is shown in color-coded images, where low or no perfusion is dark blue, and the highest perfusion levels are red. The mean flux values are obtained by using Moor LDI V5.3 image processing software (see Table of Materials).
Considering the variability of the rat population, 35 male Wistar rats were used. The mean blood flow before and immediately after urethroplasty was 603.4 and 137.6 arbitrary units (A.U.), respectively. As expected, the area showing a significant reduction (p < 0,0001) in local blood flow (in blue) corresponds to the non-vascularized graft (Figure 2).
Good tolerance to the anesthetic procedure was found in all the study animals; however, previous results obtained in the lab (data not shown) revealed that the anesthesia time could be critical to allow total animal recovery, preferentially not exceeding 45 min. Postoperatively, the rats were also free from major complications.
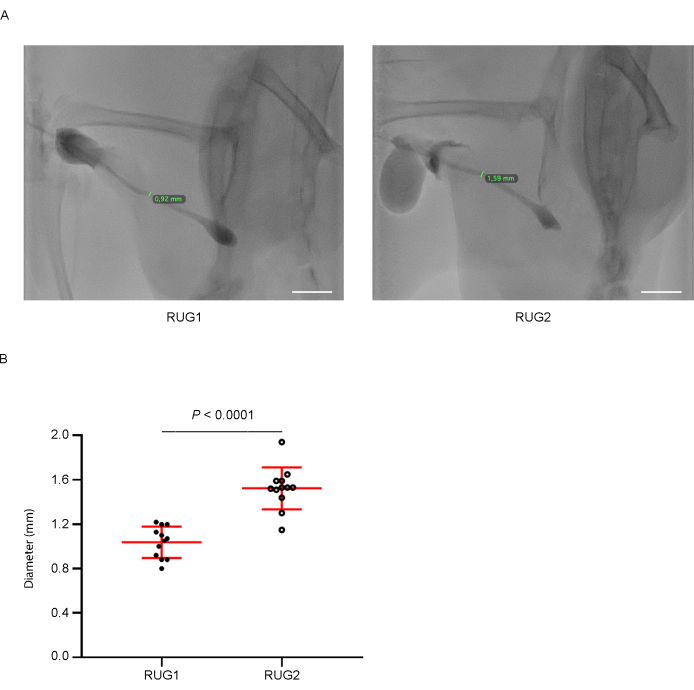
Figure 1: Retrograde urethrogram (RUG) analysis. (A) Representative images of a retrograde urethrogram before (RUG1) and 14 days after urethroplasty (RUG2). (B) Quantitative evaluation of the diameter expressed in millimeters demonstrated a significant enhancement 14 days after urethroplasty. Abbreviations: RUG1 = Retrograde urethrogram before urethroplasty; RUG2 = Retrograde urethrogram 14 days after urethroplasty. Between-group changes were assessed using a two-tailed paired t-test (n = 12). Scale bar: 10 mm. Please click here to view a larger version of this figure.
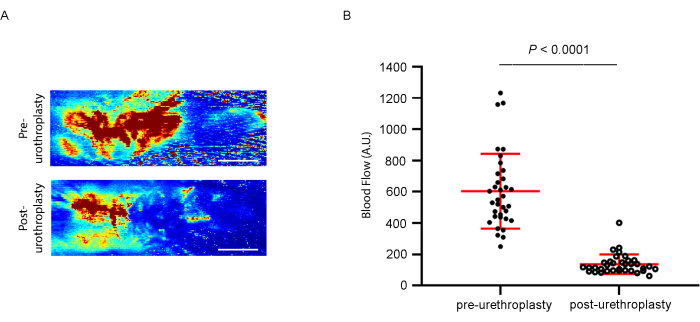
Figure 2: Laser Doppler analysis. (A) Representative images of laser Doppler flow pre- and immediately post-urethroplasty. (B) Quantitative evaluation of the blood flow demonstrated a significantly decreased blood perfusion after urethroplasty. Between-group changes were assessed by a two-tailed Wilcoxon matched-pairs signed rank test (n = 35). Scale bar: 0.5 cm. Please click here to view a larger version of this figure.
Discussion
Urethroplasty with buccal mucosa grafts is a major cornerstone in urethral reconstruction. However, innovative procedures should be developed to optimize those already described and establish new ones, such as tissue-engineered materials and biological grafts, to reduce complications and morbidity. Several procedures have been published to establish preclinical models and define surgical techniques. Souza et al.1 conducted a study including 12 New Zealand rabbits. A ventral longitudinal skin incision was performed, and the urethra was mobilized from the tunica albuginea, followed by the excision of a dorsal segment of the urethra, creating a defect. Simultaneously, a buccal mucosa graft was harvested from the cheek and placed as a dorsal onlay with a 7-0 absorbable suture. In the present study, male Wistar rats are used. Due to the smaller size, they are technically more demandable, although easier to handle. To mimic the pathophysiology of urethral strictures and the physiology of the graft takes, in this model, a urethral stricture-like disease was previously induced, as opposed to a urethral defect performed at the same operative time as the urethroplasty. Like Souza et al., running sutures to close the urethra with the graft were also used1. Nevertheless, the non-absorbable suture was used because this allows the identification of the graft circumference in further studies, such as histological ones. Martín-Cano et al.9 developed a model using Wistar rats. A sub-coronal circumferential incision was made, followed by penile degloving, allowing a good urethral exposure. The graft was harvested from the lower lip, the urethra was opened through a longitudinal ventral midline incision, and the graft was placed in a ventral onlay fashion with non-absorbable running sutures. During the procedure, a urethral catheter was placed to maintain the urethra patent. This technique described herein used the same approach of penile degloving, which allows a good exposition of the urethra and placement of a catheter to keep the urethra patent during the procedure. However, Martín-Cano et al. did not perform any previous urethral injury in the procedure, which might have influenced the natural graft take, as the tissues were healthier.
In fact, simple urethral injury and healing has been evaluated by others, like Hofer et al.10, who developed a rat model with Wistar rats consisting of a sub-coronal circumferential incision, penile degloving, and a longitudinal ventral midline incision to injure the urethra, followed by closure with a running suture. The conclusion was that, in phases of inflammation, proliferation, maturation, and remodeling analogous to dermal healing, healing of the urethra occurs. This is not limited to the site of injury, but also applies to the vast majority of periurethral tissue and the corpus spongiosum. Tavucku et al.11also described a model of urethral injury with Sprague-Dawley rats, performing a penoscrotal ventral midline skin incision to expose the urethra and then applying electrocautery current to induce urethral injury. Their data showed an increased collagen type I to collagen type III ratio, a strong indicator of fibrosis. Following this rationale, an injured tissue has more fibrosis than a healthy one, and it is expected that the graft takes are not equal in both tissues. Assuming that urethroplasties are performed in injured tissues, a major advantage of this model is that it better mimics the normal pathophysiological process. Another major advantage is the realization of a retrograde urethrogram to confirm stricture induction and later to confirm the success of urethroplasty based on urethral diameter. In fact, all animals had an improved urethral diameter after urethroplasty, showing the success of the procedure. Souza et al.1also performed retrograde urethrogram, but only at the end of the study. In this study and in Tavukcu et al.11, both urethrograms were performed, concluding the procedure's effectiveness by urethrogram analysis. Additionally, a perfusion evaluation was performed, before and after urethroplasty, which confirmed a total non-vascularized (blue) area corresponding to the buccal mucosa graft.
Nevertheless, there are several limitations related to this procedure, such as the size of the rats' urethras resulting in a demanding surgical technique, the surgery duration, and the absence of volatile anesthesia. However, it is important to consider that, although bigger animals facilitate easier technical execution, fewer molecular tools are available compared to rodents, which can be a limitation to investigating the molecular mechanisms behind innovative therapies. Another limitation is the use of a single method to induce urethral stricture, which can be due to many other causes, including trauma, infection, or congenital defects, which may lead to different pathophysiologic phenotypes. However, electrocautery was used since it allows a simple, easy, reproducible approach with consistent results.
To our knowledge, this is the first rat procedure that: (1) mimics a local pathophysiological environment previous to the graft by inducing a urethral stricture 1 week before the buccal mucosa graft placement; (2) confirms the urethral stricture and its resolution by retrograde urethrogram; (3) confirms the graft location by laser Doppler; and (4) allows an experimental model to optimize the use of grafts and develop new therapeutic strategies based on, for instance, tissue-engineered materials, in which molecular mechanisms could be investigated, with an impact on translational science to fit the clinical need.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank João Leitão, head of the Radiology department of Centro Hospitalar Universitário Lisboa Norte, and Catia Fernandes, technician of the Radiology department of Centro Hospitalar Universitário Lisboa Norte, for their cooperation in the realization of urethrograms.
Materials
| Atipamezole | OrionPharma | ANTISEDAN (atipamezole) is indicated for the reversal of the sedative and analgesic effects. | |
| Buprenorphine | RichterPharma | Buprenorphine is a derivative of the opioid alkaloid thebaine that is a more potent (25-40 times) and longer lasting analgesic than morphine. | |
| Carl Zeiss Opmi-1 FC Surgical Stereo Microscope | Carl Zeiss Microscopy, Germany | OPMI 1 FC from ZEISS symbolizes quality, precision and reliability. The manual system is easy to use and delivers high fidelity images with the legendary ZEISS optics. | |
| Carprofen | Zoetis | Carprofen is a non-steroidal anti-inflammatory drug (NSAID) of the propionic acid class. | |
| Catheter 22 G. x 1'' | B Braun | Introcan Safety | IV Catheter of fluorinated ethylene propylene (FEP) with firmer construction for arterial access. |
| Enrofloxacin | Bayer | Enrofloxacin is a fluoroquinolone antibiotic. | |
| Fentanyl | Braun | 3644960 | Fentanyl is a powerful synthetic opioid analgesic. |
| Flumazenil | Fresenius Kabi | Benzodiazepine antagonist is used for the complete or partial reversal of the sedative effects caused by benzodiazepines. | |
| High temperature cautery | Fiab | F7244 | Disposable cautery, sterile, high temperature (1200 °C), 28 mm fine tip. |
| Instillagel gel | Farco-pharma | Cellulose-based lubricant with local anaesthetic and disinfecting properties. | |
| Iopromide | Bayer | Non-ionic injectable contrast medium, with iodine. | |
| Laser Doppler imaging system (perfusion imager moorLDI2-HIR and dedicated software) | MoorLDI-V6.0, Moor Instruments Ltd, Axminster, UK | 5710 | The angiogenesis models uses Laser Doppler imaging to assess blood perfusion in the hind limbs, one of which is ligated surgically. Dedicated measurement and comparison software allows the definition of regions of interest for blood flow assessment on the ischaemic versus non-ischaemic limb to establish a "reperfusion ratio" which can be assessed as often as needed and over a number of days on the same subject. |
| Lubrithal Eye Gel | Dechra | Eye gel used in animals for prevention of dry eyes during anaesthesia. Lubricating and moisturising action on cornea and conjunctiva. | |
| Male Wistar Han IGS (Crl:WI(Han) rats | Charles River Laboratories, Spain | Twelve to fourteen-week-old | |
| Metedomidin | Virbac | 037/01/07RFVPT | Medetomidine is a synthetic drug used as surgical anesthetic. |
| Midazolam | Labesfal | Benzodiazepine medication is used for anesthesia and procedural sedation. | |
| Monoplan Angiography System | Philips Medical Systems | Azurion 7 M20 | A stationary diagnostic fluoroscopic x-ray system specifically designed to optimize the capability of users to visually and quantitatively evaluate the anatomy and function of blood vessels of the heart, brain and other organs, as well as the lymphatic system. |
| Mosquito forceps | Henry Schein | 102-4346 | Hartman-Mosquito Hemostatic Forcep Curved 3-1/2" Stainless Steel |
| Needle Holder | Henry Schein | 100-2570 | Needle holder Mayo-Hegar, stainless steel, 14 cm |
| Ophtalmic Needle Holder | Asico | AE-6143 | Needle holder barranquer most delicate without lock |
| Pentobarbital Sodium | Ecuphar | Pentobarbital Sodium is the sodium salt of pentobarbital used for euthanasia. | |
| Pointed Forceps | Aesculap | BD335R | Microforceps, 0.30 mm tip |
| Polysorb 6.0 | Medtronic (Covidien) | UL-101 | Coated Synthetic Absorbable Suture aimed to reduce the inflammatory reaction in tissues, followed by gradual encapsulation of the suture by fibrous connective tissue. |
| Providone-Iodine | Mylan | Povidone-iodine 10% is an antiseptic drug, used as a disinfectant before and after surgery. | |
| Scalpel Blade nº11 | B Braun | BB511 | Carbon steel, sterile |
| Spring Scissor | Henry Schein | 600-4826 | Surgical scissors 31 castroviejo |
| Surgical Forceps | Aesculap | BD33R | Microforceps, 0.20mm tip |
| Surgical Scissor | Aesculap | MA873R | Micro Iris Scissor, curved shrap tips |
| SurgiPro 7.0 | Medtronic (Coviden) | VP-702-X | Non-Absorbable Monofilament Polypropylene Suture indicated for use in general soft tissue ligation. |
References
- Souza, G. F., Calado, A. A., Delcelo, R., Ortiz, V., Macedo, A. Histopathological evaluation of urethroplasty with dorsal buccal mucosa: an experimental study in rabbits. International Brazilian Journal of Urology. 34 (3), 345-354 (2008).
- Andrich, D. E., Mundy, A. R. Urethral strictures and their surgical treatment. BJU International. 86 (5), 571-580 (2000).
- Bhargava, S., Chapple, C. R., Bullock, A. J., Layton, C., MacNeil, S. Tissue-engineered buccal mucosa for substitution urethroplasty. BJU International. 93 (6), 807-811 (2004).
- Fu, Q., et al. Substitution urethroplasty for anterior urethral stricture repair: comparison between lingual mucosa graft and pedicled skin flap. Scandinavian Journal of Urology. 51 (6), 479-483 (2017).
- Blain, B., et al. Vascular grafts in the rat model: an anatomic study. Microsurgery. 21 (3), 80-83 (2001).
- Schmauss, D., Weinzierl, A., Schmauss, V., Harder, Y. Common rodent flap models in experimental surgery. European Surgical Research. 59 (3-4), 255-264 (2018).
- You, H., et al. A rat orthotopic renal transplantation model for renal allograft rejection. Journal of Visualized Experiments. (180), e63464 (2022).
- Park, J. H., et al. Balloon-expandable biodegradable stents versus self-expandable metallic stents: a comparison study of stent-induced tissue hyperplasia in the rat urethra. Cardiovascular and Interventional Radiology. 42 (9), 1343-1351 (2019).
- Martín-Cano, F., et al. Histological and immunohistochemical changes in the rat oral mucosa used as an autologous urethral graft. Journal of Pediatric Surgery. 48 (7), 1557-1564 (2013).
- Hofer, M. D., et al. Analysis of primary urethral wound healing in the rat. Urology. 84 (1), e1-7 (2014).
- Tavukcu, H. H., et al. Protective effect of platelet-rich plasma on urethral injury model of male rats. Neurourology and Urodynamics. 37 (4), 1286-1293 (2018).

