Osmotic Minipump Implantation for Increasing Glucose Concentration in Mouse Cerebrospinal Fluid
Summary
This article describes a detailed protocol to increase glucose concentration in the cerebrospinal fluid (CSF) of mice. This approach can be useful for studying the effects of high CSF glucose on neurodegeneration, cognition, and peripheral glucose metabolism in mice.
Abstract
Diabetes increases the risk of cognitive decline and impairs brain function. Whether or not this relationship between high glucose and cognitive deficits is causal remains elusive. Moreover, whether these deficits are mediated by an increase in glucose levels in cerebrospinal fluid (CSF) and/or blood is also unclear. There are very few studies investigating the direct effects of high CSF glucose levels on central nervous system (CNS) function, especially on learning and memory, since current diabetes models are not sufficiently developed to address such research questions. This article describes a method to chronically increase CSF glucose levels for 4 weeks by continuously infusing glucose into the lateral ventricle using osmotic minipumps in mice. The protocol was validated by measuring glucose levels in CSF. This protocol increased CSF glucose levels to ~328 mg/dL after infusion of a 50% glucose solution at a 0.25 µL/h flow rate, compared to a CSF glucose concentration of ~56 mg/dL in mice that received artificial cerebrospinal fluid (aCSF). Furthermore, this protocol did not affect blood glucose levels. Therefore, this method can be used to determine the direct effects of high CSF glucose on brain function or a specific neural pathway independently of changes in blood glucose levels. Overall, the approach described here will facilitate the development of animal models for testing the role of high CSF glucose in mediating features of Alzheimer's disease and/or other neurodegenerative disorders associated with diabetes.
Introduction
Both type 1 and type 2 diabetes impair brain function1,2,3. For example, diabetes increases the risk of cognitive decline and neurodegenerative disorders, including Alzheimer's disease3,4. Moreover, people with diabetes have defective glucose sensing in the brain5,6. This defect contributes to the pathogenesis of hypoglycemia associated unawareness and an insufficient counter-regulatory response to hypoglycemia7,8, which can be fatal if not treated immediately.
Considering that diabetes increases glucose levels in the blood as well as in cerebrospinal fluid (CSF)9, it is important to determine whether one or both of these factors contribute to impaired brain function. Whether diabetes causes brain damage by high CSF glucose alone or in combination with other factors like insulin deficiency or insulin resistance is also an open question. Animal models of type 1 and type 2 diabetes show cognitive decline and neurodegeneration in addition to an affected energy balance and peripheral glucose metabolism10,11,12,13. However, from these models, it is not feasible to uncouple the selective effects of high CSF glucose versus blood glucose levels in mediating the complications of diabetes on brain function.
This protocol describes methods to develop a mouse model of hyperglycorrhachia to test the effects of chronically high CSF glucose levels on brain function, energy balance, and glucose homeostasis. The mouse model developed through this technique presents a tool for studies investigating the etiological role of dysregulated glucose homeostasis on neural and behavioral function.
Therefore, the proposed approach will be useful in understanding the direct effects of elevated CSF glucose levels in various pathophysiological conditions.
Protocol
All mouse procedures were approved by the Institutional Animal Care and Use Committee at the University of Rochester and were performed according to the US Public Health Service guidelines for the humane care and use of experimental animals. Six weeks old C57BL/6J male mice used for this study were commercially obtained. All the animals were group housed (5 mice per cage) in a room with a 12 h day/night cycle and were given access to food and water ad libitum. After the mice were implanted with a cannula for infusing glucose into the lateral ventricle, they were single housed to prevent any damage to the implants from other mice.
1. Assembly of osmotic minipumps
- Prepare artificial cerebrospinal fluid (aCSF) according to the following protocol. Dissolve NaCl (8.66 g), KCl (0.224 g), CaCl2.2H2O (0.206 g), and MgCl2.6H2O (0.163 g) in 500 mL of sterile water to prepare solution A. Dissolve Na2HPO4.7H2O (0.214 g) and NaH2PO4.H2O (0.027 g) in 500 mL of sterile water to prepare solution B. Then, mix solution A and B in a 1:1 ratio to prepare the aCSF.
NOTE: This formulation of aCSF contains no glucose. - To prepare the 50% glucose solution, add 50 g of glucose to 50 mL of aCSF in a beaker. Place the beaker on a hot plate and bring the temperature of the suspension to 60 °C. Mix the suspension with a magnetic stirrer until the glucose is completely dissolved.
- Add aCSF to the beaker to make the final volume 100 mL. Pass the solution through 0.22 µm pore filter to sterilize it.
- Prepare a 0.9% saline (NaCl) solution and pass the solution through the 0.22 µm filter.
- Place all the components of the minipump and infusion kit in a sterile tray and cut the tubing from the brain infusion kit to 1 inch in length with a sterile scissor. Hold the tubing with a sterile hemostat and push it on the top of one end of the cannula. Connect the other end of the tubing to the top side of the flow modulator. Cover both ends of the tubing with a small amount of glue for a stronger connection.
- Attach a 27 G needle to a 1 mL syringe and fill it with either aCSF or the glucose solution. Insert the 27 G needle into a small piece of tubing. Then, connect the tubing to the open end of the flow modulator and fill it with the corresponding solution until drops of the solution start coming out from the cannula. Remove any air bubbles trapped in the tubing.
- To fill the minipump, hold the pump upright and insert the syringe needle into the pump. Fill it with the corresponding solution until a drop of the solution starts forming at the opening of the minipump.
- Slowly insert the flow modulator into the pump while making sure not to introduce any air bubbles in the assembly.
- After the osmotic minipumps are assembled, transfer them to a 50 mL conical tube. Fill the tube with the sterile saline so that the pumps are completely submerged, while making sure that the cannula stays out of the saline solution. Four such assembled pumps can be placed in one 50 mL tube.
- Loosely place the cap on each tube and incubate the pumps at 37 °C to prime for at least 48 h.
2. Surgery to implant osmotic pumps
- Pre-surgery procedure
- Sterilize the surgical instruments in an autoclave and let them cool down for at least 30 min before use. The autoclave conditions involve steam sterilization at 121 °C for 30 min.
- Disinfect the surgery area with 70% ethanol. Wipe the knobs, ear bars, and stereotaxic frame surface.
NOTE: Surgery should be performed in an aseptic environment, keeping in mind to avoid touching non-sterile surfaces while performing surgery. Replace gloves if there is a suspicion of contamination. - Place the mouse in an anesthesia chamber to anesthetize it with 3% isoflurane mixed in air for 3-5 min. Then, remove the mouse from the chamber and place it on a sterile drape. Maintain anesthesia via a nose cone at 1.5%-2%.
- Pinch the mouse hind paw to observe any reflex and verify the depth of anesthesia.
- Inject 5 mg/kg carprofen subcutaneously for analgesia.
- Use clean clippers to clip the surgery area on the mouse head. Rub the scalp first with povidone-iodine solution and then with alcohol swabs thoroughly for at least 2 min. Repeat this process at least three times.
- Apply an eye lubricant to prevent the eyes from drying during surgery.
- Surgery
- Place the mouse on the stereotaxic frame and fit the head on the incisor bar. Cover the nose with the nose cone. Apply sterile drapes to secure the surgical area.
- Shift the flow of isoflurane to the nose cone. Continue to maintain the anesthesia at 1.5%-2% isoflurane.
- Place a heating pad under the mouse to maintain the body temperature.
- Put on new sterile gloves and, with a scalpel, make a midline incision (~10-15 mm long) on the scalp. Expose the skull surface with a spatula while using cotton tips to wipe any bleeding.
NOTE: Use a cauterizer if bleeding profusely, which is rare. - Rub with a cotton tip dipped in 3% hydrogen peroxide to expose the cranial sutures.
- Take note of bregma and lambda to navigate the skull coordinates. Set all the coordinates to zero at bregma.
NOTE: These coordinates were obtained from a mouse brain atlas14. The reference bregma-lambda distance in the atlas is 4.21 mm. To correct for age- and sex-based differences in skull size, the bregma-lambda distance for each mouse was measured and then divided by 4.21 mm to obtain a multiplication factor. For example, if the distance between bregma and lambda was 4.65 mm, division by 4.21 mm gave a multiplication factor of 1.10. All the coordinates obtained from the atlas were then multiplied with this factor to measure the actual coordinates that were used to drill the hole. In the above example, the resulting anterior-posterior (AP), and medial-lateral (ML) coordinates would be 0.55 and 1.1 mm, respectively (i.e., 1.1 x 0.5 or 1 mm). - Use an electric drill to make a hole at these coordinates: 0.5 mm posterior to bregma and 1 mm lateral to midline toward the right.
NOTE: While drilling the hole in the skull, be careful not to rupture the dura mater. Make sure the drill bit is sterile. It can be placed with other instruments in the same autoclave pouch for sterilization. - Insert a hemostat under the incised skin and push it to the place where the osmotic minipump will be implanted. Gently open and close the hemostat to make a pocket for the minipump.
- Use a sterile hemostat to pick a primed osmotic minipump from the tip of the flow modulator. Push the pump through the incised skin into the pocket.
NOTE: Be careful to not let the pump and the cannula touch any non-sterile surface. - Apply some glue on the underside of the cannula and fix it in the cannula holder. Lower the cannula slowly through the drilled hole 2 mm ventral to the skull surface. Leave it there for at least 5 min to allow the glue to solidify.
- Before inserting the cannula down into the brain, make sure that the cannula is not clogged and that the solution is flowing properly.
NOTE: Sometimes, glucose may deposit at the tip of the cannula due to a very low flow rate, blocking the flow. - With the help of a scissor, clip the top of the cannula making sure that it doesn't dislodge from the skull surface.
- Cover the cannula with a layer of dental cement for extra support.
- Use nylon sutures (5-0 size and 30 inches in length) to close the wound and apply topical antibiotic on the wound to prevent infection.
- Turn off the isoflurane and let the animal recover from the anesthesia. Then, transfer the animal to a new clean cage.
- Post-surgery care
- After surgery, monitor the mouse for at least 1 week.
- Administer 5 mg/kg carprofen subcutaneously, once every 24 hours for three days post-surgery.
- Let the wound heal completely before removing the sutures 7-10 days after surgery.
NOTE: It was observed that mice with high CSF glucose took longer to heal. Trimming nails on mouse's hind paws minimizes the chance of injury to the site of the incision due to the scratching behavior of mice.
3. Replacement of the minipumps
NOTE: Since, the minipumps used in this study last only for 4 weeks, the replacement of minipumps was also tested to extend the duration of glucose infusion, as it may be required in the case of long-term studies. This involved the following steps.
- Prepare the mice for surgery as detailed above.
- Make a small 1 cm vertical incision in the skin slightly above the pump.
- Insert a hemostat under the incised skin and pull the pump out.
- Remove the pump from the tubing and connect a new primed pump to the tubing.
- Insert the pump back inside and stitch the skin.
- Follow the same post-surgery care instructions as described above.
4. CSF collection procedure
- Preparation
- Pull 1 mm diameter glass capillaries with a micropipette puller machine to obtain 0.5 mm diameter tips. The settings are: heat = 800, pull = 15, velocity = 5, and time = 200 units. Place the pulled capillaries in a sterile box till use.
- Attach a 27 G needle to a 1 mL syringe. Insert the pulled capillary on the needle and use a small adhesive tape to secure the connection.
- Firmly fix the syringe in a capillary holder of a micromanipulator.
- Next, prepare the mouse for surgery, as described in the pre-surgery procedure above.
- Surgical procedure
- Place the mouse on the stereotaxic frame. After fixing the head on the frame, as described previously, rotate the knobs to tilt the head so that the nose is facing downward. Apply sterile drapes to secure the surgical area.
- Make a small incision on the dorsal surface, starting from the intra-aural plane to the caudal side.
NOTE: Be careful not to damage the tubing while making the incision. - Remove the support from the mouse body and let the mouse rest vertically so that the neck is fully extended dorsally.
- With the help of curved blunt forceps, gently bisect the posterior neck muscles from the midline to create a small window. Then, use a wet cotton tip applicator to gently displace the neck muscles from mid-line to the periphery.
- Observe whether the cisterna magna is exposed, which appears as a triangular window with a transparent dura membrane.
- Place the micromanipulator assembly on the stereotaxic frame next to the mouse while making sure that the capillary tip does not touch any surface.
- Gently break the capillary tip without disturbing the setup.
- While looking into the magnifying glass (magnification of 2.25x), rotate the corresponding knobs on the micromanipulator to slowly align and move the capillary tip toward the cisterna magna.
- Some resistance is felt once the capillary tip touches the cisterna magna membrane. Very slowly push the tip against the membrane with the help of the knobs.
NOTE: Be careful not to damage any blood vessels in the membrane. This step is very important to avoid any blood contamination in the CSF. - Very gently pierce the membrane. CSF will start flowing into the capillary at once due to negative pressure in the capillary.
- Leave the setup for few minutes until ~10 µL of CSF is collected in the capillary.
- Then, slowly draw the capillary out of the cisterna magna and gently press a sterile cotton tip applicator against the opening of the cisterna magna to stop CSF leakage.
- Carefully remove the capillary from the syringe and attach it to a microcap bulb dispenser. Press the bulb to transfer the CSF into a sterile microcentrifuge tube.
- Place a support under the mouse and rotate the stereotaxic knobs to level the head.
- Close the wound with nylon sutures.
- Inject 300 µL of sterile saline subcutaneously before removing the mouse from the apparatus.
- Give post-operative care to the animals as described above, until it is time to remove the sutures-~7-10 days after surgery.
5. Glucose assay
- Follow the protocol as described in the glucose colorimetric assay kit.
- Dilute the sodium phosphate buffer stock solution provided in the kit to 50 mM concentration in ultra-pure water.
- Prepare glucose standards in 0, 2.5, 5, 7.5, 10, 15, 20, 25, and 50 mg/dL ranges from the 1,000 mg/dL stock solution provided in the kit in diluted buffer solution.
- Prepare a 7 fold dilution of CSF samples in the buffer. For example, if the total CSF volume collected is 10 µL, then the seven fold diluted volume will be 70 µL.
- Pipette 15 µL of glucose standards and diluted CSF in duplicates in a 96-well plate.
- Pipette 85 µL of diluted buffer in each of the wells with standards and CSF.
- Add 6 mL of diluted buffer to the glucose calorimetric enzyme mixture vial and vortex it for a few seconds to mix thoroughly.
- Add 100 µL of the enzyme mixture preparation to each well to start the reaction.
- Seal the plate with a cover sheet and tap on the plate gently to mix the reagents.
- Place the plate in an incubator at 37 °C for 10 min. Vials with high glucose start to turn violet immediately.
- After 10 min, remove the cover and measure the absorbance at 500-520 nm using a plate reader.
- Calculate the glucose concentration in the CSF samples by interpolating their values from the standard curve.
6. Blood glucose assay
- Make a small lateral nick to the mouse tail using a razor blade and use a drop of blood to measure the blood glucose with a glucometer, according to the manual instructions.
Representative Results
Male mice were implanted with a cannula assembled to an osmotic minipump (Figure 1) to chronically infuse aCSF or a 50% glucose solution into their lateral ventricles (Figure 2). CSF was collected 10 days after the surgery (Figure 3) to validate the efficacy of this procedure. The results showed an increase in the CSF glucose levels (mean: 327.7 mg/dL) in mice infused with 50% glucose compared to that (mean: 56.5 mg/dL) in mice infused with aCSF. This is about a six fold increase in the CSF glucose levels in the experimental mice compared to their control littermates (Figure 4A). The blood glucose levels were not different between the groups (Figure 4B).
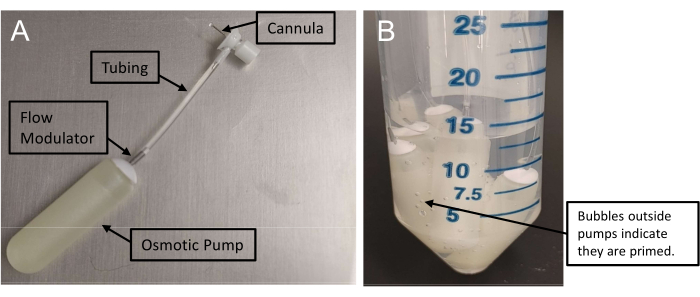
Figure 1: Assembly of osmotic minipumps. (A) Infusion assembly with a cannula connected to a minipump through tubing. These pumps require at least 48 h to prime. (B) The presence of air bubbles outside the minipumps confirms the priming. Please click here to view a larger version of this figure.
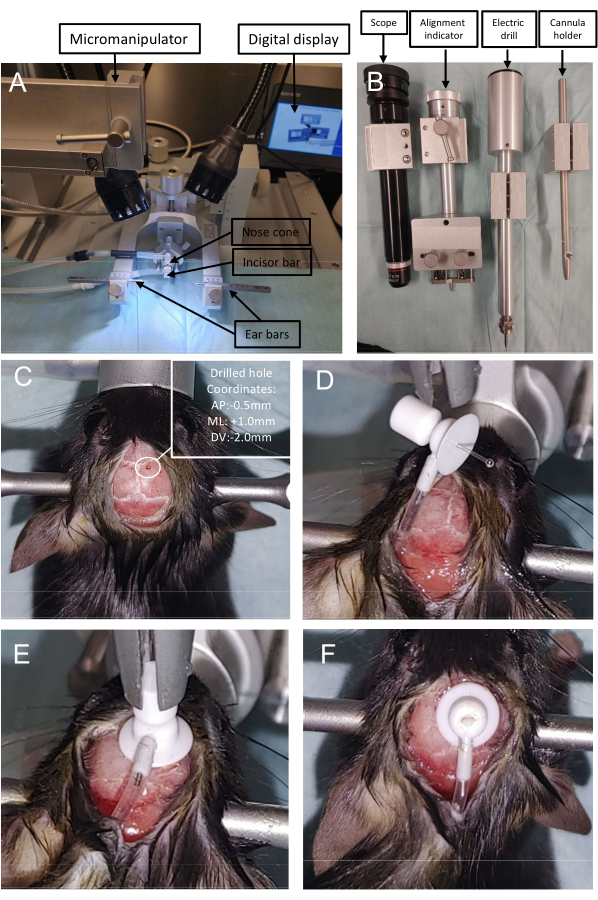
Figure 2: Stereotaxic apparatus and accessories. (A,B) Stereotaxic equipment with an attached micromanipulator and other accessories. (C) Burr hole coordinates to insert the cannula. (D) Osmotic mini pump implantation, (E,F) Insertion of the cannula in the drilled hole. Maintain aseptic conditions throughout the surgery. Please click here to view a larger version of this figure.
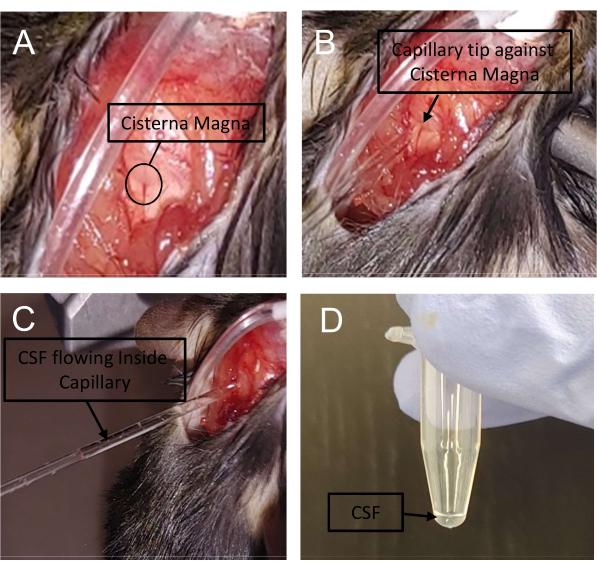
Figure 3: Cerebrospinal fluid (CSF) collection procedure. (A) The dorsal neck muscles were gently displaced with blunt forceps to expose the cisterna magna. A 1 mm capillary with a 0.5 mm diameter tip was used to (B) rupture and (C,D) collect CSF from the cisterna magna. Please click here to view a larger version of this figure.
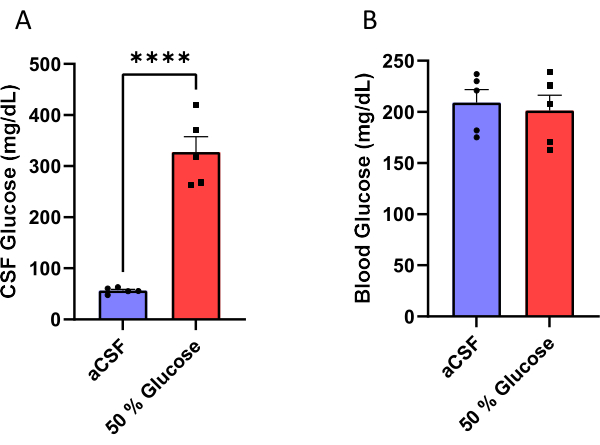
Figure 4: Measurement of glucose. (A) Increased CSF glucose (B) without affecting non-fasting blood glucose levels in mice infused with 50% glucose solution in the lateral ventricle. The efficacy of this protocol was validated by measuring CSF and blood glucose concentration 10 days after initiating the glucose infusion. Mice infused with 50% glucose solution had CSF glucose levels of 327.7 ± 30.1 mg/dL (mean ± standard error of mean) compared to mice that received artificial CSF infusion that had glucose levels of 56.5 ± 2.6 mg/dL. ****p < 0.0001, unpaired t-test. Error bars represent standard error of mean (n = 5). Please click here to view a larger version of this figure.
Discussion
This article reports a detailed protocol to increase CSF glucose in mice by using osmotic minipumps connected to a cannula implanted in the lateral ventricle. The chronic infusion of glucose in the mouse brain through this procedure will be useful in delineating the effects of long-term hyperglycorrhachia on cognition, systemic glucose metabolism, and energy balance and for better understanding the pathogenesis of diabetes complications.
Chronic diabetes causes brain damage that interrupts the communication between the brain and the peripheral organs15. Diabetes also increases the risk of neurodegenerative diseases, including Alzheimer's disease3,4. Streptozotocin (STZ)-induced type 1 diabetes has been the standard rodent model in diabetes research16; STZ damages β-cells in the pancreas, leading to type 1 diabetes-like pathology. Further, in a modified version, the use of STZ accompanied with nicotinamides can induce type 2 diabetes. Another way of developing type 2 diabetes-like phenotypes in animals is through feeding them a high fat diet16. However, in the context of studying the effect of hyperglycemia on brain function, these techniques are limited in controlling for a large number of factors (e.g., peripheral insulin/glucagon levels, and metabolic function in general). Thus, any effect of STZ-induced diabetes on brain function can only be interpreted as an associated complication, instead of pinpointing a single etiological factor. Acute injection or chronic infusion of substances in the cerebroventricular space is a technique often used to test their direct effects on brain function. Intracerebroventricular (ICV) injection of STZ has been used to develop a rodent model of Alzheimer's disease, however, it remains uncertain whether STZ-associated neural damage is due to dysregulation in glucose sensing/homeostasis or other independent mechanisms, like STZ-induced oxidative stress and DNA damage17.
The procedures described in the current protocol will be useful in developing rodent models that can answer research questions, like whether an increase in CSF glucose concentration can cause cognitive impairment. The protocol described here could be used in determining the direct effects of high CSF glucose levels on the hypothalamus and hippocampus, among other brain regions involved in nutrient sensing, metabolism, and/or cognition. This method would also clarify whether an increase in CSF glucose levels affects insulin sensitivity, insulin secretion, food intake, and/or energy balance at baseline and in response to metabolic insults. Moreover, the protocol reported here would be applicable in testing hypotheses that require longitudinal studies. For example, data could be collected before, during, and at the end of the glucose infusions to compare findings from the same animals at different times. Such a strategy would address whether complications arising from high CSF glucose level are reversible after the normal CSF glucose level is restored. In contrast, the method could also be used for hypothesis-generating studies. For example, CSF could be collected from the same animals at different times and subjected to metabolomics or proteomics analysis to identify biomarkers or any metabolic insults produced by high level of CSF glucose. Similarly, different regions of the brain could be analyzed by spatial transcriptomics to yield cell-specific information that may have been altered by high CSF glucose.
The rationale for infusing glucose-free aCSF to a sham group was to keep the CSF glucose concentration at the baseline level, so that any change in CSF glucose level induced by cannula implantation can be naturally controlled. The results in this study showed that the sham group had a CSF glucose concentration of ~60 mg/dL (~3 mM), which is in the normal CSF glucose range in mice18. The CSF glucose levels observed in individuals with type 2 diabetes are ~110 mg/dL or ~6 mM9. In the current study, ICV infusion of 50% glucose at a rate of 125µg/h elevated CSF glucose levels to ~300 mg/dL (16 mM), which is supraphysiological19. Although this supraphysiological level of CSF glucose may not be clinically relevant to the levels observed in individuals with type 2 diabetes, the results presented in this study show that the infusion of glucose in CSF can induce a chronic elevation of CSF glucose concentration in mice.
The method presented here has some limitations. It involves sophisticated mouse brain surgery that requires relevant training, skills, and experience in performing such advanced procedures. Because the catheter and minipumps are implanted for the long term, meticulous care of the mice throughout the study is necessary to monitor for health concerns or damage to the catheter assembly. A 50% glucose concentration was selected because the viscosity of a solution beyond this concentration might have affected the infusion of glucose into the ventricles. The minipumps used in this protocol had a flow rate of 0.25 µL/h, so the group of mice with 50% glucose infusion received glucose at a rate of 125 µg/h, or 3 mg of glucose per day. This dose of glucose per unit time was therefore limited by the flow rate of the minipumps.
In summary, this article reports a validated method for the chronic increase of CSF glucose in mice. The information obtained from this model will be useful in determining whether or how an increase in CSF glucose levels is involved in mediating diabetes-associated complications, such as neurodegenerative disorders, or causing peripheral metabolic insults in diabetes and obesity.
Troubleshooting
If the tubing comes off of the cannula in the mice, a small amount of glue on the cannula-tubing connection can be applied while assembling the minipump. If stitches come off and the cannula becomes visible, the incision area can be completely closed by using sutures or staples. Nails from the hind paws of the mouse should be trimmed, so that there is a lower possibility of scratching the surgery area by the mouse. Moreover, be careful not to tie off the sutures so tight that the skin will rip, as mice have delicate skin.
For fast recovery after CSF collection, the injection of 300 µL of sterile saline subcutaneously after surgery is recommended. Furthermore, keeping the maximum volume of CSF collection to 10 µL is also important.
Disclosures
The authors have nothing to disclose.
Acknowledgements
National Institutes of Health grant DK124619 to KHC.
Start-up funds and pilot research award, Department of Medicine, University of Rochester, NY, to KHC.
The Del Monte Institute for Neuroscience Pilot Research Award, University of Rochester, to KHC.
University Research Award, Office of the Vice President for Research, University of Rochester, NY, to KHC.
MUR designed and performed the method, analyzed results, prepared graphs and figures, and wrote and edited the manuscript. KHC conceived and supervised the study, analyzed results, and wrote and edited the manuscript. KHC is the guarantor of this work. All authors approved the final version of the manuscript.
Materials
| 0.22 µm syringe filter | Membrane solutions | SFPES030022S | |
| 1 mL sterile Syringe (Luer-lok tip) | BD | 309628 | |
| 1 mL TB syringe | BD | 309659 | |
| 100 mL Glass beaker | Fisher | N/a | |
| 100% Ethanol (Koptec) | DLI | UN170 | Use 70% dilution to clean the surgery area |
| 50 mL conical tube | Fisher | N/A | |
| Allignment indicator | KOPF | 1905 | |
| Alzet brain infusion kit | DURECT | Kit # 3; 0008851 | Cut tubing in the kit to 1 inch length |
| Alzet osmotic pump | DURECT | 2004 | Flow rate 0.25 µL/h |
| Anesthesia system | Kent Scientific | SomnoSuite | |
| Betadine solution | Avrio Health | N/A | |
| CaCl2 . 2H2O | Fisher | C79-500 | |
| Cannula holder | KOPF | 1966 | |
| Centering scope | KOPF | 1915 | |
| Dental Cement Liquid | Lang Dental | REF1404 | |
| Dental cement Powder | Lang Dental | REF1220-C | |
| D-glucose | Sigma | G8270 | |
| Electric drill | KOPF | 1911 | While drilling a hole avoid rupturing dura mater |
| Eye lubricant (Optixcare) | CLC Medica | N/A | |
| Glass Bead sterilizer (Germinator 500) | VWR | 101326-488 | Place instruments in sterile water to let them cool before surgery |
| Glucose Assay Kit | Cayman chemical | 10009582 | |
| H2O2 | Sigma | H1009-500ml | Apply 3% H2O2 on skull surface to make the cranial sutures visible. |
| Hair Clipper | WAHL | N/A | |
| heating pad | Heatpax | 19520483 | |
| Hemostat | N/A | N/A | |
| Isoflurane (Fluriso) | Zoetis | NDC1385-046-60 | |
| KCl | VWR | 0395-500g | |
| Magnetic stand | WPI | M1 | |
| Magnifying desk lamp | Brightech | LightView Pro Flex 2 | |
| Metal Spatula | N/A | N/A | |
| MgCl2 . 6H2O | Fisher | BP214-500 | |
| Micromanipulator (Right handed) | WPI | M3301R | |
| Micromanipulator with digital display | KOPF | 1940 | |
| Na2HPO4 . 7H2O | Fisher | S373-500 | |
| NaCl | Sigma | S7653-5Kg | |
| NaH2PO4 . H2O | Fisher | S369-500 | |
| Neosporin | Johnson & Johnson | N/A | Apply topical oinment to prevent infection |
| Parafilm | Bemis | DM-999 | |
| Rimadyl (Carprofen) 50mg/ml | Zoetis | N/A | 5 mg/kg, subcutaneous, for analgesia |
| Scalpel | N/A | N/A | |
| Stereotaxic allignment system | KOPF | 1900 | |
| Sterile 27 gauge needle | BD | 305109 | |
| Sterile cotton tip applicators (Solon) | AMD Medicom | 56200 | |
| Sterile nylon sutures (5.0) | Oasis | MV-661 | Use non-absorable suture for closing the wound |
| Sterile sharp scissors | N/A | N/A | |
| Sterile surgical blades | VWR | 55411-050 | |
| Surgical gloves (Nitrile) | Ammex | N/A | Change gloves if there is suspision of contamination |
| Tray | N/A | N/A |
References
- Moheet, A., Mangia, S., Seaquist, E. R. Impact of diabetes on cognitive function and brain structure. Annals of the New York Academy of Sciences. 1353, 60-71 (2015).
- Takeda, S., et al. Diabetes-accelerated memory dysfunction via cerebrovascular inflammation and Abeta deposition in an Alzheimer mouse model with diabetes. Proceedings of the National Academy of Sciences. 107 (15), 7036-7041 (2010).
- Arvanitakis, Z., Wilson, R. S., Bienias, J. L., Evans, D. A., Bennett, D. A. Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Archives of Neurology. 61 (5), 661-666 (2004).
- Zilliox, L. A., Chadrasekaran, K., Kwan, J. Y., Russell, J. W. Diabetes and cognitive impairment. Current Diabetes Reports. 16 (9), 87 (2016).
- Reno, C. M., Litvin, M., Clark, A. L., Fisher, S. J. Defective counterregulation and hypoglycemia unawareness in diabetes: mechanisms and emerging treatments. Endocrinology and Metabolism Clinics of North America. 42 (1), 15-38 (2013).
- Cryer, P. E., Davis, S. N., Shamoon, H. Hypoglycemia in diabetes. Diabetes Care. 26 (6), 1902-1912 (2003).
- Hwang, J. J., et al. Hypoglycemia unawareness in type 1 diabetes suppresses brain responses to hypoglycemia. The Journal of Clinical Investigation. 128 (4), 1485-1495 (2018).
- Cryer, P. E., Gerich, J. E. Glucose counterregulation, hypoglycemia, and intensive insulin therapy in diabetes mellitus. The New England Journal of Medicine. 313 (4), 232-241 (1985).
- Tigchelaar, C., et al. Elevated cerebrospinal fluid glucose levels and diabetes mellitus are associated with activation of the neurotoxic polyol pathway. Diabetologia. 65 (7), 1098-1107 (2022).
- Zheng, H., et al. Cognitive decline in type 2 diabetic db/db mice may be associated with brain region-specific metabolic disorders. Biochimica et Biophysica Acta. Molecular Basis of Disease. 1863 (1), 266-273 (2017).
- Ernst, A., et al. Diabetic db/db mice exhibit central nervous system and peripheral molecular alterations as seen in neurological disorders. Translational Psychiatry. 3 (5), 263 (2013).
- Wang, Y., Yang, Y., Liu, Y., Guo, A., Zhang, Y. Cognitive impairments in type 1 diabetes mellitus model mice are associated with synaptic protein disorders. Neuroscience Letters. 777, 136587 (2022).
- Jolivalt, C. G., et al. Type 1 diabetes exaggerates features of Alzheimer’s disease in APP transgenic mice. Experimental Neurology. 223 (2), 422-431 (2010).
- Paxinos, G., Franklin, K. B. J. . Paxinos and Franklin’s The mouse brain in stereotaxic coordinates. , (2019).
- Vinik, A. I., Maser, R. E., Mitchell, B. D., Freeman, R. Diabetic autonomic neuropathy. Diabetes Care. 26 (5), 1553-1579 (2003).
- Furman, B. L. Streptozotocin-induced diabetic models in mice and rats. Current Protocols. 1 (4), 78 (2021).
- Grieb, P. Intracerebroventricular streptozotocin injections as a model of Alzheimer’s disease: in search of a relevant mechanism. Molecular Neurobiology. 53 (3), 1741-1752 (2016).
- Kealy, J., et al. Acute inflammation alters brain energy metabolism in mice and humans: role in suppressed spontaneous activity, impaired cognition, and delirium. The Journal of Neuroscience. 40 (29), 5681-5696 (2020).
- Dougherty, J. M., Roth, R. M. Cerebral spinal fluid. Emergency Medicine Clinics of North America. 4 (2), 281-297 (1986).

