Hickman Catheter Use for Long-Term Vascular Access in a Preclinical Swine Model
Summary
A reliable and reproducible approach for the insertion and maintenance of a tunneled Hickman catheter for long-term vascular access in swine is described. Placement of a central venous catheter allows for convenient daily sampling of whole blood from awake animals and intravenous administration of medication and fluids.
Abstract
Central venous catheters (CVCs) are invaluable devices in large animal research as they facilitate a wide range of medical applications, including blood monitoring and reliable intravenous fluid and drug administration. Specifically, the tunneled multi-lumen Hickman catheter (HC) is commonly used in swine models due to its lower extrication and complication rates. Despite fewer complications relative to other CVCs, HC-related morbidity presents a significant challenge, as it can significantly delay or otherwise negatively impact ongoing studies. The proper insertion and maintenance of HCs is paramount in preventing these complications, but there is no consensus on best practices. The purpose of this protocol is to comprehensively describe an approach for the insertion and maintenance of a tunneled HC in swine that mitigates HC-related complications and morbidity. The use of these techniques in >100 swine has resulted in complication-free patent lines up to 8 months and no catheter-related mortality or infection of the ventral surgical site. This protocol offers a method to optimize the lifespan of the HC and guidance for approaching issues during use.
Introduction
The indispensable role of central venous catheters (CVCs) in patient care is owed to their convenience, favorable safety profile, and versatility1. Functions of a CVC include reliable access for total parenteral nutrition, hematopoietic stem cell transplantation, plasmapheresis/apheresis, and efficient fluid, blood, or co-drug administration2. In veterinary medicine, CVCs also minimize animal discomfort via the rapid dilution of irritant drugs and blood sampling without repeated venipuncture3. Despite their broad applications, the use of CVCs in large animal research still presents several considerable challenges4.
Percutaneous CVC placement via a guidewire or introducer catheter can be difficult for non-veterinary researchers, particularly in animals with deep venous structures5. An improper CVC installation technique may result in inadvertent placement into nearby structures, necessitating ultrasound-guided placement or a postprocedure radiography of the positioning6. However, compared to human operating rooms, ultrasounds are not readily available in many large animal research laboratories. Further, long-term use of indwelling catheters can result in line kinking, puncture, infection, or extrication by animals, with the possible disruption of timely treatment, clinical monitoring, and research outcomes4,7. Replacement of the CVC requires additional resources, including material procurement, surgical scheduling, fasting time, and radiographic access. CVC-related complications can therefore create significant technical and financial barriers or a disruption to productive translational research, particularly in swine. Contamination by food or feces, scratching against cage walls, and kicking sites of irritation may compromise a CVC, and the risk of CVC-related complications is amplified by long-term use. Thus, safe and uncomplicated maintenance of a CVC in swine requires careful consideration of CVC choice, placement, securement, protection, sanitation, and surveillance.
The Hickman catheter (HC) used in this protocol is a tunneled CVC with a polyester cuff and one to three lumens, that is commonly used for long-term intravenous access in humans and animals1,4,8,9. The tunneled catheter approach has been associated with lower complication rates and maintenance costs relative to non-tunneled variations10,11,12. The cuff reduces HC extrication by incorporating into the subcutaneous tissues surrounding the skin exit site. The multi-lumen design also enables the separation of medication administration and blood draws, thereby minimizing blood sample contamination and inaccuracy. Despite this, HC use is not without challenges, the most common of which include fracture, migration, occlusion, and infection13,14,15,16. Proper installation and maintenance of a HC are therefore indispensable skills when used in translational research. However, the current literature offers little guidance for best-practices for HC use in swine during long-term trials5,6,17.
The purpose of this study is to outline an optimized approach for HC insertion into the internal jugular vein (IJV), skin securement, and durable protection that minimizes long-term catheter-related complications and discomfort in swine. A discussion of the important considerations for HC use, potential challenges that may be encountered, and modifications that may improve the quality of this approach is included.
Protocol
All animal procedures were conducted in accordance with an animal protocol approved by the Johns Hopkins University Institutional Animal Care and Use Committee (IACUC). Strains of male and female swine undergoing HC placement include miniature swine from the Massachusetts General Hospital (MGH) swine colony, Yucatan swine, and Yorkshire-crossed swine from an agricultural vendor (20-40 kg). Swine ranged from 3-10 months of age when the HC was placed. The HC may be placed anytime relative to the animal's experimental procedure. However, it is recommended to place it beforehand to permit the collection of baseline blood values. It is also recommended to give the swine at least a 1 week acclimation period before undergoing any experimental manipulation.
1. Preoperative planning
- Prior to surgery, have a veterinarian perform a thorough clinical evaluation of all the animals.
- Fast the animals (no solid food) at least 12 h prior to the procedure. Provide water ad libitum at all times. Weigh the animals for controlled drug administration.
- On the day of surgery, sedate the animals with ketamine (20-30 mg/kg) and xylazine (2-3 mg/kg intramuscularly [IM]) mixed into a single syringe. If necessary, administer sedative agents (ketamine and xylazine) slowly intravenously [IV]. Use a sterile ophthalmic ointment on the eyes to prevent desiccation while under anesthesia.
- Place an intravenous catheter in a marginal ear vein and administer maintenance fluid at 5-10 mL/(kg∙h) of 0.9% saline or lactated ringers solution (LRS) throughout the procedure.
- When the animal is in dorsal recumbency, place an appropriately sized endotracheal tube, connect it to the anesthesia machine, and place on hand ventilation (maintained on isoflurane at 0.5-3% with 1-2 L O2/min).
- Administer preemptive analgesia (0.02 mg/kg buprenorphine IV); administer additional doses of buprenorphine intraoperatively as needed. Administer prophylactic antibiotics (20-22 mg/kg cefazolin IV) 10 min before the start of the surgery and again every 90 min intraoperatively. Administer protonix (0.5-1.0 mg/kg IV) and maropitant citrate (1 mg/kg IV) once prior to the start of surgery.
- Shave the ventral and dorsal neck, which will serve as the site for the placement of the central venous catheter. Perform preliminary preparation of the surgical area using chlorhexidine scrub.
- Confirm that the pig is within an appropriate plane of surgical anesthetic. Evaluate the anesthetic depth by testing the palpebral and jaw tone. If necessary, increase the inhalant anesthetic or administer additional sedative agents (such as ketamine) IV slowly to effect.
- Transfer the swine to the operating table.
2. Intraoperative monitoring
- While under maintenance anesthesia, continuously monitor the heart rate and electrocardiography, the noninvasive blood pressure, pulse oximetry, capnography, and the esophageal or rectal temperature, recording at least every 15 min.
- Noninvasively monitor the blood pressure via a blood pressure cuff on an extremity or the tail.
- Use a ventilator to more accurately regulate the breathing and inhalant anesthesia administration. For ventilator parameters, ensure that tidal volume is in the range of 5-10 mL/kg. Adjust the respiratory rate throughout the procedure in response to the animal's anesthetic depth; set the maximum pressure limit to 20 mmHg.
- Use a temperature-regulated warm-air blanket or pad throughout the operation to prevent hypothermia.
3. Surgical preparation
- Designate at least two people to be sterile (surgeon and assistant) and at least two people to be non-sterile (circulator and anesthetist).
- Under general anesthesia, position the swine in ventral recumbency on the operating table with its legs secured for stabilization (Figure 1).
- Aseptically prepare the surgical field with three alternating scrubs of chlorhexidine and 70% ethanol, followed by an ionophore on the surface of the skin for a minimum contact time of 10 min. Carry out a sterile prep of the following two areas:
- On the ventral side, extend from the angle of the mandible to the middle of the sternum and extend bilaterally to the lateral border of the sternocleidomastoid. Place sterile towels along the borders of the sterilely-prepped area.
- On the dorsal side, prepare the dorsolateral neck ipsilateral to the target IJV. Place sterile towels beneath the dorsal neck and overtop the dorsal field to maintain sterility while working in the ventral field.
- Place a sterile drape over the swine. Cut a rectangular area in the drape to expose the ventral field.
- On a separate sterile-draped table, connect the double-lumen HC to a long introducer piece.
- Screw claves onto luer lock adapters at the end of each lumen line (wide: red; narrow: white). Flush the red and white lines with 10 mL of 0.9% saline. Then, clamp both lines.
4. Internal jugular vein identification and preparation
- In the ventral field, make a 4 cm incision between the trachea and the medial border of the sternocleidomastoid (Figure 2). Divide the platysma and dissect the connective tissue to reveal the IJV on the lateral border of the sternocleidomastoid muscle.
- Isolate 3-4 cm of the IJV by dividing its branches with 4-0 coated and braided non-absorbable suture ties. Circumferentially dissect away from the surrounding connective tissue. Create two coated and braided non-absorbable suture ties to suspend and stabilize the IJV during catheter insertion (Figure 3).
- At the cranial end of the IJV, pass a coated and braided non-absorbable suture tie twice underneath the vessel to create a loop around it.
- At the caudal end of the IJV, pass a coated and braided non-absorbable suture tie once underneath the vessel to create a sling.
- Release traction from the suture ties. Place sterile saline-soaked gauze in the surgical site to protect the vessel and maintain the tie location.
5. Catheter exit site preparation
- Reposition the swine via lateral tilt toward the non-surgical side to expose the ipsilateral dorsal surgical field. Resecure the limbs (Figure 4).
- With a #10 blade scalpel, make a 0.5 cm puncture in the skin at the desired catheter exit site-3 cm lateral to the vertebral column and 5 cm caudal to the head (Figure 5).
6. Introduction and tunneling of the catheter
- In the ventral field, remove the wet gauze and reidentify the isolated IJV segment. Choose a target entry site to introduce the catheter subcutaneously. Ensure that this is at the same depth as the IJV, deeper than the sternocleidomastoid, and between the two coated and braided non-absorbable suture ties (Figure 6).
NOTE: Subcuticular patterns should have suture knots buried below the skin. Because the deep dermal sutures are knotted several times to ensure stability and maintenance, they occasionally occupy a larger space than desired and may be exposed through the skin. This small area of exposure is not of significant concern, and the skin should heal appropriately despite this small area of exposure. - Position the dominant hand in the dorsal surgical field and the non-dominant hand in the ventral surgical field. Hold the HC introducer in the dorsal surgical field. Suspend the remaining length of the catheter in the air above the sterile field.
- Insert the introducer into the exit puncture site with the dominant hand, pointing the tip of the device toward the non-dominant hand in the ventral field.
- Push the tip of the introducer superficially and medially to tunnel the catheter through the adipose tissue, feeling for the emergence of the tip with the nondominant hand. Once the tip emerges at the target entry site, pull the introducer and catheter through the subcutaneous tunnel until the cuff of the main line is just underneath the surface of the skin in the dorsal field.
- Cut the introducer from the line. Replace the wet gauze in the ventral surgical site.
7. Insertion of the catheter
- Reposition the swine in the supine position. Resecure the limbs, replace sterile gloves, and remove the gauze from the ventral surgical site.
- Clamp the ends of the cranial and caudal coated and braided non-absorbable suture ties. Rest the clamps on the drapes so that the IJV segment is slightly elevated.
- Cut the end of the catheter to approximately the length at which it would reach down to 1/3 of the length of the swine's sternum.
NOTE: Minimize manipulation of the catheter tip by transecting the tip with a single cut perpendicular to the line so as to avoid fragmentation or clogging of the line. Once inserted, the HC should lie within the superior vena cava, immediately cranial to the right atrium (Figure 7). - Using Adson-Brown forceps, grasp the middle of the isolated IJV segment. At that same point, make a cut halfway through the vessel with curved Metzenbaum scissors.
- While holding the IJV segment with the Adson-Brown forceps, insert the vein pick into the caudal segment of the vessel (Figure 6). While maintaining tension on the cranial suture tie, insert and thread the end of the catheter into the vessel caudally. Once the catheter is fully inserted, knot the caudal suture tie once to temporarily secure the HC.
- Test the patency of both lines outside the sterile field via blood draws and flushes using 3-5 mL of 0.9% normal saline followed by 5 mL of 100 USP units/mL heparinized saline.
- Once patency is confirmed, knot the caudal suture tie one or two times more to secure the distal IJV segment around the intravenous catheter. Knot the cranial suture tie once to occlude blood flow in the IJV segment.
- Close the ventral surgical site in layers: platysma simple interrupted sutures with a 3-0 braided absorbable suture, and subcuticular running sutures with a 3-0 monofilament absorbable suture.
8. Securing the catheter
- Undrape the swine and reposition in dorsal recumbency. Resecure the limbs.
- Secure the HC to the animal's skin at more than three points to avoid catheter detachment (Figure 8).
- Orient the HC so that it forms a "U" shape.
- Identify the points: ensure that the first point is within 2 cm of the exit site, the second point is over the forked portion of the catheter, where the red and white lines diverge, and the third point is at the top of the "U" between the first two points.
- At each point, place a ~3 cm piece of 1 inch (in) medical tape over the catheter to create a wing on each side. With a 0 synthetic, monofilament, nonabsorbable polypropylene suture, secure each wing to the skin via a single simple interrupted suture. At the second point over the forked portion, add a single interrupted suture through the gap between the two lines and ensure that the knot lies on top of the tape to prevent irritation.
NOTE: Depending on the size of the dorsal neck puncture site, a simple interrupted suture may be placed to decrease the size and minimize the risk of inadvertent catheter displacement out of the skin. Ensure that the cuff around the catheter remains subcutaneous. - Test the patency of both the white and red lines outside the sterile field via blood draws and flushes using 10 mL of 0.9% normal saline and 10 mL of heparinized saline.
- Create a protective collar.
- Wrap 4 in cotton padding around the neck three of four times in the following manner:
- Starting on the dorsal neck at the left-cranial corner, wrap diagonally toward the right-caudal corner, going above or below the red and white lines. Wrap underneath the neck toward the left-caudal corner. Wrap diagonally toward the right-cranial corner, above or below the red and white lines, and then wrap to the start point at the left-cranial corner. Alternate going above and below the red and white lines with each subsequent wrap.
NOTE: The collar should completely cover the skin attachment sites and main line of the HC. Only the red and white lines should be left out and accessible.
- Starting on the dorsal neck at the left-cranial corner, wrap diagonally toward the right-caudal corner, going above or below the red and white lines. Wrap underneath the neck toward the left-caudal corner. Wrap diagonally toward the right-cranial corner, above or below the red and white lines, and then wrap to the start point at the left-cranial corner. Alternate going above and below the red and white lines with each subsequent wrap.
- Wrap 3 or 4 in elastic adhesive tape around the neck three or four times in the same manner as the cotton padding (step 8.4.1). If the bandage covers the lines, create a slit in the bandage to accommodate them.
NOTE: Take care to not wrap this layer too tightly-a finger should be able to be easily slid under the protective collar circumferentially. - With a 0 synthetic, monofilament, nonabsorbable polypropylene suture, suture each corner at the end of the bandage to the underlying layers to maintain its position.
- With a 0 synthetic, monofilament, nonabsorbable polypropylene suture, secure the collar to the skin by tying a horizontal mattress suture lateral to the vertebral column on both the cranial and caudal end. Ensure that the knots lie on top of the bandage.
- Create a catheter pouch to protect and store the red and white lines (Figure 9).
- Cut a piece of elastic adhesive tape measuring ~100 cm in length and ~7.5 cm in width.
- Measure a ~16 cm segment of tape. Fold the tape over on itself so that the adhesive sides are facing each other to create a 16 cm flap with two layers of tape. With the remaining tape, repeat this process two more times to create three flaps of equal length in a "W" shape (Figure 10).
- Fold the remaining tail of tape over the edge of the three flaps. With the tail oriented on top, number the flaps 1-3 from top to bottom. Number the sides 1-4, starting from the tail edge and moving clockwise.
- On Side 4, remove a lengthwise sliver from Flap 2, excluding 1 cm on either end. Ensure that the width of the sliver is ~1 cm.
- Using a 0 coated and braided non-absorbable suture to perform running sutures: stitch Flaps 1 and 3 together on Side 4, Flaps 1, 2, and 3 together on Side 3, and Flaps 1 and 2 together on Side 2. Cut a 2 cm hole through the center of Flap 1.
- With Flap 1 facing down, align the hole with the point where the lines exit the collar, and orient the pouch so that the opening is caudal. Make sure that the pouch is on the midline or slightly lateral to the dorsal neck.
- Pull the red and white lines through the hole in Flap 1. Make sure that the lines lay flat between Flaps 1 and 2, with Flap 3 facing up (Figure 8).
- Using a 0 synthetic, monofilament, nonabsorbable polypropylene suture, secure the pouch to the collar with a simple interrupted suture at each corner and midway along each edge. Do not suture through the skin.
- Wrap 4 in cotton padding around the neck three of four times in the following manner:
9. Postoperative care
- Following recovery from anesthesia, return the pig to its home cage. Due to the chewing nature of swine, ensure that the pig is singly housed to prevent catheter removal by a conspecific pig. If housed next to other swine, place a barrier to prevent chewing of the catheter between the caging.
- During the immediate postoperative period, monitor the animal at least daily for signs of pain, infection, and healing. Respiratory rate, heart rate, temperature, energy, appetite, and water consumption are good health indicators during this time. Administer additional doses of an analgesic (e.g., 0.12 mg/kg buprenorphine-sustained release [SR] LAB every 48 h) if signs of pain develop. Perform catheter maintenance (step 10) and visual inspection of the surgery site daily, starting at postoperative day (POD) 1.
NOTE: Researchers who choose to adopt this Hickman catheter insertion technique can modify this procedure with multimodal analgesia; however, NSAIDs may alter study findings depending on other elements of the experimental design. This should be considered before planning the analgesia regimen. - Once the catheter's surgery site is healed, perform maintenance monitoring checks on the animal: perform weekly body weight measurements and conduct visual checks during the daily catheter maintenance. Consult a veterinarian if signs of infection, such as decreased appetite or decreased energy, develop. If complete blood counts are needed, collect blood from the red line.
NOTE: A leukocytosis characterized by a neutrophilia is often observed in animals with infection.
10. Catheter maintenance
- Designate the wider red line exclusively for blood draws and the narrower white line exclusively for medication administration. Always handle the catheter with gloved hands.
NOTE: These roles may be different depending on the study design. - Flush the red (designated blood draw) line daily to assess patency and prevent clotting as follows:
- Use aseptic technique: Wipe the clave and syringe tip with an alcohol pad in between each step. If the clave or syringe becomes unsterile, replace the contaminated material before continuing.
- Flush 1 mL of 0.9% saline into the line. Confirm the liquid is able to flush without excessive force in order to prevent pushing clots from the line into the animal.
- Pull back 2 mL of fluid. Confirm line patency by observing the liquid pulled back is dark red blood.
- If blood draw is necessary, attach an empty syringe to draw back the appropriate amount of blood.
NOTE: If excess blood is taken, blood can be returned to the animal by pushing it back through the line to decrease blood loss. This should only be done if the syringe containing blood continued to be handled using aseptic technique. Do not put contaminated blood back into the central line. - Flush the line with 5 mL of 100 USP/mL heparinized saline, or an amount necessary to flush the entire line. Clamp the line. Return the line to the protective pouch, taking care not to kink or knot the line.
- Flush the white (designed medication administration) line daily to assess patency and prevent clotting as follows:
- Use aseptic technique: wipe the clave and syringe tip with an alcohol pad in between each step. If the clave or syringe becomes unsterile, replace the contaminated material before continuing.
- Flush 1 mL of 0.9% saline into the line. Confirm the liquid is able to flush without excessive force in order to prevent pushing clots from the line into the animal.
- Administer medications using this line at a medication-specific speed and dilution. Flush the line with 1-3 mL of 0.9% saline between medications.
- Flush the line with 5 mL of 100 USP/mL heparinized saline, or an amount necessary to flush the entire line. Clamp the line. Return the line to the protective pouch, taking care not to kink or knot the line.
- Check the claves daily for signs of dysfunction, breakage, or overt contamination with blood, food, or feces. If so, replace the clave immediately. Visually inspect the Hickman pouch and protective collar daily to ensure that the sutures placed in steps 8.2 and 8.3 remain intact.
Representative Results
Over 100 swine have undergone successful HC insertion in our lab. The HC can be safely and correctly placed and secured in under 1 h with a surgeon, assistant, circulator, and anesthetist. The catheter pouch takes roughly 15-20 min to make. The technique is straightforward and easy to teach and has been performed by veterinarians, surgical residents, and medical students following supervised instructions.
HCs have remained in place without complications or revision for up to 8 months. In a recent representative cohort of 32 swine with endpoints ranging from 8 to 132 days, 78.13% of HCs remained patent until the experimental endpoint (Figure 11). Of the swine that had clinical indications that required euthanasia and diagnostic necropsy, the HC was documented with correct placement in the IJV without thrombi, debris, or signs of infection. Complication rates requiring intervention were modest: 9.38% of HCs required removal or replacement before 30 days and 12.5% required removal or replacement on or after 30 days. Additionally, 9.38% of HCs required small repairs to the original HC under sedation (Figure 11). Reasons for the line compromises were not always elucidated, but etiologies included displacement, puncture, and internal blockages (Table 1). Timely line repair and replacement have demonstrated a 100% functional success rate without significant interference with study data collection. In cases of suspected central line infection, swine underwent prompt appropriate medical treatment without further complication. There have been no HC-related mortality in swine.
Figure 1: Animal positioned in ventral recumbency. Please click here to view a larger version of this figure.
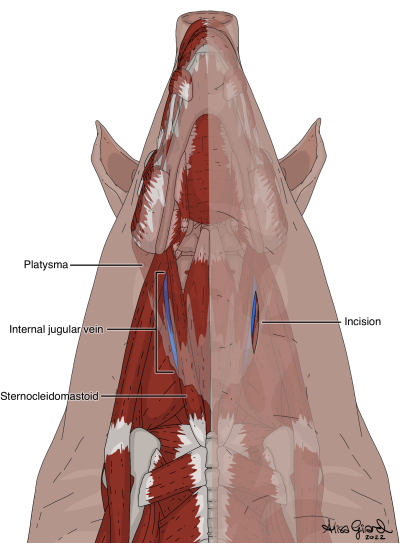
Figure 2: Ventral incision through skin and platysma to expose the internal jugular vein. Please click here to view a larger version of this figure.
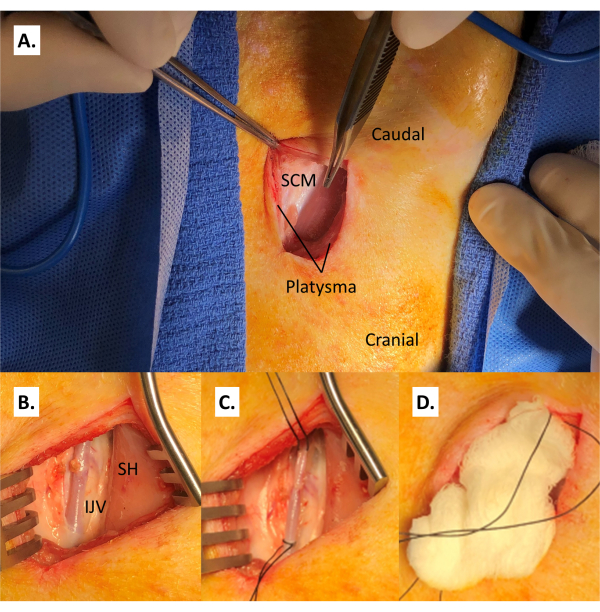
Figure 3: Accessing the internal jugular vein. (A) Incision of the platysma, revealing the sternocleidomastoid. (B) Internal jugular vein medial to the sternocleidomastoid and lateral to the sternohyoid. (C) Coated and braided non-absorbable suture ties are positioned at the caudal and cranial ends of the isolated portion of the IJV. (D) Wet gauze is placed in the incision to keep the vessel protected and maintain coated and braided non-absorbable suture positioning while repositioning the swine. Abbreviations: SCM = sternocleidomastoid; IJV = internal jugular vein; SH = sternohyoid. Please click here to view a larger version of this figure.

Figure 4: Animal positioned in lateral tilt to allow access to both the dorsal catheter exit site and the ventral incision. Please click here to view a larger version of this figure.
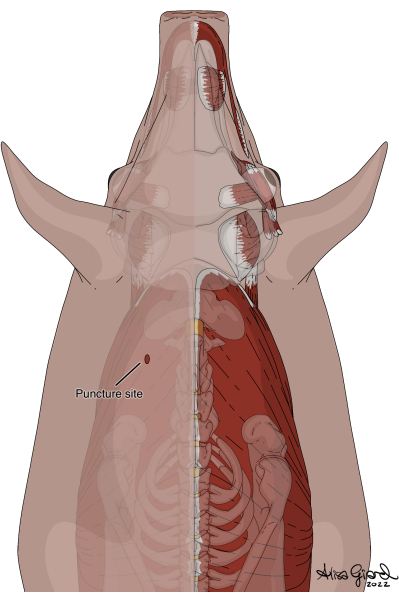
Figure 5: Dorsal puncture through the skin to serve as the catheter exit site. Please click here to view a larger version of this figure.
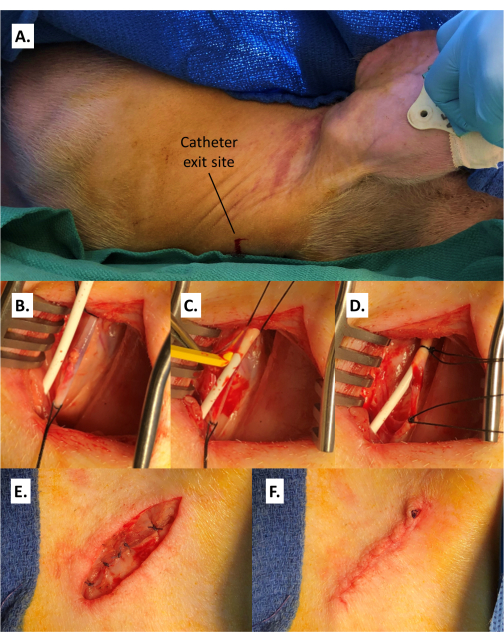
Figure 6: Placement of the catheter. (A) Puncture of the skin at the catheter exit site on the dorsolateral neck ipsilateral to the target IJV. (B) Catheter entering the surgical site lateral to the IJV. (C) Insertion of the catheter into the distal end of the IJV with the aid of a vein pick. (D) The vein is secured around the catheter distally and the proximal segment is occluded with a coated and braided non-absorbable suture tie. (E) Closure of the platysma. (F) Closure of the skin. Abbreviation: IJV = internal jugular vein. Of note, deep dorsal sutures that are extensively knotted for security may protrude through the closure; this should not interfere with wound healing. Please click here to view a larger version of this figure.
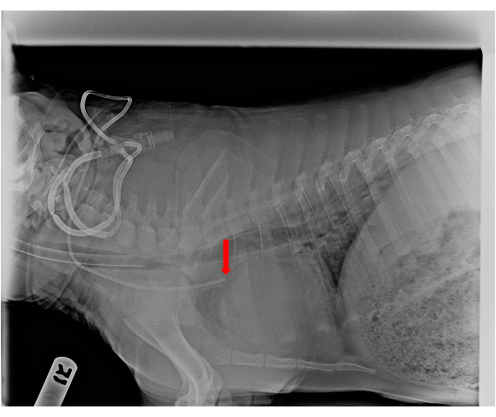
Figure 7: Lateral radiograph of Hickman catheter placement. The arrow demonstrates its correct location above the right atrium. Please click here to view a larger version of this figure.
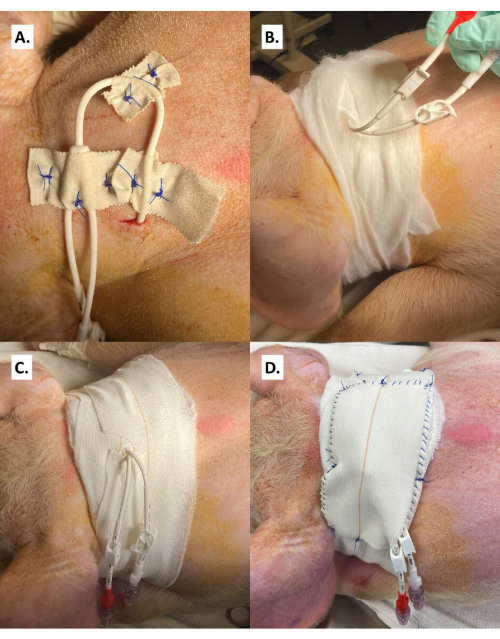
Figure 8: Securement of the catheter and bandage collar. (A) The catheter is sandwiched between layers of 1 in medical tape and at the fork where the two lumens divide. (B) Cotton roll is wrapped around the neck, alternating between cranial and caudal to the lumens. (C) The elastic adhesive bandage is wrapped around the neck with slits cut to secure the lumens centrally. (D) The two lumens are strung through the hole in the catheter pouch and the pouch is secured to the elastic adhesive bandage. Please click here to view a larger version of this figure.

Figure 9: Catheter pouch. (A) Top of the pouch. (B) Top flap (Flap 3) lifted to show middle flap (Flap 2). The deep portion of the pocket shows the cut edge of Flap 2. (C) Bottom of the pouch with a hole in the bottom flap (Flap 1) to reveal Flap 2. Please click here to view a larger version of this figure.
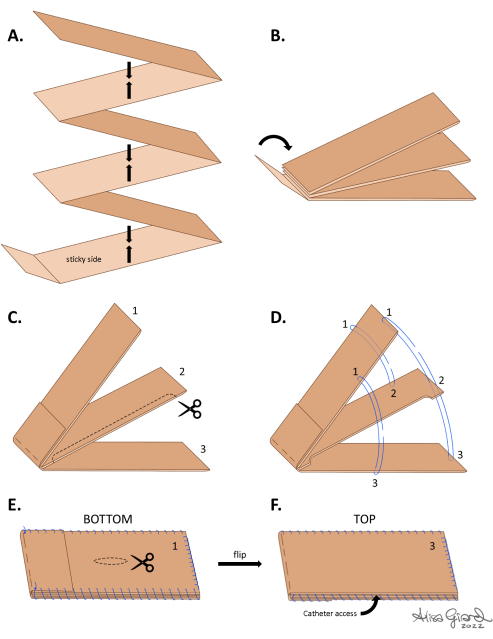
Figure 10: Design and assembly of the catheter pouch. (A) A long strip of elastic adhesive bandage tape is folded to create three flaps of equal length, with a residual loose tail. (B) The tail is folded over the end of the pouch to secure the exposed sticky folds. (C) With the tail on top, the flaps are numbered 1-3 from top to bottom. A strip of the long edge of the middle flap (Flap 2) is cut to create an inner pocket. (D) Sutures are used to join the flaps according to the numbers and suture positions around the pouch. (E) A hole is cut in the middle of Flap 3. (F) Top of the pouch. Please click here to view a larger version of this figure.
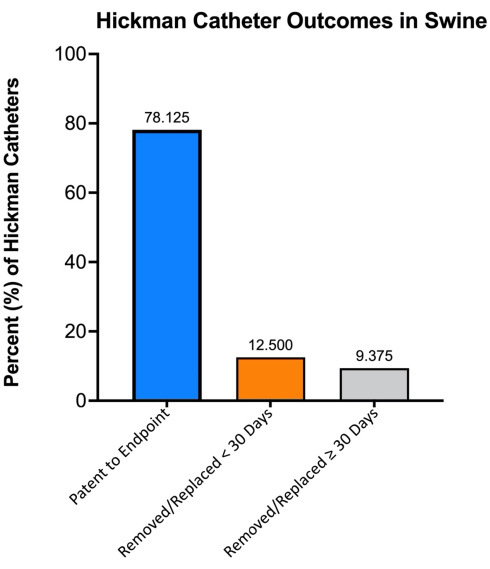
Figure 11: Hickman catheter outcomes in swine. N = 32. Experimental endpoints ranged from 8 to 132 days. Please click here to view a larger version of this figure.
Table 1: Hickman catheter outcomes and complications in swine. Abbreviation: HC = Hickman catheter. Please click here to download this Table.
Discussion
While CVCs serve a spectrum of functions in large animal research, current literature lacks a consensus approach for safe and sustainable use in long-term trials over 30 days. This protocol's stepwise procedure for HC insertion, skin securement, and storage in a handmade pouch has undergone significant adjustments for quality improvement. As such, this protocol presents a technique for HC use that permits efficient and effective intravenous access while ensuring animal welfare and minimizing complications.
Clinical and research applications of this protocol
Swine are used in many areas of biomedical research, and their use as preclinical models has increased dramatically since the early 1980s18. Notably, many anatomical and physiological characteristics of swine closely resemble humans, making them a suitable model for many diseases and a predominant laboratory animal species for surgical and interventional protocols19. The cardiovascular, integumentary, urinary, digestive, and renal systems are the most common models in swine due to the comparative overlaps with humans. Swine are one of the primary models for surgical training in laparoscopic and endoscopic techniques, organ transplantation, toxicology, pharmaceutics, and biomedical device assessment18,20,21,22. The versatility of the HC makes it an invaluable tool in these clinical and research settings. They allow reliable access for serial bloodwork and are an efficient means of intravenous fluid and medication administration4, which avoid multiple sedations that can introduce confounding variables into studies and compromise subject health. Multiple studies have also shown their utility in regularly monitoring blood drug levels, such as tacrolimus values in immunosuppression protocols23. Some of the swine included in this protocol had HCs for immunosuppressive drug level monitoring and intravenous administration of immunosuppressive and/or immunomodulatory drugs in pigs undergoing vascularized composite allotransplantation (VCA) procedures, including a heterotopic hind limb transplant model24, a swine hemiface graft dissection and transplantation model, and a renal autotransplantation model.
Critical steps in the protocol
Appropriate line length and reliable line care are essential to extend the life of the HC. The catheter must be cut where it reaches one-third of the length of the sternum (protocol step 7.4), as shorter lines are at risk of displacement and longer lines are at risk of hitting the vessel wall. To maintain patency and prevent blockages, both lines must be flushed daily with normal and heparinized saline (protocol steps 9.2-9.3). The lines should also be systematically identified by color before each use to avoid switching the roles of the red (blood draw) and white (medication administration) lines to avoid damage. For example, high negative pressure during blood draw can collapse the white line and cause clotting, whereas drug administration through the red line can cause residual drug to persist within the line or the clave, thereby artificially raising the measured drug content of drawn blood. Despite the secure dressing technique detailed in this manuscript, swine are occasionally able to gain access to the lines and cause mechanical damage, such as chewing or scratching. If one of the lines becomes unusable, the dysfunctional line should be securely occluded by tying a double knot in the line after the fork (to retain access to the remaining patent line) to prevent accidental use or dislodgement of the blockage into circulation. The remaining patent line may be used to fulfill both line roles temporarily, but the dysfunctional line should be replaced as soon as possible via a Hickman Repair Kit. It is also important to protect the lines by securely storing them in the pouch (protocol step 8.3.5) after each use. To ensure proper fixation of the pouch, the collar must be secured to the skin at both the cranial and caudal end (protocol step 8.3.4), and the pouch secured to the collar along each edge (protocol step 8.3.8). Broken sutures should be replaced expeditiously.
Protocol challenges and solutions
HC-related complications include migration, displacement, and central line infection. If it is suspected that the internal tip of the catheter has migrated distally to press against the right atrium wall, plain radiographs can help locate the catheter tip. This complication is managed with removal and replacement of the HC using the contralateral IJV. In the swine that have HCs placed according to this protocol, the use of the contralateral IJV has not resulted in any complications related to animal health or research outcomes. This is likely because swine have a robust intracranial and facial venous network that permits drainage of the head via bilateral external jugular veins, whereas the IJV has a comparatively minor contribution to head drainage25,26. In other cases where the HC is displaced from its surgical position due to loosening of the catheter or collar attachments to the skin, broken sutures should be immediately replaced. If the subcutaneous cuff exits the skin, the HC should be replaced. Regarding central line infection, signs and symptoms in swine may include lethargy, new onset of cough, decreased appetite, pyrexia, leukocytosis, and raised or unraised petechiae. Any signs of infection should be addressed immediately with antibiotics under the direction of a veterinarian, and culture of any abscess fluid should be considered if the animal is on immunosuppression treatment. Risks of central line infection are reduced by creating secure catheter pouches, regularly sanitizing and flushing the lines, employing sterile practices, and being vigilant in identifying subtle or early signs of HC dysfunction. These include line leaking, bubbles during blood draw, and increasing difficulty flushing lines.
Comparison to other CVC protocols in swine
Percutaneous methods of gaining central venous access in the external jugular vein (EJV) via palpable landmarks have been described. This provides the benefit of a reduction in soft tissue disruption and postoperative pain but can lead to complications, such as inadvertent carotid artery puncture and hematoma formation27. In contrast to percutaneous modalities, the protocol detailed in this manuscript allows for direct visualization of the target structures, which can help reduce damage to nearby tissues. Additionally, while open procedures for cannulating the EJV have been reported28, access to deeper structures such as the IJV and detailed guidance on placement and troubleshooting are limited. Another study utilized a similar approach of a paratracheal incision to access the jugular trunk, but instead used a laparoscopic suctioning device to create a subcutaneous tunnel to pass the HC and protected the external portion with a fitted jacket29. The results from this study showed a higher infection rate and thromboembolic complications in swine with tunneled HCs as compared to another group with a subcutaneous vascular access port. While the source of these complications is likely multifactorial, the protocol described in this manuscript has shown few infectious complications and helps mitigate potential causes by securing the external catheter at multiple points, creating a multilayer collar, and using a catheter pouch for line protection.
Limitations
This study presents with some limitations. While the use of three different swine strains demonstrates HC procedure success in a diverse cohort, swine have little anatomic variability and low rates of anomalous vasculature19. As such, the use of the sternocleidomastoid and sternum as surgical landmarks provided a consistent technique for incision and catheter length, respectively. The development of an optimized approach for HC placement, skin securement, and protective storage has occurred over several years in parallel with protocol-based studies in swine. Sequential modifications have been made in response to animal observation and creative troubleshooting. Therefore, a detailed report on failed modifications, or the process by which the proposed technique was established, was not included. Moreover, the data analysis does not include a control comparison group, such as swine that underwent venipuncture under sedation. Additionally, as with any procedure, this technique requires surgical experience, practice, and appropriate training of less experienced team members. The assembly of the catheter pouch may also be subject to user error. Detailed diagrams are included that aim to effectively illustrate this technique. Further, as this technique involves prophylactic antibiotics, it may not be appropriate for studies that would be impacted by antibiotic administration. Finally, this approach to HC placement and outcomes is limited to use in swine. This technique may not demonstrate the same efficacy in other large animals with varying anatomies. While HCs have been used in other species, further research is needed to adapt this technique to other animals.
Conclusions
The HC is an effective method of regular blood monitoring and intravenous drug administration in swine. This study details our best practices for HC insertion, skin securement, and durable protection that minimizes HC-related complications and animal discomfort. Through years of technique modification and troubleshooting, this protocol details an optimized approach to HC use in swine, with high reproducibility and minimal complications. Finally, guidance is offered to prevent and resolve problems that may arise during the lifetime of the HC.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to acknowledge the support of the Army, Navy NIH, Air Force, VA, and Health Affairs regarding the AFIRM II effort under award CTA05: W81XWH-13-2-0052 and CTA06: W81XWH-13-2-0053. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014, is the awarding and administering acquisition office. Opinions, interpretations, conclusions and recommendations are those of the author and are not necessarily endorsed by the Department of Defense. In addition, we would like to acknowledge support from the Department of Defense Congressionally Directed Medical Research Programs (CDMRP), Reconstructive Transplantation Research Program (RTRP), through awards W81XWH-17-1-0280, W81XWH-17-1-0624, W81XWH-17-1-0287, and W81XWH18-1-0795. We would also like to acknowledge the Department of Plastic and Reconstructive Surgery and the Johns Hopkins University School of Medicine. Additionally, we would like to acknowledge the entire veterinary staff, including Melanie Adams, Karen Goss, Haley Smoot, Kayla Schonvisky, and Victoria Manahan.
Materials
| #10 blade | Medline | MDS15110 | |
| 0.9% Sterile Sodium Chloride | Baxter | 2F7123 | |
| 0-0 Coated and Braided Nonabsorbable Suture | Covidien | S-196 | |
| 0-0 Synthetic, Monofilament, Nonabsorbable Polypropylene Suture | Ethicon | 8690H | |
| 1 inch Medical Tape | 3M | 1548S-1 | |
| 10 USP units/mL Heparin flush | Becton, Dickinson and Company | 306424 | |
| 3-0 Braided Absorbable Suture | Covidien | SL-636 (cutting needle), GL-122 (taper needle) | |
| 3-0 Monofilament Absorbable Suture | Covidien | SM-922 (cutting needle), CM-882 (taper needle) | |
| 4-0 Coated and Braided Non-absorbable Suture Ties | Ethicon | A303H | |
| 70% Ethanol | Vedco | VINV-IPA7 | |
| Adson tissue forceps | MPM Medical Supply | 132-508 | |
| Adson-Brown forceps | MPM Medical Supply | 106-2572 | |
| Air warming blanket and pad | 3M Bair Hugger | UPC 00608223595770 | |
| Backhaus towel clamp | MPM Medical Supply | 117-5508 | |
| Brown needle holder | MPM Medical Supply | 110-1513 | |
| Buprenorphine | PAR Pharmaceutical | 3003408B | |
| Cefazolin | Hikma Farmacuetica (Portugal) | PLB 133-WES/1 | |
| Chlorhexidine | Vet One | 501027 | |
| Clave | Baxter | 7N8399 | |
| Cotton Padding | Medline | NON6027 | |
| Debakey forceps | MPM Medical Supply | 106-5015 | |
| Elastic Adhesive Bandage Tape | 3M | XH002016489 | |
| Halstead mosquito forceps | MPM Medical Supply | 115-4612 | |
| Hickman Catheter | Bard Access Systems | 603710 | |
| Hickman Catheter Repair Kit, 7Fr, Red and White Connectors | Bard Access Systems | 0601690 (red), 0601680 (white), 502017 | |
| Kelly hemostatic forceps | MPM Medical Supply | 115-7014 | |
| Ketamine | Vet One | 383010-03 | |
| Lactated Ringers | Baxter | 2B2324X | |
| Maropitant Citrate | Zoetis | 106 | |
| Mayo scissors | MPM Medical Supply | 103-5014 | |
| Metzenbaum scissors | MPM Medical Supply | 132-711 | |
| Pantoprazole | JH Pharmacy | NDC 0143-9284-10 | |
| Scalpel blade handle | Medline | MDS10801 | |
| Vein Pick | SAI infusion technologies | VP-10 | |
| Veterinary Ophthalmic Ointment | Dechra | IS4398 | |
| Xylazine | Vet One | 510004 |
References
- Pontes, L., et al. Incidents related to the Hickman® catheter: identification of damages. Revista Brasileira de Enfermagem. 71 (4), 1915-1920 (2018).
- Kolikof, J., Peterson, K., Baker, A. M. Central Venous Catheter. StatPearls. , (2022).
- Central venous catheters: how, when, why? (Proceedings). DVM 360 Available from: https://www.dvm360.com/view/central-venous-catheters-how-when-why-proceedings (2011)
- Abrams-Ogg, A. C., et al. The use of an implantable central venous (Hickman) catheter for long-term venous access in dogs undergoing bone marrow transplantation. Canadian Journal of Veterinary Research. 56 (4), 382-386 (1992).
- Florescu, M. C., et al. Surgical technique of placement of an external jugular tunneled hemodialysis catheter in a large pig model. The Journal of Vascular Access. 19 (5), 473-476 (2018).
- . Central Venous Catheter Placement: Modified Seldinger Technique Available from: https://www.cliniciansbrief.com/article/central-venous-catheter-placement-modified-seldinger-technique (2015)
- Perondi, F., et al. Bacterial colonization of non-permanent central venous catheters in hemodialysis dogs. Heliyon. 6 (1), e03224 (2020).
- Faulkner, R. T., Czajkowski, W. P., Rayfield, E. J., Hickman, R. L. Technique for portal catheterization in rhesus monkeys (Macaca mulatta). American Journal of Veterinary Research. 37 (4), 473-475 (1976).
- Moss, J. G., et al. Central venous access devices for the delivery of systemic anticancer therapy (CAVA): a randomised controlled trial. Lancet. 398 (10298), 403-415 (2021).
- Dai, C., et al. Effect of tunneled and nontunneled peripherally inserted central catheter placement: A randomized controlled trial. The Journal of Vascular Access. 21 (4), 511-519 (2020).
- Wu, X., et al. Tunneled peritoneal catheter vs repeated paracenteses for recurrent ascites: a cost-effectiveness analysis. Cardiovascular and Interventional Radiology. 45 (7), 972-982 (2022).
- Onwubiko, C., et al. Small tunneled central venous catheters as an alternative to a standard hemodialysis catheter in neonatal patients. Journal of Pediatric Surgery. 56 (12), 2219-2223 (2021).
- da Silva, S. R., Reichembach, M. T., Pontes, L., de Souza, G. d. e. P. E. S. C. M., Kusma, S. Heparin solution in the prevention of occlusions in Hickman® catheters a randomized clinical trial. Revista Latino-Americana de Enfermagem. 29, e3385 (2021).
- Landoy, Z., Rotstein, C., Lucey, J., Fitzpatrick, J. Hickman-Broviac catheter use in cancer patients. Journal of Surgical Oncology. 26 (4), 215-218 (1984).
- Bawazir, O. A., Altokhais, T. I. Hickman central venous catheters in children: open versus percutaneous technique. Annals of Vascular Surgery. 68, 209-216 (2020).
- Cappello, M., et al. Central venous access for haemodialysis using the Hickman catheter. Nephrology Dialysis Transplantation. 4 (11), 988-992 (1989).
- Shastri, L., Kjærgaard, B., Rees, S. E., Thomsen, L. P. Changes in central venous to arterial carbon dioxide gap (PCO2 gap) in response to acute changes in ventilation. BMJ Open Respiratory Research. 8 (1), e000886 (2021).
- Smith, A. C., Swindle, M. M. Preparation of swine for the laboratory. ILAR Journal. 47 (4), 358-363 (2006).
- Swindle, M. M., Makin, A., Herron, A. J., Clubb, F. J., Frazier, K. S. Swine as models in biomedical research and toxicology testing. Veterinary Pathology. 49 (2), 344-356 (2012).
- Hughes, H. C. Swine in cardiovascular research. Laboratory Animal Science. 36 (4), 348-350 (1986).
- Svendsen, O. The minipig in toxicology. Experimental and Toxicologic Pathology. 57 (5-6), 335-339 (2006).
- Tumbleson, M. E., Schook, L. B. . Advances in Swine in Biomedical Research. 2, (1996).
- Jensen-Waern, M., Kruse, R., Lundgren, T. Oral immunosuppressive medication for growing pigs in transplantation studies. Laboratory Animals. 46 (2), 148-151 (2012).
- Ibrahim, Z., et al. A modified heterotopic swine hind limb transplant model for translational vascularized composite allotransplantation (VCA) research. Journal of Visualized Experiments. (80), e50475 (2013).
- Nordström, C. -. H., Jakobsen, R., Mølstrøm, S., Nielsen, T. H. Cerebral venous blood is not drained via the internal jugular vein in the pig. Resuscitation. 162, 437-438 (2021).
- Habib, C. A., et al. MR imaging of the yucatan pig head and neck vasculature. Journal of Magnetic Resonance Imaging. 38 (3), 641-649 (2013).
- Flournoy, W. S., Mani, S. Percutaneous external jugular vein catheterization in piglets using a triangulation technique. The International Journal of Laboratory Animals. 43 (4), 344-349 (2009).
- Kotsougiani, D., et al. Surgical angiogenesis in porcine tibial allotransplantation: a new large animal bone vascularized composite allotransplantation model. Journal of Visualized Experiments. (126), e55238 (2017).
- Chuang, M., et al. Comparison of external catheters with subcutaneous vascular access ports for chronic vascular access in a porcine model. Contemporary Topics in Laboratory Animal Science. 44 (2), 24-27 (2005).

