Isolation, In Vitro Expansion, and Characterization of Mesenchymal Stem Cells from Mouse Epididymal Adipose Tissue
Instructor Prep
concepts
Student Protocol
All animal experiments were conducted according to international guidelines for animal ethics and were approved by institutional committees of care and use from the State University of Santa Cruz under protocol number 021/22. Swiss male mice (6-8 weeks) were acquired from the Animal Breeding, Maintenance and Experimentation Laboratory – State University of Santa Cruz (LaBIO-UESC) Animal Research Facility, maintained in specific pathogen-free conditions, receiving water and food ad libitum with 12 h light/dark cycles.
NOTE: Other mice lineage can be used; we recommend the use of adult mice (6-8 weeks) since they have more developed adipose tissue and cells with high proliferative activity.
1. Preparation
NOTE: Before proceeding, prepare the following reagents described below. See the Table of Materials for reagent and material supplier information. Personal protective equipment such as masks, caps, lab coats, and gloves must be worn in all practical procedures.
- Epididymal adipose tissue extraction
- Reserve the surgery materials: three sets of sterile tweezers and scissors, five needles, and a beaker containing 200 mL of 70% ethanol.
- Prepare a terminal anesthetic, mixing ketamine (100 mg/kg) and xylazine (10 mg/kg).
- ADSCs culture
- Basal medium: Prepare Dulbecco's Modified Eagle Medium (DMEM) high glucose media supplemented with 10% fetal bovine serum (FBS), 50 µg/mL gentamicin, 0.25 µg/mL amphotericin B, 100 U/mL penicillin, and 100 mg/mL streptomycin.
- Digest solution: Prepare 0.15% of collagenase type II in PBS sterilized in a 0.25 µm filter.
NOTE: It is not necessary to use the four antibiotics described in the basal medium. It will depend on the cell culture conditions of the laboratory where this is performed.
- ADSCs phenotypic characterization
- Prepare 1% bovine serum albumin (BSA) in PBS.
- Dilute the anti-mouse antibodies as mentioned in Table 1.
- Prepare 2% formaldehyde in PBS.
- ADSCs osteogenic differentiation
- Osteogenic differentiation medium: Prepare DMEM supplemented with 5.67 M ascorbic acid, 10 nM dexamethasone, 0.02 M β-glycerophosphate, 10% FBS, and 1% penicillin/streptomycin.
- For Von Kossa staining, prepare the following solutions: 70% ethanol in water, 5% silver nitrate in water, distilled water, 5% sodium thiosulfate in water, and 1% eosin B in water.
- Adipogenic differentiation:
- Adipogenic differentiation medium: Prepare DMEM supplemented with 0.5 mM 3-Isobutyl-1-methylxanthine, 200 µM indomethacin, 1 µM dexamethasone, 10 µM insulin, 10% FBS, and 1 % penicillin/streptomycin.
- For Oil-red staining, prepare the following solutions: 10% formalin in deionized water, 60% isopropanol in deionized water, lysochrome (fat-soluble dye) diazo dye (Oil-Red O solution) in 60% isopropanol, and 1% hematoxylin in deionized water.
- Chondrogenic differentiation:
- Chondrogenic differentiation medium: Prepare chondrogenic medium supplemented with 10% FBS and 1% penicillin/streptomycin.
- Prepare 10% formaldehyde in PBS.
- For proteoglycans and glycosaminoglycans staining, prepare the following solutions: Heteroglycan stain (Alcian Blue 1%) in acetic acid, pH 2.5, paraffin, hematoxylin, ethanol series dilution in water (100%, 96%, 80%, and 70%).
| Antibody/Fluorophore | Dilution | Clone | Final concentration (µg/mL) | Function and cell in which it is expressed | ||
| CD34- PE | 1:100 | RAM34 | 2 | Cell adhesion factor. Hematopoietic stem cells. | ||
| CD45- APC | 1:100 | 30-F11 | 2 | Assistence in activation of leukocytes. Expressed in leukocytes. | ||
| CD71- FITC | 1:100 | C2 | 5 | Controls iron uptake during cell proliferation. Proliferating cells, reticulocytes, and precursor cells. | ||
| CD29- FITC | 1:100 | Ha2/5 | 5 | Adhesion and activation, embryogenesis, Leukocytes, dendritic cells, platelets, mast cells, fibroblasts, and endothelial cells. | ||
| CD90- PerCP | 1:100 | OX-7 | 2 | Signaling, adhesion. T lymphocyte, NK, monocyte, Hemtopoietic Stem Cells, neuron, and fibroblast. | ||
Table 1: Antibodies used for phenotypic characterization of ADSCs by flow cytometer. List of antibodies with their respective fluorochromes, dilutions, clone, and final concentration as well as their function in the cell which is expressed.
2. Methods
NOTE: The adipose tissue is distributed throughout the body in subcutaneous or intra-abdominal locations. In mice, the most common adipose tissues used for experiments include the subcutaneous epididymal, mesenteric, and retroperitoneal22. Here, the steps for obtaining epididymal adipose tissue are shown (Figure 1).
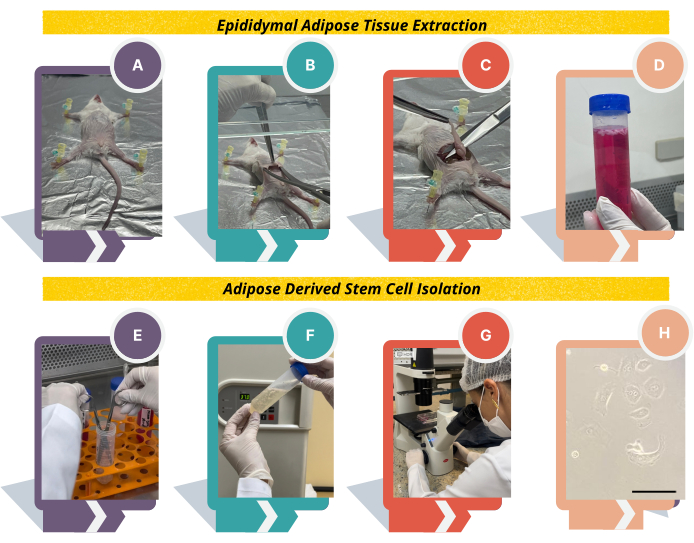
Figure 1: Experimental design to obtain adipose tissue-derived stem cells from Swiss mice. (A) Using needles, place the animal on the cork or styrofoam board; (B) Lift the skin using tweezers, make a cut in the center of the abdominal region, and detach the skin from the peritoneum; (C) identify the white adipose tissue above the epididymis; (D) transfer the tissue to DMEM medium supplemented with 10% FBS and 1% antibiotics, wash the epididymal adipose tissue thoroughly with PBS, and transfer the tissue to digest solution; (E), fragment the tissue and (F) incubate at 37 °C for 1 h vigorously shaking every 10 min; (G) after counting the cells, plate in 6-well plates and observe the cell morphology under an inverted microscope. (F) Cell morphology will change from round to fibroblast-like. Scale bar = 22.22 µm. Please click here to view a larger version of this figure.
- Extraction of epididymal adipose tissue
NOTE: Be aware that all procedures must be carried out under sterile conditions in a laminar flow cabinet in order to ensure pure and contamination-free cell culture.- Euthanize the animals using ketamine and xylazine solution (100 and 10 mg/kg, as described in step 1.1.2) by intraperitoneal route.
NOTE: Other euthanizing methods can be used23. Make sure the animal is dead by confirming the absence of a heartbeat. - Place the animal with the ventral side facing up on a cork or styrofoam board covered with aluminum foil and fix it using sterile needles (Figure 1A).
NOTE: To avoid contamination, spray 70% alcohol in the abdominal region. Alternatively, shave the hair with a scalpel. - Lift the skin using tweezers in the center of the abdominal region and make a cut with sterile scissors.
NOTE: Two sets of sterile tweezers and scissors are necessary to avoid contamination. The first one is used to open the animal, cutting the skin and peritoneal layer; the second one is used to pick up the epididymal adipose tissue. Also, reserve 150 mL of 70% alcohol in a beaker to clean scissors and tweezers between steps. - Introduce the scissors under the animal's skin and open it to detach it from the peritoneum (Figure 1B). Lift the peritoneal layer using tweezers and cut with sterile scissors.
- Use another set of sterile tweezers and scissors and identify the epididymis above the testes and the white adipose tissue above the epididymis. Pull the entire epididymal adipose tissue using sterile tweezers, approximately 2 cm in size, and cut very close to the epididymis (Figure 1C).
- Put the fat into a 50 mL sterile conical tube containing basal medium and place it on ice (Figure 1D). In the hood, proceed to the next steps as described below.
- Euthanize the animals using ketamine and xylazine solution (100 and 10 mg/kg, as described in step 1.1.2) by intraperitoneal route.
- Adipose tissue-derived stem cells (ADSCs) isolation
NOTE: Process the samples in a biological class II laminar flow hood, and personnel should wear a lab coat, gloves, and surgical mask.- Wash the epididymal adipose tissue thoroughly with PBS.
NOTE: This step is essential to remove the blood and other particles from the sample. The blood can inhibit collagenase type II enzyme and compromise the experiment. - Using a sterile tweezer, transfer the epididymal adipose tissue into a 50 mL tube containing 15-20 mL of digest solution, prepared as step 1.2.2. Fragment the tissue using sterile scissors and incubate the tissue at 37 °C for 1 h (Figure 1E, F).
NOTE: A water bath or incubator at 37 °C can be used in this step. Every 10 min, the suspension must be vigorously shaken to facilitate digestion. Do not extend the time of 1h incubation. - Inactivate the collagenase by diluting the sample 1:1 in basal medium and centrifuge the suspension at 250 x g for 10 min at 4 °C to obtain the vascular fraction of the stroma. Discard the supernatant and resuspend the pellet in 1 mL of basal medium.
- Check the viability and count the cells through the trypan blue dye exclusion method in a Neubauer chamber using a dilution of 1:10 or 1:100.
NOTE: The expected yield is around 0.5-1 million cells/g of processed adipose tissue and viability of 80%. - Plate the isolated cells (0.3-0.5 million) in 3 mL of basal medium in one well of a 6-well plate. Incubate the cells overnight at 37 °C with 5 % CO2.
- Next day: Remove the medium from the well and wash the cells with PBS. Add 3 mL of basal medium. Repeat this process every 2 days until the cells become 90% confluent.
NOTE: Always observe the morphology of the cells under the inverted microscope, changing from round to fibroblast-like (Figure 1H).
- Wash the epididymal adipose tissue thoroughly with PBS.
- Adipose tissue-derived stem cells (ADSCs) expansion
NOTE: Once the cells grow ~90% confluent, proceed to subculture as described below.- Remove the medium from the well and wash the cells with PBS. Add 0.5 mL/well of trypsin/EDTA (0.05 % trypsin; 200 mg/L EDTA) and incubate the plate for 3-5 min at 37 °C to detach the cells.
NOTE: Do not exceed 5 min in trypsin/EDTA. The complete detachment of the cells should be verified under the inverted microscope. The process can be accelerated by pipetting up and down carefully or tapping the side of the plate. - Inactivate the enzyme by diluting the sample 1:1 in DMEM supplemented with 10% fetal bovine serum (FBS) and 1% antibiotics.
- Split the cell suspension into 2 wells and add 3 mL of basal medium (DMEM supplemented with 10 % FBS and 1 % antibiotics). Incubate overnight at 37 °C with 5% CO2.
- Change medium the next day, then every 2 days until 90% confluence.
- Repeat the process until the third passage and proceed to phenotype and functional characterization.
NOTE: Always observe the morphology of the cells under the inverted microscope, changing from round to fibroblast-like.
- Remove the medium from the well and wash the cells with PBS. Add 0.5 mL/well of trypsin/EDTA (0.05 % trypsin; 200 mg/L EDTA) and incubate the plate for 3-5 min at 37 °C to detach the cells.
- Adipose tissue-derived stem cells (ADSCs) characterization
- Phenotypic characterization by flow cytometry
- Detach the cells using Trypsin/EDTA, as described in steps 2.3.1 to 2.3.2.
- Collect the cells in a conical tube (50 mL) and centrifuge at 250 x g for 10 min at 4 °C. Discard the supernatant and resuspend the pellet in 1 mL of PBS.
- Count the cells as described in step 2.2.4 and seed 0.5-1 x 106 cells in 100 µL of PBS in a 96 well-plate.
NOTE: The number of wells will depend on the color of the fluorochrome conjugate and the filters/lasers of the cytometer. - Add antibodies (Table 1) diluted in 1% BSA in PBS.
NOTE: It is not necessary to use Fc block to avoid nonspecific binding because we already use BSA to dilute antibodies. Reserve a sample of cells without receiving antibodies that will be used as a control of staining. The antibodies can be combined in the same well according to the fluorochrome dye and cytometer available in the lab. All the antibodies described in Table 1 can be added in the same well, except the CD71 FITC and CD29 FITC, which must be in different wells because of the same fluorochrome. - Incubate the plate for 30 min at 4 °C in the dark.
- Wash cells, then complete the volume to 200 µL using PBS, centrifuge the plate at 252 x g for 10 min at 4 °C, and discard the supernatant. Repeat this step one more time.
- Fix the cells by adding 200 µL per well of 2% formaldehyde in PBS. Store the cells in the dark at 4 °C until analysis in the flow cytometer.
NOTE: For best results, acquire the cells on the flow cytometer as soon as possible. The brightness of fluorescence of markers decays significantly after 48 h for most fluorochromes. So, do not exceed 48 h to read the plate. Acquisition of 30,000 events is recommended. - Use the gating strategy presented in Figure 2B to select the ASC population.
- Functional characterization: Osteogenic differentiation
- Detach the cells using Trypsin/EDTA (as described in steps 2.3.1 to 2.3.2), collect and centrifuge the cells (as described in steps 2.4.1.2 and 2.4.1.2), and count them (as described in steps 2.2.4).
- Plate, in triplicate, 2 x 105 cells per well in 6-well plates in basal medial (DMEM supplemented with 10 % FBS and 1 % antibiotics); incubate at 37 °C in 5% CO2.
NOTE: Two 6-well plates will be needed, one for each differentiation time (14 and 21 days). - Next day, change the medium to osteogenic medium (step 1.4.1).
NOTE: reserve 3 wells being cultured only with the basal medium, which will be the control for differentiation. - Culture cells for 14 and 21 days in osteogenic media and the control cells, changing media every 3 days. Proceed to Von Kossa staining (step 1.4.2) to assess the mineralized nodules at the end of each culture period.
- Aspirate the medium from each well and wash the cells twice with PBS. Fix the cells with 2 mL of 70% ethanol overnight at room temperature (RT). Wash the cells carefully in running water.
- Add 2 mL of 5% silver nitrate solution and incubate the plate in ultraviolet light for 1 h. Wash the cells with distilled water. Add 2 mL of 5% sodium thiosulfate, and incubate for 5 min. Wash with distilled water.
- Counterstain with 2 mL of eosin for 40 s. Wash with distilled water until the staining solution is removed, and visualize the staining using an optical microscope.
- Functional characterization: Adipogenic differentiation
- Detach the cells using Trypsin/EDTA (as described in steps 2.3.1 to 2.3.2), collect and centrifuge the cells (as described in steps 2.4.1.2 and 2.4.1.2), and count them (as described in steps 2.2.4).
- Plate 2 x 105 cells per well in 6-well plates in basal medial (DMEM supplemented with 10 % FBS and 1 % antibiotics); incubate at 37 °C in 5% CO2.
NOTE: Two 6-well plates will be needed, one for each differentiation time (14 and 21 days). - The next day, change the medium to adipogenic medium (step 2.5.1).
NOTE: Reserve 3 wells being cultured only with the basal medium as control. - Culture cells for 14 and 21 days in adipogenic medium, changing media every 3 days. At the end of each culture period, proceed to Oil-Red O staining (steps 2.4.3.5-2.4.3.7), an indicator of intracellular lipid accumulation.
- Aspirate the media from each well. Wash the cells twice with PBS. Fix cells in 2 mL of 10% formalin for 1 h. Wash once with PBS, and then one time with water.
- Stain with 2 mL of Oil-Red O solution in 60% isopropanol for 5 min. Wash once with distilled water.
- Counterstain with 2 mL of 1% hematoxylin diluted 1:2 in distilled water for 1 min. Wash with distilled water until the staining solution is removed, and visualize the stained samples using an optical microscope.
- Functional characterization: Chondrogenic differentiation
- Detach the cells using Trypsin/EDTA (as described in steps 2.3.1 to 2.3.2), collect and centrifuge the cells (as described in steps 2.4.1.2 and 2.4.1.2), and count them (as described in steps 2.2.4).
- Place 5 x 105 ADSCs in four polypropylene conical tubes and centrifuge at 450 x g for 5 min to form a pellet. Discard the supernatant.
- Culture the pellets in a conical tube in a chondrogenic medium (step 1.6.1) or only basal medium (control of differentiation) for 14 and 21 days at 37 °C in 5% CO2. Change medium every 3 days and avoid removing pellets.
NOTE: Each cell pellet refers to each culture time, as well as their respective controls grown only with basal medium. - At the end of each culture time point, collect pellets using fine-tip tweezers and place them in a 24-well plate. Proceed for histological sectioning and staining of proteoglycans and glycosaminoglycans (steps 2.4.4.5.-2.4.4.9).
NOTE: Each pellet is processed in a separate well. - Fix pellets in 2 mL of 10% buffered formaldehyde for 30 min. Discard the formaldehyde solution, and add 2 mL of 70% ethanol. Remove the pellet from the conical tube using a tip and embed the pellets in paraffin.
- Using a rotating paraffin microtome, make 5 µm histological sections. Incubate the sections for 15 min at 60 °C, and immerse them twice in xylene (for 5 and 15 min).
- Rehydrate the samples by immersing the histological sections in alcohol baths at decreasing concentrations (100%, 96%, 80%, and 70%) for 2 min each, then rinse with deionized water for 5 min.
- Proceed to stain the samples with Alcian blue 1% in acetic acid, pH 2.5, for 30 min.
- Counterstain with hematoxylin for 1 min, and wash with distilled water. Mount with DPX (mountant for histology) or other mount solution and visualize samples using an optical microscope.
- Phenotypic characterization by flow cytometry
Isolation, In Vitro Expansion, and Characterization of Mesenchymal Stem Cells from Mouse Epididymal Adipose Tissue
Learning Objectives
Cells extracted from adipose tissue according to the protocol presented here showed morphology matching the minimal criteria for MSCs proposed by ISCT. An overview of the protocol is shown in Figure 1. Phenotypically, ADSCs showed adherence to plastic and fibroblast-like morphology in the first days of cell culture (Figure 2A). In addition, they grew homogeneously and formed colonies. Furthermore, ADSCs showed low expression of CD34 (2.12%) and CD45 (1.81%), both hematopoietic markers, and high expression of CD71 (42.6%), CD29 (74.2%), and CD90 (55.1%), all mesenchymal cell markers (Figure 2B).
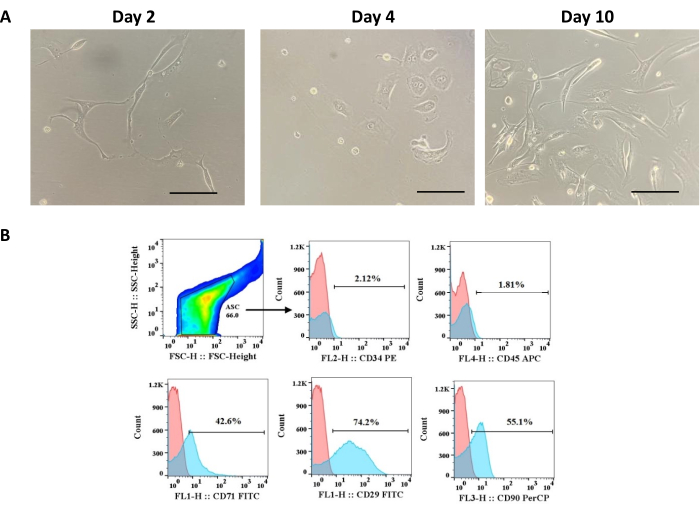
Figure 2: Phenotypic characterization of adipose tissue-derived cells. (A) ADSCs morphology changing from round to fibroblast-like on days 2, 4, and 8 of culture; scale bar = 22.22 µm. (B) Histograms for markers expressed (CD71, CD29, and CD90) or not (CD34 and CD45) by ASC. This figure has been reproduced with permission from Miranda et al.24. Please click here to view a larger version of this figure.
Functionally, ADSCs showed multipotency to differentiate into osteoblast, adipoblast, and chondroblast when cultured in conditioned media for each lineage for 14 or 21 days (Figure 3). The Von Kossa staining revealed mineralized nodules in the extracellular matrix, characteristic of the osteogenesis process (Figure 3). The Oil-red O staining evidenced lipid vacuoles in the cytoplasm of ADSCs, and the Alcian Blue staining confirmed the presence of glycosaminoglycan in the extracellular matrix (Figure 3). Together, phenotypic and functional characteristics have confirmed the population of cells extracted from epididymal adipose tissue as MSCs.
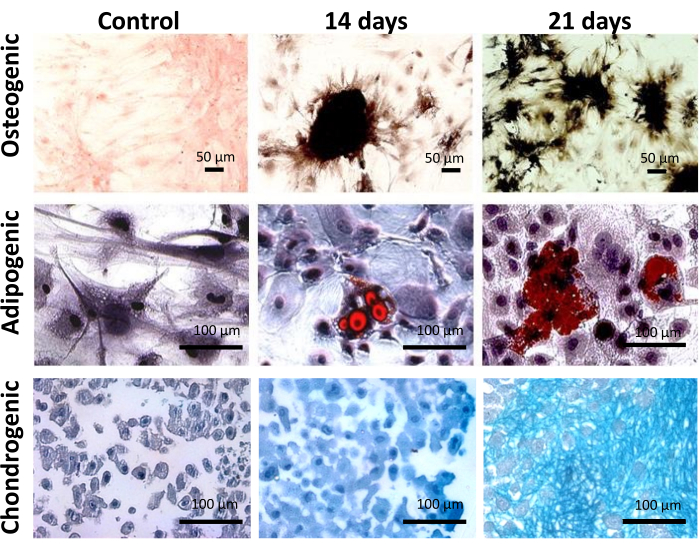
Figure 3: Functional characterization of adipose tissue-derived cells. Osteogenic, adipogenic, and chondrogenic multilineage potential of ADSC after 14 and 21 days on culture. Scale bar = 50 µm (top panel), 100 µm (middle and bottom panels). This figure has been reproduced with permission from Miranda et al.24. Please click here to view a larger version of this figure.
List of Materials
| 140 °C High Heat Sterilization CO2 Incubator | RADOBIO SCIENTIFIC CO. LTD, China | C180 | |
| 3-Isobutyl-1-methylxanthine | Sigma-Aldrich, San Luis, Missouri, USA | I7018 | |
| Acetic acid glacial | Sigma-Aldrich, San Luis, Missouri, USA | PHR1748 | |
| Alcian Blue 8GX | Sigma-Aldrich, San Luis, Missouri, USA | A9186 | BioReagent, suitable for detection of glycoproteins. 1% in acetic acid, pH 2.5 |
| Alcohol 70% | Sigma-Aldrich, San Luis, Missouri, USA | 65350-M | 70% in water |
| Amphotericin B | Sigma-Aldrich, San Luis, Missouri, USA | PHR1662 | |
| Antibodies anti-mouse anti-CD29 FITC (Clone Ha2/5) | BD Biosciences, San Diego, CA, USA | 555005 | Functions in the cell: Adhesion and activation, embryogenesis, Leukocytes, DC, platelets, mast cells, fibroblasts and endothelial cells |
| Antibodies anti-mouse anti-CD34 PE (Clone RAM34) | BD Biosciences, San Diego, CA, USA | 551387 | Functions in the cell: Cell adhesion factor. Hematopoietic stem cells |
| Antibodies anti-mouse anti-CD45 APC (Clone 30-F11) | BD Biosciences, San Diego, CA, USA | 559864 | Functions in the cell: Assists in the activation of leukocytes |
| Antibodies anti-mouse anti-CD71 FITC (Clone C2) | BD Biosciences, San Diego, CA, USA | 553266 | Functions in the cell: Controls iron uptake during cell proliferation. Proliferating cells, reticulocytes and precursors |
| Antibodies anti-mouse anti-CD90 PerCP (Clone OX-7) | BD Biosciences, San Diego, CA, USA | 557266 | Functions in the cell: Signaling, adhesion. T lymphocyte, NK, monocyte, HSC, neuron, fibroblast |
| Ascorbic acid | Sigma-Aldrich, San Luis, Missouri, USA | PHR1008 | |
| Automatic pipettes | Thermo Fisher Scientific, Waltham, Massachusetts, USA | 4700850N | Finnpipette F1 Good Laboratory Pipetting (GLP) Kits |
| Beaker | Not applicable | 1 unit | |
| Bovine serum albumin | Sigma-Aldrich, San Luis, Missouri, USA | A7906 | |
| Cell culture plates (6-well) | Merck, Darmstadt, Germany | Z707759 | 07 units sterile. TPP tissue culture plates |
| Cell culture plates (96-well. Round or V bottom) | Merck, Darmstadt, Germany | CLS353077 | 01 unit sterile. Wells, 96, Tissue Culture (TC)-treated surface, round bottom clear wells, sterile |
| Chondrogenic medium | Stem Pro Chondrogenesis Differentiation–Life Technologies | A1007101 | TGF-β2, TGF-β3, dexamethasone, insulin, transferrin, ITS, sodium-l – ascorbate, sodium pyruvate, ascorbate-2-phosphate |
| Collagenase type II | Life Technologies, California, USA | 17101015 | |
| cork or styrofoam board covered with aluminum | Not applicable | 1 unit | |
| cotton | Not applicable | 50 g | |
| Dexamethasone | Sigma-Aldrich, San Luis, Missouri, USA | D4902 | |
| Dissecting scissor | Not applicable | 03 units sterile | |
| DPX Mountant for histology | Sigma-Aldrich, San Luis, Missouri, USA | 6522 | |
| Dulbecco’s modified Eagle’s medium (DMEM) | Sigma-Aldrich, San Luis, Missouri, USA | D5523 | With 1000 mg/L glucose and L-glutamine, without sodium bicarbonate, powder, suitable for cell culture |
| Eosin B | Sigma-Aldrich, San Luis, Missouri, USA | 861006 | |
| Fetal bovine serum (FBS) | Sigma-Aldrich, San Luis, Missouri, USA | F4135 | |
| Formaldehyde | Sigma-Aldrich, San Luis, Missouri, USA | 47608 | |
| Formalin | Sigma-Aldrich, San Luis, Missouri, USA | HT501128 | |
| Gentamicin | Sigma-Aldrich, San Luis, Missouri, USA | G1397 | |
| Hematoxylin | Sigma-Aldrich, San Luis, Missouri, USA | H3136 | |
| Hypodermic Needle (0.3mm x 13mm) | Not applicable | 5 units | |
| Indomethacin | Sigma-Aldrich, San Luis, Missouri, USA | I0200000 | |
| Insulin | Sigma-Aldrich, San Luis, Missouri, USA | I3536 | |
| Isopropanol | Sigma-Aldrich, San Luis, Missouri, USA | 563935 | 70% in H2O |
| Ketamine-D4 hydrochloride solution | Sigma-Aldrich, San Luis, Missouri, USA | K-006 | 1.0 mg/mL in methanol (as free base), certified reference material, Cerilliant® |
| Neubauer chamber | Sigma-Aldrich, San Luis, Missouri, USA | BR718620 | BRAND counting chamber BLAUBRAND Neubauer pattern. With clips, double ruled |
| Nichiryo pipette tips (0.1–10 μL) | Merck, Darmstadt, Germany | Z645540 | Volume range 0.1–10 μL, elongated, bulk pack. Sterile |
| Nichiryo pipette tips (1–10 mL) | Merck, Darmstadt, Germany | Z717401 | Volume range 1–10 mL, universal, bulk pack. Sterile |
| Nichiryo pipette tips (200 μL) | Merck, Darmstadt, Germany | Z645516 | Maximum volume 200 μL, graduated, ministack. Sterile |
| Oil-Red O solution | Sigma-Aldrich, San Luis, Missouri, USA | O1391 | 0.5% in isopropanol |
| Paraffin | Sigma-Aldrich, San Luis, Missouri, USA | 107.151 | 46–48, in block form |
| Penicillin/Streptomycin | Sigma-Aldrich, San Luis, Missouri, USA | P4333 | Solution stabilized, with 10,000 units penicillin and 10 mg streptomycin/mL, 0.1 μm filtered, BioReagent, suitable for cell culture |
| Phosphate-buffered saline solution 1x (PBS). | Sigma-Aldrich, San Luis, Missouri, USA | P3813 | Powder, pH 7.4, for preparing 1 L solutions. Balanced and sterile |
| Polypropylene conical tubes (15 mL) | Falcon, Fisher Scientific | 14-959-53A | Sterile |
| Polypropylene conical tubes (50 mL) | Falcon, Fisher Scientific | 14-432-22 | 2 units sterile |
| scalpel (optional) | Not applicable | 1 unit | |
| Silver nitrate | Sigma-Aldrich, San Luis, Missouri, USA | 85228 | |
| Sodium thiosulfate | Sigma-Aldrich, San Luis, Missouri, USA | 72049 | |
| Surgical tweezer (15 cm) | Not applicable | 3 units sterile | |
| Swiss male mice (6–8 weeks) | Bioterium, Santa Cruz State University | 021/22 | |
| syringe (1 mL) | Not applicable | 1 unit | |
| Trypan Blue Dye | Sigma-Aldrich, San Luis, Missouri, USA | T8154 | 0.4%, liquid, sterile-filtered, suitable for cell culture |
| Trypsin/EDTA (ethylenediaminetetraacetic acid) | Sigma-Aldrich, San Luis, Missouri, USA | T3924 | |
| Xylazine | Sigma-Aldrich, San Luis, Missouri, USA | PHR3263 | |
| β-glycerophosphate disodium salt hydrate | Sigma-Aldrich, San Luis, Missouri, USA | G9422 | BioUltra, suitable for cell culture, suitable for plant cell culture, ≥99% (titration) |
Lab Prep
Mesenchymal stem cells (MSCs) have been extensively studied as a new therapeutic approach, mainly to stop exacerbated inflammation due to their potential to modulate the immune response. The MSCs are immune-privileged cells capable of surviving in immunologically incompatible allogeneic transplant recipients based on low expression of class I major histocompatibility complex (MHC) molecules and in the use of cell-based therapy for allogeneic transplant. These cells can be isolated from several tissues, the most commonly used being the bone marrow and adipose tissues. We provide an easy protocol to isolate, culture, and characterize MSCs from epididymal adipose tissue of mice. The epididymal adipose tissue is surgically excised, physically fragmented, and digested with 0.15% collagenase type II solution. Then, primary adipose tissue-derived stem (ADSCs) cells are cultured and expanded in vitro, and the phenotypic characterization is performed by flow cytometry. We also provide the steps to differentiate the ADSCs into osteogenic, adipogenic, and chondrogenic cells, followed by functional characterization of each cell lineage. The protocol provided here can be used for in vivo and ex vivo experiments, and as an alternative, the adipose-derived stem cells can be used to generate MSCs-like immortalized cells.
Mesenchymal stem cells (MSCs) have been extensively studied as a new therapeutic approach, mainly to stop exacerbated inflammation due to their potential to modulate the immune response. The MSCs are immune-privileged cells capable of surviving in immunologically incompatible allogeneic transplant recipients based on low expression of class I major histocompatibility complex (MHC) molecules and in the use of cell-based therapy for allogeneic transplant. These cells can be isolated from several tissues, the most commonly used being the bone marrow and adipose tissues. We provide an easy protocol to isolate, culture, and characterize MSCs from epididymal adipose tissue of mice. The epididymal adipose tissue is surgically excised, physically fragmented, and digested with 0.15% collagenase type II solution. Then, primary adipose tissue-derived stem (ADSCs) cells are cultured and expanded in vitro, and the phenotypic characterization is performed by flow cytometry. We also provide the steps to differentiate the ADSCs into osteogenic, adipogenic, and chondrogenic cells, followed by functional characterization of each cell lineage. The protocol provided here can be used for in vivo and ex vivo experiments, and as an alternative, the adipose-derived stem cells can be used to generate MSCs-like immortalized cells.
Procedure
Mesenchymal stem cells (MSCs) have been extensively studied as a new therapeutic approach, mainly to stop exacerbated inflammation due to their potential to modulate the immune response. The MSCs are immune-privileged cells capable of surviving in immunologically incompatible allogeneic transplant recipients based on low expression of class I major histocompatibility complex (MHC) molecules and in the use of cell-based therapy for allogeneic transplant. These cells can be isolated from several tissues, the most commonly used being the bone marrow and adipose tissues. We provide an easy protocol to isolate, culture, and characterize MSCs from epididymal adipose tissue of mice. The epididymal adipose tissue is surgically excised, physically fragmented, and digested with 0.15% collagenase type II solution. Then, primary adipose tissue-derived stem (ADSCs) cells are cultured and expanded in vitro, and the phenotypic characterization is performed by flow cytometry. We also provide the steps to differentiate the ADSCs into osteogenic, adipogenic, and chondrogenic cells, followed by functional characterization of each cell lineage. The protocol provided here can be used for in vivo and ex vivo experiments, and as an alternative, the adipose-derived stem cells can be used to generate MSCs-like immortalized cells.
