Modified In Vivo Matrix Gel Plug Assay for Angiogenesis Studies
Summary
The method presented here can evaluate the effect of reagents on angiogenesis or vascular permeability in vivo without staining. The method uses dextran-FITC injection via the tail vein to visualize neo-vessels or vascular leakage.
Abstract
Several models have been developed to investigate angiogenesis in vivo. However, most of these models are complex and expensive, require specialized equipment, or are hard to perform for subsequent quantitative analysis. Here we present a modified matrix gel plug assay to evaluate angiogenesis in vivo. In this protocol, vascular cells were mixed with matrix gel in the presence or absence of pro-angiogenic or anti-angiogenic reagents, and then subcutaneously injected into the back of recipient mice. After 7 days, phosphate buffer saline containing dextran-FITC is injected via the tail vein and circulated in vessels for 30 min. Matrix gel plugs are collected and embedded with tissue embedding gel, then 12 µm sections are cut for fluorescence detection without staining. In this assay, dextran-FITC with high molecular weight (~150,000 Da) can be used to indicate functional vessels for detecting their length, while dextran-FITC with low molecular weight (~4,400 Da) can be used to indicate the permeability of neo-vessels. In conclusion, this protocol can provide a reliable and convenient method for the quantitative study of angiogenesis in vivo.
Introduction
Angiogenesis, the process of formation of neo-vessels from pre-existing vessels, plays a critical role in many physiological and pathological processes, such as embryonic development, wound healing, atherosclerosis, tumor development, etc.1,2,3,4,5. This dynamic process involves several steps, including the degradation of the matrix, vascular cell proliferation, migration and self-organization to form tubular structures and the stabilization of the neo-vessels6. Promoting angiogenesis has been demonstrated to be critical in the treatment of myocardial infarction, stroke and other kinds of ischemic diseases7 while inhibiting angiogenesis has been considered a promising strategy in the treatment of cancers8 and rheumatoid diseases9. Angiogenesis has been considered an organizing principle for drug discovery10. Thus, the construction of a reliable and convenient method to assess the extent of angiogenesis is critical for mechanical research or drug discovery in angiogenesis-dependent diseases.
Several in vitro and in vivo models have been developed to evaluate angiogenesis11. Among these, two-dimensional (2-D) models, like matrix gel tube formation assay12, cannot form functional tubular structures. The animal models, such as the hind limb ischemia model13,14, can reproduce the angiogenesis process but are complex and require a laser speckle blood flow imaging system. 3D models of vascular morphogenesis, like matrix gel plug assay, provide a simple platform that can mimic the process of angiogenesis in vivo15, but the detection of angiogenesis requires immunohistochemistry or immunofluorescence staining16,17,18, which are variable and poorly visualized.
Here, we describe a protocol for a modified matrix gel plug assay where vascular cells were mixed with matrix gel and subcutaneously injected into the back of mice to form a plug. In the plug, vascular cells need to degrade the matrix, proliferate, migrate, and self-organize to finally form functional vessels with blood flow in the internal environment. Thereafter, fluorescent-labeled dextran is injected via the tail vein, to flow through the plug, and the label is visualized to indicate neo-vessels. The content of angiogenesis can be quantitatively evaluated by the length of the vessels. This method can form functional vessels that cannot be produced in 2-D angiogenesis models12, and does not need complex stain process as in ordinary matrix gel plug assay11. It also does not require expensive specific instruments like laser speckle blood flow imaging system in hind limb ischemia model13,14,19. This method is versatile, low-cost, quantifiable, and easy to perform, and can be used to determine the pro- or anti-angiogenic capability of drugs or be used in mechanical research involved in angiogenesis.
Protocol
All procedures involving animal subjects were approved by the Institutional Animal Care and Use Committee (IACUC) of Wenzhou Medical University (XMSQ2021-0057, July 19th, 2021). All reagents and consumables are listed in the Table of Materials.
1. Culture medium preparation
- 10x M199 culture medium: Dissolve M199 powder to 10x concentration with 90 mL of deionized water and add 10 mL of fetal bovine serum (FBS), then pass through a 0.22 µm filter. Store the medium at 4 °C for up to 2 months.
- Complete endothelial culture medium: Add 50 mL of FBS, 5 mL of penicillin/streptomycin, and 5 mL of endothelial cell growth supplement to 460 mL of endothelial cell medium (ECM). Store the medium at 4 °C for up to 1 month.
2. Vascular cell preparation
- Culture 1 x 105 vascular cells (primary cultured endothelial progenitor cells14, endothelial cells, or endothelial cell lines) with 8 mL of complete endothelial culture medium in 100 mm tissue culture dish at 37 °C and 5% CO2 to 70% confluency.
- Remove the culture medium and rinse the dish 2x with 1x phosphate buffered saline (PBS) to remove unattached cells and debris. Remove PBS and add 3 mL of 0.25% trypsin containing 2.21 mM EDTA, and incubate at 37 °C for 1 min.
- Neutralize the trypsin with 7 mL of complete endothelial culture medium, and gently rinse cells off the culture dish. Confirm cell detachment under a light microscope (40x magnification).
- Collect cell suspension in a 15 mL tube and centrifuge at 400 x g for 10 min. Remove supernatant and resuspend cells with 5 mL of complete endothelial culture medium.
- Count cells using a hemocytometer and move suspension containing 2 x 106 cells to a sterile 1.5 mL tube. Each plug contains 1.5 x 106 cells, additional 25% cells are used in case of waste. Each group includes at least 3 plugs.
NOTE: After trials it was found that 1.5 x 106 cells in 300 µL of matrix gel led to the proper development of angiogenesis and was therefore chosen for experimentation. - Centrifuge cell suspension at 400 x g for 5 min to pellet cells, and then remove supernatant.
3. Matrix gel preparation
- Pre-cool sterile 1.5 mL tubes containing cell pellet in a 4 °C water bath. Pre-cool 30G 1 mL insulin syringes in a 4 °C refrigerator.
- Completely thaw matrix gel in a 4 °C water bath . Do not mix or vortex. Pre-cool 10x M199 and test reagent/drug-of-interest in a 4 °C water bath.
- Mix pre-cooled matrix gel with 10x M199 containing 10% FBS and the reagent to be tested at a volume ratio of 8.8:1:0.2 to get a matrix gel and M199 mixture containing 1% FBS and the reagent.
- Resuspend cells with 400 µL of the matrix gel mixture, mix gently to avoid forming bubbles. Keep the tube on ice until mice are prepared for injection. Keep the matrix gel mixture on ice all the time to avoid coagulation.
NOTE: Here, 300 µL of matrix gel mixture containing 1.5 x 106 cells was used to form gel plug. Prepare 25% extra gel according to 25% additional cells in step 2.
4. Mouse preparation
- Anesthetize 6-8 week old male Nu/Nu mouse (18-25 g) in the chamber of the animal anesthesia device with isoflurane (3% isoflurane in 100% oxygen at a flowrate of 1 L/min). After successful anaesthetization, confirmed by the absence of righting reflex and toe pinch reflex, move the mouse out of the chamber, move the mouse out of the chamber and put an anesthesia mask on the mouse and change the concentration of isoflurane to 1.5% in 100% oxygen at a flowrate of 1 L/min.
NOTE: Provide thermal support to the animal using a heating pad throughout the procedure. - Use vet ointment on eyes to prevent dryness. Tape the limbs of the mice on the operation board in the prone position.
5. Matrix gel mixture injection
- Load 300 µL of the matrix gel mixture into a 1 mL insulin syringe with a 30G needle. Avoid bubble formation. Quickly load the matrix gel (within 2 min) to avoid solidification during this time. Place the insulin syringe loaded with matrix gel on ice immediately.
- Clean the skin on the back of mice using 75% alcohol pads. Subcutaneously inject 300 µL of matrix gel mixture into one side of the back of mice.
- Gently remove the needle from the injection site to prevent leakage of the matrix gel mixture. Check for a small hump at the injection site (Figure 1).
- Place the mouse on the heating pad for 2 min to let the matrix gel mixture coagulate and form plug.
- Repeat steps 5.2. to 5.4. to create plug on the other side of the back of the mouse. Mark the edges of the humps using a marker pen.
- Observe the mouse until it has regained sufficient consciousness to maintain sternal recumbency. Put the mouse in a separate cage until it is fully recovered. House the mice in SPF-class experimental animal laboratory at 22 ± 2 °C with a 12 h light/dark cycle for 7 days.
6. Dextran-FITC injection through the tail vein
- After 7 days of matrix gel injection, resuspend 0.5 mg of dextran-FITC with 500 µL of double distilled water (ddH2O) and then violently vortex to obtain the dextran-FITC solution at the final concentration of 1 µg/µL.
NOTE: Use dextran-FITC with molecular weight of ~150 kDa for angiogenesis assay, and use dextran-FITC with molecular weight of ~4 kDa for vascular permeability assay. - Load 50 µL of dextran-FITC solution into the 29G syringes.
- Fix the mouse on the tail vein injection instrument. Clean the tail with 75% alcohol cotton ball and gently inject 50 µL of dextran-FITC through tail vein.
- Compress the injection site with cotton swab for 1 min to stanch bleeding, and then put the mice back in the cage for 30 min.
- Repeat steps 6.3. and 6.4. until all mice have received dextran-FITC injection.
7. Matrix gel plug collection
- Intraperitoneally inject 1% pentobarbital sodium in PBS (200 mg/kg body weight) and euthanize the mice with an IACUC approved procedure.
- Cut the skin along the marked border of the plug using surgical scissors and remove the skin above the matrix gel plug.
- Collect the plug and rinse in a small beaker with 1x PBS to wash away excess blood (Figure 2A). The color of plug can roughly delineate the degree of blood abundance and the content of neo-vessels (Figure 3A).
8. Embedding matrix gel plug and section preparation
- Cover the button of tissue embedding cassette with about 0.5 mL of tissue embedding gel, then immediately place matrix gel plug on the tissue embedding gel in the desired orientation as shown in Figure 2B. Then, embed the plug with additional tissue embedding gel.
- Put the cassettes into a -80 °C freezer for 12 h to solidify.
- For thick section preparation, take out the solidified plug block and cut 12 µm thick sections using the freezing microtome according to the instruction, and mount onto microscope slides.
- Slice 5-10 sections from each plug block.
9. Quantification of angiogenesis (Figure 3)
- Acquire fluorescence images from 5 independent fields of the section with an inverted fluorescence microscope at 488 nm wavelength under 10x magnification.
- Open the image file with Image J software (https://imagej.en.softonic.com/) and click the Angiogenesis Analyze button. Then, click Analyze HUVEC Phase Contrast button, and switch to Stat Results Table to gather information. Measure all 5 acquired fluorescence images for each section.
- Analyze the total length of neo-vessels from different groups to statistically evaluate the difference in angiogenesis between these groups.
NOTE: Total master segments length indicates the total length of neo-vessels. The reagent that increases the length of neo-vessels is termed pro-angiogenic, while the reagent that decreases the length of neo-vessels is anti-angiogenic.
10. Quantification of vascular permeability (Figure 4)
- Acquire fluorescence images from 5 independent fields of the section with an inverted fluorescence microscope at 488 nm wavelength under 20x magnification.
- Open the image file with Image J software. Click the Freehand Selection button and circle the leakage area, then click on the Analyze menu and select the Measure option to get the area information of the leakage area. Use the ratio of leakage area and the area of image to indicate vascular permeability.
- Analyze the ratio of leakage area and area of the image from different groups to statistically evaluate the difference in vascular permeability between these groups.
Representative Results
Figure 1 is the flowchart depicting how to prepare the mixture of matrix gel, vascular cells, culture medium and reagent. The mixture was then subcutaneously injected into the back of Nu/Nu mice and heated using a heating pad to accelerate its coagulation to finally form gel plug.
Figure 2A is the flowchart to indicate vessels with fluorescent labeled dextran. Fluorescent labeled dextran was injected via the tail vein and circle for 30 min, so it can enter the functional vessel in the gel plug. Thereafter, the gel plugs were collected, embedded using tissue embedding gel (the orientation of matrix gel when embedded was indicated in Figure 2B). A 12 µm of thick section was sliced from the plugs (5 slices from each side) and fluorescent pictures were taken for quantitative analysis of angiogenesis and/or vascular permeability.
Figure 3 is the influence of anti-angiogenic reagent palmitate and pro-angiogenic reagent fibroblast growth factor 1 (FGF1) on angiogenesis in the gel plug. Figure 3A is the appearance of gel plugs with vehicle, palmitate or FGF1. Blood can enter the functional neo-vessel in the gel plug, which makes the plug red to varying degrees. Figure 3B is the fluorescent image of matrix gel plugs, in which the functional vessels were visualized by dextran-FITC with high molecular weight. Figure 3C is the quantitative result of vessel length of different group. Palmitate can significantly decrease the length of neo-vessels, while FGF1 treatment can obviously increase the length.
Figure 4 compares the vascular permeability in gel plugs with or without vascular endothelial growth factor (VEGF) treatment. Figure 4A is the fluorescent image of matrix gel plugs, in which the leakage area is visualized by dextran-FITC with low molecular weight and indicated by arrows. Figure 4B is the quantitative result of leakage area. The increased leakage area in VEGF treatment group revealed that VEGF can increase vascular permeability.
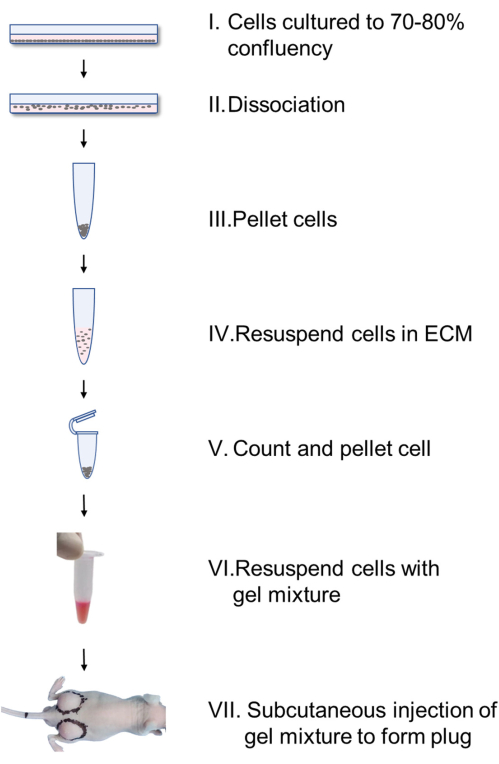
Figure 1. Flowchart showing formation of a gel plug in mice. Vascular cells cultured in monolayer culture are dissociated, pelleted, resuspended with endothelial cell medium (ECM), and counted (I-IV). After pelleting and resuspension in matrix gel mixture (containing 8.8 volume matrix gel, 1 volume 10x M199 supplemented with 10% FBS and 0.2 volume reagent), 300 µL of gel mixture was subcutaneously injected into the back of the mice (V-VII) to form plugs. Please click here to view a larger version of this figure.
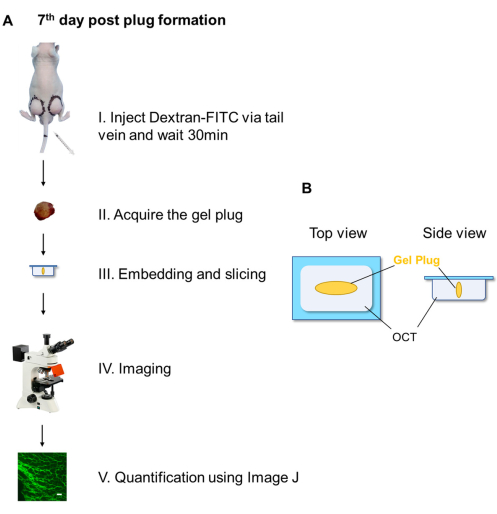
Figure 2. Flowchart showing dextran-FITC injection and quantification of angiogenesis. (A) Dextran-FITC was injected via the tail vein (I). After 30 min, gel plug was acquired, embedded and sections were sliced using freezing microtome (II, III). Thereafter, fluorescence images were taken using fluorescence microscope (IV) and angiogenesis was quantitatively analyzed using Image J software (V). (B) The diagram of matrix gel plug orientation when embedded in tissue embedding cassette. Abbreviations: OCT = optimum cutting temperature compound. Please click here to view a larger version of this figure.
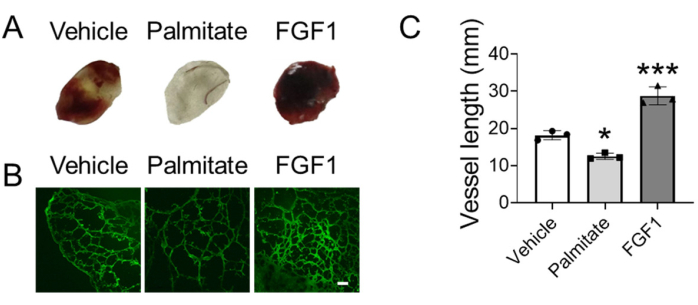
Figure 3. Evaluating angiogenesis using modified matrix gel plug assay. (A) Appearance of representative matrix gel plugs. (B) The fluorescent picture of functional vessels in matrix gel plugs indicated by Dextran-FITC. (C) The quantitative analysis of the length of functional vessels in matrix gel plugs using one-way ANOVA. *p<0.05 versus vehicle; ***p<0.001 versus vehicle. The scale bar is 100 μm. Abbreviations: FGF1 = Fibroblast growth factor 1. Please click here to view a larger version of this figure.
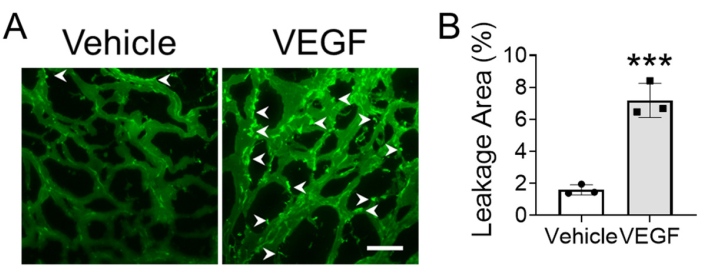
Figure 4. Evaluating vascular permeability using modified matrix gel plug assay. (A) The representative fluorescence image of vessels in gel plug, the arrows indicate leakage site. (B) The quantitative analysis of leakage area to evaluate vascular permeability using Student's t-test. ***p<0.001. The scale bar is 100 μm. Abbreviations: VEGF = vascular endothelial growth factor. Please click here to view a larger version of this figure.
Discussion
We present a reliable and convenient method for the quantitative evaluation of angiogenesis in vivo without staining. In this protocol, vascular cells were mixed with matrix gel in the presence of pro-angiogenic or anti-angiogenic reagents, and then subcutaneously injected into the back of Nu/Nu mice to form gel plug (Figure 1). After 7 days of gel plug formation, dextran-FITC was intravenously injected and circulated for 30 min. The gel plug was collected and embedded with tissue embedding gel, and 12 µm sections were sliced for photography (Figure 2). Dextran-FITC can indicate functional vessels without staining, and the length of neo-vessels can be used to quantitatively evaluate the pro-angiogenic or anti-angiogenic function of reagents. In the example presented in this protocol, palmitate treatment reduced length of neo-vessels, while FGF1 increased length of neo-vessels (Figure 3), which indicated that palmitate is anti-angiogenic, while FGF1 is pro-angiogenic. Besides that, this protocol can also be used in the evaluation of vascular permeability (Figure 4).
Caution should be exercised on the selection of the recipient mice, handling and injecting the matrix gel, collecting gel plug, and detecting the length of the vessels. Immunodeficient mice aged 6-8 weeks are recommended, although C57BL/6 mice are also workable. Considering the variability in the plug, 3-5 plugs in each group is recommended. The number of vascular cells should be even between different groups. Matrix gel should be carefully handled according to the instructions and should not be used if solidified or containing particulate matter or numerous bubbles. In the matrix gel preparation step, the final concentration of matrix gel should not be less than 80%, and vortex is not allowed as bubbles may be formed. It should be noted that the shape of gel plug may influence the reproducibility of the results, so heating pad was used to accelerate the solidification of matrix gel plug. A 37 °C animal incubator can also be used. Besides that, during gel plug embedding and section preparation, plugs and sections should be protected against bright light, and photos should be taken under a fluorescence microscope as soon as possible.
Among the various in vivo angiogenesis assays, the matrix gel plug assay is one of the most widely used methods because it is versatile, less costly, and easy to perform11. However, the staining process in ordinary matrix gel plug assay is error prone. The plug is fragile and with high water content. The dehydration and fixation steps included in the staining process may distort the gel plug, and further influence the evaluation of the length of the vessels. Besides that, the scattered vascular cells in the plug may be stained and recognized as functional vessels in ordinary matrix gel plug assay. One of the most important advantages of this modified matrix gel plug assay is that the functional vessels can be visualized without staining. FITC-labeled dextran was used to indicate vessels, and only functional vessels that have blood flow can be indicated, which contributes to the convenience and reliability of this assay.
Besides the length of neo-vessels, the permeability also affects the function of neo-vessels20. Fluorescent-labeled dextran with low molecular weight (~4,400 Da) can pass through the gap in vessels and can be used to indicate the permeability of neo-vessels. If needed, researchers can evaluate both angiogenesis and vascular permeability in the same gel plug by using both fluorescent-labeled dextran with low molecular weight and that with high molecular weight21.
Angiogenesis can be affected by the in vivo environment22, such as diabetes13,14,19, etc. This in vivo environment is hard to reproduce in these in vitro models. However, in this modified protocol, the in vivo environment can be easily reproduced by choosing proper recipient mice. Besides that, the interaction between different vascular cells also affects the process of angiogenesis. Different kinds of vascular cells, including endothelial cells, smooth muscle cells and pericytes, can be added to form neo-vessels in the plug in this protocol to investigate the contribution of different vascular cells in angiogenesis.
However, there are also some limitations for this method. The formation of neo-vessels can be affected by the shape of gel plug. Moreover, the neo-vessels formed in the gel plug is not even. There are more functional vessels at the edge of gel plug than that at the center (Figure 2A). Thus, the results may be affected by the proficiency of performer.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by Natural Science Foundation of Zhejiang Province (LY22H020005), and National Natural Science Foundation of China (81873466).
Materials
| Adhesion Microscope Slides | CITOTEST | 188105 | |
| Anesthesia System | RWD | R640-S1 | |
| Cell Counter | Invitrogen | AMQAX1000 | |
| Cell Culture Dish | Corning | 430167 | |
| Cryoslicer | Thermo Fisher | CryoStar NX50 | |
| Dextrans-FITC-150kDa | WEIHUA BIO | WH007N07 | |
| Dextrans-FITC-4kDa | WEIHUA BIO | WH007N0705 | |
| Embedding Cassettes | CITOTEST | 80203-0007 | |
| Endothelial Cell Medium | ScienCell | 35809 | |
| Endothelial Growth Supplements | ScienCell | 1025 | |
| Fetal Bovine Serum | Gibco | 10100147C | |
| Fibroblast Growth Factor 1 | AtaGenix | 9043p-082318-A01 | FGF1 |
| Fluorescence Microscope | Nikon | ECLIPSE Ni | |
| Heating Pad | Boruida | 30-50-30 | |
| Insulin Syringe | BD | 300841 | |
| Isoflurane | RWD | R510-22-10 | |
| Laboratory Balance | Sartorius | BSA124S-CW | |
| Matrigel | Corning | 356234 | Matrix gel |
| Medium 199 powder | Gibco | 31100-035 | |
| Microtubes | Axygen | MCT-150-C | |
| Optimal Cutting Temperature (OCT) Compound | SUKURA | 4583 | Tissue embedding gel |
| Palmitate Acid | KunChuang | KC001 | |
| Penicillin-Streptomycin Liquid | Solarbio | P1400 | |
| Phosphate Buffer Saline | Solarbio | P1022 | |
| Surgical Instruments | RWD | RWD | |
| Tail Vein Injection Instrument | KEW BASIS | KW-XXY | |
| Trypsin-EDTA Solution | Solarbio | T1320 | |
| Ultra-Low Temperature Freezer | eppendorf | U410 | |
| Vascular Endothelial Growth Factor | CHAMOT | CM058-5HP | VEGF |
References
- Bikfalvi, A. History and conceptual developments in vascular biology and angiogenesis research: a personal view. Angiogenesis. 20 (4), 463-478 (2017).
- Carmeliet, P., Jain, R. Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nature reviews Drug discovery. 10 (6), 417-427 (2011).
- De Palma, M., Biziato, D., Petrova, T. Microenvironmental regulation of tumour angiogenesis. Nature reviews Cancer. 17 (8), 457-474 (2017).
- Griffioen, A., Molema, G. Angiogenesis: potentials for pharmacologic intervention in the treatment of cancer, cardiovascular diseases, and chronic inflammation. Pharmacological reviews. 52 (2), 237-268 (2000).
- Viallard, C., Larrivée, B. Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis. 20 (4), 409-426 (2017).
- Craig, M., Sumanas, S. ETS transcription factors in embryonic vascular development. Angiogenesis. 19 (3), 275-285 (2016).
- Losordo, D., Dimmeler, S. Therapeutic angiogenesis and vasculogenesis for ischemic disease. Part I: angiogenic cytokines. Circulation. 109 (21), 2487-2491 (2004).
- Folkman, J. Anti-angiogenesis-new concept for therapy of solid tumors. Annals of Surgery. 175 (3), 409-416 (1972).
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nature Medicine. 1 (1), 27-31 (1995).
- Folkman, J. Opinion – Angiogenesis: an organizing principle for drug discovery. Nature Reviews Drug Discovery. 6 (4), 273-286 (2007).
- Nowak-Sliwinska, P., et al. Consensus guidelines for the use and interpretation of angiogenesis assays. Angiogenesis. 21 (3), 425-532 (2018).
- Fan, X., et al. Interleukin-1β augments the angiogenesis of endothelial progenitor cells in an NF-κB/CXCR7-dependent manner. Journal of Cellular and Molecular Medicine. 24 (10), 5605-5614 (2020).
- Dai, X., et al. Nrf2 transcriptional upregulation of IDH2 to tune mitochondrial dynamics and rescue angiogenic function of diabetic EPCs. Redox Biology. 56, 102449 (2022).
- Yan, X., et al. Liraglutide Improves the Angiogenic Capability of EPC and Promotes Ischemic Angiogenesis in Mice under Diabetic Conditions through an Nrf2-Dependent Mechanism. Cells. 11 (23), 3821 (2022).
- Koh, W., Stratman, A., Sacharidou, A., Davis, G. In vitro three dimensional collagen matrix models of endothelial lumen formation during vasculogenesis and angiogenesis. Methods in enzymology. 443, 83-101 (2008).
- Nowak-Sliwinska, P., et al. Consensus guidelines for the use and interpretation of angiogenesis assays. Angiogenesis. 21 (3), 425-532 (2018).
- Malinda, K. In vivo matrigel migration and angiogenesis assay. Methods in molecular biology (Clifton, NJ). , 287-294 (2009).
- Rezzola, S., et al. In vitro and ex vivo retina angiogenesis assays. Angiogenesis. 17 (3), 429-442 (2014).
- Dai, Q., et al. FGF21 promotes ischaemic angiogenesis and endothelial progenitor cells function under diabetic conditions in an AMPK/NAD+-dependent manner. Journal of Cellular and Molecular Medicine. 25 (6), 3091-3102 (2021).
- Gavard, J., Gutkind, J. S. VEGF controls endothelial-cell permeability by promoting the beta-arrestin-dependent endocytosis of VE-cadherin. Nature cell biology. 8 (11), 1223-1234 (2006).
- Birdsey, G. M., et al. The endothelial transcription factor ERG promotes vascular stability and growth through Wnt/β-catenin signaling. Developmental Cell. 32 (1), 82-96 (2015).
- Yan, X., et al. A Novel CXCR4 antagonist enhances angiogenesis via modifying the ischaemic tissue environment. Journal of Cellular and Molecular Medicine. 21 (10), 2298-2307 (2017).

