The Helsinki Rat Microsurgical Sidewall Aneurysm Model
Summary
Microsurgical sidewall aneurysms in rats are created by end-to-side anastomosis of an aortic graft to the abdominal aorta. We present step-by-step instructions and discuss anatomical and surgical details for successful experimental saccular aneurysm creation.
Abstract
Experimental saccular aneurysm models are necessary for testing novel surgical and endovascular treatment options and devices before they are introduced into clinical practice. Furthermore, experimental models are needed to elucidate the complex aneurysm biology leading to rupture of saccular aneurysms.
Several different kinds of experimental models for saccular aneurysms have been established in different species. Many of them, however, require special skills, expensive equipment, or special environments, which limits their widespread use. A simple, robust, and inexpensive experimental model is needed as a standardized tool that can be used in a standardized manner in various institutions.
The microsurgical rat abdominal aortic sidewall aneurysm model combines the possibility to study both novel endovascular treatment strategies and the molecular basis of aneurysm biology in a standardized and inexpensive manner. Standardized grafts by means of shape, size, and geometry are harvested from a donor rat's descending thoracic aorta and then transplanted to a syngenic recipient rat. The aneurysms are sutured end-to-side with continuous or interrupted 9-0 nylon sutures to the infrarenal abdominal aorta.
We present step-by-step procedural instructions, information on necessary equipment, and discuss important anatomical and surgical details for successful microsurgical creation of an abdominal aortic sidewall aneurysm in the rat.
Introduction
Rupture of a saccular cerebral artery aneurysm causes life threatening hemorrhage leading to stroke, permanent neurological damage, or death. Rupture can be prevented by either microsurgical clipping or endovascular aneurysm occlusion. A medical treatment to prevent aneurysm growth and rupture has not yet been established.
Experimental models for saccular aneurysms are needed to study the biology of arterial aneurysms and for the testing of novel therapeutic devices and strategies. For these purposes, several different models in different species have been developed and published1. Larger aneurysm models in pigs, dogs, and rabbits are preferably used to test endovascular innovations in complex aneurysm architecture1,2. Murine aneurysm models, on the other hand, allow testing research questions in genetically modified species3,4 and facilitate clarification of aneurysm biology at cellular and molecular level far better than larger species1. Although endovascular trans-carotid and trans-iliac device deployment is limited to bigger rats (>400-500 g) and stents smaller than 2.0 mm and 1.5 mm in diameter5,6, stents can also be placed through direct insertion into the abdominal aortic segment harbouring the experimental aneurysms. Previous work using the rat microsurgical abdominal aortic sidewall aneurysm model demonstrated its feasibility in testing novel embolic devices and its use in studying the molecular basis of aneurysm biology3,7.
Many of the currently published experimental saccular aneurysm models require expensive equipment, special environments (e.g. sterile operation rooms with fluoroscopy capabilities), interventional radiology competence, or use of expensive species. These requirements limit the widespread use of these models, and lead to the use of different models in different laboratories, which makes data comparison and meta-analysis difficult, if not impossible. A simple, robust, and inexpensive experimental model is needed as a standardized tool that can be used in a standardized manner in various labs in order to get comparable results from different institutions. For this purpose, we created the rat aorta sidewall saccular arterial aneurysm model.
The aim of this report is to present step-by-step procedural instructions, information on necessary equipment, and to discuss important anatomical and surgical characteristics for successful microsurgical creation of abdominal aortic sidewall aneurysms in the rat.
Protocol
NOTE: Male Wistar rats (mean body weight: 356 ± 44 g; 10-14 weeks old) were housed in the animal room at 22-24 °C and twelve hour light/dark cycle with free access to pellet diet, regular tap water and also received humane care in conformity with institutional guidelines. The experiments were reviewed and approved by the Committee for Animal Welfare at the University of Helsinki, Finland.
NOTE: In the following demonstration our surgical method is as follows: Anesthetize the rat by weight-adapted subcutaneous injection of medetomidine hydrochloride (0.5 mg/kg) and intraperitoneal injection of ketamine hydrochloride (50 mg/kg). Test for the lack of a toe-pinch reflex to confirm that the rat is fully anesthetized. Apply eye ointment, clip the surgical site, and clean the skin with a suitable disinfectant, for example Chlorhexidine, either in alcohol or water. Wash hands, put on protective clothing, a head cover and facemask, and sterile surgical gloves. Have a surgical assistant aid in maintaining aseptic surgery conditions and to document the surgical characteristics (as listed in Table 1). Monitor the depth of the anesthesia every 15 min during surgery by following respiratory rate, heart rate, and reaction to noxious stimulation (toe pinch test). Subcutaneous injection of buprenorphine (0.03 mg/kg) was given for postoperative analgesia and repeated if necessary every 12 hr.
1. Hardware, Consumables, and Positioning
- Keep the small animal surgery room quiet, aseptic, and maintain the room temperature at 23 +/- 3 °C. To perform the experimental aneurysm surgery the following minimal equipment is necessary:
- Use a table top surgical microscope ideally equipped with an assistant scope and digital microscope camera. Use nonporous reusable operating surface and cleanable instrument surface to protect the laboratory bench.
- Prepare consumables such as skin disinfectants, physiological saline, smaller and larger gauze swabs and suture material including 9-0 micro-sutures, a 5-0 non-absorbable, and 3-0 absorbable suture.
- Use the following standard surgical instruments: Surgical scissors, tissue forceps, soft tissue spreader or self-retaining retractor, and two Mosquito surgical clamps.
- Use a basic microsurgical instrument set that includes: Curved microneedle holder, one curved and two straight microforceps, and a straight or curved microscissor.
- Keep the microsurgical instruments in a kidney dish filled with sterile saline to keep the instrument non-sticky and clean during surgery. The kidney dish is padded with rubber mat or a surgical glove to prevent damage to the tips of the micro-instruments. Ensure that all supplies are sterile and the procedure is performed with aseptic technique according to the current recommendations for survival surgery on laboratory animals8-10.
- In addition, use a vascular clip applicator and three atraumatic temporary vascular clamps. It is important that the clamps used have a low closing power to prevent injury to the very thin wall of the rat aorta. Also prepare a ruler with half millimeter scale bars, a small colored rubber pad, and a short blunt needle.
- Place the rats in a supine position, immobilize both front and hind paws with surgical tape without applying stretch or compression to the skin, and bend their back with a thick marker or cautery pen by placing it under the lumber region of the back. It is important to obtain as much lumbar spine lordosis as possible in order to improve retroperitoneal exposure and access to the infrarenal aorta which facilitates microsurgical anastomosis. This positioning is recommended for abdominal aorta aneurysm creation but not necessary for thoracic graft harvesting.
2. Graft Harvesting
- Under general anesthesia, open the thoracic cavity (start graft ischemia time). Apply a noxious toe pinch to confirm that the rat is unresponsive before proceeding with the following steps. Cut through the midventral abdominal wall, identify the diaphragm just above the liver, and cut the connective tissue at the bottom of diaphragm to allow access to the rib cage. With large scissors, blunt side down, cut through ribs just one centimeter left and right of the rib cage midline and open up the thoracic cavity. The lungs are mobilized to the right side of the heart. Sacrifice the rats by overdosing with intracardiac injection of ketamine hydrochloride.Pulmonary trunk, left subclavian vein, left cranial vena cava, and the azygous vein interfere with the harvest of the proximal thoracic aorta.
- Caudal to the prominent veins there is a good entry point to start the dissection of the descending thoracic aorta using micro-scissors and microforceps.
- Trace the thoracic aorta back from the dorsal wall of the thorax upwards to the aortic arch by gentle blunt retraction and dissection with the Mosquito surgical clamp.
- Clamp and then cut the veins with scissors. Maintain the clamps on the veins and use as a retractor to expose the underlying aortic arch.
- Place a non-absorbable 6-0 silk ligature just above the first intercostal artery leaving the aorta.
- Cut the descending aorta just below the left subclavian artery and then below the ligature. Trimming can be done in order to get a perpendicular standardized aneurysm geometry, or if needed, to get a specific angle between the axis of the aneurysm and the aorta. Measure the graft in its width and length.
NOTE: Harvested grafts can either be immediately transplanted into recipient rats or further processed to achieve decellularization of the graft wall. Decellularized grafts can be stored at -4 degree Celsius until re-implantation at a later date (Figure 1). The decellularization of the aneurysm wall has been shown to predispose the aneurysm to enlarge11.
3. Graft Decellularization
- Freeze donor grafts in phosphate buffered saline at -4 °C.
- The next day, thaw the grafts, rinse with purified and deionized water at room temperature, and incubate for 10 hr at 37 °C in 0.1% sodium dodecyl sulfate.
- Finally, wash the sodium dodecyl sulfate-treated grafts three times with gentle agitation, refreeze in phosphate buffered saline, and keep at -4 °C until use.
4. Aneurysm Creation
- Dissection of the Abdominal Aorta
- After the animal has been anaesthetized, clip the fur from the surgical site and clean the skin with a suitable disinfectant. Test for the lack of a toe-pinch response prior to skin incision. Start the incision 1cm proximal to the genitals in midventral position (start operation time). Carefully separate the skin from the underlying muscles. End dissection one to two centimeters below the sternum.
- Carefully but firmly pull up the underlying abdominal muscles to avoid damage to the underlying organs. Extend the longitudinal cut along the linea alba upwards to the xiphoid process and end in caudal direction at the level of the bladder.
- Gently apply pressure to the bladder to empty it in order to facilitate access to the retroperitoneal space.
- Move the small intestines and the prominent cecum to the right or the left. Identify the large intestine namely the descending colon on the left bottom of the abdominal cavity.
- Cut the ligament in between the small intestine and the descending colon in cranial direction to allow wider exposure of the dorsal body wall. Place a self-retaining retractor to hold the bowels apart.
- The ideal location of end-to-side aneurysm anastomosis is found at the level between the renal and iliolumbar veins. The abdominal aorta lies retroperitoneal embedded in fat tissue. During dissection pay particular attention to the paired almost transparent ureters and testicular vessels.
- If further retraction of intestines is needed, use larger gauze swaps. The lumbar spine lordosis induced during positioning by placing a cautery pen or similar object under the lower back of the rat, reduces significantly the need of bowel retraction.
- The ventral surface of the dorsal body wall is covered with a thin parietal peritoneum. Once this is opened, visualize the aorta just underneath. During careful sharp and blunt dissection of the abdominal aorta from adjacent large veins grasp only the adventitia to avoid damage to the vessel wall.
- Infrequently, small lumbar arteries arise as segmental vessel from the dorsal surface of the abdominal aorta and interfere with preparation. Ligation and cutting of the vessel is needed to avoid retrograde oozing during aneurysm suturing. Use of curved micro-forceps can facilitate ligature placement in the depth.
- End-to-side Anastomosis
- Put a colored rubber pad underneath the abdominal aorta and upholster it with a small gauze swab. Remove loose connective tissue and adventitia at the level of the planned anastomosis site.
- Clamp the abdominal aorta distal to the anastomosis first, then proximal (start aortic clamping time). This ensures a firm filling of the vessel and facilitates subsequent arteriotomy.
- Perform the arteriotomy using either straight or curved micro-scissors. A micro-forceps holds up a very small piece of the vessel wall to cut out an elliptical shape.
- Flush the artery thoroughly with saline in both directions using a blunt tipped needle.
- Place the first two sutures of this end-to-side anastomosis at the proximal and distal end of the arteriotomy (start anastomosis time). Avoid grasping of the vessel wall with the micro-forceps whenever possible. Make sure that each suture is placed through all layers of the vessel wall.
- Perform suturing either as continuous or interrupted sutures. If interrupted suturing is chosen then place the back side nine o`clock suture first. Subsequent sutures can be spaced apart starting adjacent to the very first suture. Grasp the adventitia carefully. Avoid any squeezing/grasping of the intima.
- When the back wall is finished, check the endoluminal part of the anastomosis for misplaced sutures. Perform the same procedures in the same order on the front side. Ensure that the first of a total of three knots per suture is firm but not too tight.
- Hemostasis and Closure
- After the end to side anastomosis is completed (end anastomosis time), rinse the site with saline and remove the distal clamp first to allow for backflow (end aortic clamping time and end graft ischemia time).
- If obvious bleeding occurs from backflow an extra stitch may be needed (start time of hemostasis). In case of minor oozing, achieve hemostasis with gentle pressure over the bleeding site using a small piece of a gauze swab.
- Remove the proximal vascular clamp, rinse the anastomosis site once more, and cut off the remaining ends of the ligature at the aneurysm dome.
- Confirm patency of the aneurysm by observation of volume increase of the aneurysm during peak arterial pulse wave. Assess distal abdominal artery patency through the direct “milking test”.
- Remove the plastic sheet and the small gauze swab underneath. The pulsating blood whirl within the created aneurysm is clearly visible.
- Cover suture lines around the anastomosis with small pieces of adipose tissue or Spongostan for additional hemostasis if small oozing is still present (end time of hemostasis).
- Remove the soft-tissue spreader and gauze swabs. Place the small intestines, the cecum, and the fat mass back in their correct position.
- Close the midline abdominal muscles using 5-0, 4-0, or 3-0 resorbable or non-absorbable sutures followed by skin wound closure using continuous suture technique with 3-0 resorbable polyfilament suture (end operation time). Note: In our experience rats tolerated resorbable skin sutures better than non-absorbable monofilament sutures.
Representative Results
A pilot series comprised 14 rats. Subsequently a total of 84 animals were operated according to the presented protocol for several research projects between March and September 2012. Additional 29 animals served as donors for arterial saccular grafts. The remaining experiments were performed using pre-treated grafts harvested and stored from previous experiments using rats of the same gender, strain, weight, and age.
Body weight, overall operation time, aortic clamping time, time for anastomosis creation, time to hemostasis after anastomosis creation, graft ischemia time, and aneurysm dimensions at the time of creation (aneurysm width and length) were recorded and extracted from written case report forms. All characteristics are summarized and visualized in Table 1 and Figure 1.
With the exception of one animal that underwent a second operation due to thrombosis of the abdominal aorta distal to the anastomosis site there were no periprocedural mortality or morbidity. Mean operation time was less than 52 min (52 ± 12 min). In animals with transplantation of syngeneic aneurysms (n = 21) mean graft ischemia time was 29 ± 7 min. Overall mean aortic clamping time was 25 ± 7 min. Aneurysm dimensions revealed to be constant with low deviation of size (mean width 2.5 ± 0.2 mm and mean length 3.8 ± 0.2 mm).
The collected data underwent descriptive analysis and visualization using statistical software. Values are expressed as mean ± standard deviation (SD) and 95% confidence interval (CI).
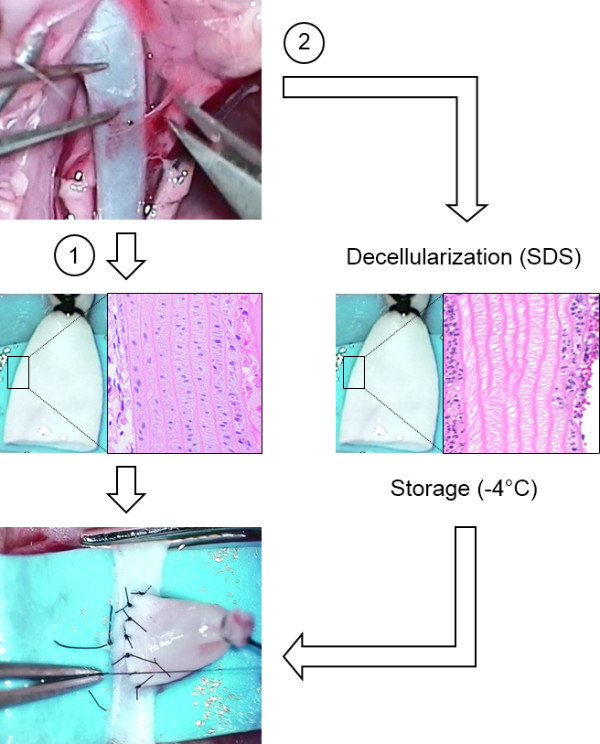
Figure 1: Non-Decellularized or Decellularized Grafts. Untreated native donor grafts from the thoracic aorta are immediately re-implanted into recipient rats (1). Grafts to be decellularized are treated with sodium dodecyl sulfate (SDS) and stored at -4 degree Celsius until re-implantation (2). The histological panel depicts longitudinal section through an untreated (left) and decellularized (right) graft wall. Hematoxylin-eosin staining.
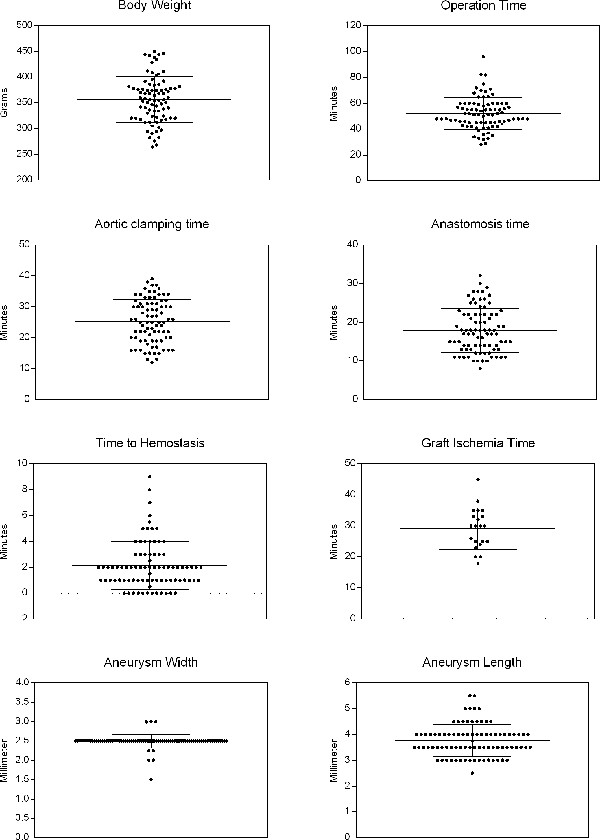
Figure 2: Surgical Characteristics. The graphs visualize the distribution of single data values (small black dots), data mean (bold long bar), and standard deviation (error bars). Please click here to view a larger version of this figure.
| Characteristic | Mean | ± SD | 95% CI upper – lower |
| Mean body weight (grams) | 363 | 47 | 350 – 373 |
| Mean operation time (minutes) | 50 | 11 | 48 – 53 |
| Mean aortic clamping time (minutes) | 25 | 7 | 23 – 27 |
| Mean anastomosis time (minutes) | 18 | 6 | 16 – 19 |
| Mean time of hemostasis (minutes) | 2 | 2 | 2 – 3 |
| Mean graft ischemia time (minutes) | 29 | 7 | 26 – 32 |
| Mean aneurysm width (millimetre) | 2.5 | 0.2 | 2.4 – 2.5 |
| Mean aneurysm length (millimetre) | 3.7 | 0.5 | 3.5 – 3.8 |
Table 1: Surgical Characteristics. SD = standard deviation; CI = confidence interval
Discussion
Progress in our understanding of the complex biology of saccular cerebral artery aneurysm depends on analysis of epidemiological and clinical data, complemented by laboratory studies on patient samples and experimental work in animal models3,12,13.
Small animals such as the rat are inherently associated with lower costs of experiments and housing, and reduced need of specialized equipment. An average total operation time of less than 60 minutes for microsurgical creation of a sidewall aneurysm in rats is much shorter than the time used for creation of more complex microsurgical venous pouch arterial bifurcation aneurysm in rabbits and dogs2,14,15. The advantages of low costs and faster methods of aneurysm creation may facilitate conduction of studies with larger number of experiments and subsequent increased statistical power. In addition, current murine models have been successfully implemented to answer research questions needing more sophisticated laboratory methodology, including transgenic animals3,4. When using mice for creation of sidewall aneurysms, one need to keep in mind that the needed interrupted 11-0 sutures require additional microsurgical skills. Performance of the presented aneurysm model in mice is also associated with higher mortality rates (30%; mainly due to complications in fluid balance and anaesthesia and smaller diameter (0.5-1 mm) of the mouse aorta)3.
Basic principles of the rat aneurysm model can be mastered in a short period of time. An introductory course in rodent microsurgery is recommended for those researchers unexperienced in performing dissections and suture techniques under an operating microscope. Highlighted key steps in the presented protocol will further simplify the procedure. Particular caution should be exercised during dissection of the abdominal aorta from adjacent large veins.
The small peripheral vasculature diameter of a medium sized rat makes trans-carotid and trans-iliac endovascular device deployment difficult5,6. However, devices can also be placed through direct abdominal aortic insertion or direct placement into the experimental aneurysm before end-to-site anastomosis7,16. Volumetric changes in neck remnants and aneurysm geometry can be follow-up with serial and non-invasive high-frequency ultrasound, micro-CT, or high resolution magnetic resonance angiography16. Previous experiments revealed high overall patency rates of 92.5% at a median follow-up of six weeks after creation without peri- or intra-procedural anticoagulation and antiaggregation3,7,16. With the exception of a single case significant growth or dilatation of the experimental aneurysms was not observed and none of them ruptured3.
However, if the harvested grafts are decellularized the aneurysms demonstrate a heterogeneous pattern of thrombosis, recanalization, growth, and eventual rupture11. Growing aneurysms in the latter study demonstrated marked adventitial fibrosis and inflammation, complete wall disruption, and increased neutrophil accumulation in unorganized intraluminal thrombus. In this way the model allows to study aneurysm growth and rupture and could be potentially used to assess biological responses induced by embolization devices in growing and rupture-prone aneurysms. None of the available aneurysm models that can be embolized ideally represent a human saccular cerebral artery aneurysm or reproduce the exact pathobiology behind aneurysm formation or rupture.
It remains a matter of debate to what extent the choice of graft (venous or arterial pouch) and choice of angioarchitecture (sidewall or bifurcation construct) influence the translation of results into clinical practice. Certainly different models are optimal for different purposes, and can be optimized to a very high level in certain institutions. The presented model will not make other models obsolete. It will remain necessary for an investigator to choose from a wide range of different technical models and animals the one that suits best the experimental goals, practical considerations, and laboratory environment.
However, some experiments should ideally be performed in a same standardized model in various institutions and labs, in order to allow better comparison of data and of devices or treatments. To date there are no guidelines for standardized testing of endovascular devices prior to clinical application, and animal models remain underused1. Standardized models will gain importance once multicenter randomized preclinical trials also emerge in this field of research.
Microsurgical aneurysm creation allows standardization of graft origin, volume-to-orifice ratio, and parent vessel to aneurysm long axis angle. The presented technique is aimed to generate standardized aneurysms with minimal variation in aneurysm dimension, location, and relation to the parent artery. This high degree of standardization and the relatively low costs make the model a good tool to test embolization materials and devices that are then tested in other more complicated and expensive models.
In conclusion the presented microsurgical sidewall rat aneurysm model is a fast, affordable, and consistent method to create experimental aneurysms that are standardized by means of size, shape, and geometric configuration of the aneurysm in relation to the parent artery.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors are solely responsible for the design and conduct of the presented study. Dr. Marbacher was supported by a grant from the Swiss National Science Foundation (PBSKP3-123454). The authors declare no conflict of interests.
Author contributions to the study and manuscript preparation include the following. Conception and design: SM, JM, JF. Acquisition of data: SM, EA, JF. Analysis and interpretation of data: SM, JF, JM. Drafting the article: SM, JF, JM. Critically revising the article: JH, MN. Statistical analysis: SM, JF. Study supervision: JF, JH, MN.
Materials
| Name of Material/ Equipment | Company | Catalog Number | Comments/Description |
| Medetomidine | Any genericon | ||
| Ketamin | Any genericon | ||
| Buprenorphine | Any genericon | ||
| Phosphate buffered saline | |||
| Sodium dodecyl sulfate (0.1%) | |||
| 3-0 resorbable suture | Ethicon Inc., USA | VCP824G | |
| 5-0 non absorbable suture | Ethicon Inc., USA | 8618G | |
| 6-0 non absorbable silk suture | B. Braun, Germany | C0761060 | |
| 9-0 nylon micro suture | B. Braun, Germany | G1118471 | |
| Spongostan | Ethicon Inc., USA | MS0002 | |
| Operation microscope | Leica , Germany | M651 | |
| Digital microscope camera | Sony, Japan | SSC-DC58AP | |
| Standard surgical instruments | B. Braun, Germany | Multiple | See protocol 1.4 |
| Microsurgical instruments | B. Braun, Germany | Multiple | See protocol 1.5 |
| Vascular clip applicator | B. Braun, Germany | FT495T | |
| Temporary vascular clamps | B. Braun, Germany | FT250T | |
| Graph Pad Prism statistical software | GraphPad Software, San Diego, California, USA | V 6.02 for Windows |
References
- Bouzeghrane, F., et al. In vivo experimental intracranial aneurysm models: a systematic review. AJNR Am J Neuroradiol. 31, 418-423 (2010).
- Marbacher, S., et al. Complex bilobular, bisaccular, and broad-neck microsurgical aneurysm formation in the rabbit bifurcation model for the study of upcoming endovascular techniques. AJNR. American journal of neuroradiology. 32, 772-777 (2011).
- Frosen, J., et al. Contribution of mural and bone marrow-derived neointimal cells to thrombus organization and wall remodeling in a microsurgical murine saccular aneurysm model. Neurosurgery. 58, 936-944 (2006).
- Marjamaa, J., et al. Mice with a deletion in the first intron of the Col1a1 gene develop dissection and rupture of aorta in the absence of aneurysms: high-resolution magnetic resonance imaging. at 4.7 T, of the aorta and cerebral arteries. Magn Reson Med. 55, 592-597 (2006).
- Oyamada, S., et al. Trans-iliac rat aorta stenting: a novel high throughput preclinical stent model for restenosis and thrombosis. The Journal of surgical research. , 166-191 (2011).
- Lowe, H. C., James, B., Khachigian, L. M. A novel model of in-stent restenosis: rat aortic stenting. Heart. 91, 393-395 (2005).
- Marjamaa, J., et al. Occlusion of neck remnant in experimental rat aneurysms after treatment with platinum- or polyglycolic-polylactic acid-coated coils. Surg Neurol. 71, 458-465 (2009).
- . with the support of the NC3Rs. Aseptic Technique in Rodent Surgery. , (2014).
- Bernal, J., et al. Guidelines for rodent survival surgery. Journal of investigative surgery : the official journal of the Academy of Surgical Research. 22, 445-451 (2009).
- Pritchett-Corning, K. R., Luo, Y., Mulder, G. B., White, W. J. Principles of rodent surgery for the new. , (2011).
- Marbacher, S., et al. Loss of mural cells leads to wall degeneration, aneurysm growth, and eventual rupture in a rat aneurysm model. Stroke. 45, 248-254 (2014).
- Frosen, J., et al. Remodeling of saccular cerebral artery aneurysm wall is associated with rupture: histological analysis of 24 unruptured and 42 ruptured cases. Stroke. 35, 2287-2293 (2004).
- Frosen, J., et al. Saccular intracranial aneurysm: pathology and mechanisms. Acta neuropathologica. , 123-773 (2012).
- Ysuda, R., Strother, C. M., Aagaard-Kienitz, B., Pulfer, K., Consigny, D. A large and giant bifurcation aneurysm model in canines: proof of feasibility. AJNR Am J Neuroradiol. 33, 507-512 (2012).
- Sherif, C., et al. Microsurgical venous pouch arterial-bifurcation aneurysms in the rabbit model: technical aspects. Journal of visualized experiments : JoVE. , (2011).
- Marjamaa, J., et al. High-resolution TOF MR angiography at 4.7 Tesla for volumetric and morphologic evaluation of coiled aneurysm neck remnants in a rat model. Acta Radiol. 52, 340-348 (2011).

