A Rat Carotid Balloon Injury Model to Test Anti-vascular Remodeling Therapeutics
Summary
The rat carotid balloon injury model described below allows researchers to evaluate drugs or therapeutics that negate injury-induced arterial hyperplasia. Detailed pre-surgical preparation, surgical procedure, and post-surgical cares of the animal are described.
Abstract
The rat carotid balloon injury is a well-established surgical model that has been used to study arterial remodeling and vascular cell proliferation. It is also a valuable model system to test, and to evaluate therapeutics and drugs that negate maladaptive remodeling in the vessel. The injury, or barotrauma, in the vessel lumen caused by an inflated balloon via an inserted catheter induces subsequent neointimal growth, often leading to hyperplasia or thickening of the vessel wall that narrows, or obstructs the lumen. The method described here is sufficiently sensitive, and the results can be obtained in relatively short time (2 weeks after the surgery). The efficacy of the drug or therapeutic against the induced-remodeling can be evaluated either by the post-mortem pathological and histomorphological analysis, or by ultrasound sonography in live animals. In addition, this model system has also been used to determine the therapeutic window or the time course of the administered drug. These studies can leadto the development of a better administrative strategy and a better therapeutic outcome. The procedure described here provides a tool for translational studies that bring drug and therapeutic candidates from bench research to clinical applications.
Introduction
Angioplasty is an endovascular procedure used to widen the narrowed or obstructed arteries resultant from pathological conditions such as atherosclerosis. One common complication of angioplasty is the post-operational neointimal hyperplasia, or restenosis, which occurs owing to surgical injuries and the subsequent inflammation-induced vascular remodeling. These conditions lead to proliferation of vascular smooth cells, and multiple pathophysiological consequences1-3. Neointimal hyperplasia re-thickens the vessel, and occurs in up to 60% post-angioplasty patients within the first year. Therefore, restenosis is a major setback of the widely used angioplasty procedure4. Although the implantation of the drug-elution stent may help to prevent restenosis, only selected candidates can undergo this costly procedure5.
Both animal and clinical studies have established that chronic inflammation generated by vascular injury and /or surgical wound serves as the main stimulus for post-angioplasty neointimal growth2,4. The rat carotid balloon injury model mimics the clinical situation and therefore serves as a valuable model system to identify cellular factors that involve in the vascular remodeling and vascular cell proliferation6-9. This model system is also a highly useful tool to evaluate and/or screen for drugs and therapeutic reagents that suppress neointimal growth in pre-clinical translational studies10-14.
Compared to the murine carotid wire injury model15 and the murine femoral artery wire injury model16, the rat carotid balloon injury model has the advantages of being sufficiently large in size for the ease of surgical procedure that facilitates reproducibility of the inflicted injury. It can provide a larger number of primary cells (e.g. vascular smooth muscle cells, endothelial cells) for additional in vitro studies to delineate the molecular mechanism governing vascular remodeling. Importantly, compared to mice, rats are also known to be a better model for physiological and toxicological studies17. Although a disadvantage or limitation of the rat model is the lack of genetic modified and gene knockout models, this disadvantage can be overcome by the availability of the rat genomic sequence and the recent development of powerful genomic editing tools such as CRISPR-CAS technology that renders possible manipulation of vast ranges of genomic sequences in different model systems18,19.
Although the rat balloon injury model has been used by multiple labs and various comprehensive protocols have been published20,21, this protocol aims at providing more details at pre-surgery preparations and may guide researchers new to this procedure to set up this surgical practice. We also emphasize the post-surgery care of the animals that allows not only post-mortem pathological and histomorphological analyses of the therapeutic effects on arterial remodeling, but also ultrasound sonographic studies in live animals13,22.
Protocol
NOTE: the use of rat balloon injury model and associated procedures including injection of recombinant sRAGE and ultrasound sonographic studies have been approved by the Animal Care and Use Committee (ACUC) of National Institute on Aging, NIH.
1. Pre-surgery Preparations
- Instrument, Surgical Platform, and Personal Protection Equipment.
- See Materials and Equipment's Table for all the surgical instruments and reagents used in this procedure. Autoclave all surgical instruments prior to surgical procedure.
NOTE: For multiple surgeries performed at the same day, use a pre-heated glass bead sterilizer to sterilize the instruments between surgeries. - Sterilize the surface of operating platform with 70% alcohol. Put on surgical gown, hair cover, surgical mask, safety glasses, and gloves. Leave a box of sterilized gloves near the surgical platform for additional use.
- See Materials and Equipment's Table for all the surgical instruments and reagents used in this procedure. Autoclave all surgical instruments prior to surgical procedure.
- Preparation of the Balloon Catheter.
- Fill 1 ml syringe with sterile water and attach the syringe to a sterile water-filled two-way stopcock. Gently push the water through the stopcock to fill the Luer-lock portion of the balloon catheter, and remove air bubbles from the opening of the catheter.
- Test balloon inflation to assure the balloon can be inflated by 0.02 ml of water. Store the tip of the balloon in a container of sterilized water before the surgery to avoid desiccation and contamination.
- Anesthesia and Animal Preparation.
- Weigh male Wistar rats (400-450 g) and check the ear tag to confirm the ID. Place the rat in an induction chamber, then turn on the vaporizer and adjust the isoflurane level to 3.5-4.0% and the oxygen level to 1.5-2.0 L. Check the depth of sedation by checking pedal withdrawal reflex such as pinching the toe or tail of the animal.
- After reaching the anesthesia (i.e. the animal lacks pedal withdrawal reflex), transfer the animal to the surgical platform and hook the rat with a nose cone that connects to an anesthesia vaporizer. Adjust isoflurane concentration to 1.5-2.0 and oxygen to 0.6-1.0 L. Periodically check the depth of sedation by pinching the toe or tail of the animal to assure that the rat is completely relaxed and immobile.
- After reaching the surgical level of anesthesia, shave the ventral neck region from the chin down to just above the sternum with animal hair clippers and swab the shaved skin with alternating 1:10 dilution of povidone-iodine and 70% ethanol 3 times.
- Place ophthalmic solution on the open eyes of the animal with a cotton swab to prevent them from drying. Moisten the tongue and mouth of the animal with drinking water to avoid drying during the operation.
- Cover the rat with a sterile surgical sheet and cut the sheet to expose the neck region. Place the animal in supine position on a heated pad on the surgical platform with the head toward the surgeon. Fix the limbs of the animal to the surgical platform using tape.
2. Surgical Procedures
- Dissection and Isolation of the Left Carotid Artery
- Use a scalpel to make a straight incision below the chin in a direction toward the tail all the way to the top of the sternum just above the rib cage. Use a 7S forceps to bluntly dissect the salivary glandular tissues under the skin to expose the muscular layer. Separate muscular tissues to gain access to carotid vasculature and the vagus nerve in the neck region.
- Bluntly dissect tissues surrounding the carotid artery, and carefully separate the vagus nerve and vascular fascia to avoid any damage. Continually dissect till the carotid artery bifurcation to expose the internal and external branches and the common carotid artery down to the sternum.
- Preparation of Arteriotomy
- For retraction and hemostasis, place a ligature at the most proximal site on the left common carotid artery, and another ligature at the location immediate distal to the bifurcation.
- Place a 4.0 silk suture at the external carotid artery as far from the bifurcation as possible to retract the artery toward the head. Permanently ligate small arteries using 4.0 silk sutures along the external carotid artery including the ascending pharyngeal, the occipital and the superior thyroid to avoid arterial leak.
- Place a 4.0 silk suture around the internal carotid artery to avoid a significant retrograde blood loss. Usually, the available length of the internal branch is minimal. To visualize the internal carotid artery, expose the internal carotid artery as much as possible by gently retracting the overlying carotid artery to the right.
- Introduction of the Balloon Catheter
- Retract the proximal suture on the common carotid artery and place an arterial clamp on the vessel to temporarily stop the blood flow. Make a small (1/3 to 1/4 of the circumference of the artery) incision on the external carotid artery at a location as distal to the suture as possible.
- Gently insert an un-inflated balloon catheter into the incision and then advance the catheter in the lumen close to the arterial clamp on the common carotid artery. Remove the clamp, and further advance the balloon catheter to the aortic arch, approximately 35-40 mm from the incision.
- Balloon Injury
- Manually inflate the catheter to a volume of 0.02 ml via the attached syringe and then lock the stopcock between the syringe and the catheter. Slowly withdraw the catheter with rotations. When the catheter is close to the arteriotomic incision, deflate the balloon. Advance the catheter back to original location.
- Repeat 2.4.1 for a total 3 times. Carefully remove the catheter from the artery lumen.
- Close Up the Surgical Wound
- Close the arteriotomy with sutures. Release the internal carotid artery to restore the blood flow. Check for arterial bleeding or leak. If bleeding occurs, apply a piece of gauze with a bit pressure to stop the bleeding.
- Remove the rest of sutures and clamps used for the procedure. Close the glandular tissue with 6.0 sutures. Close the skin with skin sutures or wound clips. Turn off isoflurane and leave the animal with pure oxygen for a few min.
3. Administration of Therapeutics, Analgesics, Post-surgery Care, and Euthanasia
- Administration of Therapeutics and Analgesics.
- Following the surgery, inject buprenorphine (0.03 mg/kg) intramuscularly. Inject 5 ml pre-warmed sterile saline subcutaneously to compensate the blood loss during the surgery. Inject sRAGE (0.5 ng/g) (therapeutics) into the peritoneum.
- Moisten the eyes again with ophthalmic ointment. Place the rat on the heat pad and monitor the animal until it regains complete consciousness to maintain sternal recumbency.
- Return the rat to a cage with clean bedding and bring the cage back to the vivarium. Place a special observation card on the cage to alert the attending veterinarians and caretakers.
- Post-surgery Cleanup
- Put used surgical material including pads, swabs, needles et al either into the trash or a special disposal container. Disinfect the surgery platform and clean the surgical instruments.
- Post-surgery Care
- Administer buprenorphine (0.03 mg/kg) twice a day (b.i.d.) during the 48 hr post-surgery period. Monitor the animal closely during the 48 hr post-surgery period for the common signs of distress such as immobility, inability to feed, hutched posture, ruffled fur, and grimace. Monitor wound conditions for redness, swelling, dehiscence, and infections.
NOTE: Additional treatments may be prescribed according to attending veterinarian's recommendation. - Remove the suture or wound clips according to attending veterinarian's recommendation.
- Administer buprenorphine (0.03 mg/kg) twice a day (b.i.d.) during the 48 hr post-surgery period. Monitor the animal closely during the 48 hr post-surgery period for the common signs of distress such as immobility, inability to feed, hutched posture, ruffled fur, and grimace. Monitor wound conditions for redness, swelling, dehiscence, and infections.
- Euthanasia
- Two weeks after the surgery, euthanize the rats to isolate the carotid arteries for post-mortem histo-morphological analyses22. Place the rat in a chamber containing 5-20% isoflurane saturated air for at least 2 min till complete cessation of respiration. Verify the death prior to dissection of the organs and tissues.
Representative Results
Two weeks after the balloon injury, the rat is euthanized and the carotid arteries are isolated for histo-morphological analysis. Both operated left carotid artery and un-operated right artery are cross-sectioned, processed, and paraffin-embedded. The paraffin samples are then further thin-sectioned, and stained with hematoxylin-eosin (H & E). Histo-morphological analyses are performed using a digital imaging analysis system. The details of artery harvest and histo-morphological analysis have been described13,23. The balloon injury-elicited neointimal growth, or the thickening of the vessel wall, if left untreated (or treated with saline as the placebo), is evident as shown in Figure 1B, compared to non-operated vessel section from the same rat (Figure 1A). The vessel section from the rat received therapeutic reagent (in this case, sRAGE) that blocks neointimal growth showed significantly reduced thickening of the vessel wall (Figure 1C). The effects of the balloon injury as well as the therapeutic treatment can also be evaluated in vivo using ultrasound sonography22, which collaborate well with the results from histo-morphological analysis (Figure 2). The details for the evaluation of the efficacy and the administration window of therapeutic sRAGE in the rat balloon injury model have been described in previous publications13,22. To evaluate therapeutic reagents that reduce or block the injury-elicited vessel wall remodeling, a group of rats (n = 8-15) are to be operated in order to obtain statistically meaningful conclusions.
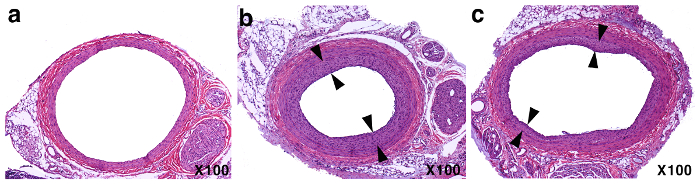
Figure 1: Representative results from the rat carotid balloon injury model. H & E staining of the carotid artery cross-sections (A) the non-operated right carotid artery section; (B) the balloon injured and placebo-treated left carotid artery section (arrows indicate the neotimal areas); (C) the balloon injured and sRAGE-treated left carotid artery section. Please click here to view a larger version of this figure.
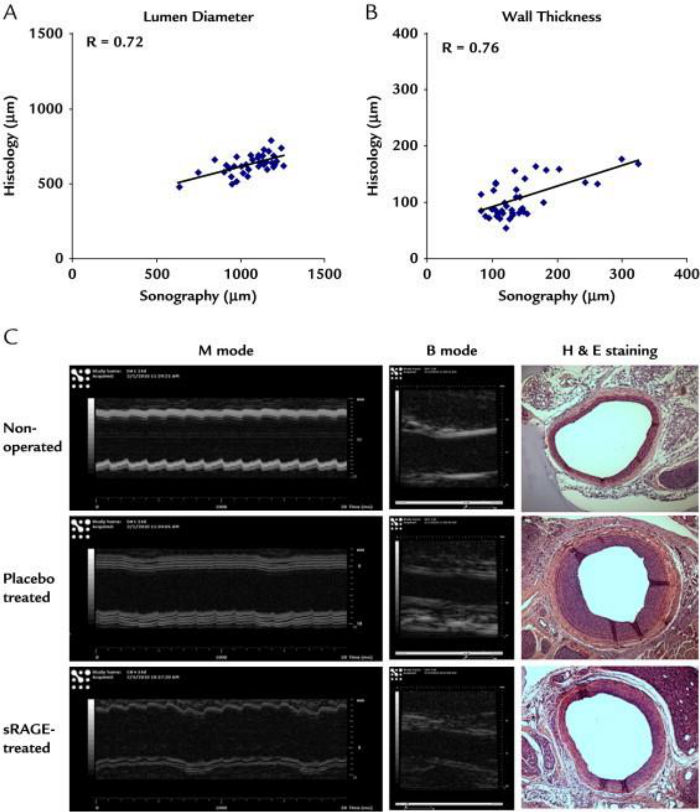
Figure 2: Correlation of ultrasound sonography and histology. sRAGE (0.5 ng/g) treated carotid vessels shown in scatter plot of data from (A) lumen diameter and (B) vessel wall thickness at 2 weeks post-injury (the non-operated, injured with sRAGE, and placebo-treated, n = 12 of each group). (C) Representative sonographic and histologic (100X) images. (Figure from reference22, with permission from the publisher). Please click here to view a larger version of this figure.
Discussion
There have been two methods used to inflate the balloon in order to generate the injury that removes the mural endothelium in the carotid arterial lumen. One is to fill the attached syringe with liquid20, and the other is to use air pressure21. We prefer to use liquid-filled syringe because the exact liquid volume (0.02 ml) will be used for each procedure. This renders a precise and reproducible inflation of the balloon, leading to a similar level of injury in each animal undergoing the procedure. Using liquid in the syringe indeed requires removal of air bubbles in the liquid. This can be achieved by pushing the bubbles out from the opening of the catheter. After removing a majority of air bubbles, any residual, tiny air bubbles will dissipate in 1-2 hr. We suggest that researchers make pre-surgery materials ready 1-2 hr prior to the procedure, and check the degree of the balloon inflation before each individual surgery to ensure the appropriate inflation.
For the surgery, the surgeon and his/her assistant(s) must wear all necessary protection gears including face masks, sterile gloves, and surgical gowns to protect both animals and researchers from infection. It is important to use fully grown, male rats with similar bodyweights (e.g. between 380-450 g) to ensure the proper size of carotid artery for the introduction of standard 2F Fogarty balloon embolectomy catheter used for this procedure. Before the surgery, check the level of isoflurane in the vaporizer to assure sufficient amount of the anesthetic that covers the entire surgery, and refill the vaporizer if necessary before each surgery starts. Check the pressure gauge of the oxygen tank to ensure sufficient oxygen for the surgery. To ensure a proper depth of anesthesia is reached, one can also monitor the respiratory rate of the rat. A normal rat has a respiratory rate of ~85 breath/min; an increase in respiratory depth and regular rhythm, and a decrease in respiratory rate signifies surgical anesthesia.
During the surgery, anesthesia must be continuously monitored and maintained by checking respiration, pedal reflex, and the response of the animal to the surgical stimulus. Of note, the surgeon should change gloves if his/her hands touch non-sterile parts of the animal during the surgery such as checking pedal reflex to prevent potential contamination and infection. In addition, the surgeon and the assistant should also carefully monitor the vital signs of the animal during the operation to prevent anesthetic overdose and take quick corrective actions to avert the situation if it occurs by turning off the isoflurane and increase oxygen level to 100%. The signs of anesthetic overdose include slow and shallow respirations, and weak and irregular pulse. We also suggest a good preparation of arteriotomy before the balloon injury including a complete separation of carotid artery sections from their adjacent tissues. This will help to avoid unexpected bleedings during the operation.
The animal under anesthesia, including during the post-surgical recovery phase, should never be left unattended. As an alternative to the regular buprenorphine (0.03 mg/kg b.i.d.) during the 48 hr post-surgery period, sustained-release buprenorphine (buprenorphine SR) can also be used. Injection of buprenorphine SR (1.0-1.2 mg/kg) immediately after the surgery will cover the post-surgical period up to 72 hr. In general, postsurgical complications are not anticipated; but a careful monitory practice ensures the welfare of the animals and is a policy by the institutional ACUC.
Because it is most important to reproduce the same level of the balloon injury in the study group of animals (8-15), adequate surgical skills and familiarity with the procedure are highly recommended. For researchers new to this procedure, we suggest first to practice the entire procedure including the preparation of arteriotomy, the balloon injury and the wound closure on rat carcasses that may be available in the institutional animal facility. Practicing the surgical procedure with veterinarians or researchers familiar with the procedure also ensures to obtain reproducible and consistent balloon injury results.
In our view, drugs and/or therapeutics that negate neointimal growth are best administered immediately after balloon injury in order to offset the inflammation at early stage. Some reagents may need to be administered continually during the subsequent days of recovery to achieve the effect. An early intervention helps to block or reduce the ensuing vascular remodeling13,22,24.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The work was supported by the intramural research program of the NIH, National Institute on Aging, and by a Priority Research Centers Program grant from the National Research Foundation (NRF-2009-0093812) funded by the Ministry of Science, Information and Communication Technology, & Future Planning, the Republic of Korea (H.T.). We thank Dr. Han-sol Park for putting “material” part together for the manuscript.
Materials
| 2 F Fogarty balloon embolectomy catheter | Edwards Lifesciences |
| Standard scalpel | Fine Science Tools |
| Small curved forceps (Large radius Dumont#7shanks curved) | Fine Science Tools |
| Large, medium and small micro-scissors | Roboz |
| Needles (20 G) | TycoHealthcare |
| Micro-surgery forceps with micro-blunted atraumatic tips | Fine Science Tools |
| Atraumatic straight small arterial clamps | Fine Science Tools |
| Retractor with maximum spread 5.5 cm long blunt teeth | Fine Science Tools |
| Silk suture (4.0 and 6.0 ) | Fine Science Tools |
| Syringe (1.0 ml) | BD |
| Curity gauze sponges | AllegroMedical |
| Cotton tip applicators sterile and non-sterile | Puritan Medical Products |
| Compact hot bead sterilizer | Fine Science Tools |
| Self-regulating heating pad | Fine Science Tools |
| ADS200 anesthesia system/ventilator | Paragon Medical |
| Isoflurane (forane), liquid form | Baxter |
| Sodium chloride 0.9% (Saline) | Hospira |
| Buprenex (buprenorphine) | Reckitt Benckiser Healthcare (UK) Ltd. |
| 70% alcohol | Fisher |
| 1: 10 Betadine | Fisher |
References
- Chaabane, C., Otsuka, F., Virmani, R., Bochaton-Piallat, M. L. Biological responses in stented arteries. Cardiovasc Res. 99, 353-363 (2013).
- Goel, S. A., Guo, L. W., Liu, B., Kent, K. C. Mechanisms of post-intervention arterial remodelling. Cardiovasc Res. 96, 363-371 (2012).
- Khan, R., Agrotis, A., Bobik, A. Understanding the role of transforming growth factor-beta1 in intimal thickening after vascular injury. Cardiovasc Res. 74, 223-234 (2007).
- Schillinger, M., Minar, E. Restenosis after percutaneous angioplasty: the role of vascular inflammation. Vasc Health Risk Manag. 1, 73-78 (2005).
- Katz, G., Harchandani, B., Shah, B. Drug-eluting stents: the past, present, and future. Curr Atheroscler Rep. 17, 485 (2015).
- Jain, M., Singh, A., Singh, V., Barthwal, M. K. Involvement of Interleukin-1 Receptor-Associated Kinase-1 in Vascular Smooth Muscle Cell Proliferation and Neointimal Formation After Rat Carotid Injury. Arterioscler Thromb Vasc Biol. , (2015).
- Lee, K. P., et al. Carvacrol inhibits atherosclerotic neointima formation by downregulating reactive oxygen species production in vascular smooth muscle cells. Atherosclerosis. 240, 367-373 (2015).
- Li, G., Chen, S. J., Oparil, S., Chen, Y. F., Thompson, J. A. Direct in vivo evidence demonstrating neointimal migration of adventitial fibroblasts after balloon injury of rat carotid arteries. Circulation. 101 (12), 1362-1365 (2000).
- Noda, T., et al. New endoplasmic reticulum stress regulator, gipie, regulates the survival of vascular smooth muscle cells and the neointima formation after vascular injury. Arterioscler Thromb Vasc Biol. 35 (5), 1246-1253 (2015).
- Deuse, T., et al. Dichloroacetate prevents restenosis in preclinical animal models of vessel injury. Nature. 509 (7502), 641-644 (2014).
- Guo, J., et al. p55gamma functional mimetic peptide N24 blocks vascular proliferative disorders. J Mol Med (Berl). , (2015).
- Oh, C. J., et al. Dimethylfumarate attenuates restenosis after acute vascular injury by cell-specific and Nrf2-dependent mechanisms. Redox Biol. 2, 855-864 (2014).
- Tae, H. J., et al. The N-glycoform of sRAGE is the key determinant for its therapeutic efficacy to attenuate injury-elicited arterial inflammation and neointimal growth. J Mol Med (Berl. 91 (12), 1369-1381 (2013).
- Zhou, Z., et al. Receptor for AGE (RAGE) mediates neointimal formation in response to arterial injury. Circulation. 107 (17), 2238-2243 (2003).
- Lindner, V., Fingerle, J., Reidy, M. A. Mouse model of arterial injury. Circ Res. 73 (5), 792-796 (1993).
- Le, V., Johnson, C. G., Lee, J. D., Baker, A. B. Murine model of femoral artery wire injury with implantation of a perivascular drug delivery patch. J Vis Exp. (96), e52403 (2015).
- Iannaccone, P. M., Jacob, H. J. Rats! . Dis Model Mech. 2 (5-6), 206-210 (2009).
- Fu, Y., Sander, J. D., Reyon, D., Cascio, V. M., Joung, J. K. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat Biotechnol. 32 (3), 279-284 (2014).
- Sander, J. D., Joung, J. K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat Biotechnol. 32 (4), 347-355 (2014).
- Tulis, D. A. Rat carotid artery balloon injury model. Methods Mol Med. 139, 1-30 (2007).
- Zhang, W., Trebak, M. Vascular balloon injury and intraluminal administration in rat carotid artery. J Vis Exp. (94), (2014).
- Tae, H. J., et al. Vessel ultrasound sonographic assessment of soluble receptor for advanced glycation end products efficacy in a rat balloon injury model. Curr Ther Res Clin Exp. 76, 110-115 (2014).
- Tae, H. J., et al. Chronic treatment with a broad-spectrum metalloproteinase inhibitor, doxycycline, prevents the development of spontaneous aortic lesions in a mouse model of vascular Ehlers-Danlos syndrome. J Pharmacol Exp Ther. 343 (1), 246-251 (2012).
- Sakaguchi, T., et al. Central role of RAGE-dependent neointimal expansion in arterial restenosis. J Clin Invest. 111 (7), 959-972 (2003).

