Measuring Biomolecular DSC Profiles with Thermolabile Ligands to Rapidly Characterize Folding and Binding Interactions
Summary
We present a protocol for rapid characterization of biomolecular folding and binding interactions with thermolabile ligands using differential scanning calorimetry.
Abstract
Differential scanning calorimetry (DSC) is a powerful technique for quantifying thermodynamic parameters governing biomolecular folding and binding interactions. This information is critical in the design of new pharmaceutical compounds. However, many pharmaceutically relevant ligands are chemically unstable at the high temperatures used in DSC analyses. Thus, measuring binding interactions is challenging because the concentrations of ligands and thermally-converted products are constantly changing within the calorimeter cell. Here, we present a protocol using thermolabile ligands and DSC for rapidly obtaining thermodynamic and kinetic information on the folding, binding, and ligand conversion processes. We have applied our method to the DNA aptamer MN4 that binds to the thermolabile ligand cocaine. Using a new global fitting analysis that accounts for thermolabile ligand conversion, the complete set of folding and binding parameters are obtained from a pair of DSC experiments. In addition, we show that the rate constant for thermolabile ligand conversion may be obtained with only one supplementary DSC dataset. The guidelines for identifying and analyzing data from several more complicated scenarios are presented, including irreversible aggregation of the biomolecule, slow folding, slow binding, and rapid depletion of the thermolabile ligand.
Introduction
Differential scanning calorimetry (DSC) is a powerful method for quantitating biomolecular binding and folding interactions1,2,3. The strengths of DSC include its ability to elucidate binding and folding mechanisms, and to yield the corresponding thermodynamic parameters2,3. Furthermore, DSC can be performed in solution under near-physiological conditions and does not require labeling of the biomolecule or ligand, e.g., with fluorophores, spin-labels or nuclear isotopes4. The instrument scans in temperature, measuring the amount of heat required to denature the biomolecule in the presence and absence of ligand. The resulting thermograms are used to extract the thermodynamic parameters governing the ligand binding and folding processes. The information provided by DSC or other thermodynamic techniques is critical to guiding the design of drugs targeting biomolecules1,5,6,7,8. However, the repeated scanning to high temperatures (~ 60 – 100 °C) can be problematic. For example, many pharmaceutically important compounds undergo rearrangement or decomposition upon sustained exposure to high temperatures9,10,11, i.e., they are thermolabile. Examination of binding interactions by DSC typically requires multiple forward and reverse scans in order to verify the reproducibility of the thermogram for thermodynamic analyses12. Thermal conversion of an initial ligand to a secondary form with altered binding characteristics leads to pronounced differences in the shape and position of successive thermograms, since the concentration of the initial ligand decreases with each scan while the thermal conversion products accumulate. These datasets are not amenable to traditional analyses.
We have recently developed a global fitting method for thermolabile ligand DSC datasets that yields the complete set of thermodynamic parameters governing the biomolecular folding and binding interactions from a single ligand-bound experiment referenced to the requisite thermogram for the free biomolecule4. The analysis reduces the experimental time and sample required by ~ 10-fold compared to standard DSC approaches. We have accounted for ligand thermal conversion by assuming this happens during the high temperature portion of each scan where the thermogram does not depend on ligand concentration. Therefore, the ligand concentration is a constant within the portion of the thermogram that is used to extract thermodynamic parameters. We additionally demonstrated how the rate constant for ligand thermal conversion can be obtained by performing one supplementary experiment with a longer high temperature equilibration period. For systems where ligand thermal conversion is less temperature-dependent (i.e., occurring appreciably at all temperatures), the analysis can be modified to include variable ligand concentrations. Here we demonstrate this procedure for the DNA aptamer MN4 in the presence of the thermolabile ligand cocaine, which rapidly converts to benzoylecgonine at high temperatures (>60 °C). Quinine is used as a negative control for ligand thermolability since it does not undergo conversion at these experimental temperatures and also binds to MN4. We describe the acquisition of thermolabile ligand DSC datasets and their analysis yielding thermodynamic and kinetic parameters of the folding, binding, and ligand conversion processes.
Protocol
1. Sample Preparation
- Purify the desired biomolecule13.
NOTE: This protocol uses purchased cocaine-binding DNA aptamer MN4 after exchanging against 2 M NaCl three times followed by three rounds of deionized water using a centrifugal filter with a 3 kDa molecular weight cut-off membrane. - Synthesize and purify, or purchase the desired thermolabile ligand13.
NOTE: MN4 binds the thermolabile ligand cocaine. MN4 also binds quinine, which is used as a negative control for ligand thermolability at these experimental temperatures. - Prepare buffers for dialysis of the purified biomolecule and dissolution of ligands (20 mM sodium phosphate and 140 mM NaCl buffer, pH 7.4, for MN4 and the ligands used here).
- Dialyze the biomolecule against at least 2 L of buffer using dialysis tubing with 0.5 – 1.0 kDa cut-off.
- Filter the final buffer (referred to as working buffer) through a 0.2 µm filter that has been thoroughly equilibrated with buffer.
- Weigh out the desired masses of the ligands and dissolve them in filtered working buffer. If the desired ligand concentrations require masses that are too small to accurately weigh, make a concentrated ligand stock solution (10x for example).
NOTE: It is crucial that all DSC experiments utilize the same working buffer for the sample and ligand, i.e., never perform an experiment where the ligand is dissolved in a different batch of working buffer than the biomolecule as this will cause buffer mismatch artifacts in the data. - Filter the biomolecule stock solution through a 0.2 µm filter that has been thoroughly equilibrated with working buffer.
- Determine the biomolecule concentration by absorbance measurements (260 nm for nucleic acids like MN4 and 280 nm for proteins).
- Store the prepared biomolecule and ligand in a 4 °C refrigerator (suitable for MN4 and the ligands used here), or at -20 or -80 °C if the biomolecule and ligands tolerate freezing and long-term storage is required. Degas the buffer, biomolecule, and ligand solutions in a table top degasser (see Table of Materials) prior to loading into the DSC.
NOTE: Degassing helps to prevent bubble formation in the DSC at higher temperatures. Bubbles cause signal artifacts that obscure DSC peak shapes and baselines.
2. DSC Preparation
- Unscrew the pressure handle from the DSC (see Table of Materials).
- Run silicon tubing from the working buffer and attach it to the front flange (metal opening) of the reference capillary.
- Create a bridge between the reference and sample capillaries by connecting the rear reference flange to the front sample flange.
- Attach a piece of silicon tubing to the rear sample flange that runs to a waste flask with a vacuum line attached.
- Turn on the vacuum line to flush the DSC with 200 mL of working buffer.
- Load the reference capillary of the DSC with working buffer. Attach roughly 3 – 5 cm sections of silicon tubing to the reference capillary flanges.
- Insert a 1 mL pipette tip into the rear flange's silicon tubing. Draw 0.8 mL of working buffer with a pipette and insert the pipette tip with buffer into the front reference flange's silicon tubing.
- Gently press the pipette plunger down to pass the working buffer through the front silicon tubing into the reference capillary and up into the rear flange's attached pipette tip. Press down the pipette plunger until the working buffer level reaches just above the front silicon tubing, then release the pipette plunger until the working buffer level reaches just above the rear silicon tubing.
- Repeat passing the working buffer back and forth to purge the volume in the reference capillary of bubbles.
NOTE: Usually, 10 passes of the solution back and forth are sufficient to clear any bubbles.
- Repeat passing the working buffer back and forth to purge the volume in the reference capillary of bubbles.
- Cap the rear pipette tip with the thumb and gently pull up on the rear pipette tip and front pipette to remove them from the reference flanges with the silicon tubing attached.
- Load the sample capillary with working buffer as in steps 2.6 – 2.9. Place a black plastic cap on the rear reference and sample flanges, leaving the front flanges uncovered.
- Attach the pressure handle.
- Open the DSC software (see Table of Materials) and pressurize the instrument by clicking the red up arrow at the top of the interface once the power reading has stabilized; the DSC power is indicated in a box at the top right of the interface along with the instrument temperature and pressure reading.
NOTE: Monitor the power reading as the DSC pressurizes. Changes in power of more than ~ 10 µW indicate bubble formation in the capillaries, which can cause artifacts in the data. The solutions must be removed and degassed further before continuing. - Equilibrate the DSC with working buffer by performing a forward and reverse scan. In the "Experimental Method" tab on the left side of the screen, ensure that the "Scanning" option is selected to run the DSC in temperature scanning mode.
NOTE: Experimental parameters are the temperature scanning range, scan rate, low and high temperature equilibration time, and number of scans.- In the "Temperature Parameters" inset under the "Experimental Method" tab, click the button for "Heating". Enter 1 and 100 °C for the lower and upper experimental temperatures, 1 °C/min for the scan rate, and 60 s for the equilibration period.
- Click the "Add Series" button under the input field for the equilibration period. Enter 2 into the "Steps to add" field in the popup window (for one heating and one cooling scan) and check the "Alternate Heating/Cooling" box. Click "OK"; the added scans appear in the lower portion of the interface. Check that the parameters for each scan are as desired.
- Start the experiment by clicking the green "play" button at the top of the interface. Navigate to the desired folder and input a file name for saving the experiment in the popup window. View the experiment progress by clicking the "Data" tab to the right of the "Experiment Method" tab.
3. Collecting Thermolabile Ligand DSC Datasets
NOTE: The minimal procedure consists of five experiments: buffer reference experiments with and without ligand (used for baseline subtraction, see Discussion), sample experiments with the free biomolecule, the ligand-bound biomolecule, and the ligand-bound biomolecule with a longer high temperature equilibration period.
- Run reference experiments for baseline subtraction of the sample data. Reload the DSC with working buffer in both capillaries and collect multiple forward and reverse scans over a suitable temperature range at 1 °C min-1 with an upper (high temperature) equilibration time of 120 s.
- Delete the previous buffer equilibration scans from the lower portion of the interface by highlighting each individually and clicking the red X to the middle right of the interface. Add the new scans by clicking the "Add Series" button, entering 20 in the field for "Steps to add", and checking the "Alternate Heating/Cooling" box. Click OK and run the experiment by clicking the green play button as above.
- Repeat steps 3.1 – 3.1.1 with working buffer containing the desired concentration of ligand in both capillaries to obtain the reference experiments for the ligand (collect two separate experiments using 120 s and 600 s high temperature equilibration times respectively, to be used in acquiring the rate constant for thermolabile ligand conversion).
NOTE: The scan rate used here ensures that the biomolecule in subsequent experiments is at thermal equilibrium in the forward and reverse scans (see Discussion). Scan rates < 0.1 – 0.2 °C min-1 lead to noisy thermograms and are not applicable in DSC experiments. The temperatures should extend from well below the melting temperature of the free biomolecule to well above the melting temperature of ligand-saturated biomolecule (~ 20 – 80 °C for MN4). Verify reproducibility of the scans (for example, 10 forward and 10 reverse scans for 20 total is sufficient). - If using multiple ligands (such as cocaine and quinine), flush the DSC with 200 mL of working buffer (repeat from step 2.5) between runs in order to remove ligand from the capillaries and prevent cross-contamination.
NOTE: It is helpful to perform a replicate experiment on the free biomolecule after a ligand-bound run in order to check if the previous ligand adsorbs strongly to the capillary walls and is not adequately removed with buffer flushing. If the thermograms for the free biomolecule appear to be shifted to a larger magnitude and higher denaturation temperature after the ligand-bound experiment, it is likely that the previous ligand is still present in the calorimeter after flushing. Remove the adsorbed ligand by incubating the capillaries with 20% Contrad-70 for 1 h at 60 °C with the plastic caps and pressure handle off. In the DSC software, change the experimental mode to "Isothermal" under the Experimental Method tab. Choose 3,600 s for the duration and 60 °C for the isothermal temperature, with zero entered for the equilibration time. Click "Add to Experimental Method". The isothermal experiment appears in the lower part of the screen. After completion, flush the instrument with 2 L deionized water and repeat from step 2.5.
- Run sample experiments using the same DSC loading procedure and experimental parameters as the reference scans. For the free biomolecule data set, ensure that the reference capillary contains the working buffer while the sample capillary contains the free biomolecule at the desired concentration in working buffer.
- For the ligand-bound experiments, ensure that the ligand is in the working buffer in the reference capillary, and the biomolecule plus ligand are in working buffer in the sample capillary. Flush the system between additions of different ligands as in step 2.5.
- Perform one additional experiment with the biomolecule bound to the thermolabile ligand where the high temperature equilibration period is increased to 600 s and all other experimental parameters are the same as step 3.2.1.
NOTE: The duration of the high temperature equilibration period for the second ligand-bound experiment is simply chosen to ensure that the ligand is more rapidly depleted than the short equilibration time experiment. If the ligand-bound peaks from the first experiment decay slowly as a function of scan number (e.g., the differences in successive peak maxima are ≤ 0.5 °C), estimate 10- to 20-fold increases in the high temperature equilibration period in order to adequately deplete the ligand during the second experiment. The accurate calculation of the rate constant for ligand conversion requires that the second experiment has swifter depletion of the ligand relative to the first. The ligand concentrations extracted from the global analysis of the two experiments will be similar and therefore unusable if the ligand depletion is not sufficiently accelerated in the second experiment.
4. Data Processing
- Open the DSC experiment files in the DSC data analysis software (see Table of Materials) and export the raw power data as spreadsheets.
- Import the spreadsheets containing the raw power data into software for data fitting.
- Baseline subtract the sample data by subtracting the buffer power data from the free and ligand-bound biomolecule experiments.
NOTE: For the thermolabile ligand experiment, the concentration of initial ligand is decreasing with each scan. Therefore, it is ideal to subtract the buffer scan 1 from sample scan 1, and so on. We have found that the buffer scans with cocaine do not change appreciably as the ligand conversion proceeds and therefore a single thermolabile ligand buffer scan can be used to subtract all of the thermolabile ligand-bound data. - Convert the baseline-subtracted sample power data to heat capacity.
NOTE: The conversion requires the biomolecule's partial specific volume, which can be estimated14,15,16. The equation for converting power to heat capacity has been described previously17.
5. Data Analysis
- Globally fit the short equilibration time thermolabile ligand-bound heat capacity dataset with a single set of baseline, ligand concentration, folding, and ligand binding parameters as described previously4.
- Repeat the global fit for the long equilibration time dataset in order to calculate the rate constant for thermolabile ligand conversion as described previously4.
Representative Results
Representative data for the thermolabile ligand DSC are shown in Figure 1. The position and height of the thermolabile ligand-bound peak successively shifts down towards that of the unbound biomolecule as the thermolabile ligand is depleted with each scan (Figure 1a). The free denaturation profile is used as a reference for the endpoint of thermolabile ligand conversion (Figure 1b). Data for MN4 bound to quinine are shown as a negative control for thermolabile ligand conversion (Figure 1b). The final thermolabile ligand scans have slightly higher transition midpoints and peak heights relative to the unbound MN4, indicating the thermolabile ligand conversion product (benzoylecgonine) has a weak affinity for MN4. The thermodynamic parameters resulting from global analysis of the datasets in Figure 1 are listed in Table 1. The folding parameters for MN4 in the presence of cocaine or quinine are identical within error, as expected since dilute small molecules are not expected to perturb the folding thermodynamics of the free biomolecule. The binding parameters are in good agreement with those from isothermal titration calorimetry (ITC)18 and reveal that MN4's preference for quinine over cocaine is driven by a more favorable binding enthalpy. In Figure 2, increasing the high temperature equilibration period yields more pronounced reductions of the thermolabile ligand concentration with each scan relative to the short equilibration period dataset. Using the optimized global fit concentration parameters from the two datasets, the rate constant for ligand conversion at the high equilibration temperature is calculated from the slope of the line in the Figure 2 inset.
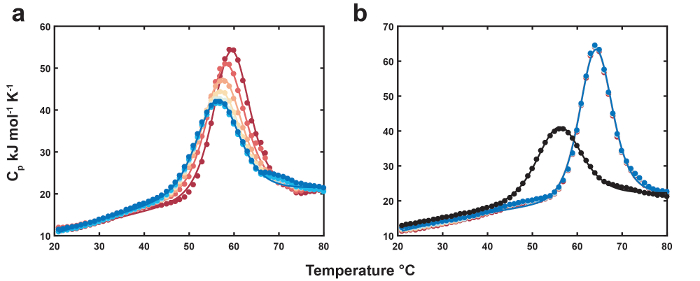
Figure 1. Thermolabile ligand DSC. (a) Thermograms of MN4 (83 µM) bound to cocaine (initial concentration 778 µM). First and last scans of MN4 in the presence of ligand are shown as dark red and blue circles while the corresponding fits are shown as colored lines. (b) Thermograms of free MN4 (83 µM, black circles) and successive scans bound to quinine (880 µM, colored circles). Fits to the free and quinine-bound datasets are shown as black and colored lines, respectively. Reproduced from reference4 with permission from the Royal Society of Chemistry. Please click here to view a larger version of this figure.
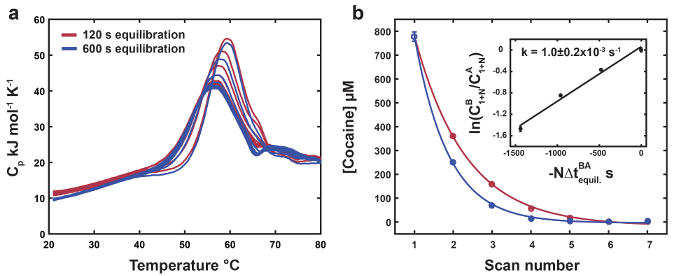
Figure 2. Measuring the Rate Constant for Thermolabile Ligand Conversion. (a) Sets of thermograms for MN4 bound to cocaine with short (120 s, dark red) and long (600 s, dark blue) equilibration times at 80 °C, respectively. (b) Concentrations of cocaine extracted from global analysis of the datasets in (a) as a function of scan number. Experimental points and exponential fits are shown as colored circles and lines respectively. The inset shows a linear fit to the previously described Supplementary Eq. 19 from Harkness et al. using the optimized global fit cocaine concentrations for the two datasets4. The rate constant for ligand thermal conversion at 80 °C is calculated as the slope of the line. The error for the rate constant for thermolabile ligand conversion is given as ± two standard deviations. Reproduced from reference4 with permission from the Royal Society of Chemistry. Please click here to view a larger version of this figure.
| Fit parameters | Cocaine added | Quinine added |
| ∆HUF | 271.3±1.8 | 272.5±4.0 |
| ∆SUF | 824.4±5.1 | 827.9±10.9 |
| a∆GUF | 21.6±0.2 | 21.6±0.9 |
| a∆HB1F | -75.2±1.6 | -101.0±4.0 |
| a∆SB1F | -154.2±5.0 | -213.7±12.0 |
| ∆CpB1F | -1.5±0.1 | -1.2±0.1 |
| a∆GB1F | -28.5±0.2 | -36.2±0.7 |
| a∆HB2F | -33.7±1.8 | – |
| a∆SB2F | -49.9±5.2 | – |
| ∆CpB2F | -2.2±0.1 | – |
| a∆GB2F | -18.6±0.3 | – |
Table 1. Thermodynamic Parameters Extracted from Global Analysis of the MN4 DSC Datasets Using Cocaine and Quinine Ligands. Parameters were calculated at 30 °C. B1F refers to cocaine- or quinine-bound folded states and B2F refers to the benzoylecgonine-bound folded state. ΔH and ΔG are expressed in kJ/mol, ΔS is expressed in J/mol/K and ΔCp is expressed in kJ/mol/K. Errors were calculated according to the variance/co-variance method19.
Discussion
Modifications and troubleshooting
The details of the global fitting analysis used in Figure 1 and Figure 2 have been described previously4. Here, we outline practical aspects of performing and analyzing DSC binding experiments with thermolabile ligands. Note that a DSC baseline obtained for the thermolabile ligand alone is subtracted from the ligand + biomolecule dataset, effectively cancelling out the heat released or absorbed by the thermal conversion process itself. The standard thermolabile ligand global fitting analysis (Figure 1 and Figure 2) assumes that the system is at thermodynamic equilibrium throughout the temperature scan and that the thermolabile ligand concentration is constant throughout each thermogram, decreasing exclusively during the high temperature equilibration period. We have previously shown that this assumption applies to cocaine-bound MN4 and is expected to hold for any thermolabile ligand/biomolecule system with kinetics similar to these.
There are, however, some situations in which the system cannot be assumed to be at thermodynamic equilibrium and/or the concentration of ligand cannot be considered constant throughout a single scan. These include i) when the ligand thermally converts rapidly relative to the temperature scan rate, ii) when the biomolecule undergoes irreversible aggregation at high temperature, iii) when the folding/unfolding rates are slow compared to the scan rate, and iv) when the ligand association/dissociation rates are slow compared to the scan rate. In these cases, the system is under kinetic rather than thermodynamic control and the analysis given in reference 4 cannot strictly be applied. Data may be simulated quantitatively following Figure 3, as described in the Supplementary File 1. In principle, these kinetics-based calculations could be used to fit non-equilibrium DSC data, potentially yielding both kinetic and thermodynamic data, however this analysis is beyond the scope of this paper. Instead, we present some representative simulated DSC data to assist the reader in identifying non-equilibrium situations.
An ideal example of thermodynamic control is shown in Figure 4a, b. DSC thermograms of the free biomolecule are superimposable (Figure 4a) and scans with the thermolabile ligand do not show hysteresis, such that the melting temperature observed on the up-scan matches the folding temperature of the previous down-scan (Figure 4b). When the thermolabile ligand converts rapidly compared to the scan rate, large distortions appear in the thermogram and the thermodynamic equations do not account for the peak shape, as shown in Figure 4c, d. This can be alleviated somewhat by increasing the scan rate. When the biomolecule aggregates in a temperature-dependent manner, DSC traces for the free biomolecule show successive decreases in magnitude (Figure 4e), while addition of the thermolabile ligand produces a pattern of decreasing thermal upshifts similar to the ideal case, but scaled by the decreasing biomolecule concentration (Figure 4f). When folding/unfolding kinetics are slow compared to the scan rate, hysteresis is apparent in DSC traces of the free biomolecule such that the apparent denaturation temperature on the up-scan is higher than the apparent renaturation temperature on the down-scan (Figure 4g). Addition of a thermolabile ligand leads to the familiar pattern of decreasing thermal upshifts, particularly for the up-scans (Figure 4h). Finally, systems with rapid folding and slow binding produce hysteresis-free DSC thermograms for the free biomolecule (Figure 4i), however data with the thermolabile ligand show hysteresis where the apparent melting temperature of the up-scan is higher than the apparent folding temperature of the previous down-scan (Figure 4j). Nevertheless, the typical pattern of decreasing thermal upshifts are apparent in both up-scans and down-scans. Non-equilibrium behavior in the case of slow folding or binding kinetics can be alleviated somewhat by decreasing the scan rate, although this runs the risk of non-negligible ligand thermal conversion occurring throughout the scan. In practice, the scan rate and upper equilibration temperature can be adjusted manually to obtain data resembling Figure 4a, b.
Limitations of the technique
Our thermodynamic analysis for DSC binding experiments with thermolabile ligands requires that the folding and binding processes are relatively rapid and that thermolabile ligand conversion is slow prior to the high temperature portion of each scan. When the lifetime of the folded and/or bound state is greater than about 30 s (koff, ku < 0.03 s-1), hysteresis becomes discernable in scans performed at 1 °C min-1. Additionally, when the ligand conversion rate constant is above approximately kc = 10-4 s-1 before the denaturation transition, there can be significant depletion of the ligand during the course of a single scan. Application of our analysis is also inappropriate when irreversible aggregation occurs. In these cases, more advanced modelling could be applied to the data. No affinity information is available if ligand conversion is so rapid that it reaches completion prior to the first denaturation transition.
Significance with respect to existing methods
Our method for the first time allows DSC to be used to measure the binding thermodynamics of high affinity, thermolabile ligands. By performing a global simultaneous analysis of all scans, thermodynamic parameters are extracted with high accuracy20. An additional benefit is that the full dataset can be collected in as little as one experiment if the thermal conversion product has no affinity for the free biomolecule. In contrast, producing a typical experimental DSC series for a non-thermolabile ligand requires ~ 7-10 total experiments.
Future applications
This approach has direct applications to characterizing tight, thermolabile inhibitors in drug discovery campaigns. Several therapeutic compounds such as antibiotics and benzodiazepines are known to be thermolabile, undergoing rapid hydrolysis at or near physiological pH and temperatures of ~ 60-70 °C11. This DSC method is well positioned to identify and characterize many more. As well, modification of the fitting protocol to account for systems under kinetic rather than thermodynamic control, as discussed above, has the potential to open the door to many more systems of biological relevance.
Critical steps within the protocol
One of the most important experimental procedures to consider is dialysis or exchange of the biomolecule and ligand into identical working buffer solutions (protocol steps 1.3-1.6). Buffer mismatch between the ligand and biomolecule solutions can lead to large artifacts in the baseline and sample scans which completely obscure the relevant folding data. Additionally, it is essential that the power reading stabilizes before the DSC is pressurized so that it can be monitored during pressurization (protocol step 2.3). If the power reading changes by more than ~ 10 µW during pressurization, bubbles have likely formed in the capillaries and can cause large artifacts in the data. In this case, the solutions need to be degassed more thoroughly.
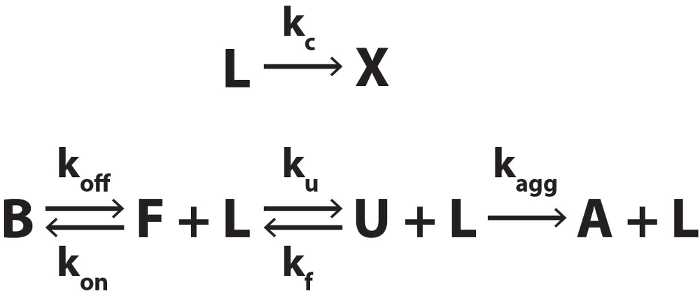
Figure 3. Biomolecular Folding, Binding to a Thermolabile Ligand, and Irreversible Aggregation. The thermolabile ligand (L) converts to product (X) with a rate constant kc. X has no affinity for the biomolecule. The bound state (B) of the biomolecule exchanges with the free folded state (F) with rate constants koff and kon. F exchanges with the unfolded state (U) with rate constants ku and kf. U irreversibly converts to the aggregated state (A) with the rate constant kagg. Please click here to view a larger version of this figure.
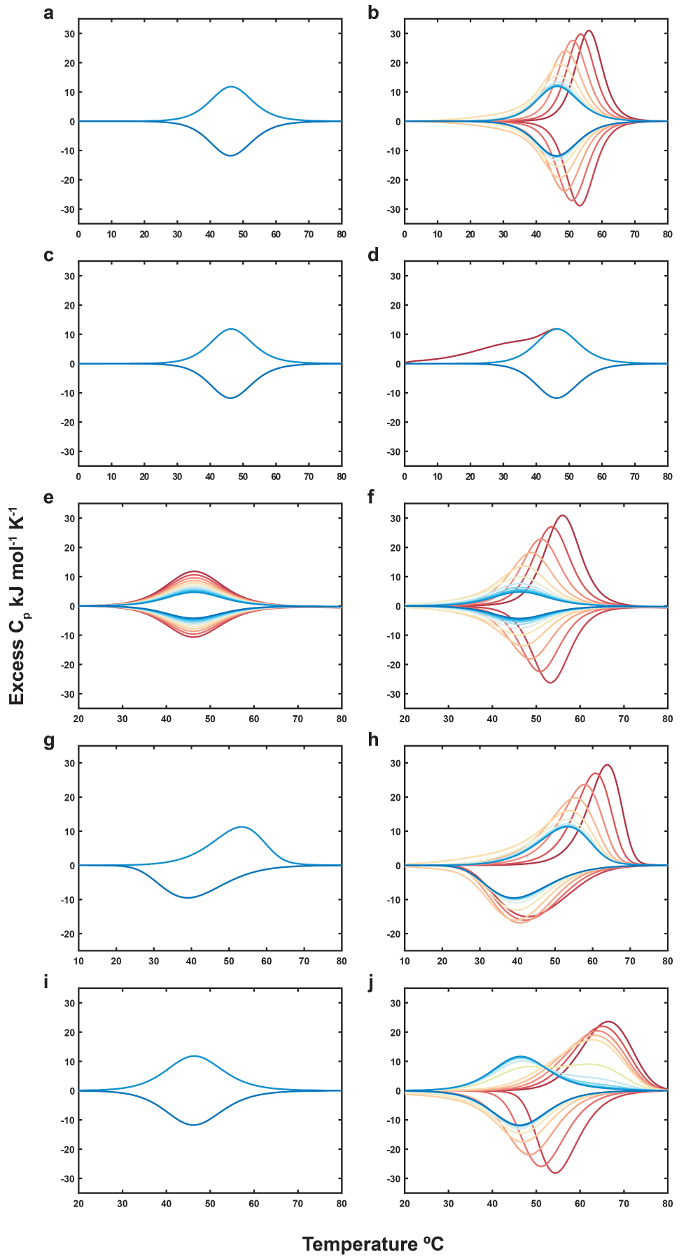
Figure 4. Computer Simulation of Equilibrium and Kinetically-controlled DSC Experiments in the Absence and Presence of a Thermolabile Ligand. (a) Equilibrium biomolecular folding. (b) Equilibrium folding, thermolabile ligand binding, and slow thermolabile ligand conversion. (c) Equilibrium biomolecular folding. (d) Equilibrium folding, thermolabile ligand binding, and fast thermolabile ligand conversion. (e) Equilibrium biomolecular folding and slow irreversible aggregation. (f) Equilibrium folding, binding, slow thermolabile ligand conversion, and slow irreversible aggregation. (g) Slow biomolecular folding. (h) Slow folding, equilibrium binding, and slow thermolabile ligand conversion. (i) Equilibrium biomolecular folding. (j) Equilibrium folding, slow thermolabile ligand binding, and slow thermolabile ligand conversion. In all panels, the first and last simulated scans are dark red and dark blue, respectively. Panels that show only light and dark blue thermograms indicate that all simulated scans overlay, and only the last two are visible in the plot. DSC experiments were simulated with 20 scans (10 melting and 10 annealing) at 1 °C min-1 scan rate, with a temperature range of 0-80 °C. Certain panels display narrower temperature ranges to allow better visualization of the simulation trends. The biomolecule and ligand concentrations in the simulations were 200 µM and 10 mM, respectively. Each experiment was simulated with a 600 s equilibration time at 0 °C before scanning and a 60 s equilibration time between each of the subsequent scans. Arrhenius parameters for equilibrium binding and folding were Aon = 5 x 10-1 M-1 s-1, Aoff = 1 x 1019 s-1, Eaon = -20 kJ mol-1, Eaoff = 120 kJ mol-1, Afold = 1 x 10-14 s-1, Aunfold = 5 x 1018, Eafold = -80 kJ mol-1, and Eaunfold = 120 kJ mol-1. Arrhenius parameters for kinetically-controlled binding and folding were Aon = 5 x 10-3 M-1 s-1, Aoff = 1 x 1016 s-1, Eaon = -20 kJ mol-1, Eaoff = 120 kJ mol-1, Afold = 1 x 10-16 s-1, Aunfold = 5 x 1016, Eafold = -80 kJ mol-1, and Eaunfold = 120 kJ mol-1. Arrhenius parameters for slow and rapid thermolabile ligand conversion were Aslow = 7.509 x 1010 s-1, Easlow = 94.65 kJ mol-1, and Afast = 1 s-1, Eafast = 10 kJ mol-1. Arrhenius parameters for slow irreversible aggregation were Aagg. = 5 x 107 s-1 and Eaagg. = 80 kJ mol-1. Excess heat capacities were calculated with ΔHUF (unfolding), ΔHBF (binding), and ΔHAF (aggregating) = 200, -140, and 50 kJ mol-1, respectively. The theoretical description of kinetically-controlled DSC experiments with thermolabile ligands and script for performing these simulations (and the necessary parameters) are available in Supplementary File 1. Please click here to view a larger version of this figure.
Disclosures
The authors have nothing to disclose.
Acknowledgements
R. W. H. V was supported by the McGill Natural Sciences and Engineering Research Council of Canada (NSERC) Training Program in Bionanomachines. A. K. M. and P. E. J. were supported by NSERC grants 327028-09 (A. K. M) and 238562 (P. E. J.).
Materials
| Sodium chloride | Chem Impex | #00829 | |
| Sodium phosphate monobasic dihydrate | Sigma Aldrich | 71502 | |
| Sodium phosphate dibasic | Sigma Aldrich | S9763 | |
| Deioinized water for molecular biology | Millipore | H20MB1001 | |
| 0.2 micron sterile syringe filters | VWR | CA28145-477 | |
| 3 kDa centrifugal filters | Millipore | UFC900324 | |
| Dialysis tubing 0.5-1.0 kDa cutoff | Spectrum Laboratories | 131048 | |
| Silicon tubing | VWR | 89068-474 | |
| Plastic DSC flange caps | TA Instruments | 6111 | |
| DNA aptamer MN4 | Integrated DNA Technologies | https://www.idtdna.com/site/order/menu | |
| Cocaine | Sigma Aldrich | C008 | |
| Quinine | Sigma Aldrich | 22620 | |
| NanoDSC-III microcalorimeter | TA Instruments | http://www.tainstruments.com/nanodsc/ | |
| DSCRun software | TA Instruments | http://www.tainstruments.com/support/software-downloads-support/instruments-by-software/ | |
| NanoAnalyze software | TA Instruments | http://www.tainstruments.com/support/software-downloads-support/instruments-by-software/ | |
| Contrad-70 | VWR | 89233-152 |
References
- Bruylants, G., Wouters, J., Michaux, C. Differential scanning calorimetry in life science: thermodynamics, stability, molecular recognition and application in drug design. Curr Med Chem. 12 (17), 2011-2020 (2005).
- Privalov, P. L., Dragan, A. I. Microcalorimetry of biological macromolecules. Biophys Chem. 126 (1-3), 16-24 (2007).
- Brandts, J. F., Lin, L. N. Study of strong to ultratight protein interactions using differential scanning calorimetry. 生物化学. 29 (29), 6927-6940 (1990).
- Harkness, R. W., Slavkovic, S., Johnson, P. E., Mittermaier, A. K. Rapid characterization of folding and binding interactions with thermolabile ligands by DSC. Chem Commun. 52 (92), 13471-13474 (2016).
- Garbett, N. C., Chaires, J. B. Thermodynamic studies for drug design and screening. Expert Opin Drug Dis. 7 (4), 299-314 (2012).
- Holdgate, G. A., Ward, W. H. J. Measurements of binding thermodynamics in drug discovery. Drug Discov Today. 10 (22), 1543-1550 (2005).
- Plotnikov, V., et al. An autosampling differential scanning calorimeter instrument for studying molecular interactions. Assay Drug Dev Technol. 1 (1), 83-90 (2002).
- Schon, A., Lam, S. Y., Freire, E. Thermodynamics-based drug design: strategies for inhibiting protein-protein interactions. Future Med Chem. 3 (9), 1129-1137 (2011).
- Periánez Parraga, L., G-L, A., Gamón Runnenberg, I., Seco Melantuche, R., Delgado Sánchez, O., Puigventós Latorre, F. Thermolabile Drugs. Operating Procedure In the Event of Cold Chain Failure. Farmacia Hospitalaria. 35 (4), 1-28 (2011).
- Murray, J. B., Alshora, H. I. Stability of Cocaine in Aqueous-Solution. J Clin Pharmacy. 3 (1), 1-6 (1978).
- Waterman, K. C., et al. Hydrolysis in pharmaceutical formulations. Pharm. Dev. Technol. 7 (2), 113-146 (2002).
- Mergny, J. L., Lacroix, L. Analysis of thermal melting curves. Oligonucleotides. 13 (6), 515-537 (2003).
- Neves, M. A., Reinstein, O., Johnson, P. E. Defining a stem length-dependent binding mechanism for the cocaine-binding aptamer. A combined NMR and calorimetry study. 生物化学. 49 (39), 8478-8487 (2010).
- Bonifacio, G. F., Brown, T., Conn, G. L., Lane, A. N. Comparison of the electrophoretic and hydrodynamic properties of DNA and RNA oligonucleotide duplexes. Biophys J. 73 (3), 1532-1538 (1997).
- Durchschlag, H., Hinz, H. -. J. Chapter 3. Thermodynamic Data for Biochemistry and Biotechnology. , 45-128 (1986).
- Hellman, L. M., Rodgers, D. W., Fried, M. G. Phenomenological partial-specific volumes for G-quadruplex DNAs. Eur Biophys J Biophy. 39 (3), 389-396 (2010).
- Farber, P., Darmawan, H., Sprules, T., Mittermaier, A. Analyzing Protein Folding Cooperativity by Differential Scanning Calorimetry and NMR Spectroscopy. J Am Chem Soc. 132 (17), 6214-6222 (2010).
- Reinstein, O., et al. Quinine binding by the cocaine-binding aptamer. Thermodynamic and hydrodynamic analysis of high-affinity binding of an off-target ligand. 生物化学. 52 (48), 8652-8662 (2013).
- Tellinghuisen, J. Statistical error propagation. J Phys Chem. A. 105 (15), 3917-3921 (2001).
- Drobnak, I., Vesnaver, G., Lah, J. Model-based thermodynamic analysis of reversible unfolding processes. J Phys Chem B. 114 (26), 8713-8722 (2010).

