Summary
Here, we present a protocol on the use of intraoperative ultrasound in spinal surgery, particularly in cases of intradural lesions and lesions in the ventral spinal canal when using a posterior approach.
Abstract
Since the 1980s, there have been several reports for the use of intraoperative ultrasound as a useful adjunct in spinal surgery. However, with the advent of newer cutting-edge imaging modalities, the use of intraoperative ultrasound in spine surgery has largely fallen out of favor. Despite this, intraoperative ultrasound continues to provide several advantages over other intraoperative techniques such as magnetic resonance imaging and computed tomography including being more cost-effective, efficient, and easy to operate and interpret. Additionally, it remains the only method for the real-time visualization of soft tissue and pathologies. This paper focuses on the advantages of using intraoperative ultrasound, especially in cases of intradural lesions and lesions ventral to the thecal sac when approaching posteriorly.
Introduction
Ultrasound is one of the most common diagnostic tools in medicine, particularly for visualizing pathology in the abdomen, limbs, and neck. However, its use to investigate cranial and spinal lesions is not currently widely utilized. In 1978, Reid was the first to report the use of ultrasound to visualize cervical cord cystic astrocytoma1. Here, scans were performed with the patient's neck flexed to allow opening of the intralaminar window. Four years later, in 1982, Dohrmann and Rubin reported the use of ultrasound intraoperatively to visualize the intradural space in 10 patients2. Pathologies identified with intraoperative ultrasound among the 10 patients included syringomyelia, spinal cord cysts, and intramedullary and extramedullary tumors. They further demonstrated the use of intraoperative ultrasound to guide catheters and probes for biopsy of tumors, drainage of cysts, and ventricular shunt catheter placement3. This allowed the real-time monitoring and precise positioning of probes/catheters, reducing inaccuracy and errors in placement. Following these initial reports, several others have published the use of intraoperative ultrasound for guiding spinal cord cyst drainage, intramedullary and extramedullary tumor resection, and syringo-subarachnoid shunt catheter placement4,5,6,7,8,9,10. Additionally, it has been shown to also increase rate of complete resection of intra-axial solid brain tumors and spinal intradural tumors11,12. Intraoperative ultrasound has also proven to be useful for the intraoperative surgical planning before manipulation of the tissue and subsequent visualization of adequate neural element decompression in patients with spine fractures7,9,13,14,15.
With the advent of newer intraoperative technology allowing clearer visualization of soft tissues, such as magnetic resonance imaging (MRI) and computed tomography (CT), intraoperative ultrasound has become less common and a less favored intraoperative imaging modality among neurosurgeons today16. However, intraoperative ultrasound can have advantages over these newer technologies in certain operative cases (Table 1). Intraoperative ultrasound has shown to demonstrate better soft tissue visualization of intradural structures when compared with intraoperative CT (iCT) or cone-beam CT (cbCT)9,17. While intraoperative MRI (iMRI) is useful where available because of the higher soft tissue resolution it provides, it is costly, time consuming and does not provide real-time images6, 16,18. An example is in the circumstance of an intradural mass ventral to the thecal sac that the surgeon is unable to directly visualize. Additionally, despite being operator dependent, from our experience, intraoperative ultrasound is fairly simple to use and can be easily read without a radiologist.
Protocol
The protocol illustrated here follows the guidelines of the human research ethics committee at Brigham and Women's Hospital.
1. Preoperative Protocol
- Assess patients with spinal pathology in clinic and determine eligibility for spinal surgery. Perform neurological assessment and obtain CT or MRI scan to identify spinal lesion.
- Include patients who have an intradural pathology such as schwannoma, ependymoma, meningioma, astrocytoma, etc.; or patients who have a ventral compressive extradural pathology, such as a ventral thoracic herniated disc, fracture fragments ventrally, or a spinal bony tumor with ventral compression.
NOTE: Pathology is determined by spinal imaging with CT or MRI. Exclusion criteria includes the patients who cannot tolerate surgery, or patients with an extremely poor prognosis.
2. Preparation for Surgery
- Do not allow the patient to consume anything by mouth after the midnight before surgery.
NOTE: The patient will be placed under general anesthesia and intubated by the anesthesiologist. - Position patient with their back exposed according to surgeon's preference for the spinal surgery.
- Sterilize the surgical area with povidone-iodine by scrubbing the area.
3. Surgery
Note: This section of the protocol follows general spine surgery techniques that can be referenced from any reputable spine surgery technique textbook19.
- Make an incision with a scalpel along the length of the spine over the appropriate vertebrae levels and continue to make a straight incision down until the bone is reached.
NOTE: The size of the incision will depend on the size of the pathology. For example, if the tumor spans two vertebral levels, then at least two vertebral levels will need to be exposed. When the bone is exposed, an X-ray with a portable X-ray machine can be performed to verify the correct vertebrae. - Perform subperiosteal dissection by electrosurgical cautery and expose the spinous process which is visualized as a bulbous bony process. Turn the cutting edge ventrally and sweep across the laminar.
- Use a combination of a Leksell bone plier and a highspeed drill to remove the bony lamina and spinous process to expose the ligamentum flavum underneath.
- Use an angled Curette and Kerrison bone punch to remove the ligamentum flavum to reveal the dura mater underneath.
- Use bipolar and hemostatic matrix to achieve hemostasis.
NOTE: The success of a good ultrasound image relies upon a clean surgical field.
4. Intraoperative Ultrasound
- Use a mobile ultrasound machine and a transducer probe with a 20 mm diameter.
NOTE: The probe should have a 10 to 4.4 MHz frequency range. Any comparable device with similar probe diameter and frequency range should suffice. - After the bony removal and dura exposure, fill the surgical field with sufficient saline solution such that the ultrasound transducer probe can be submerged.
NOTE: Generally, a range of 100-500 mL of saline solution is needed. The saline solution allows for acoustic coupling. - Turn on the ultrasound machine and place the ultrasound probe within the saline bath at the level of interest to begin acquiring images.
NOTE: It is not necessary to place the probe directly touching the dura or spinal cord. Images are acquired on the ultrasound screen in real-time and can be interpreted immediately by the surgeon. Images on the screen can be captured at any time by pressing the Freeze button and can be saved by pressing the Save button. - Acquire real time images in the longitudinal plane by placing the ultrasound probe in line with the direction of the spinal canal to visualize the spinal cord and the lesion similar to the sagittal images from the MRI.
- Acquire real time images in the transverse plane by placing the ultrasound probe perpendicular to the spinal canal to visualize the spinal cord and the lesion like the axial images from the MRI.
- Acquire real-time images to verify the location of lesions that cannot be directly visualized, to correlate with the preoperative CT or MRI images, to guide surgical tool placement, and/or to confirm the resolution of pathology.
NOTE: When needed, a small piece of sterile compressed sponge approximately 0.5 cm x 0.5 cm can be used as a hyperechoic surgical marker to be placed in the surgical field and help correlate the surgical location with the image location. This helps to locate the lesion during the surgery and also helps to identify the margin of the tumor.
5. Postoperative Follow-up
- After discharge, have the patient return to the clinic within one month for follow-up.
- Perform a neurological assessment and CT or MRI scans to confirm the resolution of the symptoms and pathology.
Representative Results
On normal spine ultrasound imaging, the dura is an echogenic layer that surrounds the anechoic spinal fluid. The spinal cord is distinguished by its homogenous appearance and low echogenicity which is surrounded by an echogenic rim. This echogenic rim is due to the density shift from the spinal fluid to the spinal cord. The central canal appears as a bright central echo, while exiting nerve roots appear highly echogenic, particularly at the cauda equina16. Intraoperative ultrasound can play an advantageous role in the intradural mass lesion resection. In a standard case, preoperative CT or MRI approximates the location of an intradural mass with respect to known adjacent structures. With this approximation, a durotomy is made, usually with the extension of the durotomy in either direction for sufficient exposure of the lesion. In cases of cauda equina tumors, the lesion can rostrally migrate with respect to preoperative imaging20. With intraoperative ultrasound, the lesion can be readily visualized prior to dural opening and durotomy can be made more appropriately and accurately to the exact location of the mass20,21. Furthermore, with intramedullary lesions where there is a need for dissection through the spinal cord to reach tumors, risk of neural damage and subsequent neurological deficit can be reduced with the use of intraoperative ultrasound to guide the surgeon22. Additionally, a sterile compressed sponge is easily identified on ultrasound a hyperechoic material without acoustic wave attenuation and can be utilized as a surgical marker to distinguish tissue planes and limits for dissection15,23. An example is seen in Figure 1, 2 and 3 where a cervical intramedullary lesion was approached via a midline myelotomy. Intraoperative ultrasound was beneficial in visualizing and delineating tumor limits, as well as determining resection and resolution of tumor mass effect.
Intraoperative ultrasound is also particularly useful in operative cases with a posterior approach to resect lesions ventral to the thecal sac, especially in the cervical and thoracic spine where the spinal cord is vulnerable to injury with manipulation. While the ventral spinal canal can be approached anteriorly for better visualization of the lesion, there are associated increases in operative time, bleeding, and morbidity. Thus, a posterior approach is preferable, and inability to visualize the lesion directly can be overcome with intraoperative ultrasound to guide the surgeon. Cases where this technique is particularly useful include resection of intervertebral disc herniations, reduction of thoracolumbar burst fractures, resection of ventral extradural tumors, and in cases of spinal canal stenosis due to ossification of the posterior longitudinal ligament where confirmation of adequate posterior decompression is needed13, 14, 24,25,26,27,28,29,30,31,32,33,34. In a symptomatic thoracic disc herniation resection by posterior approach, intraoperative ultrasound aided in evaluating decompression and ensuring all compressive disc fragments were excised (Figure 4-5). Similarly, in the case of a lumbar burst fracture, intraoperative ultrasound was useful in confirming adequate decompression and removal of all fragments (Figure 6-7).
| Imaging technology | Advantage |
| Intraoperative ultrasound | • Real-time |
| • Excellent soft tissue resolution | |
| Cone beam CT and intraoperative CT | • 3D and multiplanar reconstructions |
| • Able to be paired with navigation systems | |
| Intraoperative MRI | • Multiplanar reconsutrctions |
| • Excellent soft tissue resolution | |
| Intraoperative fluoroscopy | • Real-time |
| • 2D images of bony structures |
Table 1. Comparison of intraoperative imaging techniques
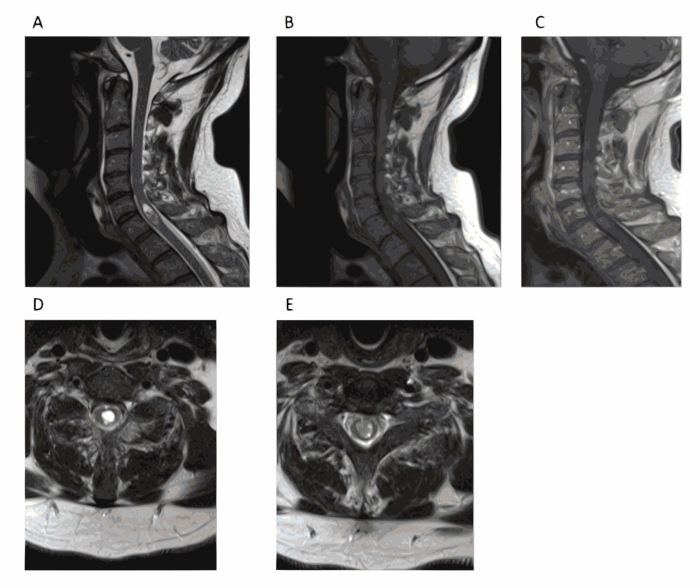
Figure 1. Preoperative images reveal an intramedullary lesion. A 54-year-old male with no significant past medical history presented with a 1-month history of fever. A cervical MRI revealed a C6 intramedullary lesion. The mass size did not change after 1 month and extensive workup did not reveal other possible causes of his fever. Patient was subsequently brought to the operating room for definitive diagnosis. (A) Sagittal T2-weighted MRI revealed an intramedullary lesion at C5-7 with fluid collection at the top of the mass. (B) Sagittal T1-weighted MRI. (C) Sagittal contrast-enhanced MRI shows scant rim enhancement. (D) Axial T2-weighted MRI at the level of the fluid collection. (E) Axial T2-weighted MRI of the lower part of the lesion. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
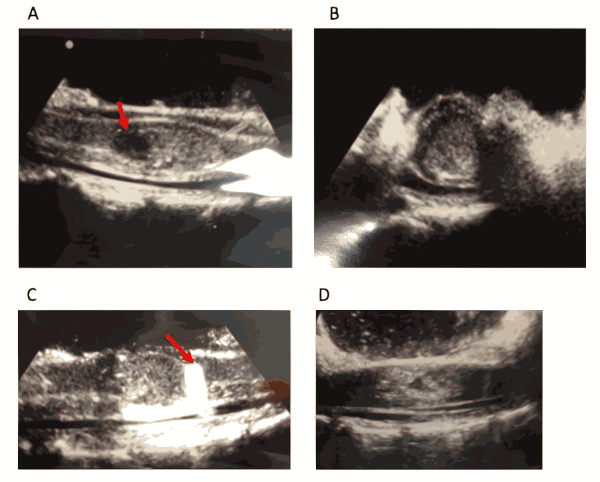
Figure 2. Intraoperative ultrasound of the spinal cord after laminectomy. Patient underwent a C5-7 laminectomy and subsequent resection of the intramedullary lesion. Intraoperative ultrasound was used to guide the surgical path through the spinal cord until the tumor could be visualized. (A) Intraoperative ultrasound correlated with preoperative MRI imaging, revealing fluid collection (white arrow). (B) Axial intraoperative ultrasound shows mass encompassing majority of the spinal cord. (C) A 0.5 cm x 0.5 cm piece of sterile compressed sponge (white arrow) was used during the operation to confirm the caudal limit of the tumor. (D) Intraoperative ultrasound post-resection confirming complete removal of tumor and resolution of mass effect. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
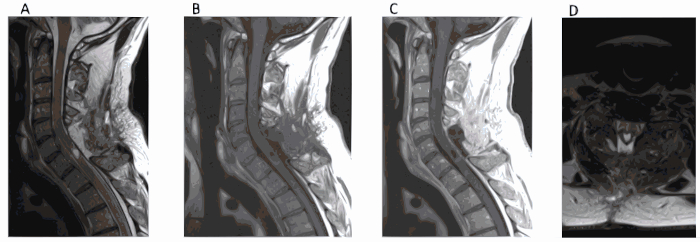
Figure 3. Post-operative resection imaging reveals complete tumor resection. Post-operatively, the patient returned to baseline and fever resolved. Pathology revealed grade II ependymoma. (A) Sagittal T2-weighted MRI 2 months post-operatively showing complete resection of the tumor. (B) T1-weighted MRI without contrast. (C, D) T1-weighted MRI with contrast. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
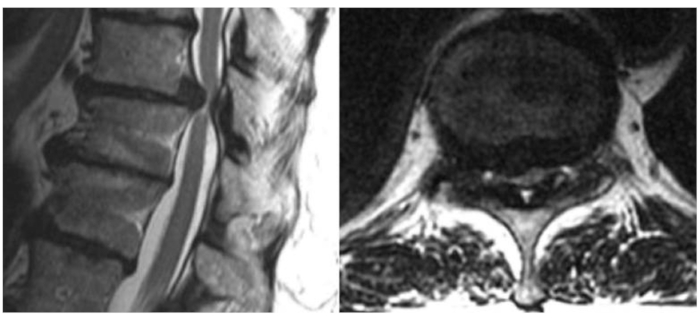
Figure 4. Preoperative MRI reveals severe spinal cord compression. A 73-year-old female presented with a history of several months of worsening gait dysfunction, spasticity, and numbness in her lower extremities. Motor strength was intact on neurological exam however she had marked clonus, 4+ lower extremity reflexes, and a wide-based staggering gait. CT and MRI revealed a large, noncalcified T10-11 intervertebral disc hernation with compression of the spinal cord. (A) Sagittal and (B) axial T2-weighted MRI revealing T10-11 disc herniation with spinal cord compression. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
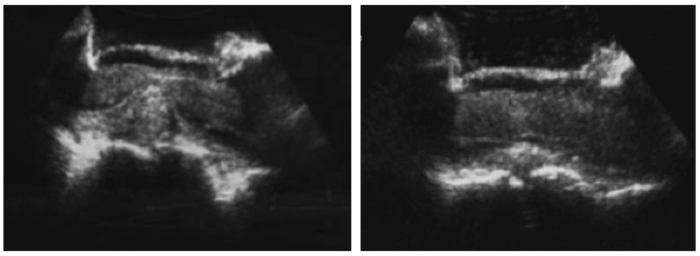
Figure 5. Intraoperative ultrasound reveals disc herniation and spinal cord compression. Patient underwent a right-sided T10-11 hemilaminectomy, facetectomy, and pedicle-sparing microdiscectomy with T9-11 fusion. (A) Intraoperative ultrasound was used to accurately determine the location of the disc herniation, (B) and to evaluate decompression and ensure complete removal of the herniated disc. Patient returned to her neurological baseline postoperatively and her prior symptoms had resolved at her 1-month follow up. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
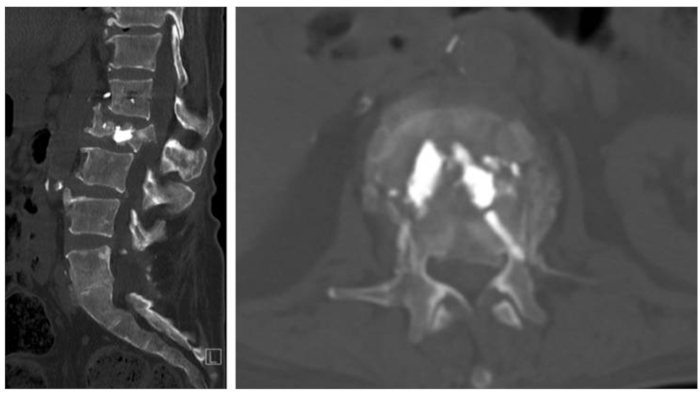
Figure 6. Preoperative CT demonstration pathological L2 burst fracture. A 57-year-old female with a history significant for metastatic appendiceal cancer and a balloon kyphoplasty at L1 and L2 a month prior for pathologic compression fractures presented with mechanical back pain and acute-onset of left anterior thigh pain. Motor strength was intact throughout, however she had decreased sensation to light touch over her left anterior thigh. (A) Sagittal and (B) axial CT revealed pathological L2 burst fracture. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
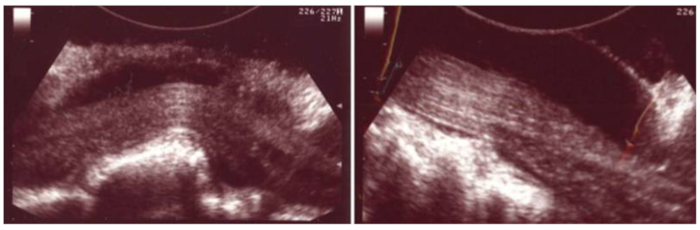
Figure 7. Intraoperative ultrasound reveals retropulsed bone fragment and subsequent complete reduction of fracture. Patient underwent an L1-L2 laminectomy, left transpedicular reduction of the fracture, and T12-L3 posterolateral fusion. Intraoperative ultrasound was used to identify any residual bone fragments. (A) A retropulsed bone fragment that was not directly visualized was seen in the ventral spinal canal displacing the thecal sac. (B) Complete reduction of the fracture and adequate decompression of the spinal canal was confirmed with ultrasound. Postoperatively, the patient returned to baseline with symptom resolution. *This figure has been modified from Vasudeva et al.35. Please click here to view a larger version of this figure.
Discussion
Intraoperative ultrasound in the spinal surgery has largely fallen out of favor with the advent of newer technology, however, it continues to provide several advantages over the other available imaging modalities such as MRI and CT6,9,16,17,18. In addition to being inexpensive, in this protocol we also show that it is simple to use and can provide visualization of structures with adequate resolution that could otherwise not be directly seen by the surgeon. It is particularly useful in cases where the surgeon is approaching a lesion located ventrally to the spinal canal in a posterior manner. Additionally, the images can be correlated with pre-operative MRI or CT images and do not require a radiologist for interpretation. Most importantly, intraoperative ultrasound remains the only imaging modality that allows for real-time image acquisition36. Ultrasound also poses no radiation risk to the patient or surgeon.
Preoperative MRI or CT images should be analyzed carefully to avoid intraoperative complications and to accurately determine the location of the initial incision. This will help ensure that the ultrasound probe will be at the precise location desired. After the initial incision is made, an X-ray can be performed intraoperatively at the incision site to confirm vertebrae location. It is critical that sufficient hemostasis is reached prior to filling the surgical field with saline to acquire clear images as blood can attenuate ultrasound waves. It is not necessary for the probe to directly touch the dura or spinal cord for images acquisition. If images are not clear upon acquisition, drain the saline solution and fill with fresh saline solution and repeat image acquisition.
The only limitations to this protocol is that it is operator dependent, however the learning curve is gentle and surgeons can become proficient after the first or second operation36.
In conclusion, intraoperative ultrasound is useful in spinal surgery and should be considered especially in cases of intradural lesions and lesions ventral to the thecal sac when approaching posteriorly. Recent introduction of contrast-enhanced ultrasound has demonstrated potential use as well, in spinal Dural arteriovenous fistulas and vascularized spinal tumors37,38. Education and the use of intraoperative ultrasound in spinal surgery should also be incorporated into residency and fellowship teaching programs. Future development in the ultrasound technology can further enhance and increase the utility of this imaging modality.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors have no acknowledgements.
Materials
| Aloka Prosound 5 mobile ultrasound machine | Hitachi | N/A | any comparable devices on the market should suffice |
| UST-9120 transducer probe. | Hitachi | UST-9120 | Has a 20mm diameter with 10 to 4.4 MHz frequency range (any comparable compatible transducer should suffice). |
References
- Reid, M. H. Ultrasonic visualization of a cervical cord cystic astrocytoma. AJR. American Journal of Roentgenology. 131 (5), 907-908 (1978).
- Dohrmann, G. J., Rubin, J. M. Intraoperative ultrasound imaging of the spinal cord: syringomyelia, cysts, and tumors–a preliminary report. Surgical Neurology. 18 (6), 395-399 (1982).
- Rubin, J. M., Dohrmann, G. J. Use of ultrasonically guided probes and catheters in neurosurgery. Surgical Neurology. 18 (2), 143-148 (1982).
- Braun, I. F., Raghavendra, B. N., Kricheff, I. I. Spinal cord imaging using real-time high-resolution ultrasound. Radiology. 147 (2), 459-465 (1983).
- Hutchins, W. W., Vogelzang, R. L., Neiman, H. L., Fuld, I. L., Kowal, L. E. Differentiation of tumor from syringohydromyelia: intraoperative neurosonography of the spinal cord. Radiology. 151 (1), 171-174 (1984).
- Juthani, R. G., Bilsky, M. H., Vogelbaum, M. A. Current Management and Treatment Modalities for Intramedullary Spinal Cord Tumors. Current Treatment Options in Oncology. 16 (8), 39 (2015).
- Knake, J. E., Gabrielsen, T. O., Chandler, W. F., Latack, J. T., Gebarski, S. S., Yang, P. J. Real-time sonography during spinal surgery. Radiology. 151 (2), 461-465 (1984).
- Montalvo, B. M., Quencer, R. M., Green, B. A., Eismont, F. J., Brown, M. J., Brost, P. Intraoperative sonography in spinal trauma. Radiology. 153 (1), 125-134 (1984).
- Montalvo, B. M., Quencer, R. M. Intraoperative sonography in spinal surgery: current state of the art. Neuroradiology. 28 (5-6), 551-590 (1986).
- Pasto, M. E., Rifkin, M. D., Rubenstein, J. B., Northrup, B. E., Cotler, J. M., Goldberg, B. B. Real-time ultrasonography of the spinal cord: intraoperative and postoperative imaging. Neuroradiology. 26 (3), 183-187 (1984).
- Mari, A. R., Shah, I., Imran, M., Ashraf, J. Role of intraoperative ultrasound in achieving complete resection of intra-axial solid brain tumours. JPMA. The Journal of the Pakistan Medical Association. 64 (12), 1343-1347 (2014).
- Ivanov, M., Budu, A., Sims-Williams, H., Poeata, I. Using Intraoperative Ultrasonography for Spinal Cord Tumor Surgery. World Neurosurgery. 97, 104-111 (2017).
- Blumenkopf, B., Daniels, T. Intraoperative ultrasonography (IOUS) in thoracolumbar fractures. Journal of Spinal Disorders. 1 (1), 86-93 (1988).
- McGahan, J. P., Benson, D., Chehrazi, B., Walter, J. P., Wagner, F. C. Intraoperative sonographic monitoring of reduction of thoracolumbar burst fractures. AJR. American Journal of roentgenology. 145 (6), 1229-1232 (1985).
- Quencer, R. M., Montalvo, B. M., Eismont, F. J., Green, B. A. Intraoperative spinal sonography in thoracic and lumbar fractures: evaluation of Harrington rod instrumentation. AJR. American Journal of roentgenology. 145 (2), 343-349 (1985).
- Sosna, J., Barth, M. M., Kruskal, J. B., Kane, R. A. Intraoperative sonography for neurosurgery. Journal of Ultrasound in Medicine: Official Journal of the American Institute of Ultrasound in Medicine. 24 (12), 1671-1682 (2005).
- Raymond, C. A. Brain, spine surgeons say yes to ultrasound. JAMA. 255 (17), 2258-2262 (1986).
- Toktas, Z. O., Sahin, S., Koban, O., Sorar, M., Konya, D. Is intraoperative ultrasound required in cervical spinal tumors? A prospective study. Turkish Neurosurgery. 23 (5), 600-606 (2013).
- . . Surgical Approaches to the Spine. , (2015).
- Friedman, J. A., Wetjen, N. M., Atkinson, J. L. D. Utility of intraoperative ultrasound for tumors of the cauda equina. Spine. 28 (3), 288-290 (2003).
- Zhou, H., et al. Intraoperative ultrasound assistance in treatment of intradural spinal tumours. Clinical Neurology and Neurosurgery. 113 (7), 531-537 (2011).
- Harrop, J. S., Ganju, A., Groff, M., Bilsky, M. Primary intramedullary tumors of the spinal cord. Spine. 34, 69-77 (2009).
- Quencer, R. M., Montalvo, B. M. Normal intraoperative spinal sonography. AJR. American journal of roentgenology. 143 (6), 1301-1305 (1984).
- Aoyama, T., Hida, K., Akino, M., Yano, S., Iwasaki, Y. Detection of residual disc hernia material and confirmation of nerve root decompression at lumbar disc herniation surgery by intraoperative ultrasound. Ultrasound in Medicine & Biology. 35 (6), 920-927 (2009).
- Bose, B. Thoracic extruded disc mimicking spinal cord tumor. The Spine Journal: Official Journal of the North American Spine Society. 3 (1), 82-86 (2003).
- Harel, R., Knoller, N. Intraoperative spine ultrasound: application and benefits. European Spine Journal: Official Publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 25 (3), 865-869 (2016).
- Lazennec, J. Y., Saillant, G., Hansen, S., Ramare, S. Intraoperative ultrasonography evaluation of posterior vertebral wall displacement in thoracolumbar fractures. Neurologia Medico-Chirurgica. 39 (1), 8-15 (1999).
- Matsuyama, Y., et al. Cervical myelopathy due to OPLL: clinical evaluation by MRI and intraoperative spinal sonography. Journal of Spinal Disorders & Techniques. 17 (5), 401-404 (2004).
- Mueller, L. A., et al. Ultrasound-guided spinal fracture repositioning, ligamentotaxis, and remodeling after thoracolumbar burst fractures. Spine. 31 (20), 739-747 (2006).
- Nishimura, Y., Thani, N. B., Tochigi, S., Ahn, H., Ginsberg, H. J. Thoracic discectomy by posterior pedicle-sparing, transfacet approach with real-time intraoperative ultrasonography: Clinical article. Journal of Neurosurgery. Spine. 21 (4), 568-576 (2014).
- Randel, S., Gooding, G. A., Dillon, W. P. Sonography of intraoperative spinal arteriovenous malformations. Journal of Ultrasound in Medicine: Official Journal of the American Institute of Ultrasound in Medicine. 6 (9), 539-544 (1987).
- Seichi, A., et al. Intraoperative ultrasonographic evaluation of posterior decompression via. laminoplasty in patients with cervical ossification of the posterior longitudinal ligament: correlation with 2-year follow-up results. Journal of Neurosurgery. Spine. 13 (1), 47-51 (2010).
- Tian, W., et al. Intraoperative 3-dimensional navigation and ultrasonography during posterior decompression with instrumented fusion for ossification of the posterior longitudinal ligament in the thoracic spine. Journal of Spinal Disorders & Techniques. 26 (6), 227-234 (2013).
- Tokuhashi, Y., Matsuzaki, H., Oda, H., Uei, H. Effectiveness of posterior decompression for patients with ossification of the posterior longitudinal ligament in the thoracic spine: usefulness of the ossification-kyphosis angle on MRI. Spine. 31 (1), 26-30 (2006).
- Vasudeva, V. S., Abd-El-Barr, M., Pompeu, Y. A., Karhade, A., Groff, M. W., Lu, Y. Use of Intraoperative Ultrasound During Spinal Surgery. Global Spine Journal. 7 (7), 648-656 (2017).
- Alaqeel, A., Abou Al-Shaar, H., Alaqeel, A., Al-Habib, A. The utility of ultrasound for surgical spinal decompression. Medical Ultrasonography. 17 (2), 211-218 (2015).
- Della Pepa, G. M., et al. Real-time intraoperative contrast-enhanced ultrasound (CEUS) in vascularized spinal tumors: a technical note. Acta Neurochirurgica. 160 (6), 1259-1263 (2018).
- Della Pepa, G. M., et al. Integration of Real-Time Intraoperative Contrast-Enhanced Ultrasound and Color Doppler Ultrasound in the Surgical Treatment of Spinal Cord Dural Arteriovenous Fistulas. World Neurosurgery. 112, 138-142 (2018).

