Analysis of Translation in the Developing Mouse Brain using Polysome Profiling
Summary
The development of the mammalian brain requires proper control of gene expression at the level of translation. Here, we describe a polysome profiling system with an easy-to-assemble sucrose gradient-making and fractionation platform to assess the translational status of mRNAs in the developing brain.
Abstract
The proper development of the mammalian brain relies on a fine balance of neural stem cell proliferation and differentiation into different neural cell types. This balance is tightly controlled by gene expression that is fine-tuned at multiple levels, including transcription, post-transcription and translation. In this regard, a growing body of evidence highlights a critical role of translational regulation in coordinating neural stem cell fate decisions. Polysome fractionation is a powerful tool for the assessment of mRNA translational status at both global and individual gene levels. Here, we present an in-house polysome profiling pipeline to assess translational efficiency in cells from the developing mouse cerebral cortex. We describe the protocols for sucrose gradient preparation, tissue lysis, ultracentrifugation and fractionation-based analysis of mRNA translational status.
Introduction
During the development of the mammalian brain, neural stem cells proliferate and differentiate to generate neurons and glia1,2 . The perturbation of this process can lead to alterations in brain structure and function, as seen in many neurodevelopmental disorders3,4. The proper behavior of neural stem cells requires the orchestrated expression of specific genes5. While the epigenetic and transcriptional control of these genes has been intensively studied, recent findings suggest that gene regulation at other levels also contributes to the coordination of neural stem cell proliferation and differentiation6,7,8,9,10. Thus, addressing the translational control programs will greatly advance our understanding of the mechanisms underlying neural stem cell fate decision and brain development.
Three main techniques with different strengths have been widely applied to assess the translational status of mRNA, including ribosome profiling, translating ribosome affinity purification (TRAP) and polysome profiling. Ribosome profiling uses RNA sequencing to determine ribosome-protected mRNA fragments, allowing the global analysis of the number and location of translating ribosomes on each transcript to indirectly infer the translation rate by comparing it to transcript abundance11. TRAP takes advantage of epitope-tagged ribosomal proteins to capture ribosome-bound mRNAs12. Given that the tagged ribosomal proteins can be expressed in specific cell types using genetic approaches, TRAP allows the analysis of translation in a cell type-specific manner. In comparison, polysome profiling, which uses sucrose density gradient fractionation to separate free and poorly-translated portion (lighter monosomes) from those being actively translated by ribosomes (heavier polysomes), provides a direct measurement of ribosome density on mRNA13. One advantage this technique offers is its versatility to study the translation of specific mRNA of interest as well as genome-wide translatome analysis14.
In this paper, we describe a detailed protocol of polysome profiling to analyze the developing mouse cerebral cortex. We use a home-assembled system to prepare sucrose density gradients and collect fractions for downstream applications. The protocol presented here can be adapted easily to analyze other types of tissues and organisms.
Protocol
All animal use was supervised by the Animal Care Committee at the University of Calgary. CD1 mice used for the experiment were purchased from commercial vendor.
1. Preparation of solutions
NOTE To prevent RNA degradation, spray workbench and all equipment with RNase decontamination solution. RNase-free tips are used for the experiment. All solutions are prepared in RNase-free water.
- Prepare cycloheximide stock solution (100 mg/mL) in DMSO and store at -20 °C.
- Prepare 2.2 M sucrose stock solution by adding 75.3 g of sucrose to RNase-free water and topping up the volume to 100 mL (for ~16 gradient preparation). The solution can be kept at -20 °C for long-term storage.
- Prepare 10x salt solution (1 M NaCl; 200 mM Tris-HCl, pH 7.5; 50 mM MgCl2).
- Prepare 60% (w/v) sucrose chase solution, containing 30 g of sucrose, 5 mL of 10x salt solution and top the volume up to 50 mL with RNase-free water.
- Optionally, add a speck of bromophenol blue or approximately 5 µL of 1% bromophenol blue in RNase-free water to the chase solution. 1.6. Store solutions at 4 °C.
2. Preparation of sucrose gradient
NOTE: Accuracy in preparation of sucrose gradients is critical in obtaining consistent and reproducible results.
- To prepare six 10-50% sucrose gradients, dilute 2.2 M sucrose solution as in Table 1.
| Sucrose solution | 10% | 20% | 30% | 40% | 50% |
| 2.2 M sucrose | 2 mL | 4 mL | 6 mL | 8 mL | 10 mL |
| 10X salt solution | 1.5 mL | 1.5 mL | 1.5 mL | 1.5 mL | 1.5 mL |
| Cycloheximide | 15 µL | 15 µL | 15 µL | 15 µL | 15 µL |
| Water | 11.5 mL | 9.5 mL | 7.5 mL | 5.5 mL | 3.5 mL |
| Total volume | 15 mL | 15 mL | 15 mL | 15 mL | 15 mL |
Table 1: Sucrose dilutions for preparations of sucrose gradients.
NOTE: Always prepare sucrose gradients in multiples of two to balance weight during ultracentrifugation.
- Wipe the metal blunt end needle with RNase decontamination solution. Briefly rinse the ultracentrifuge tubes, tubing and the syringe using RNase-free water. Air-dry the tubes such that no water drop remains in the tubes as any remaining water will alter the sucrose concentration.
- Place the metal needle on the clamp holder and the centrifuge tube on the motorized stage. To prepare gradients consistently, a home-assembled system was used that contains a motorized stage to hold the ultracentrifuge tube and a syringe pump to inject sucrose solutions (Figure 1, see step 9 for details).
- Fill a 30 mL syringe with ~16 mL of 10% sucrose (enough for six gradients), place it on the syringe pump and connect it to the needle. Make sure that there are no air bubbles in the syringe, tubing, and needle. Wipe the tip off the needle to remove any residual solution.
- Move the motorized stage up such that the tip of the needle touches the centre of the tube at the bottom.
- Set the syringe pump at a flow rate of 2 mL/min for a volume of 2.3 mL.
- After dispensing 2.3 mL of 10% sucrose solution, move down the motorized stage and repeat the process for all six gradients.
- Repeat steps 2.4-2.7 to add 20% sucrose solution to the bottom of the tubes, followed by 30%, 40% and 50% sucrose solutions similarly.
- After preparation of the gradient, seal the ultracentrifuge tube using a paraffin film.
- Leave the tubes overnight at 4 °C such that the different sucrose layers diffuse together to give a continuous gradient.
3. Tissue dissection
NOTE: Pregnant mice were euthanized by cervical dislocation preceded by anesthesia with 5% isoflurane.
- Collect CD1 mouse embryos at embryonic day 12, or other timepoints as needed, and place embryos in a Ø 10 cm plate containing ice-cold Hank's Balanced Salt Solution (HBSS) on ice to retain cell viability.
- Under the dissection scope, transfer one embryo to a Ø 6 cm plate containing ice-cold HBSS.
- Use 21-23 G needles to fix the position of the head by penetrating through eyes at an approximately 45° angle and apply force to make sure the needles are fixed on the plate (Figure 2A).
- Use No.5 forceps to remove skin and skull, from the middle to the sides.
- Use forceps to cut the olfactory bulbs and remove the meninges to expose the cortical tissues.3.6.Use curved forceps to cut the cortical tissues into 2-3 mL of neurobasal medium on ice (Figure 2B). Pool cortical tissues from different embryos as needed. Tissues from 8-10 embryos usually give ~200 µg total RNA.
4. Cell lysis
- On the day of tissue dissection, prepare fresh cell lysis buffer as described in Table 2.
| Solution | Final concentration | Volume |
| Tris-HCl (pH 7.5) | 20 mM | 100 µL |
| KCl | 100 mM | 250 µL |
| MgCl2 | 5 mM | 25 µL |
| Triton X-100 | 1% (v/v) | 500 µL |
| Sodium deoxycholate | 0.5% (w/v) | 500 µL |
| Dithiothreitol (DTT) | 1 mM | 5 µL |
| Cycloheximide | 100 µg/mL | 5 µL |
| RNase free water | Top up to 5 mL | |
| Total | 5 mL | 5 mL |
Table 2: Preparation of polysome lysis buffer.
NOTE: Supplement lysis buffer with protease and phosphatase inhibitors.
- Add cycloheximide to the neurobasal medium with dissected tissues to a final concentration of 100 µg/mL and incubate at 37 °C for 10 min. Cycloheximide blocks translation elongation and, therefore, prevents ribosome run-off 15.
- Centrifuge at 500 x g for 5 min at 4 °C and discard the supernatant.
- Wash the tissues twice with ice-cold phosphate-buffered saline (PBS) supplemented with 100 µg/mL cycloheximide.
- Add 500 µL of cell lysis buffer supplemented with 4 µL of RNase inhibitor. Pipette up and down to resuspend the tissue in lysis buffer. Use an insulin needle to gently lyse the tissue.
- Incubate the tissues on ice for 10 min with brief vortexing every 2-3 min.
NOTE: To prevent tissue degradation, ensure that all steps of tissue lysis are carried out on ice. - Centrifuge at 2,000 x g for 5 min at 4 °C.
- Transfer the supernatant to a new centrifuge tube on ice.
- Centrifuge at ~13,000 x g for 5 min at 4 °C.
- Transfer the supernatant to a new centrifuge tube on ice.
- Measure the RNA concentration in the tissue lysate using a UV-Vis spectrophotometer.
NOTE: If multiple samples are included in the experiment, dilute samples to the same concentration with extra cell lysis buffer to minimize variation.
5. Sample loading and ultracentrifugation
- Pre-cool the ultracentrifugation rotor and swing buckets at 4 °C, and set the temperature of the ultracentrifuge to 4 °C.
- Keep 20 µL of the tissue lysate as total RNA input.
- Load samples (50-300 µg RNA with an equal volume) on the top of the sucrose gradients by slowly dispensing the lysate to the walls of the ultracentrifuge tubes.
- Gently place the ultracentrifuge tubes in the swing bucket. Ensure that all diametrically opposite buckets are balanced.
- Load the swing buckets on the rotor. Set the ultracentrifuge to 190,000 x g (~39,000 rpm) at 4 °C for 90 min.
- Gently place the gradients on ice after centrifugation.
6. Fractionation and sample collection
NOTE: A home-assembled fractionating, recording and collecting system is used for the analysis and collecting samples from the gradients (Figure 3, see Device components).
- Place an empty ultracentrifuge tube on the tube piercer and gently penetrate the tube with the needle from the bottom.
- Switch on the UV monitor, and open the digital signal recording software.
- Fill a 30 mL syringe with 25 mL of 60% sucrose chase solution. Gently press the syringe such that the chase solution fills up the empty tube and go through the 254 nm UV monitor to set the baseline for detection.
- Press auto-zero on the UV-monitor to register a baseline for detection. Press play on the software to begin recording.
- Once the system establishes a baseline, pause recording on the software and retract the chase solution. Ensure that no residual chase solution remains in the system.
NOTE: Rinse the system with RNase-free water to remove residual sucrose solutions remaining from the previous run. - Load one sample tube to the tube piercer. Gently penetrate the tube with the needle from the bottom.
- Begin recording on the software. Start the syringe pump (flow rate of 1 mL/min for a volume of 25 mL) and the fraction collector to collect polysome fractions. Set fractionator settings to 30 s.
NOTE: Before each run, ensure no air bubbles in the syringe or the tubing by gently pressing the syringe to let the chase solution flow continuously through the needle. - Collect the fractions into 1.5 mL tubes (500 µL each) using a fraction collector.
- The fractions can be processed immediately or stored at -80 °C.
- After fraction collection, rinse the system with RNase-free water to remove any remnant sucrose.
7. Extraction of RNA
- To each fraction, add 10 ng of luciferase mRNA spike-in control.
- Add three volumes of guanidium hydrocholride based commercial RNA isolation reagent to each fraction.
- Vortex briefly for 15 s.
- Extract RNA using an RNA extraction kit compatible with the solution used in 7.2.
8. Reverse transcription and real-time PCR
- Measure the concentration of the RNA using UV-Vis spectrophotometer.
- Subject RNA to reverse transcription using a cDNA synthesis kit, according to manufacturer's protocol.
- Use quantitative real-time PCR (qPCR) to examine the polysomal distribution of gapdh mRNA as an example. qPCR was performed using a qPCR detection system and a qPCR mastermix reagent, with the following cycling conditions: 95 °C for 10 min, then 40 cycles of 95 °C for 15 s, 60 °C for 30 s, 72 °C for 40 s, followed by 95 °C for 60 s.
- Obtain Threshold cycle (Ct) values from the amplification plots and used to calculate fold change using ΔΔCt method.
9. Sucrose gradient making system assembly
NOTE: Follow the steps to assemble each component (as described in Table 3) of the sucrose gradient maker (Figure 1).
- Mount two vertical brackets (A2) on the base breadboard (A1).
- Use two slim right-angle brackets (B2) to mount the linear stage actuator to the base breadboard (A1).
- Use the setscrew on the Ø12.7 mm aluminum post (C2) to fix it on the tube holder base breadboard (C1) as a stand for the tube holder.
- Assemble the right-angle Ø1/2" to Ø6 mm post clamp (C3) with the post (C2) and the mini-series optical post (C4).
- Use the setscrew on the optical post (C4) to connect a small V-clamp (C5) as the tube holder.
- Put a centrifuge tube in the tube holder and adjust the angle to make it vertical. Mark the position of the tube and mount the pedestal post holder (C6) on the base breadboard (C1) to support the tube.
- On the other side of the breadboard (A1), use the setscrew of the Ø12.7 mm aluminum post (D1) to fix it and connect a mini-series optical post (D3) using a right-angle Ø1/2" to Ø6 mm post clamp (D2).
- Connect the miniature V-clamp (D4) with blunt-end needle (D5) to the post (D3). Adjust the angle of the clamp to set the needle vertical, and adjust the length of post to ensure the needle meets the centrifuge tube.
- Connect the actuator (B1) to the stepper motor driver (E1) and use the UNO R3 controller board and the joystick module from the UNO starter kit (E2) to control the actuator. A power adaptor (e.g., 9-24 V AC/DC adjustable power adaptor) can be used to drive the motor separately.
- Place the syringe pump (F) next to the gradient making station and connect the syringe with the needle.
- Control the tube holder stage up and down using the joystick.
| Component | Item |
| B1 | Linear stage actuator |
| E1 | Stepper motor driver |
| E2 | UNO project super starter kit |
| A1 | Breadboard |
| A2 | Vertical bracket |
| B2 | Slim right-angle bracket |
| C1 | Mini-series breadboard |
| C5 | Small V-clamp |
| D4 | Miniature V-clamp |
| C2 | Ø12.7 mm aluminum post |
| C4, D3 | Mini-series optical post |
| D1 | Ø12.7 mm aluminum post |
| C3, D2 | Right-angle Ø1/2" to Ø6 mm post clamp |
| C6 | Mini-series pedestal post holder base |
| D5 | Blunt end needle |
| F | Syringe pump |
Table 3: Gradient making system components.
10. Fractionating and detecting system assembly (Figure 2).
- Use a regular round jaw burette clamp to mount the optics module of the UV monitor on the tube piercer. Attach one end of the tubing (1 cm long and 0.56 mm internal diameter) to the connecter of the tube piercer and the other end to the Fluid In port of the optics module. Connect the Fluid Out port to the fractionator.
- Connect the optics module with the control unit of the UV monitor according the manual of the manufacturer.
- Use a breakout cable to connect the signal output socket to the digital converter at the ground and analog input connections, according to the manufacturer's manual.
- Connect the digital converter to a laptop using a regular USB cable, and record the converted digital signals using data acquisition software.
Representative Results
As a demonstration, the cortical lysate containing 75 µg RNA (pooled from 8 embryos) was separated by the sucrose gradient into 12 fractions. Peaks of UV absorbance at 254 nm identified fractions containing the 40S subunit, 60S subunit, 80S monosome and polysomes (Figure 4A). Analysis of fractions by western blot for the large ribosomal subunit, Rpl10 showed its presence in the 60S subunit (fraction 3), monosome (fraction 4) and polysomes (fractions 5-12) (Figure 4B). In contrast, cytoplasmic proteins Gapdh and Csde1 were not associated with ribosomes but were enriched in fractions containing free RNA (fraction 1) (Figure 4B). Consistent with the separation of proteins in different fractions, we found that gapdh and sox2 mRNAs were highly enriched in the fractions containing heavy polysomes (more than three ribosomes in fractions 7-12), suggesting that gapdh and sox2 mRNAs are efficiently translated in the developing cortex (Figure 4C,D). In contrast, rpl7 and rpl34 mRNAs were enriched in the fraction containing monosomes, suggesting repressed translation (Figure 4E,F)16. These results validated our polysome fractionation protocol.
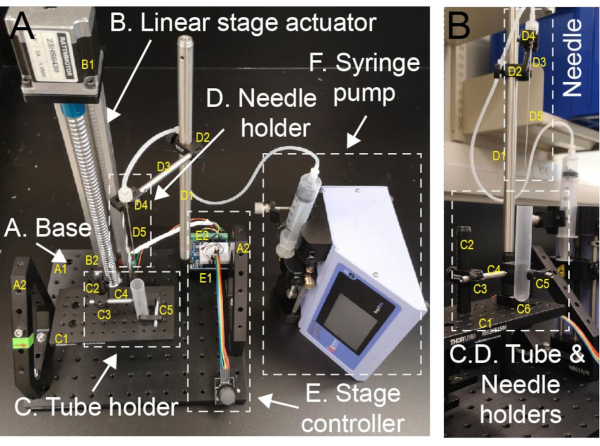
Figure 1: Setup for sucrose gradient preparation. (A) Home-assembled sucrose gradient maker consisting of a linear stage actuator, a tube holder on the motorized stage, a stage controller set, a metal blunt-end needle mounted on a needle holder, and an automated syringe pump with a syringe connected to the needle (see Table 3). (B) To prepare the sucrose gradient, the ultracentrifuge tube is placed in the tube holder. Sucrose solutions are dispensed through the blunt-end needle using a syringe pump. Please click here to view a larger version of this figure.
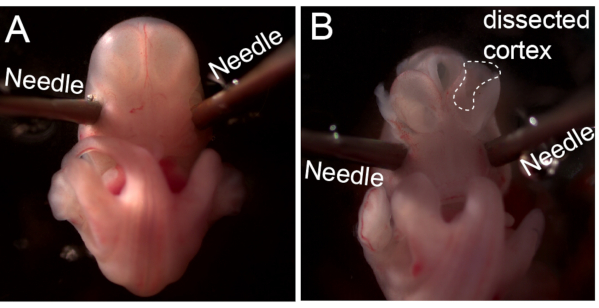
Figure 2: Dissection of the cortical tissues from the development mouse embryo. (A) Image showing an E12.5 CD1 embryo fixed to a Ø 6 cm plate using 21G-23G needles. (B) Image showing the dissected cortex after removal of the skin, skull and meninges (marked with white dashed lines). Please click here to view a larger version of this figure.

Figure 3: Setup for fractionating, recording and sample collecting. Image depicting the home-assembled fractionation, recording and sample collection system comprising of an automated syringe pump, a tube piercer, a fraction collector and a UV monitor with a digital convertor connected to a laptop for the continuous monitoring of UV absorbance. Data acquisition is handled by the commercial data acquisition software. Please click here to view a larger version of this figure.
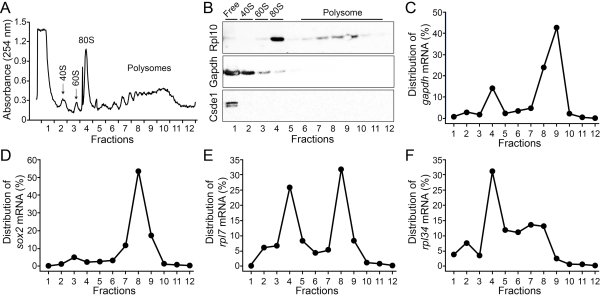
Figure 4: Polysome profiling analysis of the developing mouse cortex. (A) UV absorbance showing fractions containing free RNA (fraction 1), 40S subunit (fraction 2), 60S subunit (fraction 3), 80S monosomes (fraction 4) and polysomes (fraction 5-12). 75 µg total RNA pooled from 8 embryos was used. (B) Western blots showing the distributions of Gapdh, Rpl10 and Csde1 proteins in fractions. qPCR analysis showing the distribution of gapdh (C), sox2 (D), rpl7 (E) and rpl34 (F) mRNAs in fractions. Please click here to view a larger version of this figure.
Discussion
Polysome profiling is a commonly used and powerful technique to assess the translational status at both single gene and genome-wide levels14 . In this report, we present a protocol of polysome profiling using a home-assembled platform and its application to analyze the developing mouse cortex. This cost-effective platform is easy to assemble and generate robust, reproducible sucrose gradients and polysome profiling with high sensitivity.
It is worthy to note that the preparation of consistent and good quality sucrose gradients is critical to obtain reproducible polysome profiling results17. Changes in the volume of 10-50% sucrose solutions during gradient preparation could cause the shift of the polysome peaks and inconsistency of results in downstream analysis. Moreover, disturbing the gradient during the insertion and removal of the needle could contribute to the inconsistency of gradient preparation. Compared to other approaches and commercial devices, our home-made gradient making system used a motorized stage to reduce disturbance of gradients during preparation and a syringe pump to accurately dispense sucrose solutions, which offers a cheap and simple solution for gradient preparation.
Applying the polysome profiling platform and protocol present here to the developing mouse cortex, we found that the ribosomal protein Rpl10 and cytoplasmic proteins Gapdh and Csde1 were distributed in the correct fractions. Moreover, the highly translated gapdh and sox2 mRNAs were enriched in polysomal fractions, while translationally repressed rpl34 and rpl7 mRNAs showed less enrichement in polysomes, which validate our platform and protocol. With a similar approach, the translational status of other genes in the developing cortex can be determined individually by real-time PCR. Of note, polysome profiling has been used to analyse translational status in the developing brain at the genome-wide level. In this regard, fractions containing polysomes can be combined and extracted RNA can be analysed using deep sequencing. The platform and protocol present here can be adapted and further optimized to suit translatomic studies using other cell types and tissues. Considering the availability of cortical tissues limited by experimental conditions (e.g., <50 µg RNA), additional optimization of the protocol may provide further increases in the efficiency and reproducibility of the experiments.
While polysome profiling provides insights into the translational status of mRNAs in the developing cortex, it has limitations to analyze specific cell types at later developmental stages, when different neural cell types are present. To address this question, polysome profiling can be integrated with TRAP by pull-down of epitope tagged ribosomal protein expressed under a tissue specific promoter11.
In conclusion, the platform and protocol that we report here for sucrose gradient making and fractionation provide an economical solution to polysome profiling experiments in the context of brain development as well as other biological contexts.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by a NSERC Discovery Grant (RGPIN/04246-2018 to G.Y.). G.Y. is a Canada Research Chair. S.K. was funded by Mitacs Globalink Graduate Fellowship and ACHRI Graduate Student Scholarship.
Materials
| 1.5 mL RNA free microtubes | Axygen | MCT-150-C | |
| 10 cm dish | Greiner-Bio | 664160 | |
| 1M MgCl2 | Invitrogen | AM9530G | |
| 21-23G needle | BD | 305193 | |
| 2M KCl | Invitrogen | AM8640G | |
| 30 mL syringe | BD | 302832 | |
| Blunt end needle | VWR | 20068-781 | |
| Breadboard | Thorlabs | MB2530/M | |
| Bromophenol blue | Sigma | 115-39-9 | |
| CD1 mouse | Charles River Laboratory | ||
| Curved tip forceps | Sigma | #Z168785 | |
| Cycloheximide | Sigma | 66-81-9 | |
| Data acquisition software TracerDAQ | Measurement Computing | ||
| Digital converter | Measurement Computing | USB-1208LS | |
| Direct-zol RNA miniprep kit | Zymo | R2070 | |
| Dithiothreitol (DTT) | Bio-basic | 12-03-3483 | |
| DMSO | Bioshop | 67-68-5 | |
| Dumont No.5 forceps | Sigma | #F6521 | |
| Fraction collector | Bio-Rad | Model 2110 | |
| HBSS | Wisent | 311-513-CL | |
| Linear stage actuator | Rattmmotor | CBX1605-100A | |
| Luciferase control RNA | Promega | L4561 | |
| Maxima first strand cDNA synthesis kit | Themo Fisher | M1681 | |
| Miniature V-clamp | Thorlabs | VH1/M | |
| Mini-series breadboard | Thorlabs | MSB7515/M | |
| Mini-series optical post | Thorlabs | MS2R/M | |
| Mini-series pedestal post holder base | Thorlabs | MBA1 | |
| NaCl | Bio-basic | 7647-14-5 | |
| Neurobasal media | Gibco | 21103-049 | |
| Ø12.7 mm aluminum post | Thorlabs | TRA150/M | |
| Parafilm | Bemis | PM992 | |
| PerfeCTa SYBR green fastmix | Quanta Bio | CA101414-274 | |
| Phosphate buffered saline (PBS) | Wisent | 311-010-CL | |
| Puromycin | Bioshop | 58-58-2 | |
| Right-angle clamp | Thorlabs | RA90/M | |
| Right-angle Ø1/2" to Ø6 mm post clamp | Thorlabs | RA90TR/M | |
| Rnase AWAY | Molecular BioProducts | 7002 | |
| RNase free tips | Frogga Bio | FT10, FT200, FT1000 | |
| RNase free water | Wisent | 809-115-CL | |
| RNasin | Promega | N2111 | |
| Slim right-angle bracket | Thorlabs | AB90B/M | |
| Small V-clamp | Thorlabs | VC1/M | |
| Sodium deoxycholate | Sigma | 302-95-4 | |
| Stepper motor driver | SongHe | TB6600 | |
| Sucrose | Bioshop | 57501 | |
| SW 41 Ti rotor | Beckman Coulter | 331362 | |
| Syringe pump | Harvard Apparatus | 70-4500 | |
| Syringe pump | Harvard Apparatus | 70-4500 | |
| Triton-X-100 | Bio-basic | 9002-93-1 | |
| Trizol | Thermofisher Scientific | 15596018 | |
| Tube piercer | Brandel | BR-184 | |
| Ultracentrifuge | Beckman Coulter | L8-70M | |
| Ultracentrifuge tubes | Beckman Coulter | 331372 | |
| UltraPure 1M Tris-HCl pH 7.5 | Invitrogen | 15567-027 | |
| UNO project super starter kit | Elegoo | EL-KIT-003 | |
| UV monitor | Bio-Rad | EM-1 Econo | |
| Vertical bracket | Thorlabs | VB01A/M |
References
- Götz, M., Huttner, W. B. The cell biology of neurogenesis. Nature Reviews Molecular Cell Biology. 6, 777-788 (2005).
- Guillemot, F. Spatial and temporal specification of neural fates by transcription factor codes. Development. 134, 3771-3780 (2007).
- Fiddes, I. T., et al. Human-Specific NOTCH2NL Genes Affect Notch Signaling and Cortical Neurogenesis. Cell. 173, 1356-1369 (2018).
- Lennox, A. L., et al. Pathogenic DDX3X mutations impair RNA metabolism and neurogenesis during fetal cortical development. Neuron. 106, 404-420 (2020).
- Martynoga, B., Drechsel, D., Guillemot, F. Molecular control of neurogenesis: A view from the mammalian cerebral cortex. Cold Spring Harbor Perspective Biology. 4 (10), 008359 (2012).
- Amadei, G., et al. A Smaug2-based translational repression complex determines the balance between precursor maintenance versus differentiation during mammalian neurogenesis. Journal of Neurosci. 35, 15666-15681 (2015).
- Yang, G., Smibert, C. A., Kaplan, D. R., Miller, F. D. An eIF4E1/4E-T complex determines the genesis of neurons from precursors by translationally repressing a proneurogenic transcription program. Neuron. 84, 723-739 (2014).
- Yang, G., et al. A Glo1-Methylglyoxal pathway that is perturbed in maternal diabetes regulates embryonic and adult neural stem cell pools in murine offspring. Cell Reports. 17, 1022-1036 (2016).
- Kraushar, M. L., et al. Temporally defined neocortical translation and polysome assembly are determined by the RNA-binding protein Hu antigen R. Proceedings of the National Academy of Science U. S. A. 111, 3815-3824 (2014).
- Rodrigues, D. C., et al. Methylglyoxal couples metabolic and translational control of Notch signalling in mammalian neural stem cells. Nature Communications. 11, 2018 (2020).
- Iwasaki, S., Ingolia, N. T. The growing toolbox for protein synthesis studies. Trends in Biochemical Sciences. 42, 612-624 (2017).
- Chekulaeva, M., Landthaler, M. Eyes on translation. Molecular Cell. 63, 918-925 (2016).
- Faye, M. D., Graber, T. E., Holcik, M. Assessment of Selective mRNA Translation in Mammalian Cells by Polysome Profiling. Journal of Visualized Experiments. (92), e52295 (2014).
- Chassé, H., Boulben, S., Costache, V., Cormier, P., Morales, J. Analysis of translation using polysome profiling. Nucleic Acids Research. 45, 15 (2017).
- Schneider-Poetsch, T., et al. Inhibition of eukaryotic translation elongation by cycloheximide and lactimidomycin. Nature Chemical Biology. 6, 209-217 (2010).
- Kraushar, M. L., et al. Thalamic WNT3 secretion spatiotemporally regulates the neocortical ribosome signature and mRNA translation to specify neocortical cell subtypes. Journal of Neuroscience: Official Journal of Society of Neuroscience. 35, 10911-10926 (2015).
- Gandin, V., et al. Polysome fractionation and analysis of mammalian translatomes on a genome-wide scale. Journal of Visualized Experiments. (7), e51455 (2014).

