Standardized Rat Coronary Ring Preparation and Real-Time Recording of Dynamic Tension Changes Along Vessel Diameter
Summary
The present protocol describes the wire myograph technique for measuring vascular reactivity of the rat coronary artery.
Abstract
As a key event of cardiovascular system diseases, coronary artery disease (CAD) has been widely regarded as the main culprit of atherosclerosis, myocardial infarction, and angina pectoris, which seriously threaten the life and health of people all over the world. However, how to record the dynamic biomechanical characteristics of isolated blood vessels has long puzzled people. Meanwhile, precise positioning and isolation of coronary arteries to measure in vitro dynamic vascular tension changes have become a trend in CAD drug development. The present protocol describes the macroscopic identification and microscopic separation of rat coronary arteries. The contraction and dilation function of the coronary artery ring along the vessel diameter was monitored using the established multi myograph system. The standardized and programmed protocols of coronary ring tension measurement, from sampling to data acquisition, tremendously improve the repeatability of the experimental data, which ensures the authenticity of vascular tension records after physiological, pathological, and drug intervention.
Introduction
Coronary artery disease (CAD) has been widely recognized and concerned as a typical and representative cardiovascular disease, being the leading cause of death in both developed and developing countries1,2. As a blood and oxygen supply route for normal cardiac physiological function, circulating blood enters and nourishes the heart through two main coronary arteries and a blood vascular network on the surface of the myocardium3,4. Cholesterol and fat deposits in the coronary arteries cut off the heart's blood supply and the violent inflammatory response of the vascular system, causing atherosclerosis, stable angina, unstable angina, myocardial infarction, or sudden cardiac death5,6. In response to pathological stenosis of the coronary arteries, compensatory accelerated physiological heartbeat satisfies the blood supply of the heart itself or vital organs of the body by increasing the output of the left ventricle7. If prolonged coronary stenosis is not relieved in time, extensive new blood vessels may develop in certain areas of the heart8. At present, the clinical treatment of CAD often adopts drug thrombolysis or surgical mechanical thrombolysis and an exogenous bionic vascular bypass with frequent medication and great surgical disability9. Therefore, the functional investigation of coronary artery physiological activity is still an urgent breakthrough for cardiovascular diseases10.
There are no available technical means to detect coronary physiological activity, except for wireless telemetry systems, which can dynamically record in vivo coronary pressure, vascular tension, blood oxygen saturation, and pH values11. Therefore, considering coronary arteries' textural secrecy and complexity, accurate identification and isolation of coronary arteries are undoubtedly the best choices for exploring multiple mechanisms of CAD in vitro4.
A series multi myograph system, in particular a wire micrograph microvascular tension detector (see Table of Materials), is a very mature marketable device for recording in vitro tissue tension changes of small vascular, lymphatic, and bronchial tubes with the characteristics of high precision and continuous dynamic recording12. The said system has been extensively employed to record in vitro tissue tension characteristics of cavity structures with diameters of 60 µm to 10 mm. The continuous heating features of the platform of the wire micrograph largely offset the stimulation of the adverse external environment. Meanwhile, the constant inputs of the gas mixture and the pH values allow us to obtain more accurate vascular tension data in a similar physiological state13. However, considering the complexity of anatomical localization of rat coronary arteries (Figure 1), its isolation has been perplexing and limiting the mechanism's exploration of diversified cardiovascular disease and drug development. Therefore, the present protocol introduces the anatomical location and separation process of the rat coronary artery in detail, followed by tension measurement on the platform of the wire micrograph14.
Protocol
The animal protocol was reviewed and approved by the Management Committee from Chengdu University of Traditional Chinese Medicine (Record No. 2021-11). Male Sprague Dawley (SD) rats (260-300 g, 8-10 weeks old) were used for the present study. The rats were kept in an animal chamber and were free to drink and eat during the experiment.
1. Solution preparation
- Prepare physiological salt solution (PSS) by dissolving 118 mM of NaCl, 4.7 mM of K+, 2.5 mM of CaCl2, 1.2 mM of KH2PO4, 1.2 mM of MgCl2∙6H2O, 25 mM of NaHCO3, 11 mM of D-glucose, and 5 mM of HEPES (see Table of Materials).
- Prepare high K+ salt solution by dissolving 58 mM of NaCl, 60 mM of K+, 2.5 mM of CaCl2, 1.2 mM of KH2PO4, 1.2 mM of MgCl2∙6H2O, 25 mM of NaHCO3, 11 mM of D-glucose, and 5 mM of HEPES.
- Saturate the above two solutions and bubble with a mixed gas of 95% O2 and 5% CO2. Meanwhile, maintain the pH values of the solution between 7.38 and 7.42 with 2 mM NaOH.
NOTE: For details information on solution preparation, please see reference15.
2. Rat coronary artery dissection
- Anesthetize the rat by inhalation of 2% isoflurane. Confirm deep anesthesia by toe pinch and, if needed, administer additional anesthetics. Then immediately open the thoracic cavity to expose the heart on the portable operating table following a previously published report12.
- After dissociating and removing the heart, drain the residual blood from all the heart chambers by mildly squeezing with medical plastic forceps. Quickly place the pre-processed heart in a Petri dish containing 95% O2 + 5% CO2 saturated PSS at 4 °C, having a pH value of 7.40.
- To accurately identify the anatomical position of the coronary arteries, adjust the posture of the isolated heart under the light microscope according to the schematic diagram (Figure 2A).
NOTE: On the frontal view, the right auricle and the pulmonary artery were on the upper left and the upper right, respectively.- Cut the left and right ventricular cavities along the interventricular septum from the root of the pulmonary artery with surgical scissors and tweezers (Figure 2B).
- To dissociate the left and right coronary arteries from the myocardial tissue, dissect the right ventricle under an optical anatomic microscope to thoroughly expose the right coronary artery branch. Then identify the position of the left coronary artery by rotating the heart tissue 45° clockwise (Figure 2D).
- After removing the surrounding sticky myocardial tissue, explicitly discern the pulsing left (about 5 mm) and right (about 5 mm) coronary arteries. Separate the coronary arteries in the middle immediately and completely immerse in PSS at 4 °C. Acquire an arterial ring of about 2 mm by vertically cutting the detached artery with anatomical scissors to record the vascular tension under different stimuli (Figure 2E).
3. Suspension and fixation of arterial ring
NOTE: For details on this step, please see reference14.
- Prepare two 2 cm stainless steel wires (see Table of Materials) and pre-soak in 4 °C PSS solution saturated with 95% O2 + 5% CO2. Pass both the wires parallelly through the arterial ring along with the vessel's direction under an optical anatomical microscope and with wires of equal length exposed at both ends of the vascular cavity.
- Fix the arterial ring with the steel wire front and back in the bath of the wire micrograph filled with bubbling PSS with 95% O2 + 5% CO2. Rotate the horizontal screw knob for an appropriate front and rear spacing so that the two wires are horizontal and the arterial ring is in a natural state of relaxation.
- After installing the DMT bath on the thermostatic apparatus, open the data acquisition software (see Table of Materials) to ensure that the corresponding path signal was recorded. Set the following parameters: eyepiece calibration (mm/div): 0.36; target pressure (kPa): 13.3; IC1/IC100: 0.9; online averaging time: 2 s; delay time: 60 s. The steps of arterial ring fixation are shown in Figure 3.
4. Standardization of vascular tension in rat arterial ring
NOTE: For different cavity samples, optimal initial tension was necessary for vessels to maintain exceptional activity in vitro. For details, please see reference15.
- Achieve the optimal initial tension of the arterial ring by applying a reasonable tension along the diameter of the vessel.
NOTE: Based on the previous study16, the maximal agonist-induced tension was accomplished at the factor k value of 0.90 with the initial stretch tension of 1.16 ± 0.04 mN/mm (reference values for different vessel samples: k value, 0.90-0.95; initial tension, 1.16-1.52 mN/mm). - At this point, set the displayed vascular tension value to zero. Afterward, apply a 3 mN pull stimulus to the arterial ring by rotating the spiral axis of the bath.
- After incubation for 1 h in oxygen-saturated PSS buffer at 37 °C, pH 7.40, set the tension value to 0 mN again on the tension control panel of the wire micrograph. The setting process of the initial tension of the arterial ring is shown in Figure 4.
5. Reactivity detection of coronary artery ring
- Perform the contractile activity of the coronary artery ring with the wire myograph technique14, and validate in three separate operations by stimulating with 60 mM of K+ solution for 10 min each.
- After each stimulation, flush the bath with oxygen-saturated PSS until vascular tone returns to its initial state.
NOTE: Only when the tension fluctuation of the three parallel measurements was less than 10%, and the amplitude of each contraction was greater than 1 mN/mm, qualified and highly active arterial rings could be used for further experiments. The activity verification of the rat coronary ring is shown in Figure 5.
6. Post-surgical treatment
- After surgery, euthanize the animals following institutionally approved protocols.
NOTE: For the present study, the animals were euthanized by inhaling excess isoflurane.
Representative Results
Anatomically positioned, rat coronary arteries distributed and hidden deep in myocardial tissue were not easily recognized. By comparing the coronary arteries of humans (Figure 1A) and rats (Figure 1B), rapid and accurate separation of rat coronary arteries was conducted according to the sampling process in Figure 2. After precisely locating the right auricle, pulmonary artery, and apex from the front under an optical microscope, the myocardium was dissected along the solid black line shown in Figure 2A. About 5 mm of the interventricular branch of the coronary artery was clearly exposed to our view. After a fine separation of the sticky myocardium surrounding the ventricular septal artery, a 2 cm wire was used to traverse a 2 mm loop of the coronary artery in the direction of vascular alignment. Instantly, the detached 2 mm coronary ring was then soundly fixed in the DMT bath, as shown in Figure 3. After an initial 3 mN tension was applied to the arterial ring (Figure 4), its tension exceeded more than 2 mN by applying 60 mM K+ in parallel three times (Figure 5). Thus, the procedures above had resulted in an isolated coronary ring with excellent physiological activity.
Cumulative K+ (20, 28, 39, 55, 77, and 108 mM) or U46619 (0.01, 0.03, 0.1, 0.3, and 1 µM) were added to the bath of DMT 620M, resulting in a concentration-dependent increase in in vitro vascular tone. The next concentration of K+ or U46619 (a thromboxane A2 (TP) receptor agonist)15 was added when the vasoconstriction effect reached a plateau. The experimental results are shown in Figure 6A,B. For isolated coronary rings constricted by K+ (60 mM) and U46619 (0.3 µM), the test drug apigenin (1, 3, 10, 30, and 100 µM) caused vasodilation in a surprisingly concentration-dependent manner (Figure 6C).
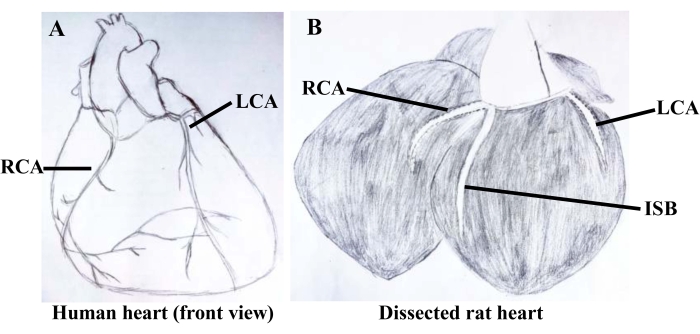
Figure 1: Freehand drawings of human and rat coronary arteries. (A) presents the characteristics of the superficial distribution of left and right coronary arteries from the front view of the human heart and is easily recognized by the naked eye. (B) demonstrates the rat left and right coronary arteries deep in the myocardium and their branching interventricular septum. Abbreviations: RCA = right coronary artery; LCA = left coronary artery; ISB = interventricular septum branch. Please click here to view a larger version of this figure.
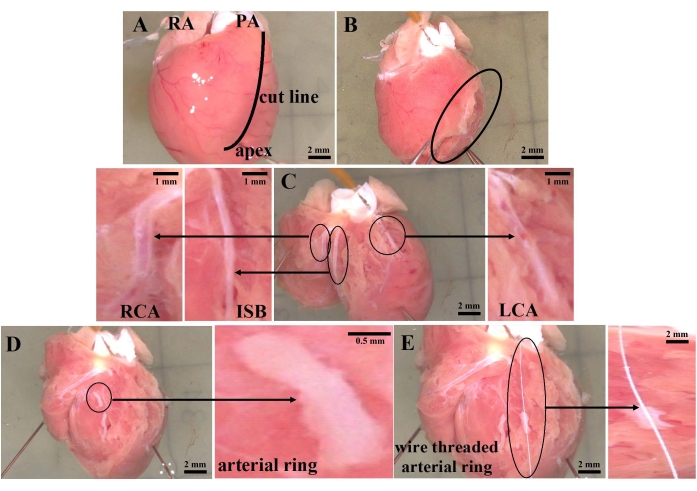
Figure 2: Diagram of coronary artery separation in rats. (A) The right auricle, pulmonary artery, apex, and anatomical line of the rat heart were observed from the front view under a light microscope. (B) The left and right ventricular lumens were incised along the septum from the root of the pulmonary artery. (C) Anatomical location of the left and right coronary arteries and their interventricular septal branch. (D) A 2 mm ring of the artery. (E) The arterial ring is fixed by wire along the direction of the vessel. Abbreviations: RA = right auricle; PA = pulmonary artery; RCA = right coronary artery; ISB = interventricular septum branch; LCA = left coronary artery. Please click here to view a larger version of this figure.

Figure 3: A schematic of arterial mounting procedure. The arterial ring with wire was transferred to (A) and clamped on the DMT bath (B). The steel wire was fixed and screwed clockwise to the upper left (C) and lower left (D). (E) The jaws apart were screwed to make space for allowing the second wire to pass through the arterial ring. (F) The second wire was parallel through the arterial ring. The steel wire was fixed and screwed clockwise to the upper right (G) and lower right (H). (I) The jaws apart were loosely screwed to leave the arterial ring in its natural state. The green lines represent the wires, and the orange cylinders represent 2 mm isolated arterial ring. Please click here to view a larger version of this figure.
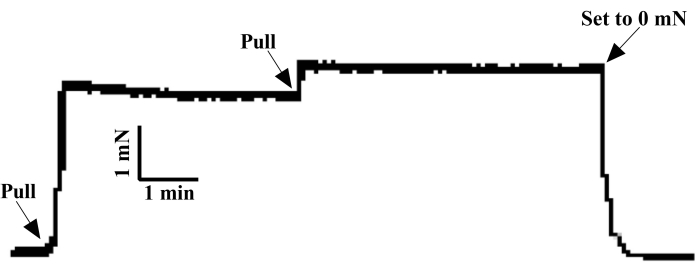
Figure 4: Normalization procedure of arterial ring. After the tension of the fixed isolated arterial ring returned to 0 mN, a 3 mN pull force was applied to the arterial ring at one time. After 5 min, the vascular tension decreased to 2.5 mN. By increasing the tension to 3 mN and holding it steady for 5 min, the tension of the coronary artery ring was initialized to 0 mN and rested for 1 h for subsequent studies on vascular tension of different stimuli. Please click here to view a larger version of this figure.
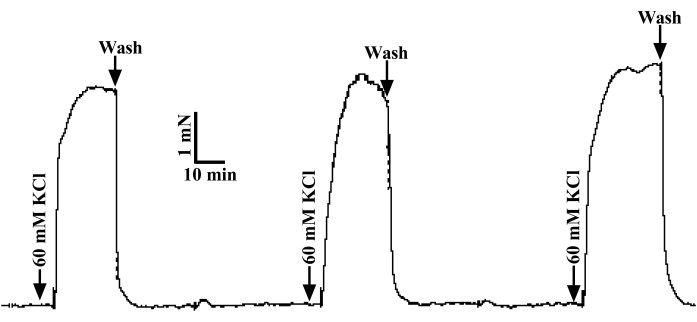
Figure 5: The testing of vascular reactivity. Three applications of 60 mM K+ stimulated the tension of the isolated coronary artery ring to more than 2 mN and the three measurements were less than 10%, suggesting a superior vascular activity. After each stimulation, the bath was gently flushed with a 37 °C oxygen saturated PSS solution until the tension was 0 mN. Please click here to view a larger version of this figure.
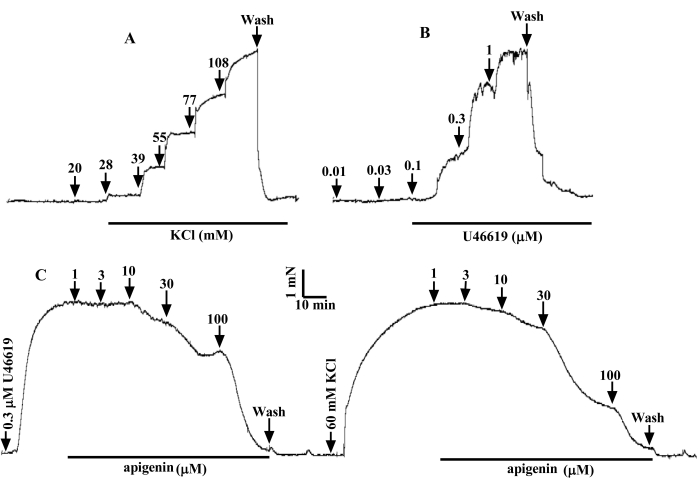
Figure 6: Representative tracer of cumulative dose contraction of rat coronary artery via K+ or U46619. As the dose of K+ (A) and U46619 (B) increased, the force increased dose-dependent. (C) referred to the relaxant effect of apigenin on 60 mM K+– and 0.3 µM U46619-contracted arterial ring in a concentration-dependent manner. Please click here to view a larger version of this figure.
Discussion
The disturbance of coronary microcirculation, which involves a wide range of patients with CAD, has been gradually recognized and concerned the basis for adequate myocardium perfusion. Considering the serious complications of sudden coronary heart disease and cardiovascular disease, timely drug prevention and treatment are extremely important for a clinical individual with CAD17. Inevitably, the secrecy of coronary artery anatomy and the complexity of its physiological structure have severely restricted the rational and scientific evaluation of the effectiveness of drugs and treatments for CAD18,19,20,21,22,23,24,25. Undoubtedly, accurate localization and isolation of active coronary arteries is a prerequisite for promoting the exploration of pathological mechanisms and evaluation of prevention and treatment measures of CAD-related diseases. The platform of the wire micrograph is suitable for unremittingly recording in vitro tissue tension with annular and cavity structures, ranging in diameter from 60 µm to 10 mm. The coronary artery ring can be attached to the chamber by two wires with a constant temperature and oxygen control. The data of vasoconstriction and relaxation after adding different drugs is inputed into the computer through the tension sensor, with data continuously acquired and documented14.
This article mainly describes the rat coronary artery's concrete position and separation process. And the dynamic process of coronary artery tension changes in rats was measured by the wire micrograph system. Given the heterogeneity of human and rat species, we must be aware of these differences when looking for and isolating rat coronary arteries. The rat coronary arteries are divided into left and right arteries with an independent interventricular septal branch. The human coronary arteries are on the surface of the heart, while the rat coronary arteries are slightly deeper. When measuring arterial ring tension, all buffered solution was saturated and bubbled with 95% O2 + 5% CO2 at 37 °C, pH = 7.40. The fixed process of the arterial ring by two wires was introduced in detail. The artery in the body is in a state of micro constriction rather than a state of complete relaxation. And the contractile function of the artery is closely related to the pull force applied to them to some extent. Therefore, it is necessary to standardize the arterial ring so that it is in an optimal preloaded state for maintaining superior vascular physiological activity in the subsequent experiment. Since high K+ condition (60 mM) can depolarize the cell membrane and activate voltage-gated Ca2+ channels, this causes the influx of extracellular Ca2+ and arterial contraction26.
In the test of vasoconstriction and dilation, the contractile effect of K+ or U46619 on rat coronary arteries was investigated. In the results, K+ or U46619 can steadily constrict the coronary arteries of rats in a concentration-dependent manner by acting on ion channels or specific receptors. K+ constricts vessels mainly by depolarizing cell membranes and opening L-type Ca2+ channels27. Meanwhile U46619, an analog of TXA2, constricts vessels primarily by activating circular nucleotide-gated channels and TXA2 receptors. Apigenin, a kind of flavonoid, widely exists in fruits, vegetables, and traditional Chinese medicines (Semen plantaginis and Chinese starjasmine)28. The results declared that apigenin could concentration-dependently dilate the contraction of coronary arteries for 60 mM K+ and 0.3 µM U46619 stimuli. At the end of the experiment, the coronary ring with favorable activity was again validated by adding 60 mM K+, causing vasoconstriction similar to that of the original stimulation. Although the study mainly focused on coronary arteries, the wire micrograph system was also applicable to other extremely small tissue vessels, lymphatics, and bronchus. In conclusion, this article principally described the location and isolation of rat coronary arteries. Meanwhile, its tension changes were measured using the wire micrograph system platform, providing an accurate and reproducible methodology for CAD exploration.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Key R&D project of Sichuan Provincial Science and Technology Plan (2022YFS0438), the National Natural Science Foundation of China (82104533), the China Postdoctoral Science Foundation (2020M683273), and the Science & Technology Department of Sichuan Province (2021YJ0175).
Materials
| Apigenin | Sangon Biotech Co., Ltd., Shanghai, China | 150731 | |
| CaCl2 | Sangon Biotech Co., Ltd., Shanghai, China | A501330 | |
| D-glucose | Sangon Biotech Co., Ltd., Shanghai, China | A610219 | |
| HEPES | Xiya Reagent Co., Ltd., Shandong, China | S3872 | |
| KCl | Sangon Biotech Co., Ltd., Shanghai, China | A100395 | |
| KH2PO4 | Sangon Biotech Co., Ltd., Shanghai, China | A100781 | |
| LabChart Professional version 8.3 | ADInstruments, Australia | — | |
| MgCl2·6H2O | Sangon Biotech Co., Ltd., Shanghai, China | A100288 | |
| Multi myograph system | Danish Myo Technology, Aarhus, Denmark | 620M | |
| NaCl | Sangon Biotech Co., Ltd., Shanghai, China | A100241 | |
| NaHCO3 | Sangon Biotech Co., Ltd., Shanghai, China | A100865 | |
| Steel wires | Danish Myo Technology, Aarhus, Denmark | 400447 | |
| U46619 | Sigma, USA | D8174 |
References
- Malakar, A. K., et al. A review on coronary artery disease, its risk factors, and therapeutics. Journal of Cellular Physiology. 234 (10), 16812-16823 (2019).
- Murray, C., et al. national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: A systematic analysis for the global burden of disease Study 2013. The Lancet. 385 (9963), 117-171 (2015).
- Zhang, Y., et al. Adenosine and adenosine receptor-mediated action in coronary microcirculation. Basic Research in Cardiology. 116 (1), 22 (2021).
- Allaqaband, H., Gutterman, D. D., Kadlec, A. O. Physiological consequences of coronary arteriolar dysfunction and its influence on cardiovascular disease. Physiology. 33 (5), 338-347 (2018).
- Minelli, S., Minelli, P., Montinari, M. R. Reflections on atherosclerosis: Lesson from the past and future research directions. Journal of Multidisciplinary Healthcare. 13, 621-633 (2020).
- Alvarez-Alvarez, M. M., Zanetti, D., Carreras-Torres, R., Moral, P., Athanasiadis, G. A survey of sub-saharan gene flow into the mediterranean at risk loci for coronary artery disease. European Journal of Human Genetics. 25 (4), 472-476 (2017).
- LaCombe, P., Tariq, M. A., Lappin, S. L. Physiology, Afterload Reduction. StatPearls [Internet]. , (2022).
- Gutterman, D. D., et al. The human microcirculation: regulation of flow and beyond. Circulation Research. 118 (1), 157-172 (2016).
- Wang, G., Li, F., Hou, X. Complementary and alternative therapies for stable angina pectoris of coronary heart disease: A protocol for systematic review and network meta-analysis. Medicine. 101 (7), 28850 (2022).
- Markousis-Mavrogenis, G., et al. Coronary microvascular disease: the "meeting point" of cardiology. European Journal of Clinical Investigation. 52 (5), 13737 (2021).
- Allison, B. J., et al. Fetal in vivo continuous cardiovascular function during chronic hypoxia. The Journal of Physiology. 594 (5), 1247-1264 (2016).
- Wenceslau, C. F., et al. Guidelines for the measurement of vascular function and structure in isolated arteries and veins. American Journal of Physiology-Heart and Circulatory Physiology. 321 (1), 77-111 (2021).
- Liu, L., et al. Comparison of Ca2+ handling for the regulation of vasoconstriction between rat coronary and renal arteries. Journal of Vascular Research. 56 (4), 191-203 (2019).
- Sun, J., et al. Isometric contractility measurement of the mouse mesenteric artery using wire myography. Journal of Visualized Experiments. (138), e58064 (2018).
- Guo, P., et al. Coronary hypercontractility to acidosis owes to the greater activity of TMEM16A/ANO1 in the arterial smooth muscle cells. Biomedicine & Pharmacotherapy. 139, 111615 (2021).
- Ping, N. N., Cao, L., Xiao, X., Li, S., Cao, Y. X. The determination of optimal initial tension in rat coronary artery using wire myography. Physiological Research. 63 (1), 143-146 (2014).
- Niccoli, G., Scalone, G., Lerman, A., Crea, F. Coronary microvascular obstruction in acute myocardial infarction. European Heart Journal. 37 (13), 1024-1033 (2016).
- Mumma, B., Flacke, N. Current diagnostic and therapeutic strategies in microvascular angina. Current Emergency and Hospital Medicine Reports. 3 (1), 30-37 (2015).
- Lanza, G. A., Parrinello, R., Figliozzi, S. Management of microvascular angina pectoris. American Journal of Cardiovascular Drugs. 14 (1), 31-40 (2014).
- Zhu, T. Q., et al. Beneficial effects of intracoronary tirofiban bolus administration following upstream intravenous treatment in patients with ST-elevation myocardial infarction undergoing primary percutaneous coronary intervention: The ICT-AMI study. International Journal of Cardiology. 165 (3), 437-443 (2013).
- Huang, D., et al. Restoration of coronary flow in patients with no-reflow after primary coronary intervention of acute myocardial infarction (RECOVER). American Heart Journal. 164 (3), 394-401 (2012).
- Fu, W. J., et al. Anti-atherosclerosis and cardio-protective effects of the Angong Niuhuang Pill on a high fat and vitamin D3 induced rodent model of atherosclerosis. Journal of Ethnopharmacology. 195, 118-126 (2017).
- Li, J., et al. Chinese medicine She-Xiang-Xin-Tong-Ning, containing moschus, corydalis and ginseng, protects from myocardial ischemia injury via angiogenesis. The American Journal of Chinese Medicine. 48 (1), 107-126 (2020).
- Wu, W., et al. Three dimensional reconstruction of coronary artery stents from optical coherence tomography: Experimental validation and clinical feasibility. Scientific Reports. 11 (1), 1-15 (2021).
- Liu, M., et al. Janus-like role of fibroblast growth factor 2 in arteriosclerotic coronary artery disease: Atherogenesis and angiogenesis. Atherosclerosis. 229 (1), 10-17 (2013).
- Hu, G., Li, X., Zhang, S., Wang, X. Association of rat thoracic aorta dilatation by astragaloside IV with the generation of endothelium-derived hyperpolarizing factors and nitric oxide, and the blockade of Ca2+ channels. Biomedical reports. 5 (1), 27-34 (2016).
- Guo, Y., et al. Anticonstriction effect of MCA in rats by danggui buxue decoction. Frontiers in Pharmacology. 12, 749915 (2021).
- Jing, Y., et al. Apigenin relaxes rat intrarenal arteries, depresses Ca2+-activated Cl− currents and augments voltage-dependent K+ currents of the arterial smooth muscle cells. Biomedicine & Pharmacotherapy. 115, 108926 (2019).

