Modified Heterotopic Abdominal Heart Transplantation and a Novel Aortic Regurgitation Model in Rats
Summary
This study demonstrates a reproducible heterotopic abdominal heart transplantation technique in rats that beginners can learn and perform. Additionally, a novel aortic regurgitation model in rats is generated by performing heterotopic abdominal heart transplantation and damaging the donor’s aortic valve using a guidewire after harvesting.
Abstract
Over the past 50 years, many researchers have reported heterotopic abdominal heart transplantation in mice and rats, with some variations in the surgical technique. Modifying the transplantation procedure to strengthen the myocardial protection could prolong the ischemia time while preserving the donor's cardiac function. This technique's key points are as follows: transecting the donor's abdominal aorta before harvesting to unload the donor's heart; perfusing the donor's coronary arteries with a cold cardioplegic solution; and topical cooling of the donor's heart during the anastomosis procedure. Consequently, since this procedure prolongs the acceptable ischemia time, beginners can easily perform it and achieve a high success rate.
Moreover, a new aortic regurgitation (AR) model was established in this work using a technique different from the existing one, which is created by inserting a catheter from the right carotid artery and puncturing the native aortic valve under continuous echocardiographic guidance. A heterotopic abdominal heart transplantation was performed using the novel AR model. In the protocol, after the donor's heart is harvested, a stiff guidewire is inserted into the donor's brachiocephalic artery and advanced toward the aortic root. The aortic valve is punctured by pushing the guidewire further even after the resistance is felt, thus inducing AR. It is easier to damage the aortic valve using this method than with the procedure described in the conventional AR model. Additionally, this novel AR model does not contribute to the recipient's circulation; therefore, this method is expected to produce a more severe AR model than the conventional procedure.
Introduction
Heterotopic abdominal heart transplantation in rats was first reported in 1964 by Abbott et al.1and has been used to study acute and chronic allograft rejection, cardiac allograft vasculopathy, ischemia-reperfusion injury, and cardiac remodeling2,3,4,5,6,7,8,9,10,11. Some modifications have been added to the procedure over the past 50 years. The fundamentals of the current procedure are as follows. The donor's ascending aorta and pulmonary artery (PA) are end-to-side anastomosed to the recipient's abdominal aorta and inferior vena cava, respectively. Although the donor's left atrium and ventricle do not receive any intracavitary flow, blood flows to the donor's coronary system; therefore, the donor's heart starts beating again after de-clamping.
Some experts with experience in hundreds or thousands of operations have reported a high success rate with short ischemia time for heterotopic abdominal heart transplantation2,3,4,5; however, it is difficult for beginners to achieve the short ischemia time from the outset. Sufficient cardioprotection is an important factor for obtaining good cardiac contraction of the donor's heart. Insufficient myocardial protection can stiffen the donor's heart. Therefore, we modified the transplantation procedure to strengthen the protection of the donor's heart. One of the aims of this study is to demonstrate a reproducible heterotopic abdominal heart transplantation procedure that beginners can easily perform since it prolongs the acceptable ischemia time.
Additionally, some researchers have reported an aortic regurgitation (AR) model in rats, which has been used to examine the effects of agents on left ventricular (LV) remodeling12,13,14,15. The conventional procedure includes the following: (1) a right lateral neck incision is made to expose the right carotid artery after anesthesia; (2) a catheter is cannulated from this vessel and advanced toward the aortic root; and (3) AR is induced by puncturing the native aortic valve under continuous echocardiographic guidance.
However, puncturing the aortic valve while holding the echocardiography probe and obtaining a good view of the ascending aorta, the aortic valve, and the catheter with an echocardiogram is challenging. Furthermore, cardiac failure following acute AR is another complication. Therefore, a novel AR model, which can be easily created and does not contribute to the recipient's circulation, has been established in this work to solve these challenges. The other aim of this study is to create an AR model by using heterotopic abdominal heart transplantation and damaging the donor's aortic valve using a guidewire after harvesting.
Protocol
All the animal procedures were conducted in accordance with "An Outline of the Act on Welfare and Management of Animals" and "Standards Relating to the Care and Keeping and Reducing Pain of Laboratory Animals" by the Ministry of the Environment, Government of Japan and the "Guidelines for Proper Conduct of Animal Experiment" by the Science Council of Japan16,17,18. The animal protocols were reviewed and approved by the Institutional Animal Care and Use Committee of the University of Tokyo (M-P19-065).
1. Heterotopic abdominal heart transplantation in rats
NOTE: Heterotopic abdominal heart transplantations were conducted in male Jcl:Wistar rats aged 7-9 weeks old. A microscope with 6.7x to 45x magnification was used to perform the procedure. The surgical instruments were autoclaved for sterilization.
- Donor heart harvest
- Anesthetize the donor rat via an intraperitoneal injection of medetomidine (0.15 mg/kg), midazolam (2.0 mg/kg), and butorphanol (2.5 mg/kg).
- Place the rat in a supine position on a heating pad over the operative board, and fix the limbs using elastic bands. Remove as much fur as possible with an electric shaver and depilatory cream. Subsequently, clean the operating area with iodine and a 70% alcohol scrub three times for disinfection.
- Make a small incision of approximately 1 cm with scissors, followed by administering heparin to the abdomen (100 U).
- Extend the abdominal incision longitudinally from above the external urethral meatus to the subxiphoid process with scissors.
- Expose the abdominal aorta with sterilized cotton swabs, and transect with scissors to unload the donor's heart.
- Incise the diaphragm along the anterior chest wall, and open the thoracic wall with a V-shaped incision using scissors. Elevate the anterior chest wall beside the head, and fix it with pins.
- Initiate topical cooling of the heart using slushed ice.
- Tape the inferior vena cava with a 5-0 silk suture.
- Remove the thymus with scissors, and subsequently identify the ascending aorta and PA originating from the heart. The ascending aorta and PA are on the right and left, respectively.
- Transect the brachiocephalic artery originating from the aortic arch with Potts scissors for additional exsanguination.
- Insert a 23 G needle into the inferior vena cava, and administer 2-3 mL of cold-modified Krebs-Henseleit cardioplegic solution. After the administration, the heart gradually stops beating. Simultaneously, ensure that the blood within the heart is drained out as much as possible.
- Ligate the inferior vena cava with 5-0 silk, and divide it distally to the ligature using Potts scissors.
- After dissecting the connective tissue between the right lung and the esophagus with scissors, ligate the hilum of the right lung with 5-0 silk, and divide it distally to the ligature with scissors.
- Ligate the right superior vena cava with 5-0 silk, and divide it distally to the ligature with Potts scissors.
- Insert one blade of the Potts scissors into the transverse sinus and divide the ascending aorta and PA en bloc as distally as possible.
- Ligate the left superior vena cava with a 5-0 silk suture, and divide it distally to the ligature and proximally to the azygos vein with Potts scissors.
- After dissecting the connective tissue between the left lung and the esophagus with scissors, ligate the hilum of the left lung with a 5-0 silk suture, and divide it distally to the ligature with scissors.
- Ligate the base of the heart with a 5-0 silk suture, and divide it distally to the ligature with scissors. This silk is used to retract the donor's heart during the anastomosis of the donor's heart. At this time, remove the donor's heart from the pericardial cavity.
- After placing the donor's heart on the plate filled with slushed ice and cold normal saline, dissect the connective tissue between the ascending aorta and the PA using micro tweezers.
- Perfuse 2-3 mL of cold-modified Krebs-Henseleit cardioplegic solution from the ascending aorta into the coronary arteries by clamping the ostium of the ascending aorta with tweezers until the red color of the coronary arteries is diminished.
- Store the donor's heart in a cold-modified Krebs-Henseleit cardioplegic solution.
- Recipient preparation
NOTE: Before harvesting the donor's heart, complete the recipient preparation.- Anesthetize the recipient rat via an intraperitoneal injection of medetomidine (0.15 mg/kg), midazolam (2.0 mg/kg), and butorphanol (2.5 mg/kg). After endotracheal intubation using a 16 G intravenous catheter, maintain the anesthesia via sevoflurane inhalation (induction at 5.0% and maintenance at 2.5% with an O2 flow rate of 0.3 L/min).
- Place the rat in a supine position on a heating pad laid over the operative board, and fix the limbs using elastic bands. Remove as much fur as possible with an electric shaver and depilatory cream. Additionally, clean the surgical site with iodine and a 70% alcohol scrub three times for disinfection.
- Using scissors, make a midline abdominal incision of approximately 6-7 cm above the external urethral meatus to the subxiphoid process.
- Retract the small intestine toward the upper-right side of the recipient with sterilized cotton swabs, and wrap it with gauze soaked with warm normal saline.
- Dissect the connective tissue between the small intestine and colon with scissors.
- Dissect the avascular area of the colon with scissors, and tape with two strip-like gauze pieces to retract the colon to the left.
- Expose the abdominal aorta and inferior vena cava with sterilized cotton swabs. Ligate the relatively large branches of both vessels with 9-0 nylon, and divide.
- Heterotopic heart transplantation
- Administer heparin to the recipient's abdomen (100 U) before initiating the donor heart transplantation.
- Clamp the abdominal aorta and inferior vena cava en bloc with side-biting clamp forceps.
- Puncture the abdominal aorta with a 90° bent 23 G needle, and extend the hole longitudinally with Potts scissors to at least the diameter of the donor's ascending aorta. Subsequently, flush the lumen with heparinized normal saline (10 U/mL) to remove clots.
- Position the sterilized small plate on the right side of the recipient's abdominal aorta and inferior vena cava. Place the donor's heart on the plate filled with slushed ice and cold normal saline, which should be replenished every 5 min.
- Tie the donor's ascending aorta to the recipient's abdominal aorta with two 9-0 nylon stay sutures at the 12 o'clock and 6 o'clock positions.
- Rotate the operative board clockwise by 90°.
- Anastomose the left side of the recipient's abdominal aorta and the donor's ascending aorta using a running 9-0 nylon suture from the caudal to the cranial direction, and tie with a 9-0 nylon stay suture at the 12 o'clock position (total of seven to eight sutures).
- Rotate the operative board counterclockwise by 180°. Translocate the donor's heart to the left of the recipient's abdominal aorta and inferior vena cava. It is easy to retract the donor's heart toward the left side with the 5-0 silk suture that was used when ligating the base of the donor's heart (step 1.1.18).
- Anastomose the right side of the recipient's abdominal aorta and the donor's ascending aorta using a running 9-0 nylon suture from the cranial to the caudal direction, and tie with a 9-0 nylon stay suture at the 6 o'clock position (total of seven to eight sutures).
- Puncture the inferior vena cava using a 90° bent 23 G needle distally to the anastomosis between the donor's ascending aorta and the recipient's abdominal aorta, and extend the hole longitudinally with Potts scissors to greater than the diameter of the donor's PA. Subsequently, flush the lumen with heparinized normal saline to eliminate clots.
- Tie the donor's PA to the recipient's inferior vena cava using a 9-0 nylon stay suture at the 6 o'clock position.
- Anastomose the left side of the recipient's inferior vena cava and the donor's PA with a running 9-0 nylon suture from the caudal to the cranial direction (total of seven to eight sutures).
- Tie the donor's PA to the recipient's inferior vena cava with a 9-0 nylon stay suture at the 12 o'clock position. Additionally, tie this stay suture to the 9-0 nylon on the left side of the anastomosis between the donor's PA and the recipient's inferior vena cava.
- Anastomose the right side of the recipient's inferior vena cava and the donor's PA using a running 9-0 nylon suture from the cranial to the caudal direction, and tie with the 6 o'clock stay suture (total of 10-12 sutures).
- Apply hemostatic agents to both the anastomoses, and place gauze around them.
- After releasing the side-biting clamp forceps, compress the anastomoses gently with sterilized cotton swabs to facilitate hemostasis. Subsequently, apply warm normal saline to the donor's heart to provide warmth. The donor's heart begins to fibrillate within a few tens of seconds and recovers to sinus rhythm after a few minutes.
- Return the small intestine to the recipient's abdomen using sterilized cotton swabs. Care should be taken to prevent compressing the donor's heart or twisting the small intestine.
- Close the abdominal wall using a running 4-0 silk suture. After administering atipamezole (0.75 mg/kg) into the abdomen, close the skin with a running 4-0 silk suture.
- Discontinue the inhalation anesthesia, and inject 1 mL of 1% lidocaine under the incision. Additionally, inject 2 mL of warm normal saline subcutaneously to compensate for blood loss.
- Warm the recipient rat using a light-emitting diode lamp. The recipient rat regains consciousness and can be extubated at approximately 30-40 min after inhalation anesthesia is discontinued.
2. Novel AR model using heterotopic abdominal heart transplantation in rats
NOTE: A novel AR model using heterotopic abdominal heart transplantation was generated using male Jcl:Wistar rats aged 7-9 weeks old. A microscope with 6.7x to 45x magnification was used to perform the procedure. The surgical instruments were autoclaved for sterilization.
- Donor heart harvest
NOTE: A modified Petri dish with a hole in the center, pliers, and a stiff guidewire are required to create the new AR model (Figure 1).- As described above, harvest the donor's heart using a similar procedure to the normal transplant in rats, except for the steps concerning the ascending aorta and PA transection (1.1.1-1.1.14, 1.1.16-1.1.17)
- Before transecting the ascending aorta and PA, remove the adipose tissue on the front of both vessels using Potts scissors. Subsequently, dissect the connective tissue between both vessels using tweezers.
- Insert one blade of the Potts scissors into the transverse sinus. Pull the Potts scissors toward the operator, and insert the blade between the ascending aorta and the PA. Subsequently, transect only the PA (Figure 2A), followed by transecting the aorta distally to the brachiocephalic artery using Potts scissors (Figure 2B).
- After ligating the base of the heart using a 5-0 silk suture and dividing it distally to the ligature with scissors, dissect the connective tissue between the ascending aorta and the PA, and perfuse cold-modified Krebs-Henseleit cardioplegic solution into the coronary arteries using a similar procedure to the normal transplant (steps 1.1.18-1.1.20).
- Fix the donor's heart with pliers (Figure 2C). Subsequently, cover the donor's heart with a modified Petri dish with a hole in the center, and immobilize the ascending aorta with a vascular clip.
- Insert a stiff guidewire into the brachiocephalic artery (Figure 2D). Push the guidewire further even after resistance is met, and pierce the aortic valve if a loss of resistance is confirmed. Perform multiple punctures if a severe AR model is to be created.
- Transect the aorta proximally to the brachiocephalic artery to remove the portion clamped with the vascular clip (Figure 2E). Subsequently, store the donor's heart in cold-modified Krebs-Henseleit cardioplegic solution using a procedure similar to the normal transplant (step 1.1.21).
- Recipient preparation
NOTE: Before harvesting the donor's heart, complete the recipient preparation.- As described above, the recipient preparation procedure is similar to that of the normal transplant (steps 1.2.1-1.2.7).
- Heterotopic heart transplantation
- As described above, the donor's heart transplantation procedure is similar to the normal transplantation (steps 1.3.1-1.3.20).
Representative Results
Regarding the normal model, good LV contraction was successfully established after de-clamping. The ischemia time of the transplanted heart and the recipient's anesthesia time were approximately 60 min and 130 min, respectively (Table 1).
Good LV contraction was also obtained after de-clamping in the new AR model. The ischemic time of the transplanted heart and the recipient's anesthesia time in the AR model were approximately 5 min and 10 min longer than the times of the normal model (Table 2). The AR model showed significantly larger LV dimensions and a thinner LV wall than the normal model (Table 3), and postoperative echocardiography detected an AR jet flow in the AR model (Figure 3). Macroscopic examination showed LV dilation and endocardial thickening (Figure 4), and the Masson's Trichrome-stained samples demonstrated fibrotic changes in the myocardium and endocardium (Figure 5). In contrast, these fibrotic changes were not found in the normal model.
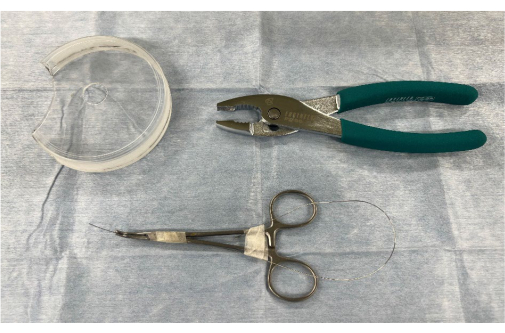
Figure 1: Surgical instruments and materials for creating an aortic regurgitation model using heterotopic abdominal heart transplantation. 1, a modified Petri dish with a hole in the center; 2, pliers; and 3, a stiff guidewire Please click here to view a larger version of this figure.
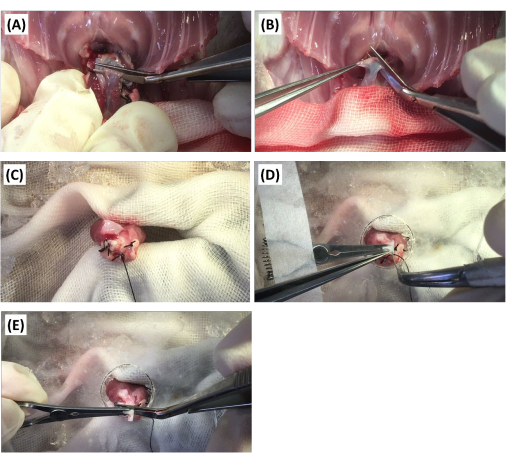
Figure 2: Surgical procedure for the creation of the aortic regurgitation model. (A) The donor's pulmonary artery is transected using Potts scissors. (B) The donor's ascending aorta is transected distally to the brachiocephalic artery with Potts scissors. (C) The donor's heart is fixed with pliers. (D) After the donor's ascending aorta is fixed with a vascular clip, the aortic valve is punctured with a stiff guidewire. (E) The aorta is transected proximally to the brachiocephalic artery using Potts scissors. Please click here to view a larger version of this figure.
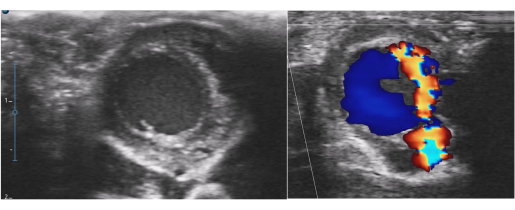
Figure 3: Postoperative echocardiography for the aortic regurgitation model. Left ventricular dilatation and a severe aortic regurgitation jet flow were detected. Please click here to view a larger version of this figure.
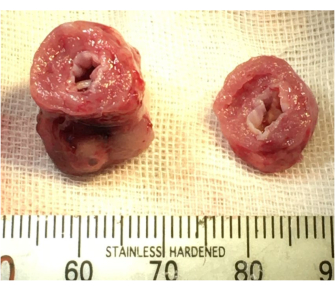
Figure 4: Macroscopic findings of the aortic regurgitation model. Left ventricular dilatation and endocardial thickening were confirmed. Please click here to view a larger version of this figure.
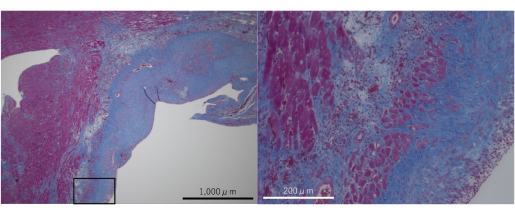
Figure 5: Masson's Trichrome-stained microphotographs of the aortic regurgitation model. Fibrotic changes were confirmed in the myocardium and endocardium. Please click here to view a larger version of this figure.
| Variables | Number | Variables | Number |
| Donor weight (g) | 236.0±40.6 | PA anastomosis time (min) | 18.8±2.7 |
| Recipient weight (g) | 294.6±43.6 | Ischemia time (min) | 59.7±4.8 |
| Clamp time (min) | 48.5±3.0 | ||
| Harvest time (min) | 16.5±2.0 | Anesthesia time (min) | 132.7±8.6 |
| Ao anastomosis time (min) | 26.9±2.7 | Extubation time (min) | 39.0±19.2 |
Table 1: Operative records of the normal model generated using heterotopic abdominal heart transplantation in rats (n = 19). Continuous variables are expressed as the mean ± standard deviation. Abbreviations: Ao = aorta; PA = pulmonary artery
| Variables | Number | Variables | Number |
| Donor’s weight (g) | 211.5±46.9 | PA anastomosis time (min) | 18.8±2.1 |
| Recipient’s weight (g) | 261.2±42.0 | Ischemia time (min) | 65.7±7.2 |
| Clamp time (min) | 49.3±4.9 | ||
| Harvest time (min) | 17.3±2.2 | Anesthesia time (min) | 143.7±14.6 |
| Ao anastomosis time (min) | 28.2±3.6 | Extubation time (min) | 28.0±14.5 |
Table 2: Operative records of the aortic regurgitation model generated using heterotopic abdominal heart transplantation in rats (n = 40). Continuous variables are expressed as the mean ± standard deviation. Abbreviations: Ao = aorta; PA = pulmonary artery
| Variables | Normal model | AR model | P value |
| LV wall (mm) | 3.05±0.50 | 2.19±0.57 | 0.002 |
| LVDd (mm) | 2.23±0.55 | 4.56±2.13 | 0.003 |
| LVDs (mm) | 1.32±0.34 | 3.30±1.79 | 0.003 |
| LV-FS (%) | 40.49±9.41 | 29.06±8.24 | 0.008 |
Table 3: Postoperative echocardiography data of the normal and AR models created using heterotopic abdominal heart transplantation in rats. Continuous variables are expressed as the mean ± standard deviation. A Student's t-test was used to compare the differences between the two groups (P < 0.05). Abbreviations: AR = aortic regurgitation; FS = fractional shortening; LV = left ventricular; LVDd = left ventricular end-diastolic diameter; LVDs = left ventricular end-systolic diameter.
Discussion
Key steps were discovered to prevent the donor heart from stiffening during implantation. First, it is vital to transect the donor's abdominal aorta before harvesting to unload the donor's heart4,7. If the donor's surgical procedure is performed without endotracheal intubation, breathing ceases after the thoracotomy, which obstructs the donor's pulmonary circulation. Consequently, the donor's heart becomes overloaded, preventing good contraction of the donor's heart after de-clamping. Second, perfusing the donor's coronary arteries with the cardioplegic solution is crucial8. Therefore, the cardioplegic solution should be perfused into the coronary arteries by clamping the ostium of the ascending aorta using tweezers after harvesting the donor's heart until the red color of the coronary arteries is diminished. Third, topical cooling of the donor's heart with slushed ice and cold normal saline is necessary. The donor's heart easily becomes warm and stiff when touching the bowel. By placing the donor's heart on a small plate, slushed ice can be placed around it. Additionally, the donor's heart can be submerged in cold normal saline during the anastomosis. Furthermore, the amount of slushed ice and cold normal saline needed can be minimized, thus preventing excessive cooling of the recipient. Moreover, using side-biting forceps to clamp the abdominal aorta and inferior vena cava is convenient7. These side-biting clamp forceps enable simultaneous clamping of the branches of both vessels and prevent backflow from these branches, thus simplifying the anastomosis of the donor's heart.
Experts with abundant experience in performing such operations can perform heterotopic abdominal heart transplantation with a short ischemia time. Plenter et al. reported a minimum ischemia time of approximately 35-45 min2. In the hands of Niimi, the ischemia time was consistently under 35 min3. Additionally, Westhofen et al. showed that the cold/warm ischemia time improved from 45 min/100 min to 10 min/20 min4. They performed heterotopic abdominal heart transplantation using mice rather than rats; however, their ischemia times were short. Therefore, completing the transplantation procedure within this short ischemia time appears difficult for beginners. This study's transplantation procedure showed approximately 60 min of ischemia time (Table 1), and all cases had good LV contraction after de-clamping because of the strengthened myocardial protection. Therefore, beginners can perform this study's procedure and achieve a high success rate.
Nevertheless, dozens of operations are required for the success of heterotopic abdominal heart transplantation. In this study, 62 operations were required to establish the transplant procedure and achieve a high success rate. Additionally, whether beginners can perform the transplant procedure easily by watching this study's video is yet to be proven. It would be useful and positive if the process could assist beginners.
Additionally, in this work, a novel AR model in rats was established using heterotopic abdominal heart transplantation and by damaging the donor's aortic valve using a guidewire after harvesting the donor's heart. Only two studies by Shimada et al. (the first author of both studies is a co-author of the present study) have reported AR models using heterotopic heart transplantation in rats19,20. Some important aspects should be considered when generating the AR model. First, the tool to puncture the aortic valve is important. The aortic valve of adult rats (approximately 200 g) is relatively sturdy; therefore, in our work, it was difficult to damage the aortic valve using a soft guidewire. In contrast, the aortic valve was easily pierced using a 23 G needle, although the risk of injury to other tissues was high, and few recipients could survive after the transplantation procedure because of bleeding. Therefore, a stiff guidewire was selected (Figure 1 and Figure 2). Second, immobilizing the donor's heart and the ascending aorta is important. Initially, the aortic valve was punctured while the ascending aorta wall was grasped using micro tweezers. However, damage to the ascending aorta wall was frequently observed due to excessive traction. Therefore, the donor's heart and ascending aorta were immobilized using a modified Petri dish with a hole in the center, pliers, and a vascular clip. (Figure 1 and Figure 2). Notably, the risk of injury to the ascending aortic wall was reduced after introducing this method.
This study's novel AR model has some benefits. First, with this model, it was possible to puncture the aortic valve more easily and in a shorter time compared to using the traditional AR model. The ischemia time in the AR model was only approximately 5 min longer than in the normal model (Table 1 and Table 2). Moreover, this method can produce various models with different degrees of AR by changing the number of punctures. Since this model does not contribute to the recipient's circulation, the recipient can survive even when the donor's heart shows severe AR (Figure 3).
Notably, the severe AR model showed fibrotic changes in the myocardium and endocardium due to severe AR jet flow (Figure 4 and Figure 5). Therefore, this model may contribute to studies on the pathomechanisms of myocardium and endocardium fibrosis and the evaluation of anti-fibrotic agents.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to thank Editage (www.editage.com) for the English language editing.
Materials
| Antisedan (atipamezole) | Nippon Zenyaku Kogyo Co., Ltd. | ||
| Domitor (medetomidine) | Nippon Zenyaku Kogyo Co., Ltd. | ||
| Dormicum (midazolam) | Maruishi Pharmaceutical Co., Ltd. | ||
| heparin | AY Pharmaceuticals Co.,Ltd. | ||
| Jcl:Wistar rats | CLEA Japan, Inc. | ||
| microscope | Orinpas Co., Ltd. | SZ61 | |
| modified Krebs-Henseleit cardioplegic solution | Merck KGaA | ||
| sevoflurane | FUJIFILM Wako Pure Chemical Corporation | ||
| SURGICEL FIBRILLAR | Johnson & Johnson K.K. | ||
| Vetorphale (butorphanol) | Meiji Animal Health Co., Ltd. |
References
- Abbott, C. P., Lindsey, E. S., Creech, O., Dewitt, C. W. A technique for heart transplantation in the rat. Archives of Surgery. 89, 645-652 (1964).
- Plenter, R. J., Zamora, M. R., Grazia, T. J. Four decades of vascularized heterotopic cardiac transplantation in the mouse. Journal of Investigative Surgery. 26 (4), 223-228 (2013).
- Niimi, M. The technique for heterotopic cardiac transplantation in mice: experience of 3000 operations by one surgeon. The Journal of Heart and Lung Transplantation. 20 (10), 1123-1128 (2001).
- Westhofen, S., et al. The heterotopic heart transplantation in mice as a small animal model to study mechanical unloading – Establishment of the procedure, perioperative management and postoperative scoring. PLoS One. 14 (4), 0214513 (2019).
- Hasegawa, T., et al. Heterotopic vascularized murine cardiac transplantation to study graft arteriopathy. Nature Protocols. 2 (3), 471-480 (2007).
- Wang, C., Wang, Z., Allen, R., Bishop, G. A., Sharland, A. F. A modified method for heterotopic mouse heart transplantation. Journal of Visualized Experiments. (88), e51423 (2014).
- Weigle, C. A., et al. An immunological model for heterotopic heart and cardiac muscle cell transplantation in rats. Journal of Visualized Experiments. (159), e60956 (2020).
- Liu, F., Kang, S. M. Heterotopic heart transplantation in mice. Journal of Visualized Experiments. (6), e238 (2007).
- Corry, R. J., Winn, H. J., Russell, P. S. Primarily vascularized allografts of hearts in mice. The role of H-2D, H-2K, and non-H-2 antigens in rejection. Transplantation. 16 (4), 343-350 (1973).
- Ono, K., Lindsey, E. S. Improved technique of heart transplantation in rats. The Journal of Thoracic and Cardiovascular Surgery. 57 (2), 225-229 (1969).
- Plenter, R. J., Grazia, T. J. Murine heterotopic heart transplant technique. Journal of Visualized Experiments. (89), e51511 (2014).
- Munakata, H., et al. Aortic conduit valve model with controlled moderate aortic regurgitation in rats: A technical modification to improve short- and long-term outcome and to increase the functional results. Circulation Journal. 77 (9), 2295-2302 (2013).
- Eskesen, K., et al. Sildenafil treatment attenuates ventricular remodeling in an experimental model of aortic regurgitation. Springerplus. 4, 592 (2015).
- Plante, E., et al. Effectiveness of beta-blockade in experimental chronic aortic regurgitation. Circulation. 110 (11), 1477-1483 (2004).
- Plante, E., et al. Left ventricular response to sustained volume overload from chronic aortic valve regurgitation in rats. Journal of Cardiac Failure. 9 (2), 128-140 (2003).
- An Outline of the Act on Welfare and Management of Animals. Ministry of the Environment, Government of Japan Available from: https://www.care.nagoya-u.ac.jp/statute/public/02OutlineAct.pdf (2007)
- Standards Relating to the Care and Keeping and Reducing Pain of Laboratory Animals. Ministry of the Environment, Government of Japan Available from: https://www.env.go.jp/nature/dobutsu/aigo/2_data/laws/nt_h25_84_en.pdf (2013)
- Guidelines for Proper Conduct of Animal Experiment. Science Council of Japan Available from: https://www.scj.go.jp/ja/info/kohyo/pdf/kohyo-20-k16-23.pdf (2006)
- Shimada, S., et al. Distention of the immature left ventricle triggers development of endocardial fibroelastosis: An animal model of endocardial fibroelastosis introducing morphopathological features of evolving fetal hypoplastic left heart syndrome. Biomed Research International. 2015, 462469 (2015).
- Shimada, S., et al. Development of a vascularized heterotopic neonatal rat heart transplantation model. European Surgical Research. 57 (3-4), 240-251 (2016).

