Exploring the Two Herb Combination Strategy to Treat Injured PC12 Cells
Summary
This protocol provides a combination strategy of two herbs to treat injured PC12 cells. The protocol provides a reference for optimizing the best application mode of traditional Chinese medicine (TCM).
Abstract
In view of the advantages of the combination of traditional Chinese medicine (TCM) in the treatment of cerebral ischemia, we studied the differences in the efficacy and mechanism between the preparation combination and the component combination in order to explore the two herb combination strategy to treat injured PC12 cells. Cobalt chloride (CoCl2) combined with a glucose-free medium was employed to induce oxidative damage of PC12 cells. Then, the optimal combination of Astragalus mongholicus (Ast) and Erigeron breviscapus (Eri) injection was selected and combined following uniform design methods after screening their safe and effective concentration on PC12 cells. Further, the component combination screened comprises 10 µM astragaloside A, 40 µM scutellarin, and 75 µM chlorogenic acid in two herbs. Then, MTT, Annexin V-FITC/PI, immunofluorescence, and Western blot analysis were used to evaluate the efficacy and mechanism of the preparation combination and the component combination on injured PC12 cells. The results showed that the optimal preparation combination for cell pro-survival was Ast injection and Eri capsule with a concentration of 6:1.8 (µM). The component combination (10 µM astragaloside A, 40 µM scutellarin, and 75 µM chlorogenic acid) was more effective than the preparation combination. Both combinations remarkably reduced apoptotic rate, the fluorescence intensity of caspase-3, and intracellular reactive oxygen species (ROS) level; meanwhile, they upregulated the expression levels of p-Akt/Akt, Bcl-2/Bax, and Nrf2. These effects were more evident in the component combination. In conclusion, both combinations can inhibit the injury induced by CoCl2 combined with a glucose-free medium on PC12 cells, thus promoting cell survival. However, the efficiency of the component combination over the preparation combination may be due to its stronger regulation of the PI3K/Akt/Nrf2 signaling pathway related to oxidative stress and apoptosis.
Introduction
Chronic cerebral ischemia, caused by cerebral hypoperfusion, is a common disease in middle-aged and elderly people1. As a long-term occult ischemia disease, it can lead to progressive or persistent neurological dysfunction. The main pathological mechanisms include cell apoptosis and oxidative damage, leading to progressive or persistent neurological dysfunction; this is the pathological basis of Alzheimer’s disease, vascular dementia, and other diseases, and seriously affects the quality of life of patients. However, there is still a lack of ideal drugs in modern medicine to treat chronic cerebral ischemia2. At the same time, the combination of Astragalus mongholicus (Ast) and Erigeron breviscapus (Eri) has been widely used in the clinical practice of traditional Chinese medicine (TCM)4. The combination strategy has remarkably improved in promoting the recovery of nerve function after cerebral ischemia, which is better than that of a single drug; however, there are large differences in the dosage ratio in the combination of the two drugs4. The effective components and mechanism of action are not well defined, which is the key issue restricting its clinical application.
Previous studies have demonstrated the synergistic effect of the preparation combination of Ast injection and Eri injection in treating cerebral ischemia in rats. It can upregulate the expression of p-Akt protein and downregulate the expression of Bcl-2 associated death promoter (BAD) protein5,6, which is one of the B-lymphoblastoma 2 (Bcl-2) family proteins and has the effect of promoting apoptosis. However, the material basis and mechanism of its synergistic effect are not clear. Further, the injury model of PC12 cells was established with CoCl2 combined glucose-free medium, and the optimal component combination in Ast and Eri has been screened and defined7.
In this study, the effective doses of Ast and Eri are screened by using the injured PC12 cell model. The optimal combination of the two drugs is screened using this model combined with the homogeneous design method. The cell model is used to further evaluate the difference in the effects and mechanisms of the preparation combination and the component combination in injured PC12 cells. This strategy aims to explore the protective effect and regulatory mechanism of the two types of combinations on injured PC12 cells through the PI3K/Akt/Nrf2 signal pathway to determine the best combination mode. This study provides a reference for optimizing the best application mode of TCM.
Protocol
1. Preparation of reagent
- Prepare the complete medium by adding 90 mL of DMEM high sugar medium, 10 mL of fetal bovine serum (FBS), and 1 mL of penicillin-streptomycin solution in a 125 mL sterile culture flask. Mix the complete medium, and store at 4 °C under closed conditions.
- Prepare CoCl2 solution by adding 0.02 g of CoCl2 powder (accurately weighed) in 10 mL of DMEM sugar-free medium (dissolved completely) to obtain a 8.4 mM stock solution. Filter the solution with a 0.22 µm bacterial filter, seal it, and store it at 4 °C away from light.
NOTE: CoCl2 is unstable and should be stored in an airtight container, protected from light; the validity period of the CoCl2 solution is 7 days. - Prepare Tris-Hcl buffered salt solution (TBST).
- Accurately weigh 8 g of sodium chloride, 0.2 g of potassium chloride, and 3 g of Tris-base. Add them into a 1 L beaker, and then add 1,000 mL of RO water to dissolve the mixture.
- Adjust the pH to 7.4, add 1 mL of Tween 20, and thoroughly mix to obtain the TBST solution.
NOTE: Tween 20 acts as a surfactant to reduce the nonspecific binding of antibodies to antigens and works best at 0.1% concentration.
- Prepare MTT (3-(4,5-Dimethylthiazol-2)-2,5-diphenyltetrazolium bromide salt) solution by weighing 0.1 g of MTT powder in 20 mL of PBS solution to obtain a 5 mg/mL stock solution. Filter the solution with a 0.22 µm bacterial filter, seal it, and store it at 4 °C away from light, placed on standby.
NOTE: MTT powder is unstable and easily absorbs moisture, so it should be stored in a closed and dry place away from light. The MTT solution expires after 7 days. MTT has a carcinogenic effect, so avoid direct contact with the skin. - Prepare the Eri capsule solution by removing the capsule shell and accurately weighing 0.18 g of the content in 10 mL of DMEM sugar-free medium to prepare a 100 µM stock solution (based on the concentration of scutellarin). Filter the solution with a 0.2 µm bacterial filter, seal it, and store it in an airtight container at 4 °C.
NOTE: Scutellarin is the main active ingredient in Erigeron breviscapus. The concentration of scutellarin in the capsules has been determined by HPLC-UV to be 2.56 mg/g, i.e., 5.54 µmol/g8. The concentration of the preparation is calculated based on the scutellarin concentration. This is convenient for evaluating the pharmacological activity of the preparation and the component. - Prepare the solution of the Ast injection by adding 5 mL of injection and 5 mL of sugar-free DMEM media in a 15 mL sterile centrifuge tube to prepare a 60 µM stock solution (based on the concentration of astragaloside A). Filter the solution through a 0.2 µm bacterial filter, and store it in an airtight container at 4 °C.
NOTE: The volume of injection is 10 mL each, and the concentration of astragaloside A in the injection has been determined by HPLC-ELSD to be 0.094 mg/mL (i.e., 120 µM)9. The preparation should be carried out in a biosafety cabinet and operated in a light-proof manner throughout. The volume of the injection solution and sugar-free medium should be measured accurately and mixed well. - Prepare astragaloside A solution by accurately weighing 7.85 mg of astragaloside A standard and dissolving it completely in 2 mL of dimethyl sulfoxide (DMSO) to prepare a 5,000 µM stock solution. Filter it with a 0.22 µm bacterial filter and store it airtight at 4 °C.
NOTE: Astragaloside A powder is insoluble in the sugar-free medium. When preparing the solution, dissolve it with the appropriate amount of DMSO to keep the final experimental concentration of DMSO below 0.1% to ensure no cytotoxicity. - Prepare the scutellarin solution by weighing 11.56 mg of scutellarin standard accurately and completely dissolving it with 5 mL of sugar-free DMEM medium to prepare a 5,000 µM stock solution. Filter it with a 0.22 µm bacterial filter and store it airtight at 4 ˚C.
- Prepare the chlorogenic acid solution by weighing 8.86 mg of chlorogenic acid standard accurately and completely dissolving it with 5 mL of sugar-free DMEM medium to prepare a 5,000 µM stock solution. Filter it with a 0.22 µm bacterial filter and store it airtight at 4 ˚C.
NOTE: Chlorogenic acid powder is unstable and easily absorbs water. It should be sealed and stored at 4 °C away from light; the exposure time should be minimized during weighing.
2. Cell viability assay
- Obtain PC12 cells and culture them in DMEM supplemented with 10% FBS, 100 U/mL of penicillin, and 100 µg/mL streptomycin.
NOTE: In this study, the cells are obtained from the Chinese Academy of Sciences. PC12 cells are adherent cells that have the general characteristics of neuroendocrine cells and are widely used in neurophysiology and neuropharmacology. - Culture the cells at 37 °C in an incubator supplemented with 5% CO2 and sub-culture them every 2-3 days. Use cells at passages 4-8 for all experiments.
- Cell culture
- Treat PC12 cells in the logarithmic growth phase (70%-80% cell confluency) with 1 mL of trypsin (0.25%) and observe the cells under a microscope. When the cells become round, stop the trypsin digestion by adding 3 mL of the complete medium.
- Transfer the cells to a sterile 15 mL centrifuge tube and centrifuge the cells at 840 x g for 5 min. Discard the supernatant, resuspend with the complete medium, and transfer the cell suspension to a 1.5 mL microcentrifuge tube. Then count the number of cells using a flow cytometer.
- Start the flow cytometry software, select the corresponding dilution multiple of the cell solution in the counting setting, and click on Density Chart after loading the cell sample from the 1.5 mL microcentrifuge tube.
- Circle the cell population away from the X-axis and Y-axis, right-click the data table below the figure, and select X, Y, Count, and Abs Count. The data under Abs Count in the data table gives the cell counting result.
- Adjust the cell concentration to 1 x 105 cells/mL, inoculate 100 µL/well in 96-well plates, and culture at 37 °C and 5% CO2 in an incubator for 24 h.
- Screening of safe concentrations of two drugs on normal PC12 cells
- Culture the cells as described in steps 2.1-2.2. Discard the supernatant, and treat the normal cells with different final concentrations of Ast injection (4, 8, 12, 16, 20, and 24 µM) and Eri capsule (0.625, 1.25, 2.5, 5, 10, and 20 µM). Treat six replicate wells of each group for 24 h.
NOTE: The drugs are added to the media to achieve the final concentration of the drug (mentioned in step 2.4.1), and then an equal volume of media is added to each well. When aspirating the supernatant, the pipette tip should be placed gently against the well wall to avoid contacting the cells at the bottom of the well. When adding different solutions, add them gently and quickly along the edges of the wells, and pay attention when changing the pipette gun head to avoid affecting the experimental results. - Discard the supernatant, add 120 µL of MTT solution (1 mg/mL) to each well, and incubate the cells for 4 h at 37 °C.
- After the incubation, discard the supernatant again and add 150 µL of DMSO to each well. Shake the plate for 10 min at a speed of 240 times/min (number of horizontal vibrations per minute) using a vortex oscillator.
- Measure the absorbance of each sample at 490 nm by using a microplate reader. Calculate the cell viability (%), which is the absorbance of the treated group/absorbance of normal the group (control) x 100.
NOTE: Ensure that the MTT solution is within the validity period; if the color has changed (to dark green), it cannot be used. When testing the absorbance of cells, a blank well (culture medium and MTT, without cells or drugs) should be set to eliminate the interference of other reagents. A volume of 150 µL of DMSO is added to each well and shaken for 10 min to dissolve the blue-purple crystals better. The absorbance should be detected as soon as possible to ensure the accuracy of the results.
- Culture the cells as described in steps 2.1-2.2. Discard the supernatant, and treat the normal cells with different final concentrations of Ast injection (4, 8, 12, 16, 20, and 24 µM) and Eri capsule (0.625, 1.25, 2.5, 5, 10, and 20 µM). Treat six replicate wells of each group for 24 h.
- Screening of effective concentrations of two drugs on injured PC12 cells
- Culture the cells for 24 h as mentioned in steps 2.1-2.2, discard the supernatant, and then treat the cells with 20 µL/well of CoCl2 (0.4 mM) combined with the sugar-free medium.
- Simultaneously treat the cells with 100 µL/well of Ast injection (final concentrations are 2, 6, 8, 10, and 12 µM) or 100 µL of Eri capsule (final concentrations are 1, 2, 3, 4, and 5 µM) at 37 °C for another 24 h. Add 120 µL/well of complete medium to the control group.
NOTE: CoCl2 induces hypoxic conditions in the cells. - Measure the absorbance of each sample by the MTT method and calculate the cell viability as described previously in steps 2.4.2 and 2.4.3.
- Screening of the optimal combination of two drugs based on the homogeneous design method
NOTE: After obtaining the effective concentration range of the Astragalus injection and breviscapus capsule, the uniform design method U7 (74) table was used to obtain six different combinations of the two drugs. Ast injection:Eri capsule: 1) 2:2.6 µM, 2) 4:5 µM, 3) 6:1.8 µM, 4) 8:4.2 µM, 5) 10:1 µM, and 6)12:3.4 µM.- Culture the cells as described above in step 2.3.1, and treat the injured PC12 cells with six proportion concentrations of Ast injection:Eri capsule as mentioned above.
- Treat the cells for 24 h and calculate the cell viability as described previously in steps 2.4.2 and 2.4.3.
- Evaluation of the protective effect of two optimal combinations on injured PC12 cells
NOTE: After obtaining the optimal preparation combination (Ast injection:Eri capsule of 6:1.8 µM) and the optimal component combination7 (10 µM astragaloside A, 40 µM scutellarin, and 75 µM chlorogenic acid), further evaluation is performed to check their protective effects on injured PC12 cells.- Culture the cells as described above in step 2.3.1, and treat the injured PC12 cells with the two types of combinations of drugs as discussed in the NOTE above.
- Treat the cells for 24 h and calculate the cell viability as described previously in steps 2.4.2 and 2.4.3.
3. Annexin V-FITC/PI assay for apoptosis rate
- Seed the PC12 cells in a 12-well plate at a concentration of 1.3 x 105 cells/well and culture them at 37 °C for 24 h.
- Group the cells: control group, model group, and two combinations of drugs. Add 1.2 mL/well of complete medium to the control group, add 1.2 mL/well of media containing 0.4 mM CoCl2 to the model group, and add 1.2 mL/well of each of the two combinations containing 0.4 mM CoCl2. Incubate the cells at 37 °C for another 24 h.
- Following drug treatment, treat the cells with 250 µL/well of trypsin, transfer the cells in a 15 mL centrifuge tube, and centrifuge at 840 xg for 5 min at room temperature (RT). Discard the supernatant and resuspend the pellet in 500 µL of the binding buffer.
- Incubate the cells in the dark with 5 µL of Annexin V-FITC and 10 µL of propidium iodide (PI) for 15 min at RT, and then check for apoptosis by flow cytometry. Set three replicate wells in each group.
NOTE: The trypsin used in this test cannot contain EDTA because EDTA is a metal ion chelating agent that competes with annexin V to bind calcium ions and affects the affinity of annexin for PI. When apoptosis occurs, phosphoserine, originally on the inner side of the cell membrane, turns outward to the cell surface, and Annexin V-FITC selectively binds to phosphoserine outside the membrane to show green fluorescence. - Click Automatic Compensation in the Start menu, select FITC, PE, PerCP, and APC in the Select channel, and select Compensation at: Height. Click OK in the Statistical Item: Median. With the same cell counting method mentioned in step 2.3.2, collect the cell samples, click the Density Map, and circle the effective cell population.
- With FITC fluorescence as the X-axis parameter, and other fluorescence parameters as the Y-axis parameter, create a density map. Click on Compensation Matrix in the Start menu and Coefficient in the overflow matrix. The density map information is displayed for the analysis of the apoptosis rate.
- Incubate the cells in the dark with 5 µL of Annexin V-FITC and 10 µL of propidium iodide (PI) for 15 min at RT, and then check for apoptosis by flow cytometry. Set three replicate wells in each group.
4. Immunofluorescence detection of caspase-3 generation
- Seed the PC12 cells onto coverslips at a density of 8 x 104 cells/coverslip and place them into 24-well plates at 37 °C for 24 h. Group the cells as mentioned above in step 3.2; set up three replicate coverslips for each group.
- Following drug treatment, rinse the coverslips twice with PBS (37 °C) for 5 min each, fix them with 4% paraformaldehyde for 15 min, and permeabilize with 0.5% Triton X-100 for 20 min at RT.
- Rinse the cells again and block them with 10% goat serum for 1 h at RT.
- Incubate each coverslip with 200 µL of primary antibody caspase-3 (a key executive protein of apoptosis) diluted at a concentration of 1:250 in 1x TBST, overnight at 4 °C. Wash with 1x TBST (three times for 3 min each), and incubate with 200 µL of secondary antibody (1:300 dilution in 1x TBST) for 1 h at RT in the dark.
NOTE: The fluorescence is easily quenched; thus, after incubation of the secondary antibody, the slides should be kept away from light. The primary and secondary antibodies should be stored at 4 °C away from light. - Add 300 µL of DAPI (0.5 µg/mL) to the coverslips (to detect the nuclei) for 10 min and wash the cells three times with 1x TBST for 5 min each.
- Observe the coverslips under the fluorescence microscope and capture images10. Perform statistical analysis of the fluorescence intensity using the imaging software.
NOTE: The slides and coverslips used should be clean and sterile. When staining, pay attention to the pH, concentration, and temperature of the dyeing solution, and avoid fluorescence quenching. The fluorescent microscope should be turned on at least 15 min in advance to preheat the mercury lamp and turned off for 30 min before being turned on again. When acquiring images, the exposure parameters of the same dye in the same batch of experiments should be consistent to ensure the accuracy of the results.
5. Immunofluorescence assay of ROS level
- Culture and treat the cells as discussed in step 4.1.
- Following drug treatment, wash each coverslip three times with PBS for 3 min, and incubate with 400 µL of DCFH-DA (10 µM) at 37 °C for 20 min.
NOTE: DCFH-DA is a universal indicator of oxidative stress. It has cell membrane permeability and no fluorescence. Once it enters the cell, it is hydrolyzed by esterase to produce 2',7'-dichlorodihydrofluorescein (DCFH) and then rapidly oxidized to produce a strong fluorescent product. - Wash the cells again with serum-free DMEM (twice for 3 min each) and immediately observe the coverslip after adding the fluorescence quenching agent under the fluorescence microscope. Calculate the relative fluorescence intensity by the imaging software.
NOTE: After loading the DCFH-DA probe, be sure to clean the residual probe that does not enter the cell, otherwise the background will become high.
6. Western blot detection of protein expression of Nrf2, p-Akt, Akt, Bcl-2, and Bax11,12
- Inoculate PC12 cells in 6-well plates at a concentration of 1 x 106 cells/well and culture at 37 °C for 24 h. Group and treat the cells as aforementioned in step 4.1, and set three replicate wells in each group.
- After drug treatment for 24 h, wash the cells three times with PBS for 5 min each and incubate on ice for 30 min in prepared lysis buffer. Following lysis, centrifuge the cells for 20 min at 16,000 x g and 4 °C and collect the supernatants.
- Detect the protein concentration and adjust according to the instructions through the Bradford assay. Dilute the samples and denature at 100 °C for 10 min.
NOTE: The lysis buffer is composed of phosphatase inhibitor, protease inhibitor, and RIPA lysate in a volume ratio of 1:1:50. In the control group, 200 µL/well of lysis buffer is required, and 100 µL/well of lysis buffer is required in the other groups. Generally, the protein concentrations of control, model, and two types of combinations groups are 0.45 mg/mL, 0.25 mg/mL, and 0.32-0.39 mg/mL respectively; their protein concentrations after dilution with the loading buffer are 0.36, 0.2, and 0.256-0.312 mg/mL, respectively. - To detect proteins with different molecular weights, load 25 µg of protein in each lane, run a 12% SDS-PAGE electrophoresis, and transfer the gel to PVDF membranes.
NOTE: During the preparation of SDS gel, it must be fully mixed without bubbles or insoluble particles; otherwise, uneven or irregular bands may occur. When transferring the membranes, ensure there are no bubbles between the PVDF membrane and the gel; otherwise, white spots will appear in the strip. In addition, this step must be performed at low temperatures, and the ice bag should be changed every 30 min. - Block the membranes with 5% nonfat milk for 1.5 h at RT and incubate with 5 mL of the corresponding primary antibodies (Nrf2 1:1,000, Akt 1:2,000, p-Akt 1:2,000, Bcl-2 1:5,000, and Bax 1:1,000) for 24 h at 4 °C. Use β-actin (1:5,000) antibody as an internal control.
- Wash the membranes with TBST (three times for 10 min each), then incubate with 5 mL of secondary HPR-conjugated antibodies (1:5,000) at RT for 2 h.
NOTE: The dilution ratio of the antibodies should not be arbitrarily changed, and the membranes should be washed with TBST sufficiently in strict accordance with the requirements to avoid the background color of the band being too black. - Finally, wash the membrane with TBST again, and treat it with chemiluminescent HRP substrate (as per the manufacturer's instructions) for protein band detection. Capture the images using the chemiluminescence imaging system and quantify the gray values of the proteins using the imaging software, with β-actin as the comparative internal control.
Representative Results
The screening of the optimal combination of Ast injection and Eri capsule is shown in Figure 1. The cell survival rate of Ast injection and Eri capsule on the normal PC12 cells is shown in Figure 1A. The cell viability was lower than 95% with Ast injection at concentrations greater than 12 µM (Figure 1A) and Eri capsule at concentrations greater than 5 µM (Figure 1A), indicating that the maximum nontoxic concentration was 12 µM and 5 µM, respectively. Their cytotoxicity was greater than that of astragaloside A and scutellarin used alone. The viability of Ast injection and Eri capsule on the injured PC12 cells induced by CoCl2 is shown in Figure 1B,C. Compared with the model group, Ast injection could improve the survival rate of the injured PC12 cells in the concentration range of 6-12 µM (p < 0.05 or p < 0.01), and Eri capsules at the concentration of 2-5 µM could improve the survival rate (p < 0.05 or p < 0.01). Ast injection and Eri capsule at the ratios of 10:1, 8:4.2, and 6:1.8 µM can significantly increase the viability of the injury PC12 cells (p < 0.01) (Figure 1D). The ratio of 6:1.8 µM exhibited the highest cell viability, indicating that it is the optimal preparation combination and pharmacological activity compared with the best component combination.
The evaluation of the protective effects of two kinds of combination on the injured PC12 cells is shown in Figure 2. Compared with the model group, the cell viability of the two combinations was significantly promoted (p < 0.001), and the component combination was superior to the preparation combination (Figure 2A). This suggests that the component combination can promote cell survival better than the preparation combination. The apoptosis rate was tested by flow cytometry (Figure 2B,C). Compared with the normal group, the percentages of early, late, and total apoptotic cells were significantly higher in the model group (p < 0.01 or p < 0.001). Compared with the model group, the percentages of apoptotic cells at each stage were significantly lower in the treatment groups (p < 0.05 or p < 0.01, or p < 0.001). The fluorescence intensity of caspase-3 protein expression in each group is shown in Figure 2D,E. Compared with the normal group, the fluorescence intensity of caspase-3 was significantly higher in the model group. Compared with the model group, the fluorescence intensity of caspase-3 was significantly lower in each treatment group (p < 0.001 or p < 0.01) and was relatively lower in the component combination group than the preparation combination group. These results suggest that the cell model can induce apoptosis, and the anti-apoptosis effect of the component combination is better than that of the preparation combination.
Western blot detected the p-Akt, Akt, Bcl-2, and Bax protein expression (Figure 3). Compared with the normal group, the expression levels of p-Akt/Akt and Bcl-2/Bax were significantly lower in the model group (p < 0.01 or p < 0.001). Compared with the model group, the expression of p-Akt/Akt and Bcl-2/Bax were significantly higher in the two combinations (p < 0.05 or p < 0.01, or p < 0.001), specifically higher in the component combination. The results suggest that the component combination is superior to the preparation combination in promoting cell survival, which is related to the stronger anti-apoptosis effect produced by upregulating the Akt/Bcl-2/Bax signal pathway.
The fluorescence of DCFH can be measured by a fluorescence microscope to determine the level of ROS in the cells (Figure 4A,B). As shown in Figure 4A, the fluorescence was almost invisible in the normal cells, and the fluorescence in the model group was significantly enhanced. The fluorescence in both combinations was significantly reduced compared to the model group. As shown in Figure 4B, the relative fluorescence intensity was significantly weaker in each treatment group compared with the model group (p < 0.001). Fluorescence had a tendency to decrease in the component combination group compared with the preparation combination group, indicating that the cell model can induce oxidative damage. The anti-oxidative damage effect of the component combination is better than that of the preparation combination.
Western blot detected the expression of the Nrf2 protein (Figure 4C). Compared with the normal group, the expression of Nrf2 was significantly reduced in the model group (p < 0.05). Compared with the model group, the expression of Nrf2 protein was significantly higher in the treatment groups (p < 0.01 or p < 0.05), specifically higher in the component combination group (Figure 4D). This suggests that the component combination is better than the preparation combination in promoting cell survival, which is related to the stronger anti-oxidative damage produced by upregulating the Nrf2 signal pathway.
In conclusion, the component combination (10 µM astragaloside A, 40 µM scutellarin, and 75 µM chlorogenic acid) could promote the survival of injured cells better than the preparation combination (6 µM Ast injection and 1.8 µM Eri capsule) through stronger regulation of signal pathways related to apoptosis and oxidative damage.
SPSS statistical software 26.0 was used for statistical analysis, and all data are expressed as the means ± standard deviation (SD). For comparisons between groups, the data were evaluated by a one-way ANOVA followed by Tukey's test. p < 0.05 was considered to indicate a statistically significant difference.
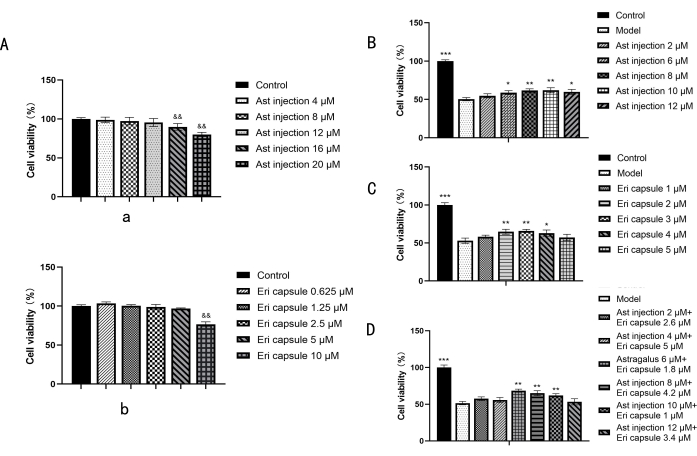
Figure 1: Effects of drugs on the viability of normal and injured PC12 cells. (A) Effects of various concentrations of Ast injection and various concentrations of Eri capsule on normal PC12 cells. (B) Screening of the effective concentration of Ast injection on injured PC12 cells. (C) Screening of the effective concentration of Eri capsule on injured PC12 cells. (D) Effects of the combinations of two drugs in different proportions on the survival rate of injured PC12 cells. Statistical values are expressed as the mean ± SD from six independent experiments. &&p < 0.01 compared with the control group. *p < 0.05 and **p < 0.01 compared with the model group. Please click here to view a larger version of this figure.
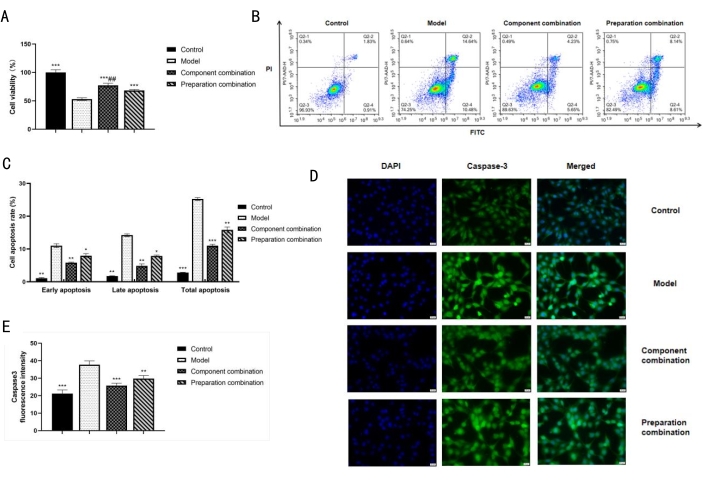
Figure 2: Effects of two combinations on the survival and apoptosis of injured PC12 cells. (A) Effect of the component combination and the preparation combination on the survival rate of injured PC12 cells. (B) Graph of apoptosis rate detected by Annexin V-PI. (C) Statistical histogram of apoptosis rate. (D) The relative fluorescence intensity of caspase-3 protein expression (400x). Scale bars: 20 µm. (E) Statistical histogram of fluorescence intensity of caspase-3 protein expression. Statistical values are expressed as the mean ± SD from three independent experiments, except for cell viability for six independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001 compared with the model group. ##p < 0.01 compared with the preparation combination group. Please click here to view a larger version of this figure.
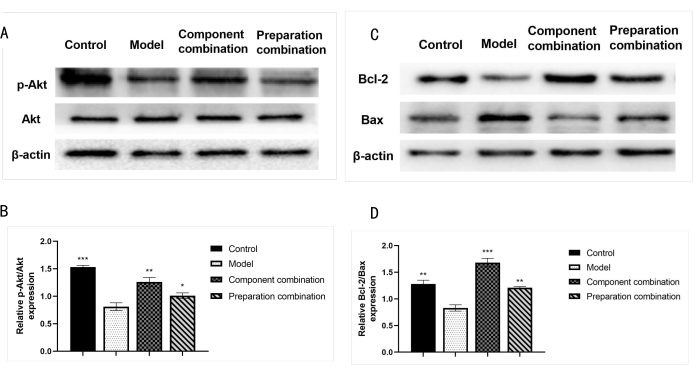
Figure 3: Effects of the two combinations on the expression levels of p-Akt, Akt, Bcl-2, and Bax. (A) Protein expression of p-Akt and Akt determined by Western blot analysis. (B) Statistical histogram of p-Akt/Akt ratio statistics in each group. (C) Protein expression of Bcl-2 and Bax determined by Western blot analysis. (D) Statistical histogram of Bcl-2/Bax ratio in each group. Statistical values are expressed as the mean ± SD from three independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001 compared with the model group. Please click here to view a larger version of this figure.
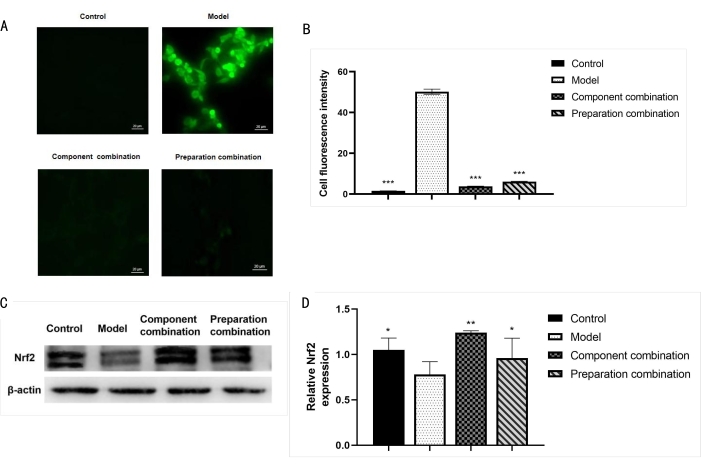
Figure 4: Effect of two combinations on the Nrf2 protein expression and ROS levels. (A) Levels of ROS were detected by fluorescence microscopy (400x). Scale bars: 20 µm. (B) Statistical histogram of ROS fluorescence intensity in each group. (C) Nrf2 protein expression determined by Western blot analysis. (D) Statistical histogram of Nrf2 protein expression. Statistical values are expressed as the mean ± SD from three independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001 compared with the model group. Please click here to view a larger version of this figure.
Discussion
There is still a lack of ideal drugs for the treatment of cerebral ischemia in modern clinical practice2. Under the guidance of supplementing qi and activating the blood circulation method, Ast, Eri, and other preparations have been used in combination in the clinical practice of TCM and have achieved good comprehensive advantages13,14,15. A large number of studies have shown that Ast can improve the permeability of the blood-brain barrier, increase cerebral blood flow, improve the ability of nerve cells to tolerate hypoxia and resist oxygen free radical damage, and can significantly improve neurological dysfunction16,17. It is particularly suitable for senile ischemic cerebrovascular disease16,17. Eri can dilate blood vessels, increase blood supply to the brain, remove ROS, and reduce lipid peroxidation18,19. However, the synergistic effect, pharmacodynamic components, and the mechanism of the combination of the two drugs are still unclear, which is a common problem restricting the clinical application of TCM.
The ischemic and hypoxic environment of chronic cerebral ischemia induces oxidative stress damage and, at the same time, activates the brain cell apoptosis signaling pathway to promote neuronal apoptosis20,21. The PI3K/Akt pathway is a classic anti-apoptotic and pro-survival signal transduction pathway22,23, among which Bcl-2 family proteins and the caspase family are the downstream executive proteins of this signal pathway. Phosphorylated Akt can directly or indirectly regulate the Bcl-2 family and inhibit activation of the downstream pathway caspase-3, thereby exerting an anti-apoptotic effect11. In addition, excess ROS generated by hypoxia can induce oxidative stress injury, while phosphorylated Akt can activate Nrf2 to eliminate excess ROS to combat oxidative stress damage12,24. Therefore, activation of the PI3K/Akt/Nrf2 pathway can effectively prevent and control oxidative stress and apoptosis, thereby reducing hypoxic-ischemic brain injury25,26.
In the study, the preparation combination was screened using the injured PC12 cell model, and the effect and mechanism in this model were compared with the component combination screened previously7. In addition, the maximum nontoxic concentration of Ast injection and Eri capsule is 12 µM (calculated by astragaloside A) and 5 µM (calculated by scutellarin), respectively, while the maximum nontoxic concentration of astragaloside A and scutellarin is 20 µM and 50 µM, respectively7. It shows that the component combination is safer than the preparation combination, with more controllable quality and comprehensive advantages of high efficiency and low toxicity.
The current cell models that can be used to simulate cerebral ischemia mainly include physical hypoxia (induced by OGD) and chemical hypoxia (induced by Na2S2O4 or CoCl2)27. Among them, both OGD and Na2S2O4 injuries have a short hypoxia time and are not suitable for simulating chronic ischemia27. CoCl2-induced hypoxia is one of the most commonly used hypoxia mimics compared to OGD and the use of other hypoxia mimics. It can lead to persistent and stable oxidative damage by ROS and produce typical apoptotic changes in different cell lines27. Therefore, in this study, CoCl2 was used for 24 h to simulate the neuronal hypoxia28,29. Its modeling concentration (0.1, 0.2, 0.4, and 0.8 mM) and the validity period (1 and 7 days) were screened. In addition, given the inevitable lack of glucose and oxygen after cerebral ischemia, the glucose-free and hypoxic environment can better simulate cerebral ischemic injury. This study showed that 0.4 mM CoCl2 combined with a glucose-free medium for 24 h could establish a stable and controllable chronic hypoxic cell model. In the follow-up study, the animal model of chronic cerebral ischemia will be used to further validate the therapeutic advantages of the component combination in vivo.
This cell model can increase the apoptosis rate, caspase-3 fluorescence intensity, and intracellular ROS level (Figure 2 and Figure 4), which can better simulate the pathological changes of chronic cerebral ischemia, such as apoptosis, oxidative stress, etc. Based on this model, it was found that the two types of combinations can inhibit apoptosis and oxidative stress damage. The effect of the component combination on promoting cell survival was significantly better than that of the preparation combination (Figure 1); both combinations could upregulate the expression levels of P-Akt/Akt, Bcl-2/Bax, and Nrf2 in different degrees (Figure 3 and Figure 4). In particular, the component combination had a stronger effect on the up-regulation of Akt/Bcl-2/Bax and Nrf2 signaling pathway. These results indicate that the component combination can better resist cell damage than the preparation combination, which is related to stronger anti-apoptosis and anti-oxidative stress.
In conclusion, these results provide a combination strategy of two herbs to treat injured PC12 cells and provides a new idea for evaluating and optimizing the combined application mode of two herbs. Overall, this study provides a reference for selecting the active component combination of TCM.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was funded by Key R & D projects of the Sichuan Provincial Department of science and technology (2020YFS0325).
Materials
| 1300 series class II biosafety cabinet | Thermo Fisher Scientific Instruments Company | 1374 | |
| 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide | Guangzhou saiguo biotech Company | 1334GR001 | MTT |
| ACEA NovoExpress 1.4.0 | ACEA Biosciences, Inc | – | |
| Akt | Wuhan Three Eagles Proteintech Group, Inc | 10176-2-AP | |
| Analytical flow cytometry | Thermo Fisher Scientific Instruments Company | 62-2-1810-1027-0 | |
| Annexin V-FITC apoptosis detection kit | Beyotime Biotechnology Company | C1062L | |
| Astragalus injection | Heilongjiang Zhenbao Island Pharmaceutical Company | A03190612144 | Ast injection |
| Astragaloside A | Chongqing Gao Ren Biotechnology Company | 84687-43-4 | |
| Bax | Wuhan Three Eagles Proteintech Group, Inc | 60267-1-Ig | |
| BCA protein quantification kit | Beyotime Biotechnology Company | P0012 | |
| Bcl-2 | Wuhan Three Eagles Proteintech Group, Inc | 26593-1-AP | |
| Carbon dioxide incubators | Thermo Fisher Scientific Instruments Company | 0816-2014 | |
| Caspase-3 | Wuhan Three Eagles Proteintech Group, Inc | 19677-1-AP | |
| Chlorogenic acid | Chongqing Gao Ren Biotechnology Company | 327-97-9 | |
| Cobalt chloride hexahydrate | Merck Biotechnology, Inc. | 7791-13-1 | CoCl2 |
| Dimethyl sulfoxide | Guangzhou saiguo biotech Company | 2020112701 | DMSO |
| Disodium hydrogen phosphate dodecahydrate | Chengdu Kolon Chemical Company | 2020090101 | |
| DMEM high sugar medium | Thermo scientific Hyclone | 2110050 | |
| DMEM sugar free medium | Beijing Solarbio life sciences Company | 2029548 | |
| ECL luminous fluid | Lianshuo Biological Company | WBKLS0500 | |
| Electronic balance | Haozhuang Hengping Scientific Instrument Co., Ltd., Shanghai, China | FA1204 | |
| Electrophoresis instrument | Bio-Rad Laboratories (Shanghai) Co., Ltd | 1658026 | |
| Erigeron breviscapus capsule | Yunnan Biogu Pharmaceutical Company | Z53021671 | Eri capsule |
| Fetal bovine serum | Four Seasons Institute of Biological Engineering | 20210402 | FBS |
| Fluorescent microscope | Olympms Corporation | IX71-F32PH | |
| Gel imager | Bio-Rad Laboratories (Shanghai) Co., Ltd | 1708265 | |
| Goat anti-mouse secondary antibody | Wuhan Three Eagles Proteintech Group, Inc | SA00001-1 | |
| Goat anti-rabbit secondary antibody | Wuhan Three Eagles Proteintech Group, Inc | SA00001- 2 | |
| IBM SPSS Statistics version 26.0 | International Business Machines Corporation, USA | – | |
| ImageJ 1.8.0 | National Institutes of Health, USA | – | imaging software |
| Immunol fluorescence staining kit | Beyotime Biotechnology Company | P0196 | |
| KCl | Chengdu Kolon Chemical Company | 2021070901 | |
| Marker | Thermo Fisher Scientific Instruments Company | 26616 | |
| Microplate reader | Perkin Elmer Corporate Management (Shanghai) Co. | HH35L2018296 | |
| Motic Inverted microscope | Nanda Scientific Instruments Co. | AE2000LED | |
| NaCl | Chengdu Kolon Chemical Company | 2014081301 | |
| Nonfat milk | Beyotime Biotechnology Company | P0216 | |
| Nrf2 | Wuhan Three Eagles Proteintech Group, Inc | 16396-1-AP | |
| P-Akt | Wuhan Three Eagles Proteintech Group, Inc | 66444-1-Ig | |
| PC12 cells | Chinese Academy of Sciences | CBP60430 | |
| Penicillin-Streptomycin solution | Thermo scientific Hyclone | SV30010 | |
| Phosphatase protease inhibitor mixture | Beyotime Biotechnology Company | P1045 | |
| Potassium dihydrogen phosphate | Chengdu Kolon Chemical Company | 2015082901 | |
| Reactive oxygen species assay kit | Beyotime Biotechnology Company | S0033S | |
| RI-PA lysis solution | Beyotime Biotechnology Company | P0013B | |
| Scutellarin | Chongqing Gao Ren Biotechnology Company | 27740-01-8 | |
| SDS-PAGE sample loading buffer, 5x | Beyotime Biotechnology Company | P0015L | |
| Sterile filter tips (0.22 µm) | Merck Biotechnology, Inc. | SLGP033RB | |
| Tris-base | Guangzhou saiguo biotech Company | 1115GR500 | TBS |
| Trypsin | Thermo scientific Hyclone | J190013 | |
| Tween-20 | Chengdu Kolon Chemical Company | 2021051301 | |
| Vortex oscillator | OHAUS International Co., Ltd., Shanghai, China | VXMNAL | |
| β-actin | Wuhan Three Eagles Proteintech Group, Inc | 66009-1-Ig |
References
- Mendelevich, E. G. Chronic cerebral ischemia, and dizziness. Zhurnal Nevrologii i Psikhiatrii Imeni SS Korsakova. 122 (3), 22-26 (2022).
- Gao, L. Expert consensus on the diagnosis and treatment of chronic cerebral ischemia with integrated traditional Chinese and western medicine. Chinese Journal of Integrated Traditional and Western Medicine. 38 (10), 1161-1167 (2018).
- Luyu, S., Heng, L., Dengke, L. Combined treatment of 45 cases of cerebral infarction with Astragalus and Dengzhan Xixin injection. Jilin Journal of Traditional Chinese Medicine. 27 (4), 23-24 (2007).
- Bei, N., et al. Clinical study of Dengzhanxixin injection combined with Astragalus injection in the treatment of acute ischemic stroke. Journal of Basic Chinese Medicine. 21 (9), 1123-1127 (2015).
- Tian, F., et al. Optimal ratio of Astragalus injection combined with Dengzhan Xixin injection against cerebral ischemia-reperfusion injury in rats by baseline equal-ratio increase-decrease design method. China Pharmacy. 30 (14), 1885-1889 (2019).
- Li, J., et al. Protective effect and mechanism of Astragalus combined with Erigeron breviscapusin the best proportion on cerebral ischemia rats. Pharmacology and Clinics of Chinese Materia Medica. 35 (2), 104-108 (2019).
- Yan, Y., et al. Study on the oxidative damage of PC12 cells with hypoxia and hypoglycemia based on the combination of Astragalus and Dengzhan Asarum based on the Nrf2/HO-1 pathway. Pharmacology and Clinics of Chinese Materia Medica. 38 (2), 159-164 (2022).
- . Chinese Pharmacopoeia, Dengzhan Asarum Granules. Volume I Available from: https://db.ouryao.com/yd2020/view.php?id=f27dcefa69 (2020)
- . Chinese Pharmacopoeia, Astragalus granules. Volume I Available from: https://db.ouryao.com/yd2020/view.php?id=fbcd0a8bf8 (2020)
- Liu, X., et al. N-linoleyltyrosine protects PC12 cells against oxidative damage via autophagy: Possible involvement of CB1 receptor regulation. International Journal of Molecular Medicine. 46 (5), 1827-1837 (2020).
- Fan, Y., et al. Gab1 regulates SDF-1-induced progression via inhibition of apoptosis pathway induced by PI3K/AKT/Bcl-2/BAX pathway in human chondrosarcoma. Tumor Biology. 37 (1), 1141-1149 (2016).
- Meng, M., Zhang, L., Ai, D., Wu, H., Peng, W. β-Asarone ameliorates β-Amyloid-induced neurotoxicity in PC12 cells by activating P13K/Akt/Nrf2 signaling pathway. Frontiers in Pharmacology. 12, 659955 (2021).
- Li, J., Chen, H. Advances in the treatment of chronic cerebral ischemia with traditional Chinese and western medicine. Xinjiang Journal of Traditional Chinese Medicine. 39 (3), 115-118 (2021).
- Yu, M., Zhan, Q., Xu, X. Discussion on the therapeutic value of traditional Chinese medicine on cognitive dysfunction caused by chronic cerebral ischemia. Chinese Medicine Modern Distance Education of China. 11 (19), 155-157 (2013).
- Wang, M., Liu, J., Yao, M., Ren, J. Advances in research on pharmacological and neuroprotective effects of traditional Chinese medicine after cerebral ischemia. China Journal of Chinese Materia Medica. 45 (3), 513-517 (2020).
- Wang, N., Sun, H., Ma, X. Effects of different doses of astragalus injection on neurological rehabilitation of stroke patients. Chinese Journal of Clinical Rational Drug Use. 9 (13), 67-69 (2016).
- Sun, Y. Q. Effects of astragalus injection on apoptosis after cerebral ischemia-reperfusion in rats. Hebei Medicine. 22 (7), 1147-1149 (2016).
- Yang, L., et al. Dengzhan Xixin injection derived from a traditional Chinese herb Erigeron breviscapusameliorates cerebral ischemia/reperfusion injury in rats via modulation of mitophagy and mitochondrial apoptosis. Journal of Ethnopharmacology. 288, 114988 (2022).
- Liao, Y., Ni, X., Zhan, C., Cai, Y. Clinical research progress of Dengzhanhua preparation in prevention and treatment of ischemic stroke and transient ischemic attack. Guangdong Medical Journal. 41 (9), 963-967 (2020).
- Li, C., Li, J., Xu, G., Sun, H. Influence of chronic ethanol consumption on apoptosis and autophagy following transient focal cerebral ischemia in male mice. Scientific Reports. 10 (1), 6164 (2020).
- El Khashab, I. H., Abdelsalam, R. M., Elbrairy, A. I., Attia, A. S. Chrysin attenuates global cerebral ischemic reperfusion injury via suppression of oxidative stress, inflammation and apoptosis. Biomedicine Pharmacotherapy. 112, 108619 (2019).
- Su, X., He, S., Chen, Z., Xie, X. Effect of breviscapine aglycone on PI 3 K/Akt conduction pathway in neonatal rats with hypoxic-ischemic brain injury. Journal of Armed Police Logistics College (Medical Edition). 27 (2), 93-96 (2018).
- Wang, X., Shi, C., Pan, H., Meng, X., Ji, F. MicroRNA-22 exerts its neuroprotective and angiogenic functions via regulating PI3K/Akt signaling pathway in cerebral ischemia-reperfusion rats). Journal of Neural Transmission. 127 (1), 35-44 (2020).
- Luca, M., Luca, A., Calandra, C. The role of oxidative damage in the pathogenesis and progression of alzheimer’s disease and vascular dementia. Oxidative Medicine and Cellular Longevity. 2015, 504678 (2015).
- Wang, Z., et al. Lupeol alleviates cerebral ischemia-reperfusion injury in correlation with modulation of PI3K/Akt pathway. Neuropsychiatric Disease and Treatment. 16 (8), 1381-1390 (2020).
- Li, H., et al. Neuroprotective effect of phosphocreatine on oxidative stress and mitochondrial dysfunction induced apoptosis in vitro and in vivo: Involvement of dual PI3K/Akt and Nrf2/HO-1 pathways. Free Radical Biology and Medicine. 120, 228-238 (2018).
- Muñoz-Sánchez, J., Chánez-Cárdenas, M. E. The use of cobalt chloride as a chemical hypoxia model. Journal of Applied Toxicology. 39 (4), 556-570 (2019).
- Tang, Y., et al. Salidroside attenuates CoCl2-simulated hypoxia injury in PC12 cells partly by mitochondrial protection. European Journal of Pharmacology. 912 (10), 174617 (2021).
- Yin, L., et al. Neuroprotective potency of Tofu bio-processed using Actinomucor elegans against hypoxic injury induced by cobalt chloride in PC12 cells. Molecules. 26 (10), 2983 (2021).

