Puncture Wound Hemostasis and Preparation of Samples for Montaged Wide-Area Electron Microscopy Analysis
Summary
A puncture wound procedure for hemostatic thrombus formation is presented here. The formed thrombi are large and are hundreds of microns in diameter. Hence, volume imaging approaches are appropriate. We suggest montaged wide-area transmission electron microscopy as a high-resolution approach available to many and detail a preparative protocol.
Abstract
Hemostasis, the process of normal physiological control of vascular damage, is fundamental to human life. We all suffer minor cuts and puncture wounds from time to time. In hemostasis, self-limiting platelet aggregation leads to the formation of a structured thrombus in which bleeding cessation comes from capping the hole from the outside. Detailed characterization of this structure could lead to distinctions between hemostasis and thrombosis, a case of excessive platelet aggregation leading to occlusive clotting. An imaging-based approach to puncture wound thrombus structure is presented here that draws upon the ability of thin-section electron microscopy to visualize the interior of hemostatic thrombi. The most basic step in any imaging-based experimental protocol is good sample preparation. The protocol provides detailed procedures for preparing puncture wounds and platelet-rich thrombi in mice for subsequent electron microscopy. A detailed procedure is given for in situ fixation of the forming puncture wound thrombus and its subsequent processing for staining and embedding for electron microscopy. Electron microscopy is presented as the end imaging technique because of its ability, when combined with sequential sectioning, to visualize the details of the thrombus interior at high resolution. As an imaging method, electron microscopy gives unbiased sampling and an experimental output that scales from nanometer to millimeters in 2 or 3 dimensions. Appropriate freeware electron microscopy software is cited that will support wide-area electron microscopy in which hundreds of frames can be blended to give nanometer-scale imaging of entire puncture wound thrombi cross-sections. Hence, any subregion of the image file can be placed easily into the context of the full cross-section.
Introduction
The formation of a puncture wound thrombus that leads to bleeding cessation is one of the most essential events in life1. Yet despite that essentiality, knowledge of what occurs structurally during thrombus formation, be it in a vein, an artery, an atherosclerotic event, or an occlusive clot, has been limited by resolution and imaging depth. Conventional light microscopy is limited in depth when compared to a fully formed puncture wound thrombus, 200 to 300 µm in Z1, and in resolution level when compared to the size of platelet organelles and their spacing, often less than 30 nm2. Two-photon light microscopy can yield the needed depth of imaging but does not improve resolution significantly. The most recent advances in light microscopy, for example, super-resolution techniques, are still resolution limited, in practice ~20 nm in XY and twice that in Z, and depth limited, no more than conventional light microscopy. Furthermore, super-resolution light microscopy, like much of research light microscopy, is based on fluorescence microscopy, a technique that is inherently biased to a small set of candidate proteins for which good antibodies exist or good tagged constructs3. In conclusion, conventional scanning electron microscopy can, at most, visualize the surface of the forming platelet-rich thrombus.
To overcome these technical limitations to characterizing thrombus structure, we had three goals. First, reproducibly produce a defined puncture wound in a mouse vein or artery that could then be readily stabilized in situ by chemical fixation. Second, apply a preparative procedure that emphasizes membrane preservation, a goal consistent with the aim of defining the position of individual platelets within the forming thrombus. Third, use an unbiased visualization technique that, in a single image, could be scaled between nanometer to near millimeter scale.
Montaged, wide-area electron microscopy was chosen as a major end visualization technique for a single important reason: in electron microscope imaging, one sees a vast array of features within a cell that outlines its organelles and features within the organelles. Small objects such as ribosomes can be recognized. This range of features is seen because the electron-dense heavy metal stains, uranyl, lead, and osmium, that are used for electron microscopy to yield contrast bind to a wide range of molecules. In an electron microscope image, one sees much of what is there, while with immunofluorescence and protein tagging approaches, one only sees what lights up. This means, for example, the antigen sites present on a given individual protein species. In the case of a tagged molecule, often a protein, it is the site(s) where that protein is. All other molecules are dark and not lit up. However, is this choice of electron microscopy practical? A puncture wound thrombus has a size of 300 by 500 µm and, at a pixel size of 3 nm, that is an image of 100,000 by 167,000 pixels. A high-quality electron microscope camera has 4000 by 4000 pixels. That means that approximately 1000 frames must be stitched/blended to give a single image. That is a possibility that has been present in most electron microscopes manufactured in the last 15 years. The microscope stage is computerized, and the images can be stitched together with a computer. That is the rationale that led to choices underlying the formulation of the presented protocol.
Conclusively, we present below a series of steps that give a reproducible wound in the vein or artery of mice that then, following in situ fixation steps and later embedding steps, can be visualized by montaged, wide-area transmission electron microscopy at nm scale and in the stitched image visualized at near mm scale, the scale of the actual in situ fixed thrombus. Scalability of this kind is required for understanding thrombus formation as both a problem in hematology/health and as a developmental biology system in which platelets are the major cell type. These advances deliver a major virtue of electron microscopy, namely, one sees what is there, not only what lights up. For a detailed protocol on the preparation of samples for serial block face scanning electron microscopy (SBF-SEM), the reader is directed to a recent article by Joshi et al.4.
Protocol
Experiments were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Arkansas for Medical Sciences. Here, 8 – 12-week-old, wild-type male and female C57BL/6 mice were used. These mice are young adults with little accumulation of fat. The same procedures are applicable to mice mutants for various proteins important to hemostasis, such as von Willebrand factor or platelet glycoprotein VI (GPVI)5,6. All equipment, surgical instruments, reagents, and other materials used are shown in Figure 1 and listed in the Table of Materials.
1. Jugular vein/femoral artery puncture wound1,7
- Anesthetization of the mouse
- Place the mouse in the induction chamber and turn the isoflurane vaporizer to a high flow rate setting (500 mL/min, 3% isoflurane). Isoflurane is chosen because it has little effect on blood vessel diameter.
CAUTION: Long-term exposure to isoflurane can cause adverse health effects. The use of a low-flow vaporizer, anesthesia gas capture canisters, and good room ventilation can greatly reduce exposure. - When the mouse appears asleep, pinch one of the mouse's toes to see if the mouse reacts. If so, place the mouse back in the induction chamber for a few minutes and then try the pinch test again. If the mouse does not react, place it in a supine position on the heating plate (previously covered with aluminum foil).
- Turn the vaporizer down to a low flow rate (100 mL/min, 1.75% isoflurane). Place the mouse's nose in the nose cone. Extend each limb and tape down with small pieces of adhesive tape.
- Insert the rectal temperature probe and set the temperature to 37 °C on the temperature controller. Tape down the temperature probe cord to prevent the probe from accidentally being pulled out.
- Place the mouse in the induction chamber and turn the isoflurane vaporizer to a high flow rate setting (500 mL/min, 3% isoflurane). Isoflurane is chosen because it has little effect on blood vessel diameter.
- Mouse incision
- Using the clippers, shave the mouse's neck and chest (for jugular vein) or inner right leg (for femoral artery). Wipe away the fur clippings with an alcohol pad. If needed, go over the area again with the clippers to remove any remaining fur.
- Wipe the area again with a fresh alcohol pad to clear away all the clippings. It is very important that the area is as clean as possible.
- Using scissors and blunt forceps, lift and cut the skin over the right jugular vein. Cut a round window in the skin large enough to expose the jugular vein and some of the surrounding tissue.
- Cleaning of the jugular vein (same principles apply to the femoral artery)
- Using the blunt dissection technique, gently push aside adipose and lymph nodes as well as connective tissue.
NOTE: Some general cleaning around the vessel takes place at this step; however, careful, thorough cleaning will take place under the microscope after the jugular vein is fixed in paraformaldehyde for 24 h. - Fill a 20 mL syringe with room temperature normal saline. Load the syringe into the syringe infusion pump. Attach the 27G butterfly needle and associated tubing to the syringe.
- Set the syringe pump to a flow rate of 0.5 mL/min. The gentle stream of saline will wash away blood from the punctured vein/artery. Position the stream of saline a few mm to the side of the puncture site, not directly over it.
- Using the blunt dissection technique, gently push aside adipose and lymph nodes as well as connective tissue.
- Puncturing the jugular vein/femoral artery
- Start the timer. Obtain a 30G hypodermic needle (nominal 300 µm diameter) for the jugular vein and a 33G hypodermic needle (200 µm diameter) for the femoral artery. The use of a smaller needle gives only a slightly longer bleeding time for the higher-pressure arterial situation.
- Turn the bevel facing up. Position the needle at a 25° angle over the jugular vein/femoral artery. Note the time.
- Quickly puncture the vessel, making sure to pierce the top of the vessel only. Be careful not to puncture the bottom of the vessel as well. Note the time when the bleeding stops completely.
- Wait for the appropriate amount of time to obtain thrombus formation (Figure 2A) before starting perfusion fixation (i.e., either 1, 5, 10, or 20 min to obtain different stages of thrombus development).
- Performing transcardial perfusion fixation
- Set up the peristaltic pump before the surgery begins. Place two 50 mL conical tubes in a test tube rack. Fill one tube with 4% paraformaldehyde fixative solution prepared in sodium cacodylate buffer solution (0.2 M sodium cacodylate, 0.15 M sodium chloride at pH 7.4) for jugular puncture studies and fill the other tube with sodium cacodylate buffer solution.
NOTE: The conditions detailed here are sufficient for the low-pressure case of the jugular and are then modified for the higher-pressure case of an artery. For the higher-pressure system studies with arteries, i.e., the femoral artery, the paraformaldehyde fixative is supplemented with 0.1% glutaraldehyde, and external drip fixation is combined with perfusion fixation. - Place the uptake end of the pump tubing into the tube containing the buffer solution. Attach a 27G butterfly needle with associated tubing to the output end of the pump tubing and place the needle in a clean beaker.
- Set the peristaltic pump to a flow rate of 10 mL/min. Turn on the perfusion pump.
- When buffer solution begins to flow from the needle into the beaker, turn the pump off. The tubing and needle are now primed for the transcardial perfusion.
- Expose the chest cavity by making a midline cut in the skin from the top of the sternum to a few millimeters past the bottom of the sternum using dissecting scissors and blunt forceps. Make transverse incisions along the base of the rib cage, starting at the midline and cutting from medial to lateral on each side.
- Immediately before starting the transcardial perfusion, quickly cut open the rib cage and reflect each side to expose the heart.
- Turn on the peristaltic pump. Place the tip of the 27G butterfly needle into the base of the left ventricle.
- While holding the needle secure in the left ventricle, cut a slit in the right auricle using sharp dissecting scissors to allow blood and perfusion fluid to flow out. Note the time on the timer.
- Perfuse with buffer solution for 3 min to flush the blood out of the circulatory system. While still holding the needle secure in the left ventricle, turn off the peristaltic pump and move the uptake end of the peristaltic pump tubing over to the 50 mL tube containing the fixative solution.
- Note the time on the timer. Turn the peristaltic pump on again and perfuse with the fixative solution for 7 min. Turn the peristaltic pump off.
- Take the needle out of the left ventricle and place it in a waste beaker. Place the uptake end of the pump tubing into a beaker of distilled water.
- Turn the peristaltic pump on and let approximately 20 mL of water flow through the tubing and out the 27G needle into the waste beaker to clean the tubing. Be sure to let all the water exit the tubing and needle before turning the pump off.
- Set up the peristaltic pump before the surgery begins. Place two 50 mL conical tubes in a test tube rack. Fill one tube with 4% paraformaldehyde fixative solution prepared in sodium cacodylate buffer solution (0.2 M sodium cacodylate, 0.15 M sodium chloride at pH 7.4) for jugular puncture studies and fill the other tube with sodium cacodylate buffer solution.
2. Collection of samples for electron microscopy
- After perfusion fixation supplemented with an initial 2 min external fixative in the case of femoral artery puncture wounds, carefully dissect the portion of the jugular vein/femoral artery containing the puncture site and thrombus (Figure 2A). With sharp scissors, make a diagonal cut across the distal end of the vessel segment and make a straight cut across the proximal end of the vessel segment.
NOTE: Making cuts in this manner helps to orient the vessel later and to identify the direction of blood flow in the vessel. - Submerge the vessel segment in fresh, room temperature, 4% paraformaldehyde in sodium cacodylate buffer solution (0.2 M sodium cacodylate, 0.15 M sodium chloride at pH 7.4) and place at room temperature for 1 h.
- Transfer the fixed tissue to 4 °C for 24 h. The next day, take a 35 mm culture dish containing a ~5 mm thick silicone mat (prepared in advance according to the manufacturer's instructions). Pour enough sodium cacodylate buffer solution into the dish to submerge the fixed vein segment.
- While viewing under a light microscope (~15x magnification), carefully transfer the tissue to the 35 mm dish containing buffer solution and pin each end of the vessel to the silicone mat using stainless steel minutien pins and fine forceps.
- Clean the jugular vein/femoral artery segment carefully using a pair of micro scissors and very fine forceps.
- For jugular vein only: Remove one of the pins and, using the micro scissors, cut the vessel wall opposite the puncture/thrombus site lengthwise. Gently open the vessel and, using 4 to 5 pins (each cut in half with wire cutters), pin it to the silicone mat with the intraluminal side facing up.
- Aspirate the buffer solution, quickly replace it with 1% paraformaldehyde solution in sodium cacodylate buffer solution and place the lid on the dish. Do not let the tissue dry out at any time. Always keep it submerged in fluid.
- Store the tissue at 4 °C until it is time to process it for electron microscopy (Figure 2B). Process some of the jugular vein/femoral artery puncture wound samples for imaging by SBF-SEM4 and others for imaging by WA-TEM8,9. The WA-TEM steps are described in detail below.
CAUTION: Carry out all work involving formaldehyde, propylene oxide, OsO4, and 2,4,6-Tris(dimethylaminomethyl)phenol (DMP-30) in a chemical fume hood. These are hazardous chemicals. Exposure to OsO4 can cause blindness and death.
NOTE: All rinse and stain volumes are at least 2x the volume of the sample. The 1x phosphate-buffered saline (PBS) can be substituted for the 0.1 M sodium cacodylate buffer. During the fixation procedure, it is very important never to let the samples dry out, as this would significantly affect the ultra-structure. Use polypropylene tubes.
3. Preparation of sample for montaged wide-area transmission electron microscopy (WA-TEM)
NOTE: This step represents a decision point at which the investigator is committing to preparing for WA-TEM. This preparative procedure does not support SBF-SEM. For SBF-SEM volume EM, all staining must be done before embedding in plastic. Please see4 for an SBF-SEM preparation protocol.
- Wash the tissue sample in the 35 mm dish 2x with Hank's balanced salt solution (HBSS) or PBS at room temperature (RT). Keep the sample pinned, allowing for tracking the orientation of the sample through these preparative steps.
- Fix with 0.8 mL of 0.05% malachite green/ 2.5% glutaraldehyde/ 0.1 M sodium cacodylate (pH 6.7 – 7.0) for 20 min on ice.
- Remove fixative and wash sample 4x for 5 min each with 0.1 M sodium cacodylate. Remove 0.1 M sodium cacodylate.
- Postfix with 0.8 mL of 0.8% K3Fe(CN)6 or K4Fe(CN)6 / 1.0% OsO4 K3Fe(CN)6 in 0.1 M sodium cacodylate. Place a cover over the dish to keep the light out (OsO4 is light-sensitive). Stain for 20 min to 1 h at RT.
- Remove the OsO4 and rinse 4x for 5 min each with 0.1 M sodium cacodylate buffer. Remove sodium cacodylate buffer and stain with 1% tannic acid for 20 min on ice.
- Remove tannic acid and wash for 5 min with 0.1 M sodium cacodylate buffer 1x and 2x for 5 min each with molecular-grade distilled (DI) water.
- Stain with 0.5% uranyl acetate (UA) for 1 h RT or overnight at 4 °C in the dark.
- Remove the uranyl acetate and rinse 5x for 5 min each with molecular-grade distilled water. Before the last wash and while the sample is still submerged in DI water, cut the silicone mat around the pinned sample with a razor blade. Lift this small piece of silicone mat out of the plate and place it into a polypropylene tube.
NOTE: It is important the sample remains pinned to the silicone piece while in the tube. Switching to polypropylene tubes in this step is necessary because the propylene oxide used in subsequent steps will degrade the polystyrene of the 35 mm plates. - After removing the last buffer, briefly rinse 1x with 25% ethanol. Discard the 25% ethanol and incubate with 25% ethanol for 3 min on ice.
- Remove the 25% ethanol. Briefly rinse 1x with 50% ethanol. Discard the 50% ethanol and incubate with 50% ethanol for 3 min on ice.
- Remove the 50% ethanol and briefly rinse 1x with 75% ethanol. Discard the 75% ethanol and incubate with 75% ethanol for 3 min on ice.
- Remove the 75% ethanol and briefly rinse 1x with 95% ethanol. Discard the 95% ethanol and incubate with 95% ethanol for 3 min on ice.
- Remove the 95% ethanol and wash 3x, at least 10 min each time, with 100% ethanol (200 proof). Remove 100% ethanol and add propylene oxide (PO). Rinse 3x at least 10 min each, with PO.
- Prepare the resin mix as described below.
- Warm the resin components at 60 °C to liquify fully. Thoroughly mix 12.5 mL of Embed-218, 7.5 mL of Araldite 502, and 27.5 mL of DDSA together in a 50 mL tube at 60 °C. This mixture can be kept for 6 months at 4 °C. Warm it to 60 °C before using. Aliquot the required amount prior to use.
- Embed the samples as described below.
- Place an aliquot of the resin mix in a tube and add 20 µL/mL of DMP-30 activator. Mix thoroughly by rotating at 60 °C; the color should darken.
- Dilute this aliquot of activated resin by 50% in PO and allow the resin and PO to mix completely.
- Remove the last PO rinse from the sample-containing tube and replace it with the 50% resin/PO mixture, nearly filling the tube completely. Rotate the sample overnight at room temperature.
- The following day, mix a new aliquot of resin (1 mL per sample) with 20 µL/mL of DMP-30.
- Place the sample in a polypropylene dish with a small volume of 50% resin/PO mix from the sample tube under a dissecting microscope (~6x magnification) and carefully take the pins out of the tissue. Transfer only the tissue to another polypropylene dish containing a small amount of 100% resin with a DMP-30 activator.
- Fill a silicone embedding mold with the 100% resin with the DMP-30 activator and carefully place the sample in the mold. Place mold in oven and bake at 60 °C for at least 48 h . The prepared sample is shown in Figure 2B.
- Perform montaged wide-area transmission electron microscopy (WA-TEM) imaging as described in10,11. See Figure 3 for a depiction of the process going from an initial puncture to a 3-dimensionally rendered 1-min thrombus. The inclusion of a detailed protocol for montaged WA-TEM electron microscopy imaging is not included here and is beyond the scope of the present protocols on sample preparation.
NOTE: WA-TEM is an underappreciated capability of most modern transmission electron microscopes with a computerized stage. Shareware software supporting these functions10,11 is available from the group of Dr. David Mastronarde, University of Colorado, Boulder (http://bio3d.colorado.edu/SerialEM/, http://bio3d.colorado.edu/imod/.)
Representative Results
Quantitation of drug effects on puncture wound bleeding time
Puncture wound bleeding times provide a physiological model of a drug risk that can be readily carried out in mice. Outcomes that come from a puncture wound experiment are predictive. Here, we show a dabigatran dose-response bleeding curve. Dabigatran, a thrombin inhibitor, is used as an oral direct-acting anti-coagulant, a so-called DOAC12. The jugular vein puncture wound model was used to assess the risk inherent in different doses of dabigatran through potential prolonged bleeding through delayed thrombus formation in mice. A varying dosage of 15, 50, or 150 μg/kg of dabigatran was administered intravenously (i.v.) 20 min before the jugular vein puncture. We show the bleeding time means at various concentrations of dabigatran (Figure 4). At dose level, a significant prolongation of bleeding time is observed, p <0.051. This indicates a significant risk factor in the use of the drug in humans. Experimentally, the drug at these doses has a significant effect on the ultrastructure of the forming thrombus.
Assessing platelet activation state in a forming puncture wound thrombus
The WA-TEM approach provides both an overview of a wound area in a single plane when viewed at lower magnification and much detail at higher magnification (Figure 5). The details obtained can be directly placed within the context of the overview. We have used this approach to assess the recruitment of discoid-shaped platelets to a forming puncture wound thrombus. Circulating platelets are discoid, hence the name, and contain a full set of secretory granules. A key question has been how circulating platelets can be recruited to the growing thrombus without disturbing the free-flowing properties of the remaining platelets. Our work points to a Tether and Activate hypothesis in which circulating platelets are recruited to the thrombus, one by one, through a tethering process as discoid platelets containing a full set of organelles2. The evidence for this comes from WA-TEM of jugular vein thrombi 1 min post-puncture. Following sectioning to yield full thrombus slices from the middle of the still bleeding wound, we imaged 500-800 frames, 4,000 by 4,000 pixels, 3.185 nm pixel size in a rectangular XY raster grid across the complete dimension of the thrombus in cross-section using an automated stage and a transmission electron microscope using SerialEM software10. Frames were then stitched together using eTomo software, part of the IMOD program suite11. This results in a single image of varying size, for example, 120,000 pixels by 90,000 pixels that be full screen at low zoom, 0.02, or at detail at higher zoom, 0.25 to 1.0 using 3DMOD software, again a part of the IMOD program suite (Figure 5). In net, the software-driven analysis found that peripherally bound, thrombus-associated discoid platelets within the puncture hole were spaced apart as beads on a string. A short distance within the thrombus, the discoid shape was less distinct, and the platelets were more tightly associated with one another. Deeper within the thrombus, platelets were rounded, tightly packed, and partially degranulated. A sequence of morphology suggested the initial binding of platelets to the thrombus through a long tether molecule, likely von Willebrand factor, and that any further platelet activation steps were restricted to the interior of the forming thrombus. Hence, platelet recruitment to the thrombus could occur without disturbing the circulation of blood within the jugular vein.
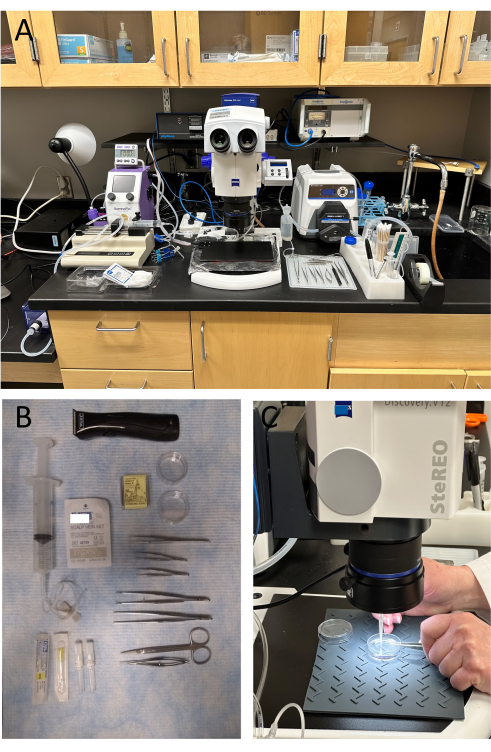
Figure 1: Surgical set-up to perform a jugular vein/femoral artery puncture wound thrombus experiment. Surgical equipment (A) and instruments (B) are carefully arranged for efficiency of movement since timing is critical during the experiment. (C) After perfusion fixation, the portion of the vessel containing the puncture wound thrombus is pinned to a silicone mat in a 35 mm culture dish while viewing under the light microscope. Please click here to view a larger version of this figure.
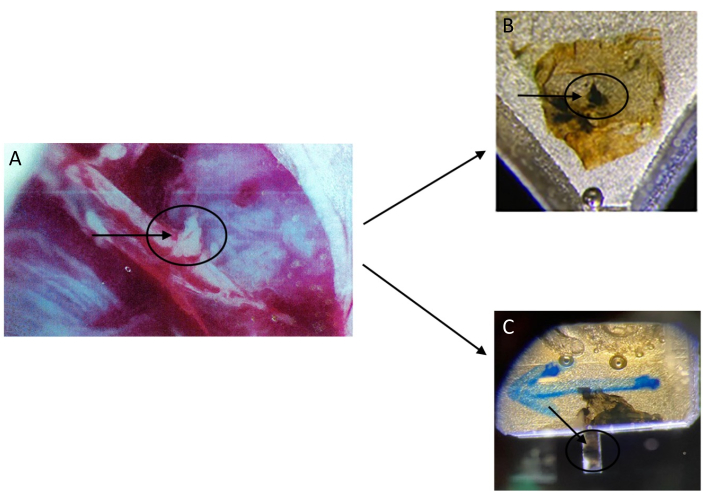
Figure 2: Going from a blood vessel to plastic embedded samples that will be sectioned for either WA-TEM or SBF-SEM. (A) A femoral artery with a puncture wound and accumulating extra-vascular thrombus formation (circle and arrow) is shown. (B) A thrombus (circle and arrow) is embedded in plastic for sectioning for WA-TEM with a microtome for subsequent 2D imaging. (C) A thrombus (circle and arrow) is embedded in plastic, trimmed to give a short stick, less than 1 mm, for attachment to a pin within an SBF-SEM imaging chamber. Please click here to view a larger version of this figure.
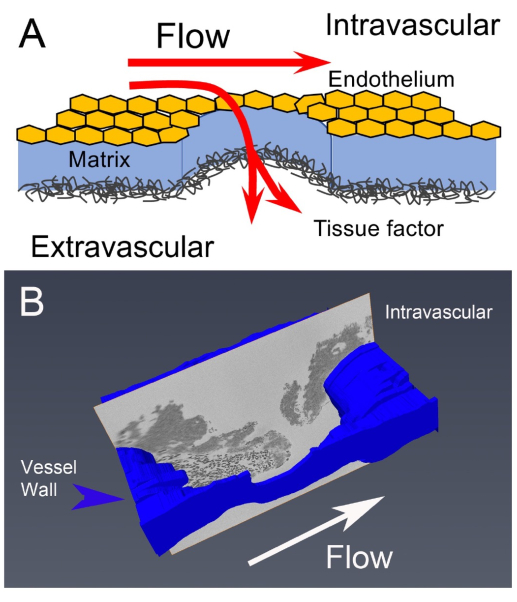
Figure 3: Puncture wound to thrombus formation: A schematic of preparative flow from a puncture wound (A) to an image slice of a 1 min post-puncture jugular vein thrombus (B). The vessel wall is labeled in blue on the image slice, the small black dots near the vessel wall are red blood cells, and the thrombus platelet aggregates appear as dark gray areas. This figure has been modified from1. Please click here to view a larger version of this figure.
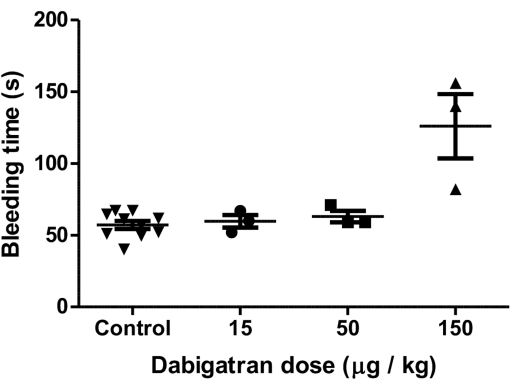
Figure 4: An example of data, in graphic form, generated in a jugular vein puncture wound experiment. A dabigatran dose-response bleeding curve is shown. This figure has been modified from1. The bleeding time means at various concentrations of dabigatran are shown along with error bars indicating plus or minus standard deviation. Statistical analysis using the student's t-test indicated a p-value of <0.05. Please click here to view a larger version of this figure.

Figure 5: WA-TEM images of a slice stitched together to form a montage. (A) Individual high-resolution frames are collected and then stitched together for the purpose of placing features in a larger context. (B) By zooming up, important details can be brought out in context (poly: polymorphonuclear leukocyte, endo: endothelium, and col: collagen fibers). Please click here to view a larger version of this figure.
Discussion
We present a detailed puncture wound procedure for producing hemostatic thrombi in jugular veins and femoral arteries, their in situ perfusion fixation, and sample processing for montaged wide-area transmission electron microscopy. The overall procedures are useful for generating hemostatic thrombi for ultrastructural analysis and for comparing bleeding times in experimental mice, for example, mice treated with different types and dosages of pharmaceuticals. It is also useful for comparing bleeding times in control wild-type mice to bleeding times in knockout mice (i.e., knockouts for various glycoproteins in platelets that interact with adhesive ligands) and subsequent high-resolution electron microscopy. The protocol can be readily adapted to immunofluorescence analysis as the primary readout with the loss of features that make electron microscopy a strong choice. On the other hand, immunofluorescence is a prime tool for locating proteins within a structure. As with any technical protocol, there are some steps that are more critical than others and there are steps within the protocol that can be treated as choice points important to subsequent analysis.
The critical steps, choice points for subsequent analysis, and limitations are described as follows. The actual needle puncture step: The puncture wound is made with a syringe needle held at an angle of 25° to the vein or artery. This minimizes the chance of puncturing across both sides of the vein or artery. A 90° angle on the other hand maximizes the chances of not scraping the intravascular endothelial layer or damaging the extravascular collagen matrix on the outside of the blood vessel but carries a greater risk of puncturing both sides of the vein or artery. The choice is one of experience, hands, and confidence.
The primary fixation here is done with perfused paraformaldehyde, a small molecule containing a single reactive aldehyde group. Because it contains a single chemically reactive group, paraformaldehyde has little tendency to cross-link cellular components and little tendency to destroy antigenicity and hence, in sum, is considered to be a comparatively weak fixative to glutaraldehyde, a common fixative for electron microscopy, which contains two reactive aldehydes and hence produces much more molecular cross-linking13. This is an in situ step designed to preserve the structure. The perfusion steps clear non-thrombi-trapped red blood cells from the preparation. That gives a preparation that is free of most red blood cells and makes it easy to focus on platelet properties. We have explored using in situ fixation with a glutaraldehyde/paraformaldehyde mix applied extravascularly. This procedure is effective, particularly in a high flow/pressure situation. However, it does fix circulating red blood cells in place. This can complicate subsequent segmentation analysis of electron micrographs for the shape and composition of the platelet aggregates forming the puncture wound thrombus. The use of glutaraldehyde greatly complicates alternative immunofluorescence procedures because of the high level of autofluorescence generated by the fixative.
The pinning/splaying of the fixed blood vessel out on a silicone pad is crucial to locating the thrombus for embedding purposes. In this step, the orientation of the sample with respect to blood flow is tracked and known for subsequent steps. The puncture hole and formed thrombus can be readily located in the open blood vessel. In contrast, the thrombus is difficult to find in excised, intact blood vessels. That would be especially true in a sample rich in fixed circulating red blood cells. Being able to track where the needle in the haystack is located is crucial.
Post-splaying the blood vessel open and photographing where the formed thrombus, puncture hole, is located presents a choice point. In the overall protocol, the presented subsequent steps are specific to preparing the sample for WA-TEM. At this step, other protocol choices could be made. On the one hand, the sample could be processed for immunofluorescence microscopy rather than electron microscopy. On the other hand, the sample could be processed for serial block face scanning electron microscopy (SBF-SEM or another volume electron microscopy approach) instead of WA-TEM. The processing steps for SBF-SEM are decidedly different because to prepare for this procedure, all heavy metal staining steps for electron microscopy must be done pre-embedding4.
The heavy metal staining steps in this protocol are designed to highlight membranes versus ribosomes and chromosomes, biological complexes rich in nucleic acid. Other heavy metal staining protocols could be used, for example, Storrie and Attardi14 and Liu et al.15.
Attention to differences in veins and arteries is important. The higher pressure of the artery versus the vein must be offset16,17. For example, a smaller diameter needle is used to produce the femoral artery wound versus the jugular wound. This compensates for the higher pressure, giving nearly equal bleeding cessation time. In the jugular vein puncture wound procedure, preference is given to using paraformaldehyde as a fixative under hypertonic conditions because paraformaldehyde, as a small, single aldehyde-containing fixative, acts rapidly but is a weak fixative that tends to leave antigenicity intact. That means that the protocols described here can be complemented by antibody-mediated localization studies at either the light or the electron microscope level18,19. In the case of the femoral artery puncture wound, glutaraldehyde, a stronger cross-linking fixative, is used in conjunction with paraformaldehyde in a 0.1 M sodium cacodylate buffer, pH 7.4, to stabilize and fix arterial thrombi. This modification is necessitated to offset the higher blood pressure of an artery versus a vein. In the arterial case, the sodium cacodylate concentration is half the concentration used in the fixative solution for jugular vein thrombi.
The sample processing procedure for electron microscopy presented here is tailored to WA-TEM and gives limited-volume electron microscopy information. WA-TEM produces a limited series of high-resolution images, 3.185 nm pixel size or smaller, across full thrombi cross sections either parallel or perpendicular to flow1. A combination of glass and diamond knives is used to manually produce a series of cross sections at an estimated 10%, 25%, 50%, 75%, and 90% into the thrombus. Achieving full thrombus cross sections requires imaging hundreds of frames with a 4,000 x 4,000 pixels electron microscope quality camera. The frames are then stitched together. Nearly 400 – 800 frames are required to achieve a full cross-section image. WA-TEM can be complemented by SBF-SEM. SBF-SEM produces a series of sequential images of the block face as it is progressively cut away in small steps with a diamond knife enclosed within the vacuum of the SEM imaging chamber1. These images can be put together to give a full 3-dimensional rendering of the forming thrombus. Because the microtome is inside the SEM imaging chamber, all staining steps for SBF-SEM must be done pre-embedding. The combination of these two electron microscope approaches allows one to image at ~3 nm or more resolution across a limited number of cross-sections, WA-TEM, or across the full depth of the near millimeter size puncture wound thrombus, SBF-SEM. Viewing the stitched WA-TEM images in 3DMOD software10 allows rapid manipulation between various zoom levels so that even high-resolution views can be placed in context within the full thrombus width cross-section.
In conclusion, we present a proven protocol for producing puncture wound thrombi in mice, fixing them in situ, and processing them for electron microscopy in a manner where the thrombus can be readily located within the vein or artery and its orientation with respect to blood flow traced. Various decision points are presented so the user can make choices as to how the samples will be analyzed. We focus on preparation for electron microscopy that highlights membranes versus other components in the cell. In the end, the portion of the protocol that is most general to other investigations is the appreciation by the authors that wide-area transmission electron microscopy (WA-TEM) is a montaging approach that supports nanoscale knowledge of cells, i.e., platelets within a large structure, near millimeter size, here the forming thrombus. The resulting ability to place detail within the overall context is the central strength of combining the various steps into a full procedure.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors extend thanks to colleagues at the University of Arkansas for Medical Sciences (Jerry Ware and Sung W. Rhee), the University of Pennsylvania (Tim Stalker and Lawrence Brass), the University of Kentucky (Sidney W. Whiteheart and Smita Joshi), and the National Institute of Bioimaging and Bioengineering of the National Institutes of Health (Richard D. Leapman and Maria A. Aronova) from whom we have learned much. The authors express appreciation to the American Heart Association and the National Heart Lung and Blood Institute of the National Institutes of Health (R01 HL119393, R56 HL119393, R01 155519 to BS and subawards from NIH grants R01 HL146373 and R35 HL150818) for financial support.
Materials
| 0.9% Normal Saline Solution | Medline | BHL2F7123HH | |
| 27G x 3/4 EXELint scalp vein set | Medline | NDA26709 | |
| 30G x 1/2 EXELint hypodermic needles | Medline | NDA264372 | |
| 33G x 1/2 EXELint specialty hypodermic needles | Medline | NDA26393 | |
| 50 mL Conical Tubes | Fisher Scientific | 06-443-20 | |
| Alcohol Prep Pads (70% Isopropyl Alcohol) | Medline | MDS090670Z | |
| Aluminum Foil | Fisher Scientific | 01-213-100 | |
| Animal Heating Plate | Physitemp Instruments | HP-1M | |
| Araldite GY 502 | Electron Microscopy Sciences | 10900 | |
| Axiocam 305 Color R2 Microscopy Camera | Carl Zeiss Microscopy | 426560-9031-000 | |
| BD Luer-Lok Syringes, 20 mL | Medline | B-D303310Z | |
| Calcium Chloride | Fisher Scientific | C79-500 | |
| Cell Culture Dishes 35mm x 10mm | Corning Inc. | 430165 | |
| Cotton Tipped Applicators | Medline | MDS202055H | |
| DMP-30 Activator | Electron Microscopy Sciences | 13600 | |
| Dodecenyl Succinic Anhydride/ DDSA | Electron Microscopy Sciences | 13700 | |
| Dressing Forceps, 5", curved, serrated, narrow tipped | Integra Miltex | 6-100 | |
| Dressing Forceps, 5", standard, serrated | Integra Miltex | 6-6 | |
| EMBED 812 Resin | Electron Microscopy Sciences | 14900 | |
| Ethyl Alcohol, anhydrous 200 proof | Electron Microscopy Sciences | 15055 | |
| Fisherbrand 4-Way Tube Rack | Fisher Scientific | 03-448-17 | |
| Fisherbrand Digital Timer | Fisher Scientific | 14-649-17 | |
| Fisherbrand Single Syringe Infusion Pump | Fisher Scientific | 7801001 | |
| Gauze Sponges 2" x 2"- 4 Ply | Medline | NON26224H | |
| Glutaraldehyde (10% Solution) | Electron Microscopy Sciences | 16120 | |
| Isoflurane Liquid Inhalant Anesthesia, 100 mL | Medline | 66794-017-10 | |
| Jeweler-Style Micro-Fine Forceps, Style 5F | Integra Miltex | 17-305 | Need 2 pairs. |
| L/S Pump Tubing, Silicone, L/S 15; 25 Ft | VWR | MFLX96410-15 | |
| L-Aspartic Acid | Fisher Scientific | BP374-100 | |
| Lead Nitrate | Fisher Scientific | L-62 | |
| Malachite Green 4 | Electron Microscopy Sciences | 18100 | |
| Masterflex L/S Easy-Load II Pump Head | VWR | MFLX77200-62 | |
| Masterflex L/S Variable Speed Digital Drive | VWR | MFLX07528-10 | |
| MSC Xcelite 5" Wire Cutters | Fisher Scientific | 50-191-9855 | |
| Osmium Tetroxide 4% Aqueous Solution | Electron Microscopy Sciences | 19150 | |
| Paraformaldehyde (16% Solution) | Electron Microscopy Sciences | 15710 | |
| Physitemp Temperature Controller | Physitemp Instruments | TCAT-2LV | |
| Potassium Ferrocyanide | Sigma-Aldrich | P-8131 | |
| Propylene Oxide, ACS Reagent | Electron Microscopy Sciences | 20401 | |
| Pyrex Glass Beakers | Fisher Scientific | 02-555-25B | |
| Rectal Temperature Probe for Mice | Physitemp Instruments | RET-3 | |
| Scotch Magic Invisible Tape, 3/4" x 1000" | 3M Company | 305289 | |
| Sodium Cacodylate Buffer 0.2M, pH 7.4 | Electron Microscopy Sciences | 11623 | |
| SomnoFlo Low Flow Electronic Vaporizer | Kent Scientific | SF-01 | |
| SomnoFlo Starter Kit for Mice | Kent Scientific | SF-MSEKIT | |
| Stainless Steel Minutien Pins | Fine Science Tools | 26002-10 | |
| Stereomicroscope steREO Discovery.V12 | Carl Zeiss Microscopy | 495015-9880-010 | |
| Sylgard 184 Silicone Elastomer | World Precision Instruments | SYLG184 | silicone mat |
| Tannic Acid | Electron Microscopy Sciences | 21700 | |
| Thiocarbohydrazide (TCH) | Sigma-Aldrich | 88535 | |
| Uranyl Acetate | Electron Microscopy Sciences | 22400 | |
| Vannas Spring Micro Scissors | Fine Science Tools | 15000-08 | |
| Von Graefe Eye Dressing Forceps, 2.75", Curved, Serrated | Integra Miltex | 18-818 | Need 2 pairs. |
| Wagner Scissors | Fine Science Tools | 14068-12 | |
| Wahl MiniFigura Animal Trimmer | Braintree Scientific | CLP-9868 | |
| Zen Lite Software | Carl Zeiss Microscopy | 410135-1001-000 |
References
- Rhee, S. W., et al. Venous puncture and wound hemostasis results in a vaulted thrombus structured by locally nucleated platelet aggregates. Comm Biol. 4 (1), 1090 (2021).
- Pokrovskaya, I. D., et al. Tethered platelet capture provides a mechanism for restricting circulating platelet activation in the wound site. Res Pract Thromb Haemost. 7 (2), 100058 (2023).
- Yadav, S., Storrie, B. The cellular basis of platelet secretion: emerging structure/function relationships. Platelets. 28 (2), 108-118 (2017).
- Joshi, S., et al. Ferric chloride-induced arterial thrombosis and sample collection for 3D electron microscopy analysis. J Vis Exp. (193), e64985 (2023).
- Guerrero, J. A., et al. Visualizing the von Willebrand factor/glycoprotein Ib-IX axis with a platelet-type von Willebrand disease mutation. Blood. 114 (27), 5541-5546 (2009).
- Kato, K., et al. The contribution of glycoprotein VI to stable platelet adhesion and thrombus formation illustrated by targeted gene deletion. Blood. 102 (5), 1701-1707 (2003).
- Tomaiuolo, M., et al. Interrelationships between structure and function during the hemostatic response to injury. Proc NatlAcad Sci U S A. 116 (6), 2243-2252 (2019).
- Cocchiaro, J. L., et al. Cytoplasmic lipid droplets are translocated into the lumen of the Chlamydia trachomatis parasitophorous vacuole. Proc Natl Acad Sci. 105 (27), 9379-9384 (2008).
- Pokrovskaya, I. D., et al. Chlamydia trachomatis hijacks intra – Golgi COG complex – dependent vesicle trafficking pathway. Cell Microbiol. 14 (5), 656-668 (2012).
- Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J Str Biol. 152 (1), 36-51 (2005).
- Kremer, J. R., Mastronarde, D. N., McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J Str Biol. 116 (1), 71-76 (1996).
- Chan, N. C., Eikelboom, J. W., Weitz, J. J. Evolving treatments for arterial and venous thrombosis: role of the direct oral anticoagulants. Circ Res. 118 (9), 1409-1424 (2016).
- Kiernan, J. A. Formaldehyde, formalin, paraformaldehyde, and glutaraldehyde: what they are and what they do. Microscopy Today. 8 (1), 8-13 (2000).
- Storrie, B., Attardi, G. Expression of the mitochondrial genome in HeLa cells: XV. Effect of inhibition of mitochondrial protein synthesis on mitochondrial formation. J CellBiol. 56, 819-831 (1973).
- Liu, S., Pokrovskaya, I. D., Storrie, B. High – pressure freezing followed by freeze substitution: an optimal electron microscope technique to study Golgi apparatus organization and membrane trafficking. Meth Mol Biol. 2557, 211-223 (2023).
- Klarhofer, M., et al. High-resolution blood flow velocity measurements in the human finger. Mag Res Med. 45 (4), 716-719 (2001).
- Chaudry, R., Miao, J. H., Rehman, A. Physiology, cardiovascular. StatPearls. , (2022).
- Pokrovskaya, I. D., et al. SNARE – dependent membrane fusion initiates a-granule matrix decondensation in mouse platelets. Blood Adv. 2 (21), 2947-2958 (2018).
- Pokrovskaya, I. D., et al. 3D ultrastructural analysis of a-granule, dense granule, mitochondria, and canalicular system arrangement in resting human platelets. Res Pract Thrombosis Haemostasis. 4 (1), 72-85 (2019).

.
