Morpholino Injection: A Method to Knockdown Target Gene Expression to Study Function in Zebrafish
Abstract
Source: Hyde D.R., et al. In vivo Electroporation of Morpholinos into the Regenerating Adult Zebrafish Tail Fin. J. Vis. Exp. (2012)
The protocol describes targeted knockdown of gene expression in zebrafish by morpholino injection in the tail.
Protocol
1. Morpholino Injection
- The day prior to injecting the morpholino, make an injection plate (Figure 1). Make sure to cut out a notch at one end of the well, which helps stabilize the fish for the microinjection procedure.
- Dilute the fluorescein-tagged morpholino to the proper concentration (recommend starting with 1.2 mM) and place in a 65 °C water bath, for 5 minutes.
- Load the needle with the morpholino (note: depending on whether you back-fill or front-load your needle will determine whether you load the needle first, or cut the tip first).
- Cut the tip off the needle at an angle.
- Follow the directions of your micro-injection system to inject an approximately 5 nl bubble of morpholino per injection. Larger volumes of morpholino could disrupt the tissue.
- Anesthetize the fish and place on injection plate. Remove all excess liquid and orient the needle to the appropriate location, just distal to the bony ray (Figure 2). Using a microscope and microinjection apparatus appropriate for morpholino injections (see Table of Specific Reagents), inject the morpholino into the fin from anterior to posterior, as shown in Figure 2. Injecting from posterior to anterior causes the fin tissue to roll up during the injection and is very difficult.
- Insert the needle gently into the regenerative tissue, just distal to each bony ray (marked "1" in Figure 2) and push distally until located in the blastema (marked "2" in Figure 2). Be careful not to push the needle through the entire fin. When the needle is correctly localized, inject the morpholino (i.e. click one time). At 1.2 mM, the fluorescein-tagged morpholino solution appears yellow-green, even in normal light conditions. With each injection, a yellow "puff" of morpholino solution can be visualized, which helps localize the injection to the blastema. Approximately 75 nl (10-15 clicks) of morpholino solution should be injected per bony ray. Pause ~ 1 sec between clicks. We only inject the dorsal side, using the ventral as electroporation-only control.
Representative Results
Figure 1. A. Schematic of the injection plate, which is made from agarose and contains a small well to hold the fish during microinjection of the morpholino.
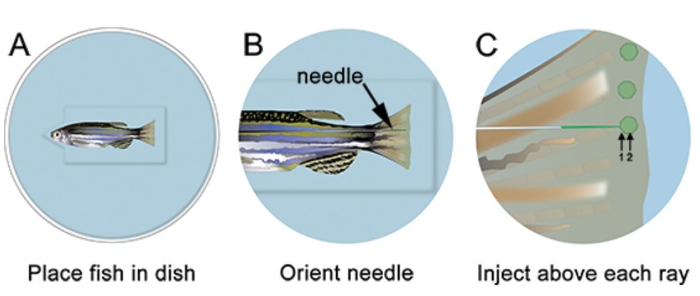
Figure 2. Schematic of morpholino microinjection. A. Place the fish in the dish with the head of the fish in the notch cut out of the well, which will help the fish stay stable. B. At low magnification, arrange the needle so that it is close to the regenerating tissue of the fin. C. At higher magnification, inject the morpholino distal to each bony fin ray (i.e. in each blastema). The needle should enter the tissue just distal to the bony ray (1), and then continue to the location of the blastema (2). Note: the green circles in the schematic are only meant to show the location of the injection. The morpholino can briefly be visualized as a green/yellow "puff" following each injection; however, this does not persist as shown in the schematic.
Materials
| Morpholino | GeneTools, LLC | Morpholino should be custom designed to your protein of interest | |
| Micro-injection pump | World Precision Instruments, Inc. | PV830 | Pneumatic PicoPump Many different microinjection systems could be used |
| Micro-manipulator | World Precision Instruments, Inc. | MMJR | Right-handed (MMJL for lefthanded) |
| Micro-injection needles, 1.0 mm outside diameter | World Precision Instruments, Inc. | 1B100F-4 | These are borosilicate glass capillaries, pulled into a needle |
| Needle holder | World Precision Instruments, Inc. | 5430-ALL | Pico Nozzle Kit; make sure to inset the 1.0 mm pipette gasket |
| Needle puller | Sutter Instrument Co. | P-97 | Other micropipette/needle pullers should also work |
| Microscope | Leica, Nikon, Zeiss | Number varies depending on the manufacturer | Any stereomicroscope with 20X optics and the ability to work with micromanipulators |
| 2-Phenoxyethanol | Sigma-Aldrich | 77861-1L | Anesthesia; dilute 1:1000 in fish system water for procedure, 1:500 for euthanasia |

