Imaging Intracellular Ca2+ Signals in Striatal Astrocytes from Adult Mice Using Genetically-encoded Calcium Indicators
Summary
The properties and functions of astrocyte intracellular Ca2+ signals in the striatum remain incompletely explored. We describe methods to express genetically encoded calcium indicators in striatal astrocytes using adeno-associated viruses of serotype 2/5 (AAV2/5), as well as procedures to reliably image Ca2+ signals within striatal astrocytes in situ.
Abstract
Astrocytes display spontaneous intracellular Ca2+ concentration fluctuations ([Ca2+]i) and in several settings respond to neuronal excitation with enhanced [Ca2+]i signals. It has been proposed that astrocytes in turn regulate neurons and blood vessels through calcium-dependent mechanisms, such as the release of signaling molecules. However, [Ca2+]i imaging in entire astrocytes has only recently become feasible with genetically encoded calcium indicators (GECIs) such as the GCaMP series. The use of GECIs in astrocytes now provides opportunities to study astrocyte [Ca2+]i signals in detail within model microcircuits such as the striatum, which is the largest nucleus of the basal ganglia. In the present report, detailed surgical methods to express GECIs in astrocytes in vivo, and confocal imaging approaches to record [Ca2+]i signals in striatal astrocytes in situ, are described. We highlight precautions, necessary controls and tests to determine if GECI expression is selective for astrocytes and to evaluate signs of overt astrocyte reactivity. We also describe brain slice and imaging conditions in detail that permit reliable [Ca2+]i imaging in striatal astrocytes in situ. The use of these approaches revealed the entire territories of single striatal astrocytes and spontaneous [Ca2+]i signals within their somata, branches and branchlets. The further use and expansion of these approaches in the striatum will allow for the detailed study of astrocyte [Ca2+]i signals in the striatal microcircuitry.
Introduction
Astrocytes are ubiquitous and abundant glial cells of the brain. It is well established that astrocytes serve vital support and homeostatic roles including buffering of K+ concentration in the extracellular space, uptake of neurotransmitters as well as providing nutrients. However, recent studies show that they also display [Ca2+]i signals, which occur spontaneously and are increased by neuronal activity1. The existence of astrocyte [Ca2+]i signaling has been increasingly thought to trigger their communication with neurons, and as such has been interpreted as a form of “Ca2+ excitability” within astrocytes. The available data over the last two decades suggest two settings in which astrocytes and neurons may communicate, perhaps in a bidirectional manner. First, astrocytes often respond with an increase in [Ca2+]i when activated by neurotransmitters and neuromodulators released from neurons2. Second, [Ca2+]i increases within astrocytes cause the release of signaling molecules from astrocytes that in turn may affect neurons and blood vessels. Evidence suggests that molecules released from astrocytes lead to changes in the functions of synapses, circuits and ultimately behavior3-5 via astrocyte-to-neuron signaling. However, this remains a rapidly developing research area, and it has been argued that a better and detailed understanding of astrocyte [Ca2+]i is needed to resolve some of the current uncertainties6.
In past work, it was demonstrated that bulk loading of organic Ca2+ indicator dyes into astrocytes fails to reliably detect [Ca2+]i signals within entire astrocytes in culture and in situ7-10. These findings have been discussed by us and others6,11,12. The emerging picture is that [Ca2+]i signals within astrocyte processes (e.g., branches and branchlets), which are the primary sites for interactions with neurons and blood vessels, have rarely been explored in detail. Recently, the use of genetically encoded calcium indicators (GECIs) such as cytosolic GCaMP3, GCaMP5G and GCaMP6 and plasma membrane tethered versions (e.g., Lck-GCaMP3) has permitted the study of [Ca2+]i signals in small compartments of astrocytes such as thin processes, near the plasma membrane and within entire territories7,8. However, GECIs have one disadvantage over organic Ca2+ indicator dyes and that is the requirement for genetic methods to deliver the encoding genes selectively to astrocytes in vivo for periods of weeks for the GECIs to be appropriately expressed. Expression in vivo is typically achieved using transgenic mice, knock-in mice or with virus based delivery approaches. In the present JoVE article we report methods and procedures employed to deliver GECIs to striatal astrocytes using adeno-associated viruses. We focus on cyto-GCaMP3 as an example, but the same basic procedure works for any other GECI or fluorescent protein based reporter.
Protocol
All animal protocols were in accordance with the US National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee at UCLA.
1.1) Prepare Micropipette and AAV2/5 Virus Loading
- Use fine borosilicate glass micropipettes for the injection of the virus. Pull the micropipette using a two-step pulling program with a vertical puller. Bevel the pipette at an angle of 40° by using a pipette grinder. The pipette that is ideal for use will have a tip diameter of 20 – 40 μm and a shank length of 6 – 7 mm. Autoclave the pipette and surgical instruments. The drill is sterilized by wiping with 70% ethanol.
- Store the virus AAV2/5-GfaABC1D.cyto-GCaMP3.SV40 (1.5 x 1013 gc/ml), at -80 °C in 10 μl aliquots. Just before surgery, take out the aliquots out of the freezer and store on ice until used for filling the micropipette.
- Fill the micropipette with virus using a syringe pump. Tightly connect one end of a piece of tubing, through an appropriately sized tubing compression fitting, to a glass syringe with a reinforced plunger. Attach the autoclaved micropipette to the other end of the piece of tubing through an appropriately sized removable needle compression fitting.
- Before loading the viral vector, remove air bubbles from the pumping system including tubing, syringe and micropipette by filling the system with mineral oil colored with Sudan red IV (~1 mg/50 ml) in a 1 ml syringe with a needle (27 G).
- Place a clean piece of parafilm under the glass micropipette, and dispense 5 – 10 μl of the viral vector onto the parafilm.
- Suck the viral vector by moving the plunger of the syringe backward at 0.5 μl/min, and mark the boundary between the vector and the oil on the glass pipette.
1.2) Anesthesia, Hair Cutting, Head Fixation and Preparation for Craniotomy
- Put a mouse (P49-63, C57BL/6) into the chamber filled with N2O and O2, and 5% isoflurane. Assess the depth of anesthesia by loss of purposeful movement and a slowed respiratory rate (~60 – 90/min). Cut hair on the head when the mouse is anesthetized.
- Fit the mouse in the stereotaxic frame, with its head secured by blunt ear bars and its nose placed into an anesthesia and ventilation system. Maintain continuous isoflurane at 2 – 3%. Apply artificial tears ointment to both eyes since mice lose their blink reflex under anesthesia.
- Administer 0.05 ml of Buprenorphine (0.1 mg/ml) by subcutaneous injection for pain relief.
- Clean surgical area with 10% povidone iodine and 70% alcohol three times, alternating between the two solutions and starting with povidone iodine. Start from the front and wipe toward the back to remove any already cut hairs.
- Make a skin incision on top of the head which starts between the eyes and ends between the ears.
1.3) Craniotomy and Microinjections
- Remove the periosteum by swiping with a cotton swab and then dry the surface of the skull with dental cotton balls. Make a mark around the injection site, and drill around the edge of the mark using a small steel burr powered by a high speed drill.
- Remove the bone and clean the surface with saline.
- Place the pipette into the left dorsal lateral striatum with the following coordinates (mm): anterior-posterior +0.8, medial-lateral: +2.0, dorsal-ventral from the pial surface: -2.4 13. Note that these coordinates mean that the tip of the microinjection needle sits just above the dorsolateral striatum (Figure 1A, B), meaning that it only slightly penetrates within this nucleus. Avoid damaging blood vessels. If the pipette happens to be right on a blood vessel, slightly adjust the coordinates by ~0.1 – 0.2 mm.
- Start injection with injection rate set at 0.2 μl/min, and typically inject 1 to 1.5 μl of virus.
- Leave the pipette in place after the injection for 10 min, and then withdraw the pipette slowly.
- Finish by closing the surgical wound with continuous suture external nylon suture.
1.4) Recovery
- Put the mouse in a clean cage on a heating pad for 24 hr. Do not return a mouse that has undergone surgery to the company of other animals until fully recovered. Do not leave the mouse unattended until it has regained sufficient consciousness to maintain sternal recumbency.
- Add Trimethoprim Sulfamethoxiazole (5 ml per 500 ml water, containing 40 mg trimethoprim and 200 mg sulfamethoxiazole) in the water for a week. Administer Buprenorphine two times per day for up to 3 days after surgery.
- Maintain the mouse on a 12 hr light-dark cycle, fed and watered daily, and is weighted once per day up to 3 days after surgery. In these 3 days, any mouse with a loss of more than 10% body weight is considered as compromised, and will be euthanized instead of being subjected to experimentation.
- Separately, change the mouse cage twice per week, and monitor for general health at least once per day. Typically, the mouse is used within 2 – 3 weeks after the stereotaxic microinjections.
2. Acute Brain Slice Preparation for Confocal Ca2+ Imaging
2.1) Preparation of Cutting and Recording Solutions.
- Prepare cutting solution comprising (mM): 194 sucrose, 30 NaCl, 4.5 KCl, 10 D-glucose, 1 MgCl2, 1.2 NaH2PO4, and 26 NaHCO3, saturated with 95% O2 and 5% CO2.
- Prepare recording solution comprising (mM): 124 NaCl, 4.5 KCl, 1 MgCl2, 10 D-glucose, 2 CaCl2, 1.2 NaH2PO4, and 26 NaHCO3; pH 7.3 – 7.4, 290 – 295 mOsm, saturated with 95% O2 and 5% CO2. Fill the brain slice holding beaker with recording solution, and keep it at 34 °C.
2.2) Slicing
- Two to three weeks after stereotaxic microinjections, deeply and terminally anesthetize the mouse, and then decapitate it.
- Extract the brain, and use a blade to remove the uninjected right hemisphere, and mount the left one onto the vibratome tray using super glue. Fill the vibratome tray with ice cold cutting buffer, and keep saturating it with 95% O2 and 5% CO2.
- Cut coronal or sagittal striatal brain slices at 300 μm thickness. Usually, 4 – 5 coronal, or 6 – 7 sagittal striatal slices can be collected.
- Transfer the slices to the slice holding beaker warmed at 34 °C, and keep them there for 30 min before storing them at room temperature for subsequent recording.
- Incubate the brain slices in oxygenated recording buffer at room temperature for at least 30 min before [Ca2+]i imaging.
3. Immunohistochemistry (IHC)
- Two weeks after virus injection, perfuse the mouse transcardially with PBS followed by 10% formalin in PBS.
- Remove, post-fix overnight, and cryoprotect the brain in buffered 30% sucrose for at least 2 days.
- Prepare 40 μm sections using a cryostat microtome.
- Rinse sections 3 times with PBS at 10 min interval.
- Incubate sections for 1 hr in PBS with 10% normal goat serum and with 0.5% Triton X-100.
- Incubate sections overnight at 4 °C in primary antibody prepared in PBS with 0.5% Triton X-100. The primary antibodies used were as follows: chicken anti-GFP 1:1,000, mouse anti-S100β 1:400, mouse anti-NeuN 1:600, mouse anti-glutamine synthetase 1:300.
- Rinse sections 3 times with PBS at 10 min intervals.
- Incubate sections with the secondary antibodies prepared in PBS with 10% normal goat serum for 2 hr at room temperature. The second antibodies were as follows: goat anti-mouse Alexa546 1:500, goat anti-chicken Alexa488 1:500.
- Mount the sections on glass slides, dry and apply several drops of antifade mounting medium, then slowly cover the sections with a glass coverslip. Store the slides at 4 °C.
- Take images on a confocal microscope with a 40X oil immersion lens with a numerical aperture of 1.3.
4. Confocal [Ca2+]i Imaging
NOTE: [Ca2+]i imaging was done using a confocal microscope with a 40X water immersion objective lens with a numerical aperture of 0.8.
- Between 1 and 5 hr after slicing, place slice in the recording chamber and superfuse with oxygenated recording buffer at room temperature with flow rate of 1 – 2 ml per min. Then place a platinum harp with nylon strings on top of the slice to minimize movement during the experiment.
- Visualize and locate the dorsal striatum with bright field luminescence under a 10X water immersion objective.
- Switch to the 40X water immersion objective lens, and turn on the 488 nm line of the Argon laser. By changing the focal plane, find cells located about 20 μm below the slice surface, displaying basal fluorescence in soma, branches and branchlets. Of note, compromised cells usually display fluorescence level above average, which should be avoided.
- Use ‘frame scan’ mode to start scanning. Usually, the laser intensity adjusted to 0.5 – 5% of the maximum output is sufficient to image [Ca2+]i with cyto-GCaMP3. For monitoring [Ca2+]i dynamics in the whole territory of astrocytes, the ‘frame scan’ with a scan rate of 1 sec per frame or faster is recommended, but this needs to be decided depending on the experiment in question.
- If necessary, to image [Ca2+]i in processes, use digital magnification of 2 – 3 fold to monitor 1 – 2 astrocytes in the whole field of view.
- To avoid saturated pixels during recording, use ‘HiLo’ lookup mode to pseudocolor the cells. Always start with a “pilot recording” for about 5 min, to test if red color appears as indication of saturation. If so, lower the laser output power, or lower the PMT/Gain/offset values and perform another pilot recording to test the pixel values are in the non saturated range. Typically, the combined parameters used for imaging astrocyte [Ca2+]i in the striatum are as follows: laser output power 0.5 – 5% (of 10 mW), PMT 550 – 650 Volts, Gain 2.0 – 3.5X, and offset 0 – 5%.
Representative Results
For astrocyte specific expression of cyto-GCaMP3 in the striatum, we used adeno-associated virus (AAV) of the 5 serotype, and the GFAP GfaABC1D promoter (Figure 1A), which has been previously shown to drive robust GCaMP3 and reporter gene expression in hippocampal and cortical astrocytes8,14. Two weeks after virus microinjection into the mouse striatum, the mouse (~10 weeks old) was perfused and IHC was performed on thin brain sections to evaluate cyto-GCaMP3 expression in the striatum (Figure 1B). We detected cyto-GCaMP3 using GFP antibodies and also stained the slices with a known astrocyte marker, S100β. Cyto-GCaMP3 expression was only found in S100β positive cells. No expression was found in neurons visualized with NeuN (Figure 2A and 2B), suggesting astrocyte specific expression. Importantly, cyto-GCaMP3 expression was detected in ~60% of S100β positive cells (Figure 2C).
Astrocytes respond to brain insults such as injury, ischemia and infection by displaying changes that have become known as astrocyte reactivity, which represents a spectrum of potential changes from mild to severe15. We sought to evaluate if virus microinjection caused overt astrocyte reactivity. It was previously shown that reduced expression levels of Glutamine Synthetase (GS) were associated with the reactive astrocytes16,17. Therefore, GS expression in the striatum of WT and virus injected mice was compared. We found no significant changes in GS expression in striatal astrocytes following virus injection (Figure 3A – C), indicating no overt astrocyte reactivity using GS levels as a metric. This general approach could be expanded in future work to evaluate astrocyte reactivity using other markers of reactivity. However, note that GFAP levels may not be a suitable way to measure reactivity in striatal astrocytes, because GFAP is not expressed at detectable levels in most striatal astrocytes under basal conditions18. In summary, by using IHC we conclude cyto-GCaMP3 expression was robust and specific to astrocytes in the striatum, and that virus injection did not cause astrocyte reactivity as assessed by GS expression level changes.
We next prepared acute brain slices from virus injected mice to image [Ca2+]i in striatal astrocytes. Acute 300 μm thick sagittal or coronal slices were prepared, and after a period of recovery the slices were placed in a recording chamber on a confocal microscope for [Ca2+]i imaging using the 488 nm line of an Argon laser. Striatal astrocytes expressing cyto-GCaMP3 could easily be identified in most (usually all) of the striatal slices (~6 – 7 slices per mouse), as they showed visible green fluorescence in cells with clearly bushy morphology (Figure 4). A typical 5 min recording at 1 Hz from a representative field is shown in Figure 4A; at least 10 astrocytes displaying cyto-GCaMP3 could be imaged and are plotted in Figure 4A as Regions of Interest (ROI). Higher magnification imaging with an additional digital zoom of 2.0 – 3.0 was necessary to identify single astrocytes (Figure 4B). Under these circumstances, entire astrocyte territories were revealed by cyto-GCaMP3 expression, as shown in Figure 4B. Spontaneous Ca2+ signals were easily measured in the somata as well as in the branches and branchlets of astrocytes6.
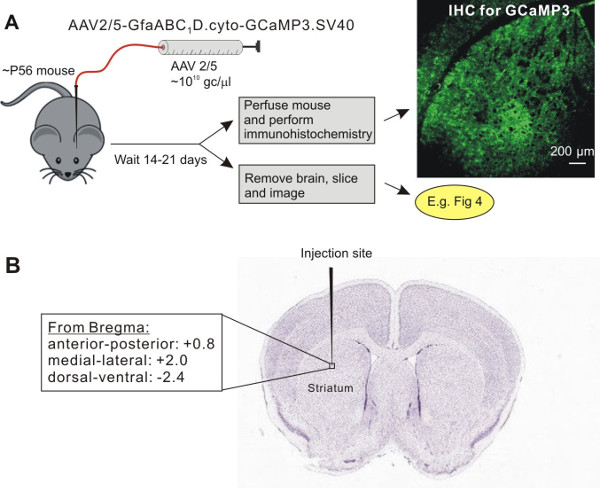
Figure 1: Viral delivery of GECIs (e.g. cyto-GCaMP3) by AAV2/5 into the adult mouse striatum. A. Schematic illustrates the protocol for AAV2/5 microinjections into the dorsolateral striatum. B. Shows the approximate position of the microinjection needle in relation to the striatum and the coordinates used for stereotaxic injections. The image of a Nissl-stained slice in panel B was downloaded from ALLEN BRAIN ATLAS.
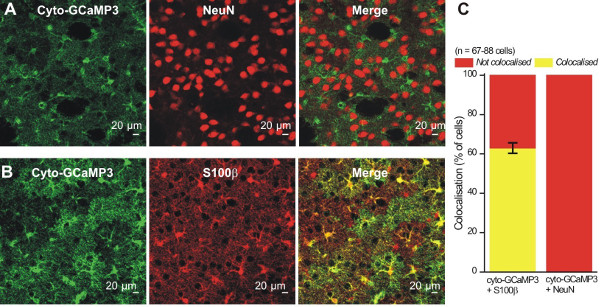
Figure 2: Cyto-GCaMP3 expression was specific to striatal astrocytes. A – B. Representative images showing that cyto-GCaMP3 expression colocalizes with a marker for astrocytes (S100β) (B), but not with a marker for neurons (NeuN) (A). C. Summary bar graph for colocalization experiments like those shown in A and B.
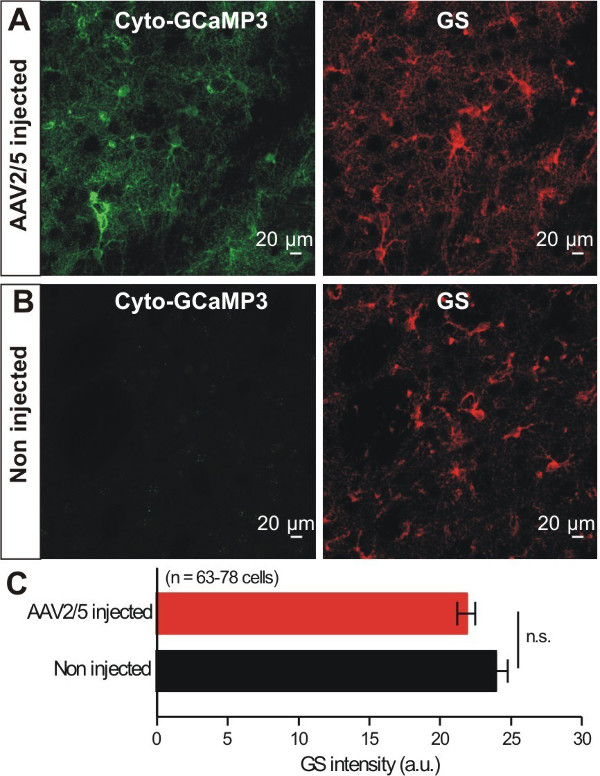
Figure 3: Cyto-GCaMP3 expression within striatal astrocytes did not cause changes in the expression of GS. A – B. Representative images of GCaMP3 and GS IHC from mice that had received AAV2/5 for GCaMP3. Images for control non injected mice are also shown. C. Bar graph summarizes results from experiments such as those in A and B (n.s. indicates not significant using an unpaired Student’s t test, p <0.05).
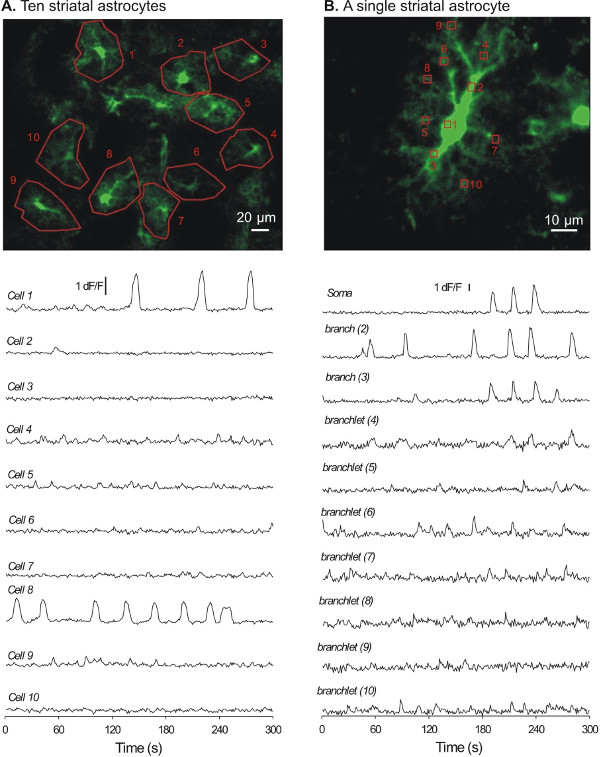
Figure 4: Representative examples of [Ca2+]i signals recorded from striatal astrocytes with cyto-GCaMP3. A. A flattened z-stack image showing 10 astrocytes (numbered 1 – 10).The traces below show [Ca2+]i signals recorded over 5 min from the 10 cells shown in A. B. A flattened and zoomed-in z-stack image for a single astrocyte.The traces below show [Ca2+]i signals recorded over 5 min for the single cell shown in B.
Discussion
The methods described herein have allowed us to express cyto-GCaMP3 in striatal astrocytes in vivo for subsequent [Ca2+]i imaging in situ. This method has advantages over using transgenic or knock-in mice, including robust expression of the targeted protein, rapidity and flexibility of experimental implementation and anatomical specificity. The expression of GCaMP3 using AAV2/5 was found to be specific and robust. The combination of GFAP GfaABC1D promoter with AAV of the serotype 5 is critical to achieve the specificity. One limitation of the technique described here is that virus infection and surgery procedure may cause astrocyte reactivity, especially with high-titer viruses19. Importantly, in the current study, AAV2/5 microinjections with a relative low titer did not cause decreases in GS that have been associated with astrocyte reactivity19. Similar controls have been reported for hippocampal astrocytes8. However, it is important to emphasize the necessity of these controls for each brain area to be studied, not least because astrocytes are heterogeneous20, and because they likely perform different functions in distinct regions of the brain.
One of the important factors that may affect the robustness of the targeted gene expression using virus microinjection is the coordinates in use, which varies among different mouse strains and changes over the development of the animals. Only C57BL/6 mice of ~8 weeks old were used throughout this study. It has been also shown that viral expression of GFP in striatal neurons and astrocytes in rats could be detected 4 days post injection, reached a plateau by 2 – 4 weeks post injection, and then remained stable for 9 months post injection 21. The time course of cyto-GCaMP3 expression has not been precisely determined here, but at least two weeks for virus expression are recommended.
The approach of using AAV2/5 mediated expression of GECIs in striatal astrocytes can now be used to better understand astrocyte [Ca2+]i signaling in the striatum. Specifically, cyto-GCaMP3 expression level is sufficiently high to permit imaging of [Ca2+]i signals in small populations of astrocytes (~10 cells) and within the entire territories of single astrocytes. The detailed analysis of [Ca2+]i signals is still ongoing, but to date [Ca2+]i signals have been detected from distal processes as far as ~50 µm away from the soma (Figure 4). Detailed experiments on astrocyte [Ca2+]i signals and their correlative and causative functions within the striatum are feasible. In the future, refined approaches (i.e., promoters) are needed to deliver GECIs to genetically defined populations of astrocytes so that one can explore calcium signaling within diverse populations of astrocytes20.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
The majority of the work and the personnel involved were supported by NIH grant NS060677 and partly by NIH grants MH099559 and MH104069 (to BSK). Some of the work was also supported by the CHDI Foundation.
Materials
| Syringe Pump | Harvard Apparatus | 704506 | |
| Glass Capillaries | World Precision Instruments | 1B100-4 | |
| Micropipette puller | Narishige | PC-10 | |
| Micropipette grinder | Narishige | EG-40 | |
| pZac2.1 GfaABC1D.cyto-GCaMP3 | Addgene | 44331 | a plasmid sent to UPenn Vector Core for virus packaging |
| I mL syringe | BD | 309628 | |
| syringe needle | BD | 305109 | |
| AAV2/5 virus | UPenn vector core | NA | |
| Sudan red IV | Sigma-Aldrich | 67386 | |
| Mineral oil | CVS Pharmacy | 152355 | |
| Cryostat | Leica | CM3050 S | |
| Stereotaxic instrument | David Kopf Instruments | 900LS | |
| High Speed Rotary Micromotor Kit | FOREDOM | K.1070 | |
| Paraformaldehyde | Santa cruz biotechnology | sc-281692 | |
| Super Glue | Krazy®Glue | KG925 | |
| Microslicer | Ted Pella | DTK-Zero 1 | |
| Confocal microscopes | Olympus | FV300 and FV1000 | |
| Normal goat serum | Vector | S-1000 | |
| chicken anti-GFP | Abcam | ab13970 | |
| mouse anti-s100β | Sigma-Aldrich | S2532 | |
| mouse anti-NeuN | Millipore | MAB377 | |
| mouse anti-glutamine synthetase | Millipore | MAB302 | |
| goat anti-mouse-Alexa546 | Invitrogen | A11003 | |
| goat anti-chicken-Alexa488 | Invitrogen | A11039 | |
| Microscope Slides | Fisher Scientific | 12-550-15 | |
| Cover Glass | Fisher Scientific | 12-548-5J | |
| Mounting Medium | Vector | H-1000 |
Referenzen
- Agulhon, C., et al. What is the role of astrocyte calcium in neurophysiology. Neuron. 59, 932-946 (2008).
- Khakh, B. S., North, R. A. Neuromodulation by extracellular ATP and P2X receptors in the CNS. Neuron. 76, 51-69 (2012).
- Gourine, A. V., et al. Astrocytes control breathing through pH-dependent release of ATP. Science. 329, 571-575 (2010).
- Florian, C., Vecsey, C. G., Halassa, M. M., Haydon, P. G., Abel, T. Astrocyte-derived adenosine and A1 receptor activity contribute to sleep loss-induced deficits in hippocampal synaptic plasticity and memory in mice. J Neurosci. 31, 6956-6962 (2011).
- Shigetomi, E., Jackson-Weaver, O., Huckstepp, R. T., O’Dell, T. J., Khakh, B. S. TRPA1 channels are regulators of astrocyte basal calcium levels and long-term potentiation via constitutive D-serine release. J Neurosci. 33, 10143-10153 (2013).
- Tong, X., Shigetomi, E., Looger, L. L., Khakh, B. S. Genetically encoded calcium indicators and astrocyte calcium microdomains. Neuroscientist. 19, 274-291 (2013).
- Shigetomi, E., Kracun, S., Sofroniew, M. V., Khakh, B. S. A genetically targeted optical sensor to monitor calcium signals in astrocyte processes. Nat Neurosci. 13, 759-766 (2010).
- Shigetomi, E., et al. Imaging calcium microdomains within entire astrocyte territories and endfeet with GCaMPs expressed using adeno-associated viruses. J Gen Physiol. 141, 633-647 (2013).
- Shigetomi, E., Khakh, B. S. Measuring near plasma membrane and global intracellular calcium dynamics in astrocytes. J Vis Exp. 26, (2009).
- Reeves, A. M., Shigetomi, E., Khakh, B. S. Bulk loading of calcium indicator dyes to study astrocyte physiology: key limitations and improvements using morphological maps. J Neurosci. 31, 9353-9358 (2011).
- Li, D. D., Agulhon, C., Schmidt, E., Oheim, M., Ropert, N. New tools for investigating astrocyte-to-neuron communication. Frontiers in Cellular Neuroscience. 7, (2013).
- Davila, D., Thibault, K., Fiacco, T. A., Agulhon, C. Recent molecular approaches to understanding astrocyte function in vivo. Front Cell Neurosci. 7, 272 (2013).
- Paxinos, G., Franklin, K. . The Mouse Brain in Stereotaxic Coordinates. , (2012).
- Perea, G., Yang, A., Boyden, E. S., Sur, M. Optogenetic astrocyte activation modulates response selectivity of visual cortex neurons in vivo. Nat Commun. 5, 3262 (2014).
- Sofroniew, M. V., Vinters, H. V. Astrocytes: biology and pathology. Acta Neuropathol. 119, 7-35 (2010).
- Eid, T., et al. Loss of glutamine synthetase in the human epileptogenic hippocampus: possible mechanism for raised extracellular glutamate in mesial temporal lobe epilepsy. Lancet. 363, 28-37 (2004).
- Eid, T., Williamson, A., Lee, T. S., Petroff, O. A., de Lanerolle, N. C. Glutamate and astrocytes–key players in human mesial temporal lobe epilepsy. Epilepsia. 49 Suppl 2, 42-52 (2008).
- Tong, X., et al. Astrocyte Kir4.1 ion channel deficits contribute to neuronal dysfunction in Huntington’s disease model mice. Nat Neurosci. 17, 694-703 (2014).
- Ortinski, P. I., et al. Selective induction of astrocytic gliosis generates deficits in neuronal inhibition. Nat Neurosci. 13, 584-591 (2010).
- Zhang, Y., Barres, B. A. Astrocyte heterogeneity: an underappreciated topic in neurobiology. Curr Opin Neurobiol. 20, 588-594 (2010).
- Reimsnider, S., Manfredsson, F. P., Muzyczka, N., Mandel, R. J. Time course of transgene expression after intrastriatal pseudotyped rAAV2/1, rAAV2/2, rAAV2/5, and rAAV2/8 transduction in the rat. Mol Ther. 15, 1504-1511 (2007).

