Acetylcholine Re-Challenge After Intracoronary Nitroglycerine Administration
Summary
This protocol presents the acetylcholine rechallenge after nitroglycerine as an add-on procedure to spasm provocation testing. The purpose of this technique is to unmask co-existing microvascular spasm in patients with epicardial spasm and to assess the protective efficacy of nitroglycerine on a per-patient level to guide medical therapy.
Abstract
Coronary artery spasm (CAS) can be diagnosed in a large proportion of patients with recurrent angina with non-obstructive coronary artery disease (ANOCA) using acetylcholine (ACh) spasm provocation testing. CAS can further be divided into different subtypes (e.g., focal, diffuse epicardial, or microvascular spasm), each with different pathophysiological mechanisms that may require tailored drug treatment. The evidence behind the role of nitrates in the setting of each CAS subtype is lacking, and the effectivity can vary on a per-patient basis. In order to assess on a per-patient level whether nitroglycerine (NTG) can prevent inducible spasm, the vasospastic ACh dose can be readministered after NTG administration as part of the spasm provocation test. The preventive effect of NTG is assessed by evaluating improvements in the severity of induced symptoms, ischemic ECG changes, and by reassessing the site and mode of spasm on angiography. This technique can therefore be used to assess the nitrate responsiveness on a per-patient level and unmask co-existing microvascular spasm in patients with epicardial spasm that is prevented with NTG. The NTG rechallenge, therefore, allows to further guide targeted therapy for CAS and provide new insights into the pathophysiological mechanism behind vasospastic disorders.
Introduction
Coronary artery spasm (CAS) can be diagnosed in a large proportion of patients with recurrent angina and non-obstructive coronary artery disease (ANOCA) by means of spasm provocation testing with acetylcholine(ACh)1,2,3,4.The recent CorMicA trial demonstrated that identification and concurrent tailored treatment of CAS persistently improve the patient's quality of life and reduce the burden of angina5. Usually, once CAS is diagnosed, it is regarded as one distinct disease and treated with anti-vasospastic medication, such as calcium antagonists and nitrates1. Although, CAS can be divided into different subtypes with different pathophysiological mechanisms that may require tailored drug treatment6. CAS can occur on an epicardial level, either focally or diffusely, throughout the epicardial coronary arteries or on a microvascular level. The former is defined as vasospastic angina (VSA) and the latter as microvascular angina (MVA) due to microvascular spasm according to the COronary VAsomotor DISorders study group (COVADIS)3,4. Moreover, combinations of endotypes of CAS may co-exist that can further complicate tailored treatment. This is especially important as the occurrence of microvascular spasm may be masked during spasm provocation when a simultaneous epicardial spasm occurs.
As a consequence, treatment of these patients in clinical practice can be cumbersome and initiates a period of trial and error based treatment with various anti-vasospastic or anti-anginal medications. Nitrates, in particular, are often initiated as first-line treatment in short-acting form as rescue medication for acute anginal attacks or in the long-acting form as maintenance therapy. The evidence behind the role of nitrates in the setting of each CAS subtype is lacking, and the effectivity can vary on a per-patient basis. Especially in the case of microvascular or diffuse distal epicardial spasm, the effect of NTG is controversial7,8. Furthermore, the therapeutic efficacy of chronic NTG treatment has to be weighed against potential side effects, such as severe headaches and a worsened exercise capacity9,10,11.
Recently Seitz et al. demonstrated the clinical usefulness of the ACh rechallenge technique after NTG administration as an add-on procedure to the spasm provocation test12. This is performed after a positive spasm provocation test by readministering the vasospastic dose of ACh in a similar fashion as the vasospastic dose itself 3 min after NTG administration. To this end, the COVADIS criteria are revisited in order to evaluate the preventive effect of NTG, e.g., improvement in symptoms, ischemic ECG changes, and reassessment of the site and mode of spasm by angiography3,4. Moreover, prevention of epicardial spasm during rechallenge can unmask the co-existence of microvascular spasm.
The purpose of the rechallenge after NTG, therefore, is two-fold: (1) to assess the preventive effect of NTG on the re-occurrence of spasm on a per patients level in order to improve clinical outcomes and tailor treatment immediately after diagnosis that is made during spasm provocation and (2) to assess the co-existence of microvascular spasm in patients with epicardial coronary artery spasm10,13.
A previous publication by Ong et al. extensively covered the spasm provocation test14. In our institute, we use a variation of this protocol where the ACh dosages are administered in 60 s instead of 20 s. The purpose of this paper is to describe the NTG rechallenge as an add-on procedure to the ACh spasm provocation test. This technique can be performed with each type of protocol, as demonstrated by Seitz et al. since the results of the NTG rechallenge did not differ among the participating centers that used different protocols.
Protocol
Intracoronary ACh testing has been approved by the local ethics committee of the Academic Medical Centre, and the protocol follows the guidelines of Amsterdam UMC for human research.
1. Preparation of the ACh stock solution
- Mix the 20 mg of ACh with the 2 mL of solvent provided with the package (Table of Materials).
- Add 1 mL of the ACh solution to 499 mL of 0.9% NaCl to create the stock solution, corresponding to a 20 µg/mL dose.
- Fill a sterile dish with 50 mL of the stock solution, which will be used to prepare the syringes under sterile conditions.
2. Preparation of the syringes containing ACh for intracoronary injection
- Prepare five 10 mL syringes under sterile conditions for the intracoronary injection according to the Supplementary Material.
NOTE: It is advised to prepare each syringe sequentially after administration and not all at once to avoid confusion when administering the syringes
3. Diagnostic coronary angiography
- Anesthetize the puncture site of the right radial artery (usually 2 mL of lidocaine) or the area of the puncture site of the right femoral artery (usually 15 mL of lidocaine) when the femoral artery is the preferred route of catheterization.
NOTE: By puncturing the skin with the needle, the successfulness of the local anesthesia can be assessed. - Puncture the artery with a cannula using the Seldinger wire technique, then insert a wire through the cannula. After removing the cannula, insert a sheath (6F) over the wire. Perform coronary angiography under sterile conditions.
- Advance the wire into the ascending aorta through the sheath and subsequently position the diagnostic catheter (6Fr) above the aortic valve. Withdraw the wire and attach the contrast syringe to the catheter.
NOTE: The contrast agent used is iodixanol Injectable Contrast Medium. - Position the tip of the diagnostic catheter for the left coronary artery in the left main coronary artery. Confirm the position of the catheter by injecting 2 mL of the contrast agent.
- Exclude the presence of obstructive coronary artery disease by perming coronary angiography. Inject ~5-10 mL of the contrast agent to visualize the coronary arteries in different views.
NOTE: Most often, the following projections provide the best overview: LAO 40° and RAO 35° for the right coronary artery and LAO 45°/CRAN 25°, RAO 30°/CRAN 30° and RAO 20°/ CAUD 30° for the left coronary artery.
4. Preparations for Doppler flow assessment
NOTE: Here, ComboWire was used as the Doppler guidewire with the ComboMap system (Table of Materials)
- Connect the Doppler guidewire to the system and zero the pressure on the console and the aortic pressure on the hemodynamic system.
- Advance the Doppler guidewire through the guiding catheter into the ostium of the coronary artery with the pressure sensor of the Doppler guidewire placed just distally of the tip of the guiding catheter. Press normalization (Norm) on the system to equalize the two pressures.
- After normalization, reintroduce the wire via the microcatheter into the proximal or mid LAD and acquire a stable flow signal.
NOTE: The Doppler guidewire can alternatively be introduced more distally into the LAD or in the CX when acquiring an adequate signal in the proximal LAD is not feasible, such as in the case of tortuous anatomy or when another position is otherwise required, for instance, proximal from a myocardial bridge. - Document the position of the wire using fluoroscopy.
- Start ACh testing as described in section 5 after exclusion of any relevant epicardial stenosis (≥50%) by visual assessment.
5. Intracoronary administration of ACh
- Administer the first syringe containing 2 µg of acetylcholine as described in the Supplementary Material into the LCA through the guiding. Inject this in 60 s with continuous monitoring of the ECG, average peak velocity (APV), and the patient's symptoms (e.g., angina and/or dyspnea).
- Flush the ACh that remains in the guiding into the left coronary artery at a similar injection speed to the injection itself with contrast using the contrast syringe.
- Perform coronary angiography of the left coronary artery by injecting 10 mL of the contrast agent into the catheter in the same projection as done for the baseline coronary angiography. Bookmark and print a 12-lead ECG after every dose of ACh or when symptoms and/or ECG changes occur.
- If diagnostic criteria are not met, continue with the administration of the second syringe containing 20 µg of acetylcholine as described in the Supplementary Material using the same technique as described in steps 5.1.1-5.1.3. Pause for 3 min between every dose.
- If diagnostic criteria are not met after the second dose, continue with the administration of the third syringe containing 100 µg of acetylcholine as described in the Supplementary Material using the same technique as described in steps 5.1.1-5.1.3. Pause for 3 min between every dose.
NOTE: At dose 3 most patients will report some symptoms, ischemic ECG changes can occur, and some epicardial diameter reduction may occur. In some cases, the speed of the manual injection will have to be slowed down. For instance, at doses 3 and 4, bradycardia may occur, and the speed of the injection will have to be slowed down. An extended injection over 1-3 min may also be feasible. - If diagnostic criteria are not met after the third dose, continue with the administration of the fourth syringe containing 100 µg of acetylcholine as described in the Supplementary Material using the same technique as described in steps 5.1.1-5.1.3. Pause for 3 min between every dose.
NOTE: At dose 4 most patients will report some symptoms, ischemic ECG changes can occur, and some epicardial diameter reduction may occur. In some cases, the speed of the manual injection will have to be slowed down. For instance, at doses 3 and 4, bradycardia may occur, and the speed of the injection will have to be slowed down. An extended injection over 1-3 min may also be feasible.
- When diagnostic criteria according to COVADIS are not met after step 5.1.6 in the LCA, continue with RCA as the target vessel. Inject 80 µg of ACH (10 mL, syringe #RCA) in 60 s into the right coronary artery while continuously monitoring the ECG and the patient's symptoms.
NOTE: Clinical practice on testing of the RCA can vary among centers as routine testing of the RCA is advocated according to the JCS guidelines as multi-vessel epicardial coronary vasospasm is associated with worse prognosis2,15. In contrast, vasospasm provocation protocols from European and United states centers do not routinely test the RCA16. Acetylcholine can induce bradycardia and can be resolved by decreasing the speed of the injection to prevent prolonged bradycardia and/or asystole.- Flush the remaining ACh in the guiding into the left coronary artery at a similar injection speed to the injection itself with contrast using the contrast syringe. Perform coronary angiography of the RCA (perform angiography in the same projection as the baseline angiography).
- Inject 200 µg of intracoronary NTG into each target vessel after the test, or when severe symptoms (e.g., angina and/or dyspnoea)), ischemic ECG changes or epicardial spasm occurs. Continuously monitor the APV and patient's symptoms to monitor the effect of NTG.
- Perform coronary angiography of the target vessel after 1 min or when the APV has returned to baseline, and no symptoms are present to document reversion of spasm
6. Rechallenge
- In the case of a positive diagnosis, wait for 3 min after NTG administration until the patient is free of symptoms, ECG changes have resolved, and APV values have returned to baseline before starting the rechallenge.
- Inject 10 mL of the syringe containing the acetylcholine dose that previously induced vasospasm as described in the Supplementary Material into guiding. Inject this in 60 s with continuous monitoring of the ECG, APV and the patient's symptoms (e.g., angina and/or dyspnea).
- Flush the remaining ACh in the guiding into the left coronary artery at a similar injection speed to the injection itself with contrast using the contrast syringe. Image the target vessel in the same projection as done in the baseline angiography after injection of the 10 mL.
- Perform coronary angiography of the left coronary artery by injecting 10 mL of contrast into the catheter in the same projection as done for the baseline coronary angiography. Bookmark and print a 12-lead ECG after every dose of ACh or when symptoms and/or ECG changes occur.
- Inject 200 µg of intracoronary NTG into each target vessel after the test or when severe symptoms (i.e., angina and/or dyspnea), ischemic ECG shifts, or epicardial spasm occurs.
- Perform coronary angiography of the target vessel after 1 min or when the APV has returned to baseline, and no symptoms are present to document reversion of spasm
Representative Results
Interpretation of the ACh-test and rechallenge are based on criteria defined by the COVADIS study group4. A positive diagnosis for CAS is defined as (i) reproduction of the previously reported symptoms such as chest pain, shortness of breath, or other symptoms and (ii) the induction of ischemic ECG changes (ST-segment elevation or depression, or U-waves) in reaction to ACh. (Figure 2). It is therefore important to register a 12-lead-ECG continuously throughout the test and monitor for ischemic changes during ACh administration or when symptoms are reported. Finally, the distinction between epicardial and microvascular spasm is made by comparing coronary diameter reduction in reaction to the spasm provocating dose and NTG (Figure 2 and Figure 4). When epicardial vasoconstriction of >90% is evident on angiography, epicardial spasm can be diagnosed, and when this is <90% occurs, this is considered to be a microvascular spasm. Furthermore, the epicardial spasm may occur within the confines of one isolated coronary segment (focal spasm) or in ≥2 adjacent coronary segments (diffuse spasm)4.
When performing an ACh rechallenge, it can be useful to obtain a VAS score at the initial spasm provocation and rechallenge to quantify the improvement in symptoms. Improvement in provoked ischemic ECG changes, changes in APV, and severity of vasoconstriction on angiography can help as an objective measure of the preventive effect of NTG (Figure 2, Figure 3, and Figure 4).
Continuous assessment of Doppler flow provides valuable information during spasm provocation. Most importantly, it provides a safety feature as flow alterations most often occur prior to ECG changes (Figure 5). This improves operator awareness and patient safety.
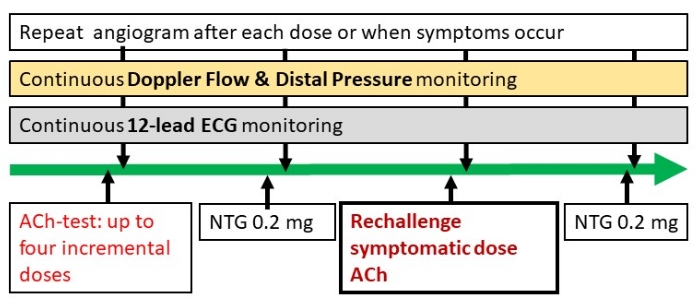
Figure 1: Flow chart protocol. The ACh rechallenge can be applied to any protocol version and can therefore be performed according to local protocol. After NTG administration, the spasm provocative dose can be readministered to perform the rechallenge. Please click here to view a larger version of this figure.
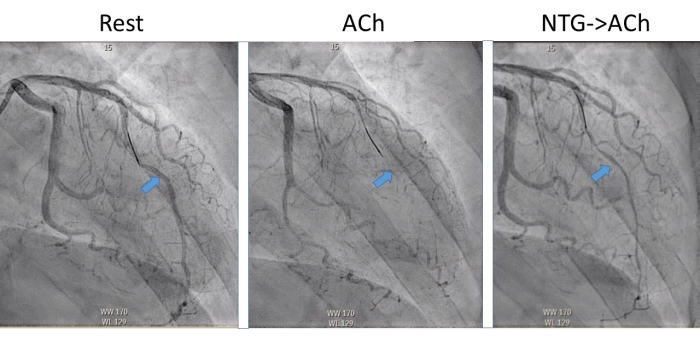
Figure 2: Coronary angiographical evaluation of the NTG rechallenge. The coronary angiography performed during rest shows that no significant lumen reduction is present in the LAD depicted by the arrow (Rest). At the fourth dose (ACh) <90%, epicardial lumen reduction occurs together with ECG changes and recognizable symptoms and therefore meets the diagnostic criteria for epicardial vasospasm. The last image depicts the preventive effect of NTG when the coronary artery is rechallenged after intracoronary administration of NTG. Now some vasoconstriction occurs although no >90% lumen reduction and the severity of anginal complaints are reduced. Please click here to view a larger version of this figure.
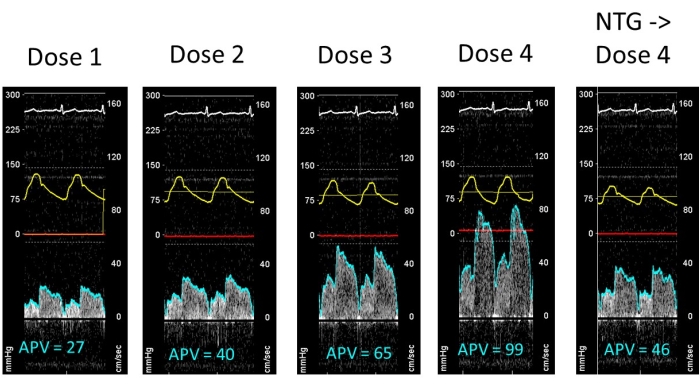
Figure 3: Example of APV changes during spasm provocation. At the fourth dose, epicardial vasoconstriction causes very high APV values, whereas the preventive effect of NTG causes lower APV values because the epicardial coronary artery does not constrict. Please click here to view a larger version of this figure.
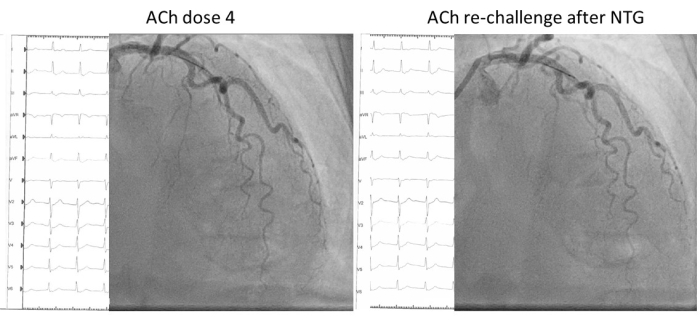
Figure 4: Example of a non-responder. In this example, the patient experiences recognizable anginal symptoms and ischemic ECG changes at the fourth acetylcholine dose without epicardial vasospasm consistent with the diagnosis of microvascular spasm according to COVADIS (left). After nitroglycerine administration, when symptoms have disappeared and ECG changes have normalized, the rechallenge with the same ACh dose commenced. Besides some epicardial vasodilation, the patient experienced symptoms and ischemic ECG changes of comparable severity compared to the spasm provocative dose. Please click here to view a larger version of this figure.
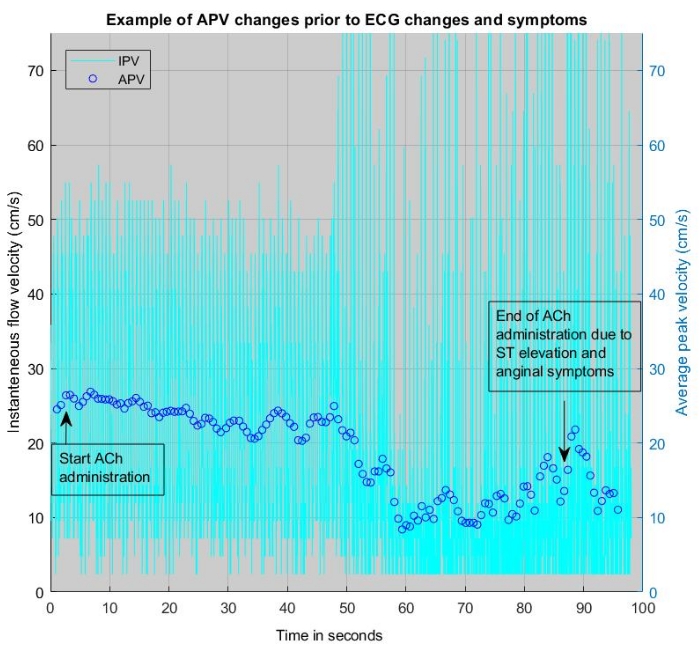
Figure 5: Example of APV changes during ACh infusion where a change in APV precedes symptoms or ECG changes and can be heard as a change in pitch of the acoustic signal. This example is from a 3 min infusion with acetylcholine. Please click here to view a larger version of this figure.
Supplementary Material: Instruction for preparing the syringes for ACh spasm provocation. Please click here to download this File.
Discussion
The usefulness of the ACh after NTG rechallenge has shown to be two-fold: (1) to unmask the co-existence of microvascular spasm in patients with epicardial spasm and (2) to assess the preventive efficacy of NTG on a per-patient level in order to guide medical therapy12. Regardless of the result of the spasm provocation test, intracoronary NTG is always routinely administered into the target vessel after the test or when severe symptoms, ischemic ECG changes, or epicardial spasm occur. Adding the ACh rechallenge after spasm provocation will only minimally extend the overall length of the total procedure. ACh rechallenge technique can be performed with each type of protocol as demonstrated by Seitz et al. as sub-analysis revealed that the results of the preventive effect of NTG during rechallenge did not differ among centers that used different protocols12.
An ACh rechallenge after NTG can provide valuable information regarding the preventive effect of NTG on a per-patient level to guide medical treatment in the outpatient clinic. For instance, when NTG prevents the occurrence of vasospasm during rechallenge and improvement in symptoms, nitrate-based medications are more likely to have a substantial clinical benefit to the patient. Whereas when NTG does not or only minimally prevent the re-occurrence of spasm, this will most likely also translate into a decreased clinical effectivity of nitrate-based therapies, and other vasospastic medication should be considered. Most patients with epicardial spasm are good NTG responders, whereas, in a large proportion of patients with microvascular spasm, the effect of NTG is limited in preventing spasm12.
Intracoronary NTG of 200 µg can be safely administered in most patients; however, should the blood pressure be low, a dose of 100 µg can be considered keeping in mind that a lower dose could influence the response. In some patients, additional NTG administration and time are needed to revert spasm and start the rechallenge. Be aware of a possible drop in blood pressure when administering additional dosages of NTG. Once the patient is free of symptoms, the ACh rechallenge can commence. Taking into account the short half-life of ACh, we advise waiting 3 min before readministering the vasospastic dose of ACh to allow any spasm to resolve. In addition, a return of APV to baseline levels may also be a sign that vasospasm has revolved.
When performing an Ach rechallenge, we suggest using a Doppler flow wire during the procedure because this allows to better assess all vascular domains. As discussed in another contribution, collection testing with adenosine using Doppler flow velocity measurements is advocated to further assess impaired vasodilation in ANOCA17. Furthermore, the endothelial function can be measured during ACh administration with a Doppler flow wire. The use of a Doppler flow wire also provides valuable information during spasm provocation. Most importantly, it provides a safety feature as flow alterations most often occur prior to ECG changes (Figure 5), improving operator awareness and patient safety. It is important to note that when using a microcatheter to stabilize the flow or combowire in spasm provocation, only 6 Fr catheters should be used to allow for rapid backflow and NTG administration.
In the future, the ACh rechallenge technique could be applied to vasoactive medication other than NTG that can also be administered intracoronary with immediate effect. The advantage of this is that the rechallenge can be performed in one setting, whereas with medication without immediate effect this would require two procedures. The main limitation of this technique is that it is not performed blindly or in a cross-over manner with saline.
Nevertheless, rechallenging the coronary circulation with the vasospastic dose of ACh allows to further guide targeted therapy for CAS and provide new insights into the pathophysiological mechanism behind vasospastic disorders on a per-patient level.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
None
Materials
| Cannula (various manufacturers) | BBraun | 4206096 | |
| ComboMap system | Volcano-Philips | Model No. 6800 (Powers Up) | |
| ComboWire XT Guide Wire | Volcano-Philips | 9515 | Doppler guidewire |
| Diagnostic catheter | Boston scientific | 34356-661 | H749343566610/ MODEL-6F MACH 1 JL3.5 |
| Diagnostic catheter | Boston scientific | 34356-686 | H749343566860/MODEL – 6F MACH 1 JR4 |
| FINECROSS MG Coronary Micro-Guide Catheter | Terumo | NC-F863A | |
| Intracoronary NTG | hameln pharma gmbh | RVG 119982 | |
| Lidocaine HCL | Fresenius Kabi | RVG 51673 | |
| Miochol-E Acetylcholine chloride | Bausch & Lomb | NDC 240208-539-20 | |
| Sheath Radialis | Teleflex | AA15611S | |
| Syringe- 10 mL | BBraun | 4606108V | |
| Visipaque | GE Healthcare | RVG 17665 | Iodixanol injectable contrast medium |
Referenzen
- Knuuti, J., et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. European Heart Journal. 41 (3), 407-477 (2020).
- JCS Joint Working Group. Guidelines for diagnosis and treatment of patients with vasospastic angina (Coronary Spastic Angina) (JCS 2013). Circulation Journal: Official Journal of the Japanese Circulation Society. 78 (11), 2779-2801 (2014).
- Ong, P., et al. International standardization of diagnostic criteria for microvascular angina. International Journal of Cardiology. 250, 16-20 (2018).
- Beltrame, J. F., et al. International standardization of diagnostic criteria for vasospastic angina. European Heart Journal. 38 (33), 2565-2568 (2017).
- Ford, T. J., et al. 1-Year Outcomes of angina management guided by invasive coronary function testing (CorMicA). JACC: Cardiovascular Interventions. 13 (1), 33-45 (2020).
- Bairey Merz, C. N., Pepine, C. J., Walsh, M. N., Fleg, J. L. Ischemia and no obstructive coronary artery disease (INOCA): Developing evidence-based therapies and research agenda for the next decade. Circulation. 135 (11), 1075-1092 (2017).
- Sun, H., Fukumoto, Y., Ito, A., Shimokawa, H., Sunagawa, K. Coronary microvascular dysfunction in patients with microvascular angina: analysis by TIMI frame count. Journal of Cardiovascular Pharmacology. 46 (5), 622-626 (2005).
- Kiyooka, T., Kobayashi, Y., Ikari, Y. A case of vasospastic angina in which the ergonovine provocation test with intracoronary isosorbide dinitrate and nicorandil was effective in the diagnosis of microvascular spasm. Cardiovascular Intervention and Therapeutics. 29 (4), 344-349 (2014).
- Takahashi, J., et al. Prognostic impact of chronic nitrate therapy in patients with vasospastic angina: multicentre registry study of the Japanese coronary spasm association. European Heart Journal. 36 (4), 228-237 (2015).
- Ferrari, R., et al. Expert consensus document: A ‘diamond’ approach to personalized treatment of angina. Nature Reviews Cardiology. 15 (2), 120-132 (2018).
- Redfield, M. M., et al. Isosorbide mononitrate in heart failure with preserved ejection fraction. The New England Journal of Medicine. 373 (24), 2314-2324 (2015).
- Seitz, A., et al. Acetylcholine rechallenge: A first step towards tailored treatment in patients with coronary artery spasm. Journal of the American College of Cardiology: Cardiovascular Interventions. 15 (1), 65-75 (2022).
- Crea, F., Lanza, G. A. Treatment of microvascular angina: The need for precision medicine. European Heart Journal. 37 (19), 1514-1516 (2016).
- Ong, P., Athanasiadis, A., Sechtem, U. Intracoronary acetylcholine provocation testing for assessment of coronary vasomotor disorders. Journal of Visualized Experiments: JoVE. (114), e54295 (2016).
- Han, S. H., et al. Impact of multi-vessel vasospastic angina on cardiovascular outcome. Atherosclerosis. 281, 107-113 (2019).
- Feenstra, R. G. T., et al. Principles and pitfalls in coronary vasomotor function testing. EuroIntervention: Journal of EuroPCR in Collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. , (2021).
- Seitz, A., Beck, S., Pereyra, V. M., Bekeredjian, R., Sechtem, U., Ong, P. Testing acetylcholine followed by adenosine for invasive diagnosis of coronary vasomotor disorders. Journal of Visualized Experiments: JoVE. (168), e62134 (2021).

