Measurement of Endothelium-Dependent Vasorelaxation in the Mouse Thoracic Aorta Using Tensometric Small Volume Chamber Myography
Summary
The present protocol describes the concepts and technical application of the tensometric myograph technique using a multi-chamber myograph system in the experimental ex vivo assessment of mouse aortic endothelial function.
Abstract
Small volume chamber tensometric myography is a commonly used technique to evaluate the vascular contractility of small and large blood vessels in laboratory animals and small arteries isolated from human tissue. The technique allows researchers to maintain isolated blood vessels in a tightly controlled and standardized (near-physiological) setting, with the option of adjusting to various environmental factors, while challenging the isolated vessels with different pharmacological agents that can induce vasoconstriction or vasodilation. The myograph chamber also provides a platform to measure vascular reactivity in response to various hormones, inhibitors, and agonists that may impact the function of smooth muscle and endothelial layers separately or simultaneously. The blood vessel wall is a complex structure consisting of three different layers: the intima (endothelial layer), media (smooth muscle and elastin fibers), and adventitia (collagen and other connective tissue). To gain a clear understanding of the functional properties of each layer, it is critical to have access to an experimental platform and system that would allow for a combinational approach to study all three layers simultaneously. Such an approach demands access to a semi-physiological condition that would mimic the in vivo environment in an ex vivo setting. Small volume chamber tensometric myography has provided an ideal environment to evaluate the impact of environmental cues, experimental variables, or pharmacological agonists and antagonists on vascular properties. For many years, scientists have used the tensometric myograph technique to measure endothelial function and smooth muscle contractility in response to different agents. In this report, a small volume chamber tensometric myograph system is used to measure endothelial function in the isolated mouse aorta. This report focuses on how small volume chamber tensometric myography can be used to evaluate the functional integrity of the endothelium in small segments of a large artery such as the thoracic aorta.
Introduction
For the last few decades, the small chamber myography system has been used to measure the reactivity of different layers of blood vessel walls in response to various pharmacological agents and neurotransmitters in an ex vivo, real-time setting. Vascular reactivity is a major component of a healthy functional blood vessel and is critical for the regulation of blood flow and perfusion in peripheral and cerebral vasculature1. Within the blood vessel wall, the interaction between endothelial and smooth muscle layers is a major determinant of vascular tone, which is also constantly impacted by structural changes in the connective tissue layer surrounding the blood vessel wall (adventitia).
The endothelial layer controls vasomotion by releasing a few vasodilatory factors, including nitric oxide (NO), prostacyclin (PGI2), and endothelium-derived hyperpolarizing factor (EDHF), or by producing vasoconstrictive agents such as endothelin-1 (ET-1) and thromboxane (TXA2)2,3,4. Among these factors, NO has been extensively studied, and its important regulatory roles in other critical cellular functions such as inflammation, migration, survival, and proliferation have been highly cited in scientific literature2,5.
In the field of vascular biology, chamber myography has provided vascular physiologists and pharmacologists with a valuable and reliable tool to measure endothelial function in a tightly controlled semi-physiologic system1. Currently, there are two different myograph systems available to scientists: wire (or pin) tensometric (isometric) myography and pressure myography. In a wire myography system, the blood vessel is stretched between two wires or pins, allowing for the isometric measurement of force or tension development in the wall of the blood vessel, while pressure myography is a preferable platform for measurements of vascular reactivity in small resistance arteries, where changes in blood pressure are considered the main stimulus for changes in vascular tone and vasomotion. There is a general agreement that, for small resistance arteries such as mesenteric and cerebral arteries, pressure myography creates a condition that is closer to the physiological conditions in the human body. The small chamber myograph can be utilized for vessels with very small diameters (200-500 µm) to much larger vessels such as the aorta.
While the wire myograph is a powerful system for recording blood vessel tension under isometric conditions, the pressure myograph is a more appropriate system for measuring changes in vessel diameter in response to changes in isobaric conditions. The diameter changes in the vessel in response to changes in pressure or flow are much larger in a small muscular artery (arteriole) compared to large elastic arteries such as the aorta. For these reasons, the pressure myograph is considered a better tool for small blood vessels with substantial vasoreactivity1. One of the other practical strengths of multi-chamber small volume chamber tensometric myography is that one can discern the contribution of different mechanisms to vascular reactivity by studying multiple (up to four) segments of the same artery and from the same animal to reduce variability and produce robust and conclusive data. It is also relatively easy to set up and maintain technically. Vessels of almost any size can be studied with a wire myograph. It is a more cost-effective solution for assessing vascular function and is a good alternative to pressure myography in experiments where the length of the dissected vessel is too short for the pressure myograph protocol.
This report provides a detailed protocol for the assessment of endothelial function in the isolated mouse thoracic aortic ring using mounting pins in the small volume chamber tensometric myography technique using the DMT-620 multi-chamber myograph system (DMT-USA). This protocol utilizes a 6-month-old male C57BL6 mouse with an average weight between 25-35 g. Fortunately, this protocol can be applied to various animal types and weights, considering the broad range of vessel types and diameters that this protocol can be used for.
Protocol
All surgical procedures and animal care were approved by the Institutional Animal Care and Use and Care Committee (IACUC) of Midwestern University (IACUC# AZ-3006, AZ-2936).
1. Buffer preparation
NOTE: Although the HEPES physiological salt solution (HEPES-PSS) buffer is stable at 4 °C for 7 days, it is recommended that all buffers are freshly made on the day of each experiment. All other reagents and agonists must be prepared freshly for each experiment. The HEPES-PSS buffer used in this protocol is a well-established buffer for ex vivo vascular studies that has been shown to be cytoprotective for more than 12 h while preserving the vessel's vasodilatory responses-the main focus of this experimental protocol6,7.
- Prepare HEPES-PSS solution (pH 7.4) as follows: Mix 10 mmol/L HEPES, 6 mmol/L glucose, 1.5 mmol/L CaCl2, 130 mmol/L NaCl, 4 mmol/L KCl, 4 mmol/L NaHCO3, 1.2 mmol/L MgSO4, 1.2 mmol/L KH2PO4, and 0.03 mmol/L EDTA.
- Prepare HEPES-PSS high K+ buffer. This is identical to the HEPES-PSS solution, except that it contains 5 mmol/L HEPES, 65 mmol/L NaCl, 10 mmol/L glucose, 1 mmol/L MgCl2, 80 mmol/L KCl and does not contain MgSO4 and EDTA.
2. Myograph unit preparation
- Turn on and set the water bath to 37 °C. Ensure an appropriate water level.
- Place two beakers labeled appropriately into the water bath, one with 600 mL of HEPES-PSS solution and one with 150 mL of high K+ solution.
- Aerate a beaker of 300 mL of HEPES-PSS solution with carbogen gas (5% CO2 and 95% O2) for at least 10 min.
- Add 30 mL of the aerated HEPES-PSS solution to a 50 mL centrifuge tube, label appropriately (thoracic cage, mouse identification number), and place it on ice. Keep the remaining aerated HEPES-PSS solution on ice to be used during the aortic dissection process.
- Turn on the four-chamber myograph unit; add 6 mL of HEPES-PSS solution to each myograph chamber, and set the heat to 37 °C. Allow the solution in each chamber to be aerated (with carbogen mix) for 30 min and check to ensure that the chambers have reached the desired 37 °C (use this waiting period to dissect the mouse aorta).
- Turn on the myograph data acquisition hardware and computer.
3. Mouse aorta isolation
- Anesthetize the experimental mouse with 5% isoflurane inhalation and euthanize by cervical dislocation (a 6-month-old male C57BL/6J mouse is used for this demonstration).
- Lay the mouse on a surgical board in the supine position and secure the appendages to the board using surgical tape.
- Spray the abdominal region with 70% alcohol so that the fur is completely wet and any loose/dry hairs do not enter the incision. Gently wipe away excess solution.
- Use forceps to locate and isolate the abdominal skin just inferior to the xiphoid process of the sternum.
- Create tension by lifting the skin straight up. Make a blunt cut using scissors, and remove the superficial skin, exposing the thoracic cavity.
- Lift the xiphoid process and make lateral incisions just inferior to the process along the subcostal margins.
- Extend the lateral incisions cranially to remove the anterior portion of the ribcage.
- Remove the thoracic cage by making a blunt cut through the spine, below the diaphragm, and in the neck.
NOTE: Researchers may also remove the aorta directly from the animal, but care should be taken to protect the vessel and flush the clotted blood from the aorta. This protocol uses the example of removing the thoracic cage and carefully cleaning and dissecting the aorta. With practice, this method can be performed quickly and efficiently and is beneficial for preserving the extra tissue for other studies such as immunohistochemistry, histology, and western blotting. - Transfer the thoracic cage to a clear silicone elastomer-coated Petri dish filled with ice-cold aerated HEPES-PSS buffer and pin it on both sides.
NOTE: Use a silicone elastomer kit (Table of Materials). Mix base and curing agent (10:1 by weight), then pour into a glass Petri dish, and allow to cure for 24 h at 25 °C. - Place the Petri dish under a stereo zoom microscope, gently cut and remove the heart and attached aorta from the rib cage, and transfer to a clean, clear silicone elastomer-coated dish (Figure 1) .
- Then, gently remove fat and connective tissue and any clotted blood from the aorta. Using sharp, small scissors, dissect and isolate the whole aorta starting from the arch area to the bottom part of the descending aorta. Remember that the aortic arch is not suitable for myography experiments but can be used for histological studies.
- Use cold aerated HEPES-PSS solution throughout the dissection. Change the solution every 10 min or when visibility is compromised, whatever comes first. It is recommended that each aortic ring is mounted onto the myograph chamber within 20 min post dissection (max 60 min).
4. Mounting of the aortic segments onto the myograph chambers
- Cut the dissected aorta into four segments of 2 mm each using sharp small surgical scissors.Use the mini-ruler within the dish as a reference.
- Place the myograph chamber under the stereo zoom microscope to easily visualize the pins and set the micrometers such that the pins are nearly touching (Figure 2).
- Make sure both tissue-holding pins are properly aligned.
- Pay attention and avoid pinching the aorta while mounting the segments to prevent damage to the endothelium.
- In chambers already containing 5 mL of warmed and aerated HEPES-PSS buffer, carefully slide each of the 2 mm aortic segments onto the two mounting pins using forceps. Be very gentle while sliding the aortic segments over the pins. The endothelial layer is very fragile and comes off very easily from the luminal side.
- Slowly move the pins apart by rotating the micrometer counter-clockwise so the aortic segment does not slide off the pins when placing the chamber back in the myograph unit (Figure 3).
- Use the previously calibrated eyepiece graticule of the dissecting microscope to measure the true and accurate length of the mounted aortic ring by positioning the beginning of the ruler at one end of the segment (marked as tissue end α1) and making a note of the measurement at the other end of the aortic segment in ocular divisions (marked as tissue end α2)8. These values will be used during the normalization steps in section 5.
- Return each myograph chamber to the unit, and start aerating the chambers at 37 °C for 30 min.
NOTE: The four segments of the aorta can be used as replicates for the same treatment, or each segment of the aorta can be used simultaneously for different experiments.
5. Normalization
NOTE: A normalization procedure is necessary to ensure that the experimental conditions are properly standardized and the collected data is reliable and reproducible. The "IC1/IC100", or "Normalization Factor", is defined as the ratio of the internal circumference of the artery at which it is possible to record the maximum response to a vasoconstrictor (e.g., 60 mM KCl) divided by the internal circumference at which a transmural wall pressure of 100 mm Hg (i.e., IC100) is recorded. Therefore, by multiplying the IC100 by this ratio, we can determine the internal circumference of the artery at which an optimal response (i.e., IC1) can be established.
- Set the micrometer such that the pins are nearly touching.
- Set forces to zero for all myograph chambers and let it equilibrate for another 1-2 min.
- Make a note of the first diameter reading from the micrometer. This is the position where the gap between two pins is considered zero (needed for step 5.7.).
- Open the data acquisition software Normalization Settings under the DMT drop-down menu; a new screen will open with the following settings and default values:
Eyepiece calibration (mm/div): 0.36
Target pressure (kPa): 13.3
IC1/IC100: 0.9
Online averaging time (s): 2
Delay time (s): 60
Play sound on delay completion (check box)
Sounds (with drop-down selection menu or Browse function) - Depending on the number of segments, choose the correct number of channels available on-screen and start the chart recording.
- Use the drop-down menu to select the channel of interest; the normalization screen will appear.
- Enter the constant values into the windows as follows: Tissue end-points α1; Tissue end-points α2; Pin diameter: 40 µm; Micrometer reading value from the analog micrometer scale.
- In order to record the first point (the initial value of X or X0), click on the Add Point button. Following a delay of 60 s, the values for the force and the effective pressure (ERTP) corresponding to this micrometer will be displayed, and the box for the micrometer reading becomes active and available.
- Start stretching the vessel by turning the micrometer in a counter-clockwise direction. Use the micrometer reading box to enter the value and click the Add Point button (there will be a delay time of 60 s again).
- Keep stretching the vessel and continue to add micrometer values until the value of Micrometer X1 is seen, which is the calculated micrometer setting used to stretch the vessel to its IC1.
- Set the micrometer to the X1 value.
NOTE: The normalized (optimal) tension for a 6-month-old C57BL6 mouse is 6 mN. - After normalization, let the tissue rest and equilibrate for 30 min. There is no need to change the HEPES-PSS solution in the chambers at this point.
NOTE: Each chamber is capable of holding 8 mL of solution. This protocol is written with the use of 5 mL, which allows for full submergence of the vessel and mounting pins. - Use the rest and equilibration time to prepare the following.
- Prepare the working stock of acetylcholine serial dilutions in distilled water (RO water) from lowest to highest concentrations as follows: 50 nM, 100 nM, 500 nM, 1 µM, 5 µM, 10 µM, 50 µM, 100 µM, 500 µM, and 1 mM. Keep all the tubes on ice for the duration of the experiment.
- Prepare a 100 mM working solution of N-nitro-L-arginine methyl ester (L-NAME) in double-distilled water and keep the solution on ice for the duration of the experiment.
- Prepare the sub-maximum dose of phenylephrine (10 µM) determined in a separate set of experiments.
NOTE: The equilibration period is necessary for the blood vessel segment to adjust to the new environment in the myograph chamber, reset ion gradients, and achieve a stable level of passive tension prior to being subjected to different pharmacological and mechanical challenges.
6. Measurement of endothelium-dependent vasorelaxation in aortic rings
- At the end of the 30-min equilibration period, drain the chambers and add 5 mL of fresh, warm, aerated HEPES-PSS solution to each chamber. Drain one chamber at a time to minimize tissue exposure to air. Draining of the chambers is performed using a vacuum pump set to 60 cmHg and the myograph valves set to a 6 s delay to ensure the removal of the 5 mL of solution.
- If needed, readjust the force measurements to read the optimal tension of 6 mN. It is very critical to readjust the force to optimal tension during every incubation period, as well as prior to each new set of experiments. Rest the tissue for another 15-20 min.
- Open the data acquisition software, change the tracking numbers from 50:1 to 500:1, and press Start. At this stage, the registered force for each chamber can be seen in software.
- Immediately prior to the start of a new experiment, be sure to add the appropriate label on the data acquisition software.
- Drain the chambers one more time before adding 5 mL of high K+ solution to each chamber. This is suitable for testing the viability of aortic tissue and the integrity of the smooth muscle contractile response to membrane depolarization before using the tissue for any other experimental protocol. This helps determine whether the blood vessel segment is usable for further experimentation.
- Drain the chambers again as soon as the contractile response to the high K+ solution reaches a plateau for force generation (Figure 4).
NOTE: Do not leave the high K+ solution in the chambers for more than 3 min, as it would damage the tissue. - Wash the tissue with HEPES-PSS solution 3x. Add 5 mL of high K+ solution to each chamber. Drain the chambers as soon as the contractile response to high K+ solution reaches a plateau for force generation and wash the tissue 3x with HEPES-PSS before allowing the tissue to rest for another 15 min.
NOTE: In some laboratories, the blood segments are subjected to a high K+ solution three consecutive times to ensure the variability of the blood vessel before initiating the myograph experiments. In this protocol, the newly isolated aortic segments are subjected to a high K+ solution twice before using the tissue for further experiments. It is important to keep this consistent across all experiments. - Use the average of the two registered peaks of force development (contraction force) in response to a high K+ solution to normalize the registered values in response to other vasoconstrictor and vasodilator agents used in the experiment.
- Rest the tissue for 15-20 min.
- Pre-contract the aortic segments with the vasoconstrictor agent phenylephrine (PE) at an already established sub-maximum dose (10 µM final concentration in the chamber).
NOTE: The sub-maximum dose of phenylephrine (10 µM) was determined in a separate set of experiments, where aortic segments were subjected to increasing concentrations of phenylephrine solution at final concentrations of 1 nM, 5 nM, 10 nM, 50 nM, 100 nM, 500 nM, 1 µM, 5 µM, 10 µM, and 50 µM. As a result, it was determined that a final concentration of 10 µM of phenylephrine could generate the sub-maximum contraction force (90% of the maximum force) in 2 mm aortic segments, which is right before the contraction force reaches a plateau. The reason for using the sub-maximum concentration of phenylephrine is to avoid issues related to aortic tissue saturation or desensitization to the vasoconstricting agonist. - Allow the PE-induced contraction curve to reach a plateau for tension (force) development (Figure 5).
- At a plateau of tension development to phenylephrine, perform the acetylcholine dose-response curve by adding 5 µL of increasing doses of acetylcholine working stock solutions in 3 min intervals in order to establish final concentrations of acetylcholine in the myograph chamber as follows: 50 pM, 100 pM, 500 pM, 1 nM, 5 nM, 10 nM, 50 nM, 100 nM, 500 nM, and 1 µM (Figure 6).
NOTE: After adding each dose of acetylcholine, wait for at least 3 min (for the tension to reach a plateau) before adding the next dose. - At each step, pipette the acetylcholine stock solution into the chamber very slowly and far from the aortic rings to avoid any tissue disturbance.
- After completing the dose-response experiment, drain the chambers and wash the aortic segments with warm and aerated HEPES-PSS solution 3x to remove any remaining residue of the drug.
- Rest the tissue for 30 min before starting the next experiment steps. The viability of the aorta is checked before each experimental step using HEPES-PSS high K+ solution. If all the preparation and experimental steps are followed correctly, the aortic tissue viability will remain the same for the duration of the entire experiment (4-6 h).
7. Effects of general inhibitors of NO production on endothelium-mediated vasorelaxation
- Use the same aortic tissue for this part of the experiment after carefully washing and resting the segments for at least 30 min.
NOTE: Use the 30 min resting time to prepare a 100 mM working solution of N-nitro-L-arginine methyl ester (L-NAME) in double-distilled water and keep the solution on ice for the duration of the experiment. - After resting the aortic segments for 30 min, drain the chambers and add fresh, warm high K+(60 mM KCl) solution to each chamber.
NOTE: It is important that, for each part of the experiment, the aortic tissue is challenged with a high K+ solution at least once. The recorded contraction peak will be used for normalizing the data collected during that part of the experiment. - Drain the chambers again as soon as the contraction response to high K+ solution reaches a plateau and wash the tissue with HEPES-PSS solution 3x.
- In order to assess the contribution of NO production to the observed acetylcholine-induced vasorelaxation (see section 6.), at this stage, preincubate the aortic segments with a general inhibitor of NO production (L-NAME) by adding 10 µL of the prepared 100 mM working stock solution of L-NAME to each myograph chamber (containing 5mL buffer solution) in order to achieve a final concentration of 200 µM in each myograph chamber.
NOTE: L-NAME is a non-selective and potent inhibitor of all isoforms of NOS (an enzyme responsible for NO production), and, therefore, it is considered an effective tool to block the production of NO in the blood vessel wall9. - Allow the aortic segments to rest during the pre-incubation time with L-NAME (30 min).
- Do not wash at this point.
- Without removing the L-NAME, add the sub-maximum dose of phenylephrine (10 µM final concentration) to the chamber to induce aortic contraction.
NOTE: Due to the inhibitory action of L-NAME on endogenous NO production, the newly registered peak for phenylephrine-induced aortic contraction will be much higher than the initial peak that was recorded prior to incubating the tissue with L-NAME. This is expected due to L-NAME's effect on basal NO production in the aortic wall, which leads to higher force generation (due to higher contractile force generation by the smooth muscle). - Wait until the phenylephrine-induced contraction curve reaches a plateau for tension (contractile force) development.
- At this point, add acetylcholine to the myograph chamber to achieve a final concentration of 500 nM (this is the sub-maximum concentration of acetylcholine that was established during the experiments described in section 5. of the protocol).
- Wait for a few minutes to register any possible changes in force development until the formed plateau is stable (Figure 7).
NOTE: Due to the inhibitory action of L-NAME on the endothelial layer's ability to produce NO in response to acetylcholine, it is not expected to see any changes in registered force generation in response to phenylephrine, as the aortic endothelial NOS (eNOS) is not able to generate NO in response to acetylcholine application. - Drain the chambers and wash the tissue with warm, aerated HEPES-PSS solution 3x.
8. Contribution of the endothelial layer to aortic vasorelaxation
- In order to underscore the role of the endothelial layer in aortic vasorelaxation, test acetylcholine-induced vasorelaxation in endothelium-denuded aortic segments, in which the endothelial layers are removed.
- Position the myograph chamber under the microscope and gently pass a small wire (the same wire that is used for wire myography can be used here) through the lumen of the aorta. Gently move the wire through the lumen (gentle circular movement) for a short period of time. This is enough to remove the endothelium or intima.
- Cut the aorta into 2 mm aortic rings and mount those rings onto the myograph chamber as described before.
- Set the optimal tension at 6 mN and allow the tissue to rest for 30 min in aerated, warm HEPES-PSS buffer
- At the end of the 30 min equilibration period, drain the chambers and add 5 mL of fresh, warm, aerated HEPES-PSS solution to each chamber.
- Zero the force for all myograph chambers by turning the micrometer in a counter-clockwise direction.
- Slowly rotate the micrometer counter-clockwise to increase the distance between the pins until the registered force reaches the desired optimal tension for the mouse aorta (6 mN for C57BL/6J mouse aorta). Rest the tissue for another 15-20 min at the optimal tension (6 mN).
- Drain the chambers 1x more before adding 5 mL of high K+ solution to each chamber.
- Drain the chambers again as soon as the contraction response to high K+ solution reaches a plateau and wash the tissue with HEPES-PSS solution 3x. Rest the tissue for 15-20 min
- Pre-contract the aortic segments with the vasoconstrictor agent phenylephrine (10 µM final concentration in the chamber).
NOTE: Due to the removal of the endothelium, the basal NO production in the aortic wall is significantly diminished; therefore, the peak for phenylephrine-induced contraction (the peak of force generation) is expected to be higher in aortic tissue lacking the endothelial layer. - When the phenylephrine-induced force reaches a plateau, add the sub-maximum concentration of acetylcholine to the myograph chamber to achieve a final concentration of 500 nM.
NOTE: If the removal of the endothelial layer is done properly, there will not be any changes in the registered phenylephrine-induced force, as the acetylcholine-induced NO production by the endothelium will have been diminished due to the removal of the endothelial layer. The recorded trace will be very similar to the one observed in the presence of L-NAME (section 7.). - Wait for 3 min to ensure that the registered plateau for phenylephrine-induced force development is not changing before ending the experiment (Figure 8).
NOTE: If the application of acetylcholine causes any drop in phenylephrine-induced contraction while in the plateau, this is an indication that the endothelial layer has not been completely removed.
Representative Results
The tensometric small chamber myography protocol explained here is the standard method for measuring vascular reactivity in small and large arteries and allows for simultaneous measurements of vascular reactivity in up to four blood vessel segments from the same experimental small laboratory animal. In this report, we specifically use the system to measure endothelial function in the isolated mouse aorta (Figure 1). In this protocol, isolated aortic segments are mounted onto a small organ chamber (Figure 2) between two small stainless steel pins (Figure 3). The myograph chamber can hold up to 8 mL of buffer solution and provide a semi-physiologic environment for the isolated vessels for the duration of the experiments. It is very important that, prior to each experiment, the viability of each isolated segment is tested and verified. The standard protocol to establish the integrity and viability of each isolated vessel segment is to challenge the tissue with a high concentration of potassium chloride to induce smooth muscle membrane depolarization. In the scenario that the isolated vessel is healthy and responsive, we would be able to record the contractile force generation on the display (Figure 4). The peak of the recorded force is later used to normalize the force generation for the same segment in response to the agonists used during the protocol (e.g., phenylephrine). In order to measure endothelium-mediated vasorelaxation, it is necessary to pre-contract the aortic tissue with the sub-maximum concentration (10 µM) of phenylephrine, which causes smooth muscle-mediated contraction and force generation (Figure 5). When the phenylephrine-induced contraction reaches a plateau (Figure 6), increasing doses of acetylcholine are applied in multiple steps to achieve the maximum vasorelaxation in the isolated segment (Figure 6). The level of vessel relaxation is an indirect measurement of endothelium-mediated nitric oxide production. To further confirm that acetylcholine-induced vasorelaxation in aortic rings is due to the production of nitric oxide, aortic segments are pre-treated with a general inhibitor of nitric oxide production (200 µM of L-NAME) for 30 min prior to phenylephrine application. As shown in Figure 7, L-NAME is able to completely block acetylcholine-induced vasorelaxation in the pre-contracted aorta, highlighting the fact that acetylcholine induces aortic vasorelaxation through increasing nitric oxide production. On the other hand, the removal of the endothelial layer from the aortic segments also blocks acetylcholine-induced vasorelaxation, underscoring the role that the endothelium plays in blood vessel relaxation (Figure 8).
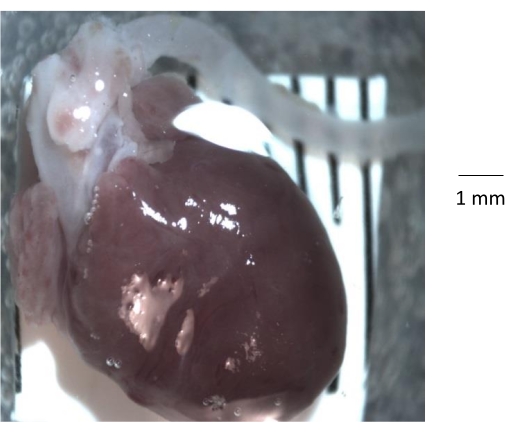
Figure 1: Gross anatomic view of the heart, aortic root, and descending aorta isolated from a 6-month-old control mouse. After removing the rib cage from the mouse, the heart and aorta are isolated from the rib cage and transferred to a clean silicone elastomer-coated Petri dish. Prior to isolating the aorta, it is important to remove all the fat and connective tissue and any blood clot from the lumen of the aorta. Please click here to view a larger version of this figure.

Figure 2: A representative image of a chamber of the myograph unit showing the 200 µm mounting pins. As shown, the two pins inside the myograph chamber are barely touching. Prior to using the chamber, it is critical to make sure that the pins are properly aligned. Please click here to view a larger version of this figure.

Figure 3: Anchoring the aortic segments onto the myograph chamber. A 2 mm mouse aortic segment isolated from a 6-month-old C57BL/6 mouse is held by two pins inside a myograph chamber. This is achieved by gently sliding the aorta onto the two mounting pins using forceps. The red dotted box shows the zoomed-in image of the 2 mm aortic segment that is mounted between two pins inside the myograph chamber. Please click here to view a larger version of this figure.
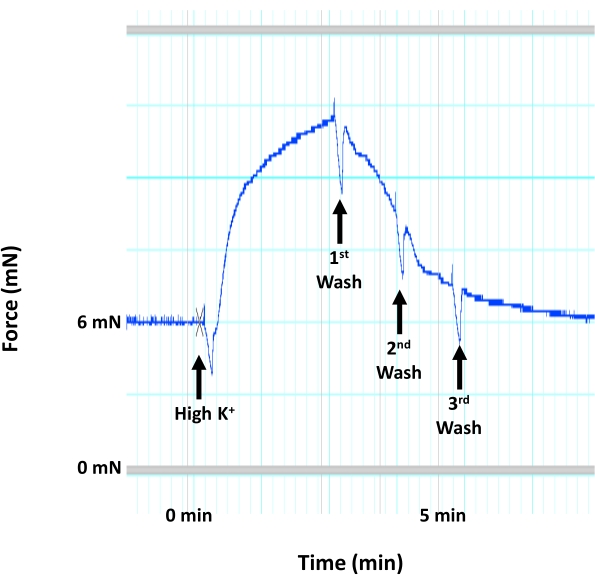
Figure 4: Aortic contraction due to smooth muscle membrane depolarization. Representative image showing the trace for mouse aortic contraction (force generation) in response to a high concentration of K+ (60 mM KCl) that would induce smooth muscle membrane depolarization and contraction within the medial layer of the aorta. The application of high K+ solution is followed immediately by three consecutive washes using warm, aerated HEPES-PSS solution. Please click here to view a larger version of this figure.
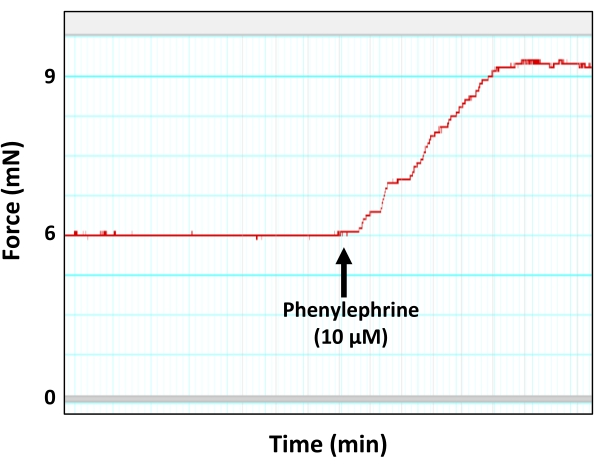
Figure 5: Aortic contraction in response to the vasoconstricting agent phenylephrine. Representative myograph trace showing force generation (contraction) by the aortic ring in response to the sub-maximum concentration of phenylephrine (10 µM). As shown, the peak of phenylephrine-induced contraction eventually reaches a plateau. Please click here to view a larger version of this figure.
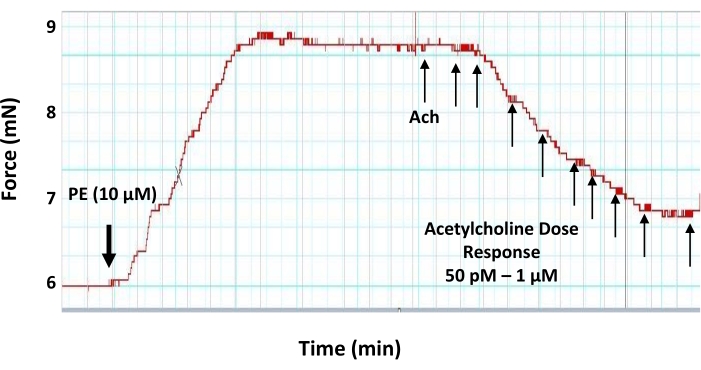
Figure 6: Dose-response effects of acetylcholine on the pre-contracted aortic ring. Representative myograph trace showing the dose-response (50 pM-1 μM) vasodilatory effect of the vasodilator neurotransmitter acetylcholine on a 2 mm pre-contracted aortic ring. The aortic ring is pre-contracted with 10 µM of phenylephrine prior to the application of acetylcholine. The first dose of acetylcholine is added when the phenylephrine-induced tension reaches a plateau. Please click here to view a larger version of this figure.
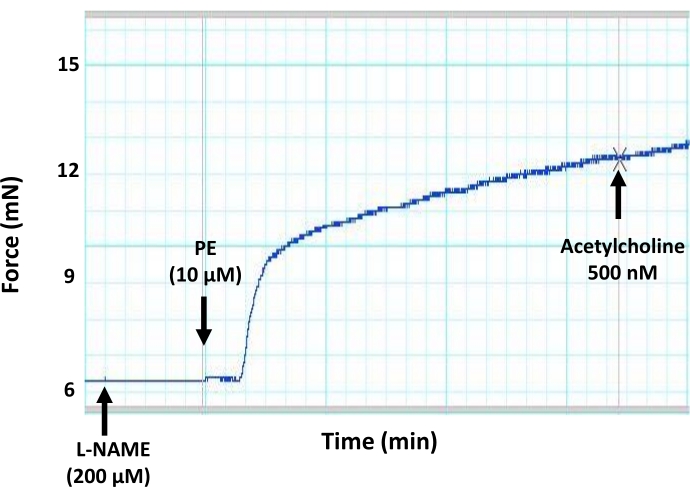
Figure 7: Effects of a general inhibitor of NO production (L-NAME) on endothelium-mediated vasorelaxation in mouse aorta. Representative myograph trace showing that the preincubation of aortic segments with a general inhibitor of NO production (L-NAME, 200 µM final concentration) blocks acetylcholine-induced vasodilation in a pre-contracted aortic ring. This is due to the inhibition of NO production by the endothelium due to the inhibitory action of L-NAME on eNOS. Acetylcholine was added to the pre-contracted aortic segment at the sub-maximum concentration of 500 nM. Please click here to view a larger version of this figure.
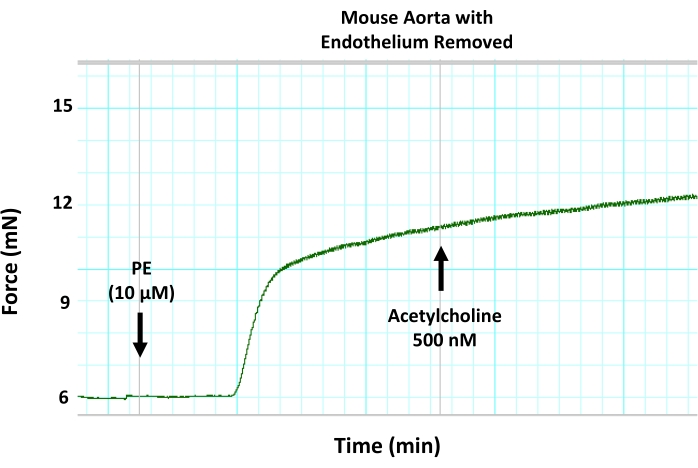
Figure 8: Effects of mechanical endothelium removal on endothelium-mediated vasorelaxation in mouse aorta. Representative myograph trace showing that removing the endothelium from aortic segments using wire denudation blocks acetylcholine-induced vasodilation in a pre-contracted aortic ring. This is due to the inhibition of endothelium-mediated vasorelaxation. Acetylcholine was added to the pre-contracted aortic segment at the sub-maximum concentration of 500 nM. Please click here to view a larger version of this figure.
Discussion
The field of vascular biology heavily relies on tools that help researchers to assess the functional and structural integrity of the blood vessel wall. It also demands special attention on the direct and indirect interactions between the three layers of blood vessels: the intima, media, and adventitia. Among those three layers, the intima is formed by a monolayer of endothelial cells and has a very important function in regulating vascular health and hemostasis.
It is well established that any damage to the endothelial layer can negatively affect its ability to release NO and other vasodilatory factors, leading to dysregulation of vascular function, which is observed in various vascular disorders such as atherosclerosis, aneurysm, and vasculitis10,11,12. In order to understand the underlying mechanisms that control normal endothelial function and assess the vasodilatory function and integrity of the endothelium within the vascular wall, it is imperative to utilize a standard experimental system that mimics the in vivo physiological conditions.
For large arteries, such as the aorta, the small chamber tensometric (isometric) myography is vastly recognized as a reliable tool that creates the best available, near-physiologic conditions for the blood vessel in an ex vivo setting. The system also allows for maintaining the viability of tissue for a considerably long period of time (up to 6-8 h) in the laboratory setting, making the technique a valuable and versatile tool. One other advantage is that the myograph chamber allows the blood vessel rings to be kept and reused for back-to-back different experiments, thus making it a cost-effective approach while reducing the need for high numbers of experimental mice. Up to four blood vessel segments can be tested simultaneously using a four-chamber myograph system, increasing consistency while reducing variations across experiments.
Various pharmacological and mechanical tools can be used to study the function of the endothelial layer in blood vessels. The major marker of a functional endothelium is the normal production of NO, which is known as the most important vasodilatory agent produced and released by the endothelial layer. Endothelial dysfunction is mainly associated with a significant drop in NO production and has been shown to be involved in the progression of different vascular disorders such as hypertension, thrombosis, and atherosclerosis.
Within the vascular bed, NO production is mainly controlled by changes in blood flow and pressure, or by other intracellular events that may lead to changes in cytoplasmic calcium concentration or the activation of signaling pathways in response to hormones and growth factors13,14. Changes in NO production are considered one of the early and reliable markers of endothelial dysfunction, and they are usually detectible early during the progression of cardiovascular disorders. Regardless of the disease model, vascular biologists are very much interested in tools and assays that allow for measuring endothelial function. It is especially important that one can differentiate between the contribution of various layers of the blood vessel using a platform that mimics the physiological conditions.
In a small chamber myograph, researchers can utilize pharmacological and mechanical tools to measure endothelial function in a tightly controlled environment. Inside the myograph chamber, an artificial environment is created that can support the normal function of the blood vessel. In such an artificial environment, since the isolated blood vessel segments are not supported by the surrounding connective tissue and other organs, it is important to determine the optimal passive tension at which the isolated segments can generate the maximum possible contraction in response to vasopressors. At the optimal tension, one can measure the normal maximum contractile response to vasoconstricting agents such as phenylephrine or norepinephrine to test the structural and functional integrity of the smooth muscle layer of the blood vessel wall. In the laboratory, it was determined that the passive tension of 6 mN is a proper tension for 2 mm mouse aortic segments15. However, the optimal passive tension must be determined for different types of arteries in different species16.
In addition, before performing any experiments with isolated blood vessels, it is imperative to test the viability of isolated rings to make sure they meet the inclusion and exclusion criteria for viable and usable tissue. This is usually achieved by subjecting the isolated rings to high concentration K+ solution (60 mM KCl). This results in depolarization of the smooth muscle membrane due to the opening of voltage-gated calcium channels (VGCC), leading to smooth muscle contraction and aortic vasoconstriction. This method is used to validate the viability of the aortic segments before using those segments for further experiments.
On the other hand, vasodilating agents such as acetylcholine can be used to test the functional properties of the endothelial layer. If the endothelium layer is intact and functional, then a sub-maximum concentration of acetylcholine can induce relaxation in a pre-contracted blood vessel segment17. The magnitude of acetylcholine-induced relaxation is an indication of the level of NO release from the endothelial layer. Any damage to the endothelial layer (mechanical or functional) will have an impact on NO production and the vessel vasodilatory response. In this protocol, the provided data shows that acetylcholine can induce relaxation in the pre-contracted mouse aorta in a dose-dependent fashion, with sub-maximum relaxation achieved at the final concentration of 500 nM (Figure 6).
In some experiments, researchers are interested in measuring the smooth muscle direct response to the vasodilator NO. Since the focus of such experiments is on smooth muscle function only, there is a protocol in place that would allow researchers to bypass the endothelium contribution by removing (denuding) the endothelium layer from the blood vessel segment. The endothelium can be removed through various methods, including air, rolling between fingers, or removal by wire. In such experimental settings, the denuded blood vessel (with no endothelial layer) is subjected to NO donors such as nitroglycerin and sodium nitroprusside. This allows for determining whether the smooth muscle cyclic GMP-protein kinase G signaling pathway that responds to NO is intact and functional7,18. This manuscript explained how the removal of the endothelial layer in the isolated aortic segment completely blocks acetylcholine vasodilatory effects, highlighting the importance of NO release in blood vessel relaxation and vasodilation (Figure 8).
While the wire and tensometric myograph techniques have wide utility in vascular biology experiments, it is worth noting the potential limitations. Specifically, the tensometric system has tissue size limitations (i.e., smaller vasculature). In addition, the ex vivo nature of this technique does not allow for manipulation of the intraluminal pressure and flow for more precise mimicry of in vivo vascular function and hemodynamic parameters. Pressure myograph setups would, in fact, account for these variables and simulate real-time vascular dynamics while also allowing for the use of smaller resistance arteries. In addition, wire myograph experiments cannot fully replicate physiological conditions or accurately model the interactions between circulating blood, vessel walls, and surrounding tissue, which also play important roles in regulating vascular function in the body.
Regardless of the experimental protocol used in the myography experiments or the selected vasoconstrictor/vasodilator in the experiment, the small chamber myography system provides a reliable, reproducible, and stable semi-physiologic platform for measuring blood vessel reactivity and functional integrity. Although the focus of this report was only on the basic measurement of endothelial function, the myography system can be used to assess many other functional properties of the blood vessel, such as the stress/strain relationship, vascular wall strength, rupture point, and passive and active contraction to name a few. This highlights the value of the myograph system as a reliable tool in the field of vascular biology.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
This work was supported by funding from the National Institutes of Health (R15HL145646) and Midwestern University College of Graduate Studies.
Materials
| Acetylcholine | SigmaAldrich | A6625-100G | |
| CaCl2 | SigmaAldrich | C4901-1KG | |
| Carbogen gas | Matheson | H103847 | |
| Dissecting scissors | FST | 91460-11 | |
| DMT 620 Multi chamber myograph system | DMT | DMT 620 | Multi chamber myograph system |
| Dumont forceps | FST | 91150-20 | |
| EDTA | SigmaAldrich | E5134-10G | |
| Glucose | SigmaAldrich | G8270-1KG | |
| HEPES | SigmaAldrich | H7006-1KG | |
| KCl | SigmaAldrich | P9541-1KG | |
| KH2PO4 | SigmaAldrich | P5655-1KG | |
| LabChart | ADI instruments | Data acquisition software | |
| Light source | Volpi | 14363 | |
| L-Name | Fischer Scientific | 50-200-7725 | |
| MgSO4 | SigmaAldrich | M2643-500G | |
| Microscope | Leica | S6D | stereo zoom microscope |
| NaCl | SigmaAldrich | S5886-5KG | |
| NaHCO3 | SigmaAldrich | S5761-500G | |
| Organ bath system | DMT | 720MO | |
| Phenylephrine | SigmaAldrich | P6126-10G | |
| Pump | Welch | 2546B-01 | |
| Software | ADI instruments | LabChart 8.1.20 | |
| Spring Scissors | FST | 15003-08 | |
| Sylgard 184 Kit | Electron Microscopy Services | 24236-10 | silicone elastomer kit |
| Tank Regulator | Fischer Scientific | 10575147 | |
| Water bath system | Fischer Scientific | 15-462-10 |
Referenzen
- Wenceslau, C. F., et al. Guidelines for the measurement of vascular function and structure in isolated arteries and veins. American Journal of Physiology-Heart and Circulatory Physiology. 321 (1), 77-111 (2021).
- Deanfield, J. E., Halcox, J. P., Rabelink, T. J. Endothelial function and dysfunction: Testing and clinical relevance. Circulation. 115 (10), 1285-1295 (2007).
- Lerman, A., Zeiher, A. M. Endothelial function: Cardiac events. Circulation. 111 (3), 363-368 (2005).
- Rajendran, P., et al. The vascular endothelium and human diseases. International Journal of Biological Sciences. 9 (10), 1057-1069 (2013).
- Galley, H. F., Webster, N. R. Physiology of the endothelium. British Journal of Anaesthesia. 93 (1), 105-113 (2004).
- Orita, H., et al. In vitro evaluation of phosphate, bicarbonate, and Hepes buffered storage solutions on hypothermic injury to immature myocytes. Cardiovascular Drugs and Therapy. 8 (6), 851-859 (1994).
- Liu, Y. H., Bian, J. S. Bicarbonate-dependent effect of hydrogen sulfide on vascular contractility in rat aortic rings. American Journal of Physiology. Cell Physiology. 299 (4), 866-872 (2010).
- Griffiths, K., Madhani, M. The use of wire myography to investigate vascular tone and function. Methods in Molecular Biology: Atherosclerosis. 2419, 361-367 (2022).
- Pfeiffer, S., Leopold, E., Schmidt, K., Brunner, F., Mayer, B. Inhibition of nitric oxide synthesis by NG-nitro-L-arginine methyl ester (L-NAME): Requirement for bioactivation to the free acid, NG-nitro-L-arginine. British Journal of Pharmacology. 118 (6), 1433-1440 (1996).
- Bacon, P. A. Endothelial cell dysfunction in systemic vasculitis: New developments and therapeutic prospects. Current Opinion in Rheumatology. 17 (1), 49-55 (2005).
- Gallo, G., Volpe, M., Savoia, C. Endothelial dysfunction in hypertension: Current concepts and clinical implications. Frontiers in Medicine. 8, 798958 (2021).
- Mikolajczyk, K., et al. The important role of endothelium and extracellular vesicles in the cellular mechanism of aortic aneurysm formation. International Journal of Molecular Sciences. 22 (23), 13157 (2021).
- Vallance, P., Hingorani, A. Endothelial nitric oxide in humans in health and disease. International Journal of Experimental Pathology. 80 (6), 291-303 (1999).
- Tousoulis, D., Kampoli, A. M., Tentolouris, C., Papageorgiou, N., Stefanadis, C. The role of nitric oxide on endothelial function. Current Vascular Pharmacology. 10 (1), 4-18 (2012).
- Gibson, C., et al. Mild aerobic exercise blocks elastin fiber fragmentation and aortic dilatation in a mouse model of Marfan syndrome associated aortic aneurysm. Journal of Applied Physiology. 123 (1), 147-160 (2017).
- Xiao, X., Ping, N. N., Li, S., Cao, L., Cao, Y. X. An optimal initial tension for rat basilar artery in wire myography. Microvascular Research. 97, 156-158 (2015).
- Chung, A. W., Yang, H. H., Yeung, K. A., van Breemen, C. Mechanical and pharmacological approaches to investigate the pathogenesis of Marfan syndrome in the abdominal aorta. Journal of Vascular Research. 45 (4), 314-322 (2008).
- Zhong, C., et al. Age impairs soluble guanylyl cyclase function in mouse mesenteric arteries. International Journal of Molecular Sciences. 22 (21), 11412 (2021).

