Amplitude-Integrated EEG in Infants at Risk of Hypoxic-Ischemic Encephalopathy: A Feasibility Study in Road and Air Transport in Western Australia
Summary
Here, we present a feasibility study to assess a portable amplitude-integrated electroencephalogram (aEEG) recording system during the transport of infants with suspected hypoxic ischemic encephalopathy (HIE).
Abstract
Infants at risk of HIE require early identification and initiation of therapeutic hypothermia (TH). Earlier treatment with TH is associated with better outcomes. aEEG is frequently used when making the decision whether to commence TH. As this is often limited to tertiary centers, TH may be delayed if the infant requires transport to a center that provides it. We aimed to provide a method for the application of amplitude-integrated electroencephalogram (aEEG) and to determine the feasibility of acquiring clinically meaningful information during transport. All infants ≥35 weeks, at risk of HIE at referral, were eligible for inclusion. Scalp electrodes were placed in the C3-C4; P3-P4 position on the infant's scalp and connected to the aEEG amplifier. The aEEG amplifier was, in turn, connected to a clinical tablet computer with EEG software to collect and analyze aEEG information. Recordings were reviewed by the chief principal investigator and two independent reviewers (blinded) for background trace and artifact. Predefined criteria for data quality were set to movement artifacts and software impedance notifications. Surveys were completed by healthcare staff and parents for acceptability and ease of use.
Introduction
Impaired oxygen delivery or blood flow to the brain around the time of birth can cause brain injury (hypoxic ischemic encephalopathy; HIE). This is a leading cause of death and disability in full-term babies1. HIE is reported to have a worldwide incidence of 2 per 1000 deliveries, which is likely to be higher in low to middle-income countries. In Western Australia, approximately 50 babies are born each year who have a clinical diagnosis of HIE (32308 live births in 2022, an incidence of approximately 1.5 per 10002).
HIE is diagnosed with criteria based on the circumstances of birth, changes in the infant's blood acid balance (from umbilical cord or baby after birth), and changes in neurological status3. The treatment for moderate or severe HIE is whole-body hypothermia of 33-34 °C. The earlier this is commenced, the likely greater the benefit to the baby's neurological outcome3,4,5. Amplitude-integrated EEG (aEEG) is an essential part of assessment and monitoring, which in many regions is only available in tertiary centers. Electrodes (EEG safety DIN connector or DIN 42802 with 1.5 mm diameter pin) are inserted into the scalp to measure and record brain electrical activity. Electrodes are then connected to an amplifier, which is in turn connected to a clinical tablet computer to display and record brainwave activity. aEEG can provide information on brain wave activity and be used to assist with the diagnosis of HIE6. The combination of neurological examination and aEEG may enhance the ability to identify infants with moderate or severe HIE7,8,9.
The Newborn Emergency Transport Service of Western Australia (NETS WA) provides intensive care transport to approximately 1100 sick babies per year. Healthcare staff in local hospitals can contact NETS WA through a free phone number and are immediately connected via a conference call with a neonatal specialist doctor and nurse to request a transfer. The NETS team travels either by specialized road ambulance or fixed-wing aircraft with a purpose-built intensive care transport equipment cot. On arrival of the transport team to the referring hospital, the baby is assessed, and treatment is continued or escalated. The team then transfers the infant to a tertiary neonatal intensive care unit (NICU) in the state capital (Perth). In Western Australia, owing to the often-large distances between referring hospitals and centralized specialist services10 (a maximum distance of 1381 miles), aEEG may not applied for up to many hours after birth, leading to a delay in treatment.
Many monitoring systems have been designed for the intensive care unit but owing to their size and power requirement, they are impractical for the transport environment. Ward-based equipment is prone to movement artifacts, which may record insufficient quality data in the transport environment. Ambulatory EEG amplifier technology, designed primarily to be worn by patients at home, can connect to a computer tablet with EEG detection software and be applied to a newborn during transport11. This can be carried by the neonatal transport team to a baby with suspected HIE. The software is capable of being viewed remotely via a portable internet uplink and web-based viewer. Currently, there are no reports on the suitability or readability of aEEG in the transport environment.
Applying and monitoring aEEG in the transport of infants with HIE has not been previously reported, and there are no existing protocols for its use. There is no evidence to support feasibility or utility in the transport environment. We present a protocol for aEEG use in neonatal transport. It aims to assess whether aEEG in babies with HIE who require transport after birth is feasible and can provide readable clinical information.
Protocol
The study was approved by the Child and Adolescent Health Service (CAHS) Human Research Ethics Committee (HREC, Approval Number RGS0000004988) and adhered to the tenets of the Declaration of Helsinki.
1. Identification of patients for inclusion in the study
- At the time of referral determine the infant's eligibility to be enrolled in the study. Any infant with suspected HIE requiring transport is eligible for the study.
- Use the standardized diagnostic criteria12 to identify suspected HIE infants:
- Check if the gestational age is >35 weeks.
- Check if it is <6 h post birth.
- Check for evidence of asphyxia as defined by the presence of the following four criteria:
- Check for any acute perinatal event that may result in HIE (e.g., abruption of placenta, cord prolapse, severe FHR abnormality).
- Check if the Apgar score is <6 at 10 min or if there is a continued need for resuscitation with positive pressure ventilation +/- chest compressions at 10 min of age.
- Check if the cord pH is <7.0 or BE is >12 mmol/L within 60 min of birth (if able to do gas).
- If cord pH is not available, check if arterial pH is <7.0 or BE is >12 mmol/L within 60 min of birth (if able to do gas).
2. Obtaining consent from the parents
- On arrival, approach parents for verbal consent for the use of the device and a survey on the acceptability of its use.
- Provide the parent information sheet, which contains all the necessary information for informed consent.
- Ensure the parent(s) understand the information and have the opportunity to ask any questions.
- Obtain written consent within 72 h of the verbal consent.
3. Requesting consent from the transport doctor and nurse
- Request consent from the transport doctor and nurse to send them surveys about their experience using aEEG in transport.
4. aEEG application
- Insert aEEG electrode pins into the baby's scalp per standard guidelines13.
- Measure and locate the anatomical landmarks: Sagittal suture and ear tragus.
- Align the electrode measuring tape (Figure 1) vertically on the head and parallel to the face.
- Match the letters/symbols on the positioning aid until the same letter/symbol in both locations (Figure 1).
- Mark the positions with a surgical pen on each side of the tape (Figure 2).
- Let the hair part naturally or part it vertically away from the marked position using sterile water and swab sticks. Dry it with gauze.
- Clean skin with 1% chlorhexidine/alcohol swabs and let it dry.
- Hold the skin tight and insert the needle at 30° angle, with the sensor wire upwards.
- Secure the subdermal needle electrode in place using steristrips and the Chevron method14 (Figure 3).
- Repeat the previous steps for the other needle insertions.
- If the scalp is very hairy, use skin preparation swabs around the needle insertion site and allow drying prior to taping.
- Point the needles and wires in the same direction.
- Place the reference electrode on the chest (anterior or posterior), the back of the shoulder, or the nape area of the neck.
- Connect the electrodes to the aEEG amplifier using the C3/C4 and P3/P4 configurations (see Figure 4).
5. Setting up EEG amplifier and tablet
- Connect the amplifier to the tablet computer using an ethernet cable; an overview of the connections can be seen in Figure 5.
- Turn on the tablet and log in.
- Enter the Acquire Pro application. The default neonatal configuration will be automatically applied.
- Change the default settings of impedance from 5 KΩ to 10 KΩ.
- Press Start Recording, and a patient ID will automatically be created.
- Press the EEG tab to observe the live trace.
- Secure the clinical tablet to the cot. Transport the infant in either a Mansell neocot or a Voyager transport incubator (Figure 6 and Figure 7). The location of the tablet on the cot depends on which cot is taken on the transport (see Figure 8).
- If using an aircraft, dismount the tablet from the cot and hold it in the hands.
6. aEEG monitoring during transport.
- During the transport, note any movements that may contribute to artifact, clinical change, or seizure on the tablet software. Use buttons that can be pressed on the tablet to label important events, including seizure, patient care, loading/unloading, take-off, and landing.
- For continuous clinical events such as seizures, a label will be added for the duration they occur. When the event finishes, press the button a second time. For non-continuous events, press the button only once for a time stamp.
- On arrival at the receiving hospital, transfer the baby to the intensive care bed. Disconnect scalp electrodes from the study amplifier and transfer them onto the NICU aEEG monitor, where the recording can be continued per unit protocol.
NOTE: Treating clinicians must be blinded to the aEEG trace and acute decisions must not be made based on the traces.
7. Data interpretation
- Record demographic and transport information in addition to electroencephalogram.
- Download aEEG traces using the Centrum application.
- Ensure that the aEEG traces are reviewed by the CPI and two independent assessors with between 5-20 years' experience of reading neonatal aEEG.
- Blind the assessors to the clinical information and traces.
- Separate traces into 15 min segments and review the number of artifact segments. A segment of the trace is considered an artifact if two of the three 'experts' agree. An arbitrary value of 100 µV has been chosen at which movement artifact is said to occur.
- Download raw data from each trace at a rate of 8 Hz from the C3-C4 electrodes.
NOTE: Impedance (Z) is the resistance of current flow and measures the quality of skin electrode placement and lead motion artifact. Low, equal, and stable impedance ensures optimal aEEG. The software system records impedance as a discrete notification set at >10 KΩ. The magnitude and number are recorded for each trace. - Complete staff and parent surveys after the transport using an electronic data capture tablet application.
NOTE: The study aims to recruit and collect data from 20 patients with suspected HIE.
8. Analysis
- Record and present demographic, transport, and aEEG descriptives using median (interquartile range; IQR).
- Present the artifact as a percent of the total trace. Describe movement artifact as the mean (standard deviation; SD) percent of the total trace.
- Present Z as a mean number (range) and Z magnitude as median (IQR) as kiloohms (KΩ).
- Define traces as total and movement artifacts free and absent of Z 80% of the time.
NOTE: Representative results presented in this methodology are expressed as a median and interquartile range and comparison of medians using the Mann-Whitney U test.
Representative Results
Between 1st September 2022 and 5th June 2023, 25 babies were transported in Western Australia with possible HIE and were eligible for inclusion. Of the total, 20 infants were consented, recruited, and successfully had aEEG applied during their transport. A total of 5 patients were not recruited and 3 were missed for consent. In one case, the device had started an automatic software update and was unusable; in one case, the baby was not at risk of HIE and was deemed ineligible for recruitment. Of the 20 infants, 12 (12/20; 60%) were male and 8 (8/20; 40%) were female. A total of 13 infants (13/20; 35%) were primarily transported by fixed-wing aircraft and 7 (7/20; 65%) by road. In total, 38 h of aEEG traces were recorded. The median postnatal age at which the recording commenced was 5.7 h (3.9-6.5 h) h. Infants primarily transported by fixed-wing aircraft compared to the road had aEEG commenced at 8.9 h (6.2-10.8 h) vs. 4.2 h (3.0-5.5 h); p = 0.0048. The median duration of EEG data recorded per patient was 1.9 h (1.6-3.1 h), with a range of 0.9-6.4 h. There were no impedance notifications in 16/20 recordings. Four recordings had a mean (range) of 4 (1-6) impedance notifications per recording. Despite this, all recordings had information that could be analyzed; examples are shown in Figure 9. The technology was acceptable to parents and staff.
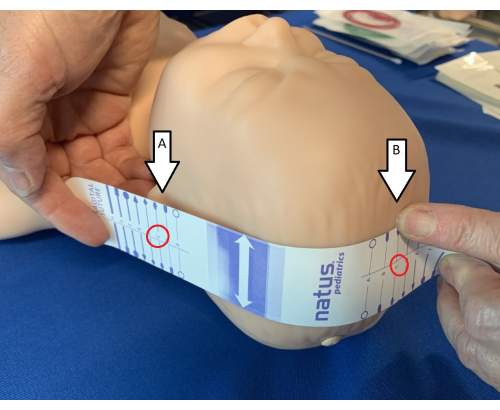
Figure 1: Identification of landmarks for measuring tape. Using the electrode measuring tape, identify the ear tragus (A) and sagittal suture (B) and ensure the tape is lined up so that the letters match (in the mannequin shown in the figure, it is "C"). Please click here to view a larger version of this figure.
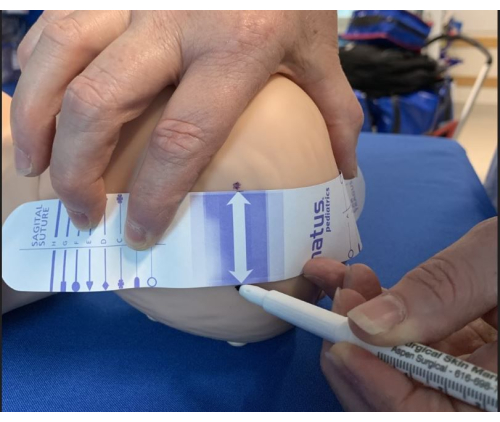
Figure 2: Identifying sites for electrode placement. Once the measuring tape is appropriately positioned to landmarks, mark electrode sites on both sides of the arrow using a marker pen. Please click here to view a larger version of this figure.
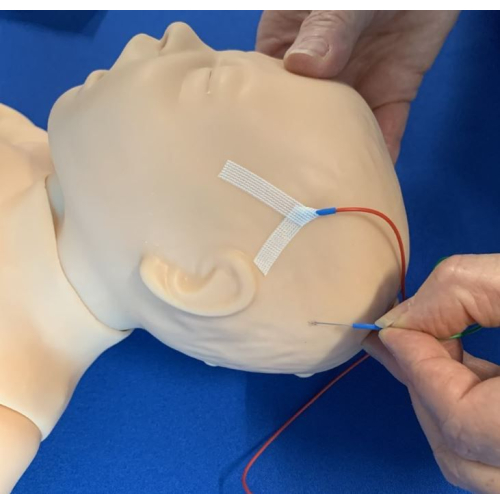
Figure 3: Insertion of electrodes. The electrodes were inserted subcutaneously and positioned caudally, taping with the steristrips Chevron technique. Please click here to view a larger version of this figure.
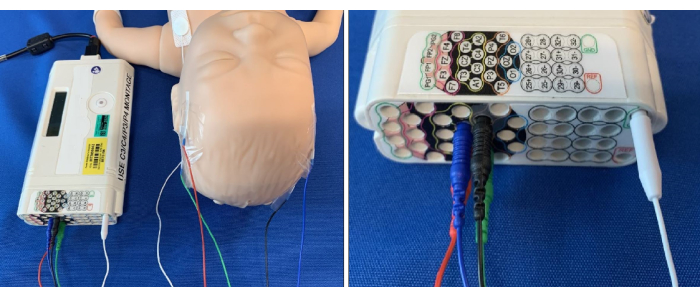
Figure 4: Electrodes connected to the corresponding site on the aEEG amplifier. Please click here to view a larger version of this figure.
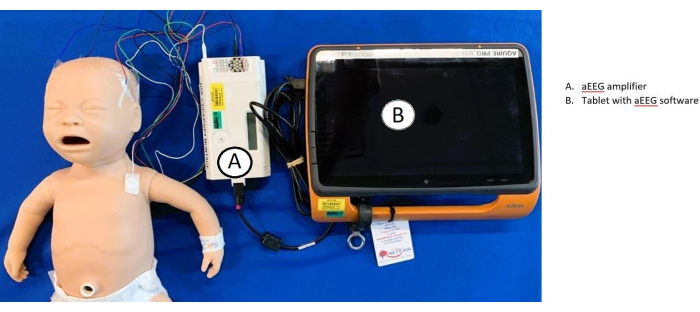
Figure 5: Overview of aEEG set-up and connectivity. Electrodes are connected to the baby and (A) amplifier. The amplifier is then connected to the (B) tablet, which uses aEEG software. Please click here to view a larger version of this figure.

Figure 6: Mansell cot. Please click here to view a larger version of this figure.

Figure 7: Voyager cot. Please click here to view a larger version of this figure.

Figure 8: Tablet secured to the cot and visible to the transport team. (1) aEEG tablet with continuous trace on the screen, (2) Trackit T4 aEEG amplifier, (3) aEEG scalp electrodes on the mannequin, (4) Mansell cot, and (5) vitals monitor. Please click here to view a larger version of this figure.
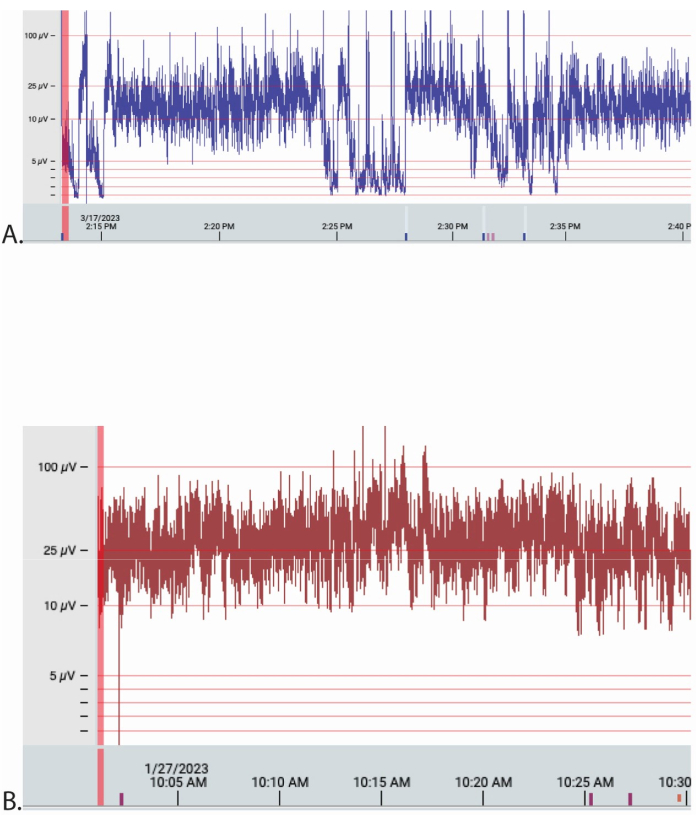
Figure 9: Examples of aEEG traces from 2 patients extracted from the online Stratus EEG Analysis software. The Y-axis is electrical amplitude (µV), and the X-axis is date and time in 5 min increments. The time base was 30 mm/s for each, and the sensitivity was 70 µV/cm for both traces. The traces are montages of the C4-C4, P3-P4 as interpreted by 'experts'. (A,B) Patient A was transported by road and patient B by air. Please click here to view a larger version of this figure.
Discussion
This novel study describes the application and early data acquisition of aEEG in infants with suspected HIE who require transport soon after birth. aEEG application during neonatal transport has not been reported previously. aEEG has been used under novel circumstances, such as remote monitoring under telehealth neurocritical care consultation in Brazil and the Paediatric ED15,16.
The aEEG needle application is similar to that used in ‘static’ neonatal intensive care17 and is positioned on the scalp to achieve optimum signal for the aEEG. The positioning of this device in the neonatal transport environment may differ depending on the configuration of the transport cot. There were no issues with the needle positioning in the scalp during transport in the 20 cases; no additional trauma or bleeding was noted. It was noted on one occasion that a needle became displaced, but this is no more common than would be in a ‘static’ NICU environment in a normally moving baby. All aEEG recordings were made using an ethernet cable attached to the clinical tablet to the amplifier. This device has Bluetooth wireless capability, the next phase of method refinement. This needs to be tested in the aircraft environment.
The main limitation of aEEG remains the subjective and interpretative nature of the output, and the risk of misinterpretation cannot be underestimated18. In our methodology, we used a combination of expert consensus review, trace time > 100 µV, and the number and magnitude of impedance. Traces were separated into 15 min segments to simplify the interpretation between ‘experts’. Each section was assessed for background pattern, and the whole trace was assessed. No portion was not assessed. The aEEG interpretation of the baseline was considered as an artifact or not if 2/3 ‘experts agreed. This consensus approach was used owing to the lack of objective assessment in aEEG interpretation and to assure traces were robustly readable. The normal range of human brain activity on the aEEG scale is 10-25 µV; >100 µV was chosen as prolonged portions above this value are more likely to be related to non-biological activity (i.e., external movement). Impedance was measured as a standard to understand opposition to the electrical signal. We believe the combination of these measures provides a holistic overview of the aEEG trace.
There were some limitations of these methods. This study was initially designed to include video as well as an EEG recording. At the time of this study, the camera equipment was not available. We were unable to assess the functionality of video recordings in transport. To refine the method further, a video camera will be located on the cot to record baby movement alongside the EEG in the future. This may support seizure identification during transport. aEEG currently cannot be objectively reviewed. We believe the proxy measures we used to demonstrate feasibility are robust. As machine learning and artificial intelligence progress, more objective methods may be available and integrated into the software to provide definitive scoring in the future19.
The current tablet does not have mobile capabilities. The Newborn Emergency Transport Service of Western Australia (NETS WA) is upgrading to a 5G sim-operated device, which can transmit data via the software cloud to the receiving hospital. Specialists can then access and review aEEG traces while the transport is in progress, except during flights, where only the senior registrar can view these.
The method is unique and has not been reported previously; it has similarities to the technique in ‘static’ NICU with some important differences. The technique has important clinical and research applications as the study of brain function in transported babies is very limited. Early clinical decision-making and prognosis are important in perinatal asphyxia, and babies who are not born in tertiary institutions should have this opportunity also. Applying aEEG in transport may allow the study of babies with HIE (in early identification and prognosis), seizure presentation, and monitoring over distance. It may promote the optimal neuromonitoring of babies who require sedation during transport.
In summary, aEEG in neonatal transport is both feasible and produces sufficient data for clinical interpretation. Having the ability to make early clinical decisions in time-critical conditions (such as HIE) is important. Future studies should focus on how the new technology can improve patient outcomes utilizing real-time aEEG traces and include video monitoring for seizures during transport.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
We thank the Perth Children’s Hospital Foundation for their generous support in purchasing the EEG equipment used in this project. We thank Gardar Thorvardsson (Kvikna) and Ieesha Sparks (Temple Medical and Scientific) for their support in the technical aspects of the study project.
Materials
| Stratus EEG Centrum, Acquire Pro Software for Microsoft Windows |
Stratus Software Solutions LLC Kvikna Medical Lyngháls 9 110 Reykjavik Iceland |
Version 4.2 | |
| Trackit T4PCU24+8 | Lifelines Neuro 7 Clarendon Court Over Wallop StockBridge Hants, UK |
SN: T4-170046 Issue: 4 C169 |
|
| Ultra Subdermal Needle Electrode | Natus 3150 Pleasant View Road Middleton WI 53562 USA |
019-476600 | 14 mm x 0.38 mm SST Needle DIN 42802 connector |
Referenzen
- Kurinczuk, J. J., White-Koning, M., Badawi, N. Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Hum Dev. 86 (6), 329-338 (2010).
- . Midwives Notification System, Government of Western Australia, Department of Health Available from: https://www.health.wa.gov.au/Reports-and-publications/Western-Australias-Mothers-and-Babies-summary-information/data?report=mns_birth_y (2023)
- Shankaran, S., et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med. 353 (15), 1574-1584 (2005).
- Thoresen, M., et al. Time is brain: Starting therapeutic hypothermia within three hours after birth improves motor outcome in asphyxiated newborns. Neonatology. 104 (3), 228-233 (2013).
- Walløe, L., Hjort, N. L., Thoresen, M. Start cooling as soon as possible. Acta Paediatr. 108 (4), 771 (2019).
- Skranes, J. H., et al. Amplitude-integrated electroencephalography improves the identification of infants with encephalopathy for therapeutic hypothermia and predicts neurodevelopmental outcomes at 2 years of age. J Pediatr. 187, 34-42 (2017).
- Shalak, L. F., Laptook, A. R., Velaphi, S. C., Perlman, J. M. Amplitude-integrated electroencephalography coupled with an early neurologic examination enhances prediction of term infants at risk for persistent encephalopathy. Pediatrics. 111 (2), 351-357 (2003).
- Hellstrom-Westas, L., Rosen, I., Svenningsen, N. W. Predictive value of early continuous amplitude integrated eeg recordings on outcome after severe birth asphyxia in full term infants. Arch Dis Child Fetal Neonatal Ed. 72 (1), F34-F38 (1995).
- Davies, A., Wassink, G., Bennet, L., Gunn, A. J., Davidson, J. O. Can we further optimize therapeutic hypothermia for hypoxic-ischemic encephalopathy. Neural Regen Res. 14 (10), 1678 (2019).
- Davis, J. W., et al. Outcomes to 5 years of outborn versus inborn infants <32 weeks in western australia: A cohort study of infants born between 2005 and 2018. Arch Dis Child Fetal Neonatal Ed. 108 (5), 499-504 (2023).
- Schomer, D. L. Ambulatory EEG monitoring, reviewing, and interpreting. J Clin Neurophysiol. 38 (2), 77-86 (2021).
- . Neonatology. Hypoxic ischaemic encephalopathy (HIE) Available from: https://cahs.health.wa.gov.au/~/media/HSPs/CAHS/Documents/Health-Professionals/Neonatology-guidelines/Hypoxic-Ischaemic-Encephalopathy.pdf?thn=0 (2018)
- . Neonatology. Monitor: Low impedance needle electrodes Available from: https://cahs.health.wa.gov.au/-/media/HSPs/CAHS/Documents/Health-Professionals/Neonatology-guidelines/Brainz-Monitor-Low-Impedance-Needle-Electrodes.pdf?thn=0 (2021)
- Found, P. W. H., Baines, D. B. Efficacy of securing cannulae with different taping methods. Anaesth Intensive Care. 28 (5), 547-551 (2000).
- Stephens, C. M., et al. Electroencephalography quality and application times in a pediatric emergency department setting: A feasibility study. Pediatr Neurol. 148, 82-85 (2023).
- Variane, G. F. T., et al. Remote monitoring for seizures during therapeutic hypothermia in neonates with hypoxic-ischemic encephalopathy. JAMA Netw Open. 6 (11), e2343429 (2023).
- Bruns, N., et al. Application of an amplitude-integrated EEG monitor (cerebral function monitor) to neonates. J Vis Exp. 127, 55985 (2017).
- Dilena, R., et al. Consensus protocol for eeg and amplitude-integrated eeg assessment and monitoring in neonates. Clin Neurophysiol. 132 (4), 886-903 (2021).
- Moghadam, S. M., et al. An automated bedside measure for monitoring neonatal cortical activity: A supervised deep learning-based electroencephalogram classifier with external cohort validation. Lancet Digit Health. 4 (12), e884-e892 (2022).

.
