Murine Dermal Lymphatic Endothelial Cell Isolation
Summary
This paper describes a method for magnetic bead-based isolation of murine endothelial cells from dermal lymphatic capillaries. The isolated lymphatic endothelial cells can be used for downstream in vitro experiments and protein expression analysis.
Abstract
The lymphatic system participates in the regulation of immune surveillance, lipid absorption, and tissue fluid balance. The isolation of murine lymphatic endothelial cells is an important process for lymphatic research, as it allows the performance of in vitro and biochemical experiments on the isolated cells. Moreover, the development of Cre-lox technology has enabled the tissue-specific deficiency of genes that cannot be globally targeted, leading to the precise determination of their role in the studied tissues. The dissection of the role of certain genes in lymphatic physiology and pathophysiology requires the use of lymphatic-specific promoters, and thus, the experimental verification of the expression levels of the targeted genes.
Methods for efficient isolation of lymphatic endothelial cells from wild-type or transgenic mice enable the use of ex vivo and in vitro assays to study the mechanisms regulating the lymphatic functions and the identification of the expression levels of the studied proteins. We have developed, standardized and present a protocol for the efficient isolation of murine dermal lymphatic endothelial cells (DLECs) via magnetic bead purification based on LYVE-1 expression. The protocol outlined aims to equip researchers with a tool to further understand and elucidate important players of lymphatic endothelial cell functions, especially in facilities where fluorescence-activated cell sorting equipment is not available.
Introduction
The lymphatic system plays a pivotal role in human physiology. It is considered a vital homeostatic factor, that facilitates important functions, such as the maintenance of tissue-plasma fluid balance, immune surveillance, and lipid absorption1, as well as recently-identified functions, such as the repair ability of the heart2 and regenerative capacity of bone and hematopoiesis3. Despite the significant role of the lymphatic system, several aspects of the role of the lymphatics, as well as the molecular mechanisms governing certain physiological parameters and responses remain elusive. Moreover, lymphatic vascular defects are triggering or deteriorating factors in certain pathophysiological conditions. Well-known examples are the primary and secondary lymphedemas, depending on the genetic or non-genetic origin of the disease, respectively, while the role of the lymphatic system on primary tumor growth and metastatic dissemination is pivotal, as it can act as a conduit for metastasis and modulator of immune functions1.
The lag in the study and knowledge of the lymphatic compared to the blood vasculature is mainly due to the later discovery of the lymphatic system in comparison with the blood vascular system and the delay in the identification of lymphatic-specific molecular markers. This has greatly improved in recent decades, leading to the alteration of the conventional picture of the lymphatics at steady state and in diseased conditions1. Similar to angiogenesis, lymphangiogenesis is the formation of new lymphatic vessels from pre-existing ones and plays a pivotal role in the development of a wide spectrum of diseases4,5,6. However, unlike angiogenesis, the investigation of lymphangiogenesis has been limited to mostly in vivo models focusing on developmental vessel formation and structural defects in pathological models. The isolation of the lymphatic endothelial cells is important for in vitro studies, and this can be provided by the presented protocol.
Several protocols for lymphatic endothelial isolation from mice have been developed, which require the use of fluorescence-activated cell sorting7. The advantage of the cell sorter, where available, is the higher degree of purity, contrary to bead-based isolation, with the latter providing a higher yield. The protocol utilizes the expression of lymphatic endothelial hyaluronan receptor 1 (LYVE-1), a lymphatic endothelial marker, important for cell trafficking within the lymphatic vessels4,8. Since LYVE-1 expression is limited to the lymphatic capillaries and not the collecting lymphatic vessels, the technique is suitable for the isolation of lymphatic endothelial cells specifically from the lymphatic capillaries, contrary to other lymphatic markers, such as podoplanin, which are expressed uniformly in all lymphatic endothelial cells9. For the protocol, other important lymphatic markers that were not transmembrane, such as Prox1, were excluded.
As the physiological outcome was lymphangiogenesis, which is usually initiated at the lymphatic capillaries, LYVE-1 was selected as a target due to its selectivity in these cells and because the selected antibody provided a high yield. The enzymes used were collagenase type II, which is generally known to be more effective in collagen dissociation than type I10, and dispase, a broader protease, regularly used for dermis-epidermis separation11; however, other enzymes for tissue separation have been reported12,13 and can be used. The goal of this protocol is to describe a method for dermal lymphatic endothelial cell isolation from lymphatic capillaries of adult mice with high yield using magnetic bead purification, especially in places where fluorescence-activated cell sorting is not readily available. The method is suitable for applications where the purity of lymphatic endothelial cells can be compromised for downstream applications.
Protocol
Animal studies were performed according to the approved protocols by the Institutional Animal Care and Use Committee (IACUC) of Texas Tech University Health Sciences Center (TTUHSC) for the experiments at TTUHSC and by the Veterinary Administration of the Prefecture of Western Greece according to Directive 2010/63 for the experiments at the University of Patras. Diligently follow waste disposal regulations when disposing of animal waste materials.
1. Isolation of mouse dermis
- Day 1
NOTE: On Day 1; get the following materials and equipment ready: anesthesia chamber, animal hair clipper, isoflurane, fine forceps, 0.2 µm syringe filters.- Prepare a 2 mg/mL Dispase solution in DMEM with 1x Antibiotic-Antimycotic Solution and filter-sterilize it.
NOTE: The use of medium or PBS can be replaced by HBSS, as it has been successfully used in other reported protocols12. The 1x Antibiotic-Antimycotic Solution can be substituted by the addition of penicillin (100 I.U./mL), streptomycin (100 µg/mL), and amphotericin (2.5 µg/mL). - Anesthetize the mouse by placing the mouse in an anesthesia chamber with isoflurane administration (0.5-2% isoflurane, flow: 1 L/min). After the mouse stops moving, transfer it out of the cage, and continue anesthesia adjusting a nose cone with a similar isoflurane flow. Toe-pinch the animal to confirm sufficient anesthesia levels and constantly monitor the mouse to ensure deep anesthesia is preserved (no motility). Clip approximately 9 cm2 of the dorsal and ventral fur (mouse back and abdomen respectively) longitudinally along the midline, using a hair clipper. Let the mouse recover from anesthesia and return it to the cage.
NOTE: Hair removal (depilation) cream can be used instead. Make sure the fur is completely removed in the skin areas to be isolated while maintaining the integrity of the skin. Try to clip as extended skin area as possible. To ensure that potential inflammation from the hair removal is resolved, cell isolation must take place the following day. To increase the number of isolated cells pool 2 to 4 mice per group.
- Prepare a 2 mg/mL Dispase solution in DMEM with 1x Antibiotic-Antimycotic Solution and filter-sterilize it.
- Day 2
NOTE: Keep the following materials and equipment ready: euthanasia chamber, biosafety cabinet, CO2 incubator, ethanol, fine scissors, fine forceps, blunt forceps, 100 mm tissue culture plate, Dispase, DMEM, 1x Antibiotic-Antimycotic Solution.- Euthanize the mouse by CO2 administration and place it in a supine position, exposing the clipped area. Spray 70% ethanol as a disinfectant.
NOTE: From this step onwards, all steps must be performed under sterile conditions (in a biosafety cabinet). - Place a sterile 100 mm Tissue Culture Plate on ice.
- Make a 0.5 inch superficial cut at the abdomen and cut longitudinally along the midline to the neck and the lower abdomen. Make lateral cuts to be able to flap open the skin.
NOTE: The incision size and the skin area isolated depend on the mouse size. - Using blunt forceps, carefully separate the skin from the surrounding tissues. Do this going around the entire torso of the mouse, cutting the skin laterally around the neck and the lower abdomen.
- Carefully place the skin into the 100 mm Tissue Culture Plate, with the dermis side facing down, keeping the skin as stretched as possible. Cover the plates and keep them on ice. Complete the isolation of all mice from the group.
- After the skin is extracted from all mice, incubate plates for 10 min at room temperature to flatten the explant.
- Add 6 mL of Dispase solution to the plate, making sure the skin is completely immersed and incubate overnight with mild agitation at 4 °C.
NOTE: Instead of Dispase, trypsin digestion can also be performed12, followed by Liberase instead of Collagenase Type II treatment (below).
- Euthanize the mouse by CO2 administration and place it in a supine position, exposing the clipped area. Spray 70% ethanol as a disinfectant.
2. Cell isolation from the dermis
- Day 3
NOTE: Keep the following materials and equipment ready: centrifuge, CO2 incubator, biosafety cabinet, scissors, DMEM, 1x Antibiotic-Antimycotic Solution, Collagenase Type II, PBS, 50 mL conical tube, 100 mm and 60 mm tissue culture plates, DNAse I solution, Collagen Type I, 70 µm and 40 µm cell strainers, 0.2 µm syringe filters, LEC medium.- Prepare 500 mL of 1x PBS (1.37 M NaCl, 27 mM KCl, 100 mM Na2HPO4, 18 mM KH2PO4, pH 7.4) and autoclave for 20 min at 15 psi or filter-sterilize. Alternatively, use commercially available sterile PBS.
- Prepare 10 mL of 2 mg/mL Collagenase Type II solution in DMEM with 1x Antibiotic-Antimycotic Solution and filter-sterilize it.
NOTE: Perform the next steps under a sterile hood to decrease the chances of sample contamination. - Use a pipette to remove the liquid from the dish.
- Use forceps to separate the dermis (bottom of the skin) from the epidermis (top) and remove any obvious adipose tissue. Transfer the dermis to a clean Tissue Culture plate.
- Add 1 mL of DMEM with 1x Antibiotic-Antimycotic Solution in the clean Tissue Culture Plate and tilt it so that the liquid accumulates on one side of the plate.
- Mince the dermis into 1-2 mm2 pieces and transfer them to a new 50 mL conical tube.
NOTE: This step must ideally be completed within 4-5 min, as longer processing is not expected to provide higher yield or cell survival. - Add 10 mL of the Collagenase Type II solution.
- Add 50 µL (25 units/mL of the solution) of DNAse I solution (reduces cell clumping) and digest the dermis solution for 2 h at 37 °C with continuous agitation (use a rotating/shaking incubator at 30-50 rpm).
NOTE: Check periodically to identify whether most of the dermis has been digested. If not, increase the digestion time (1 extra h maximum) and agitation speed, if possible. - Coat 60 mm Tissue Culture Plates with 2 mL of 10 µg/mL Collagen Type I in 0.02 M Acetic Acid for >20 min at 37 °C. Wash 2x with sterile PBS.
- Filter the cell suspension using a 70 µm cell strainer and centrifuge at 250 × g for 5 min. Discard the supernatant.
- Resuspend in 10 mL of DMEM with 1x Antibiotic-Antimycotic Solution.
- Filter the cell suspension using a 40 µm cell strainer and centrifuge at 250 × g for 5 min. Discard the supernatant.
- Resuspend in 1 mL of complete LEC media (Endothelial Cell Growth Base Medium with Supplement).
- Plate the cells in the collagen-coated 60 mm Tissue Culture Plate in complete LEC media. This will be Day 0 of cell culture.
- Replace the media after every 2 days by washing cells with 1x PBS twice and adding fresh LEC media.
- Repeat step 2.1.15 until the cells are 80-90% confluent.
NOTE: If the purification is performed before the cells are 80-90% confluent, fewer lymphatic endothelial cells will be isolated. This process takes about 7 to 10 days.
3. Magnetic Bead Coating with LYVE-1 antibody
NOTE: One day before the purification step, get the following materials and equipment ready: 4 °C Refrigerator, Rotor, magnetic separator, PBS, Anti-Rabbit IgG Magnetic Beads, microcentrifuge tubes, 0.2 µm syringe filters, Magnetic Bead Coating Solution, LYVE-1 antibody.
- Prepare the Magnetic Bead Coating Solution: 20 mL of PBS with 0.1% BSA and 2 mM EDTA and filter-sterilize it. Store it at 4 °C.
NOTE: More than a few days of storage is not recommended; check for precipitates or contamination before use. - Pipette 500 µL of Anti-Rabbit IgG Magnetic Beads to a microcentrifuge tube and add 1 mL of ice-cold Magnetic Bead Coating Solution.
- Use a magnetic separator to wash the beads.
- Repeat the washes 2x with 1 mL of the ice-cold Magnetic Bead Coating Solution.
- Resuspend the beads in 500 µL of Magnetic Bead Coating Solution and make aliquots of 150 µL in microcentrifuge tubes.
- Add 15 µg of the LYVE-1 antibody to each tube (15 µL from 1 mg/mL antibody stock concentration).
- Incubate overnight at 4 °C with continuous agitation.
4. Lymphatic endothelial cell purification
NOTE: Get the following materials and equipment ready: CO2 incubator, shaker, timer, parafilm, DMEM, 1x Antibiotic-Antimycotic Solution, BSA, PBS, Magnetic Bead Coating Solution, trypsin-EDTA, 100 mm or 60 mm tissue culture plates, 0.2 µm syringe filters, Collagen Type I, Acetic acid, LEC medium.
- Prepare 50 mL of DMEM with 1x Antibiotic-Antimycotic Solution and 0.1% BSA and filter-sterilize it.
- Coat 60 mm Tissue Culture Plates with 10 µg/mL Collagen Type I in 0.02 M Acetic Acid for >20 min at 37 °C. Wash 2x with sterile PBS.
- Wash the precoated beads with 1 mL of Magnetic Bead Coating Solution, using a magnetic separator.
- Resuspend the beads in 150 µL of DMEM with 0.1% BSA.
NOTE: The beads can now be stored at 4 °C for up to 1 month. - Wash the cells 2x with PBS.
- Add 3 mL of DMEM with 0.1% BSA (for a 60 mm Tissue Culture Plate) and 50 µL of the antibody-conjugated beads to the cells.
NOTE: If the cells from two or more mice are pooled, use a 100 mm Tissue Culture Plate and add 6 mL of DMEM with 0.1% BSA. - Seal the dish with parafilm.
- Place the dish on a shaker agitating at 10 RPM at RT and incubate for exactly 10 min.
NOTE: Longer incubation will result in non-specific binding. To ensure that the beads interact with the entire plate surface, manually rotate the plate after every few minutes as it continues to shake. - Discard the medium and wash the cells once with PBS. Quickly inspect the cells to verify the presence of LEC colonies on the plate.
- Trypsinize the cells: use 1 mL of trypsin-EDTA and incubate for 3 min at 37 °C. Inspect the cells for successful cell detachment.
- Neutralize the solution with 2 mL of complete LEC media and transfer the solution to a 5 mL sterile polypropylene tube.
- Using a magnetic separator wash cells 4 x 3 mL of DMEM with 0.1% BSA to remove unbound cells.
- Resuspend the remaining bead-bound cells in complete LEC media and plate onto the previously Collagen-coated 60 mm Tissue Culture Plates.
- Incubate the cells at 37 °C and change the media every other day after two PBS washes each time.
- Culture the cells till they reach confluency (it normally takes 7 to 10 days).
- At that point, check cells under the microscope for confluency and proceed with preparing cells for flow cytometry. If a high F4/80+ percentage is identified, then repeat the purification step with LYVE-1 (Step 4: Lymphatic Endothelial Cell Purification) again or ideally another lymphatic marker (i.e., podoplanin14).
NOTE: The high expression of the protein targeted by the antibody on the magnetic bead-purified cell population has been demonstrated15,16. However, for LYVE-1, it is important to perform a flow cytometry control for the identification of the percentage of other LYVE-1+ cells, mostly macrophages. LYVE-1 is only expressed in subsets of macrophages that are associated with tumors and wounds, whereas in healthy skin, LYVE-1 expression is more restricted to lymphatic endothelial cells17. Thus, the F4/80 screening is not mandatory, but rather dependent on the experimental context. The mice used for demonstration did not carry tumors or wounds, just potential inflammation from hair removal, but we included the F4/80 staining as a baseline of the percentage of LYVE-1+ macrophages.
5. Flow cytometry
NOTE: Get the following materials and equipment ready: centrifuge, CO2 incubator, timer, hemocytometer, EDTA solution, 1x Antibiotic-Antimycotic Solution, BSA, PBS, trypsin-EDTA, polypropylene tubes, 0.2 µm syringe filters, 70% ethanol, F4/80 FITC-conjugated antibody.
- Prepare PBS containing 0.1% BSA and filter-sterilize it.
- Prepare 1 mM EDTA solution in PBS.
- Trypsinize the cells: incubate the cells with 1 mL of 1 mM EDTA solution for 5 min at 37 °C.
NOTE: Incubation with EDTA solution is a milder method of cell detachment, which preserves better the transmembrane proteins and is preferred, but it requires longer incubation periods. Alternatively, trypsin-EDTA solution can be used for 1 min incubation at 37 °C, but this can affect antibody affinity for flow cytometry applications. - Neutralize the solution with 2 mL of complete LEC media and transfer the solution to a 5 mL sterile polypropylene tube.
- Centrifuge the samples at 250 × g for 5 min.
- Wash with PBS.
- Repeat the centrifugation and PBS wash.
- Resuspend the cells in PBS containing 0.1% BSA and count them using a hemocytometer or alternative method. Aliquot ~1 × 106 cells into polypropylene tubes with a final volume of 100 µl. From this step onwards, perform all the steps at 4 °C.
NOTE: The cells can be fixed before staining or stained while alive. Fixation can be suboptimal for flow cytometry, as some antibodies may not detect the antigens after fixation; therefore, we recommend running flow cytometry immediately after the previous step and skipping the fixation step. If that is not possible, fixation prior to the flow cytometry experiment will allow a delay in the flow cytometry step. If fixation is to be performed, the performance of the antibodies in the context of fixed cells should be evaluated in advance. - Perform fixation (optional step; if the cells are not to be fixed, proceed with immunostaining): resuspend the cells in ice-cold 70% ethanol and gently mix for 10 min on ice.
NOTE: Ethanol fixation can permeabilize the cells and lead to the release of GFP; however, paraformaldehyde-based fixation can destroy dyes used for flow cytometry and ideally can be avoided. - Incubate for 15 min at room temperature.
- Wash cells 2x by centrifugation at 250 × g for 5 min in 1x PBS.
- Proceed to the next step or resuspend the cells in 0.5-1 mL of 1x PBS and store at 4 °C.
6. Immunostaining
NOTE: Handle the cells gently and avoid extensive pipetting, when possible, to maintain the cell membrane integrity and minimize cell death, ensuring that the cells are not forming clumps. From the isolated endothelial cells, prepare the following groups: unstained cells and stained cells with another endothelial cell marker (i.e., VEGFR3, podoplanin), F4/80, and both. Include commercially available mouse lymphatic endothelial cells and macrophages, if available, as positive controls. For demonstration purposes, and due to the endogenous tdTomato fluorescence (PE+), we have used FITC-conjugated F4/80 staining.
- Dilute the primary antibody in PBS with 0.1% BSA according to the recommended or optimized dilution. Add 0.5 µg (1 µL of antibody solution) of the F4/80 FITC-conjugated antibody to the 100 µLof the cell suspension.
- Resuspend the cells after the addition of the diluted fluorescent-conjugated antibody. Mix by flicking the polypropylene tube to ensure that there are no clumps and all cells are properly coated.
- Incubate for 1 h at room temperature (fixed cells) or 30 min on ice (live cells) in the dark.
- Wash cells 2x by centrifuging them at 250 × g for 5 min at 4 °C (for live cells) or at room temperature (for fixed cells), using PBS with 0.1% BSA in the dark.
- For non-fluorescently-conjugated antibodies
- Dilute the fluorochrome-conjugated secondary antibody in the incubation buffer. For Alexa Flour antibodies, use a 1:500 dilution.
- Add the secondary antibody to 100 µL of the cell suspension solution.
- Incubate for 30 min at room temperature (fixed cells) or on ice (live cells).
- Wash twice by centrifugation at 250 × g for 5 min at 4 °C (for live cells) or at room temperature (for fixed cells) in the incubation buffer.
- Resuspend cells in 0.5 mL of 1x PBS and analyze them using a flow cytometer.
- Follow these gating strategy steps to identify the macrophage population within the LYVE-1-expressing cells:
- Consider forward and side scattering to distinguish the cellular population.
- Consider forward scattering for cell size area and height to distinguish the single cells (Singlets).
- Take into account F4/80 (FITC) expression and RFP (PE) expression to identify macrophages (F4/80+) within the tdTomato (PE)-expressing isolated murine cells (due to the presence of a fluorescent tdTomato reporter in the specific mice, described below).
Representative Results
The method was developed to identify the protein expression of a small GTPase, RhoA, the coding gene of which was floxed in both alleles and deleted under the effect of the tamoxifen-inducible lymphatic endothelial-specific promoter Prox1-CreERT2 18. The isolated LECs can be cultured for up to four generations, after which they become senescent. They have been frozen, thawed, and successfully stimulated with angiopoietin-2. Due to the limited number of cell passages, we have not tried multiple freeze-thaw cycles. The typical cell yield upon isolation from one mouse provides a 40-50% confluency (~1.5 × 106 cells) of a 60 mm dish, and the proliferation is slow, as the dish reaches confluency after 5 days when we perform the LYVE-1-based purification.
After the first purification, the LYVE-1+ cells have a 15% confluency of the 60 mm dish (~4 × 105 cells). A second round of purification is optional, and the data and images provided are all with one round of purification. There are a number of cells that are not selected, which carry magnetic beads, and it seems that the number of beads attached was not sufficient for their selection. The addition of DNAse in parallel to dispase digestion is always recommended, as mentioned earlier, although we did not observe a noticeable increase in the yield. For the demonstration experiment in the present manuscript, we used mice carrying the tdTomato fluorescent reporter, which we had previously used in combination with floxed genes and different promoters16,18. The purpose of the use of these mice was the (red; PE+) fluorescent view of the isolated cells.
The different steps of the tissue processing are demonstrated in Figure 1A, with images of the extended skin of the mice prior to the overnight treatment with Dispase (Figure 1B), the removal of the epidermis from the dermis the following day (Figure 1C), and the mincing of the dermis for the isolation of the cells right after (Figure 1D). Upon the purification of the LYVE-1+ cells via the incubation with the magnetic beads, the cells are easily identified with several beads attached to them (Figure 2) at low confluency, where single cells are shown 24 h after isolation, (Figure 2A–C) and at high confluency, 7 days after (Figure 2D). Low-magnification images from the mixed cell population isolated from the mouse dermis are shown (Figure 2E).
Indicative flow cytometry data are presented from the optional analysis to identify the percentage of LYVE-1+ macrophages in the isolated lymphatic endothelial cell population. Figure 3 shows the flow cytometry gating strategy, with 5% of the selected LYVE-1+ cells being positive for F4/80 (macrophages). This percentage is increased from 3% of the F4/80+ cells in the isolated (non-purified) murine skin cells (not shown). The endogenous red fluorescence of the transgenic mice allows complementary monitoring via fluorescence. As the cells proliferate, the density of the magnetic beads attached is decreased, and an optional purification step against other lymphatic markers such as VEGFR3 or podoplanin14 can provide a more purified lymphatic endothelial cell population.
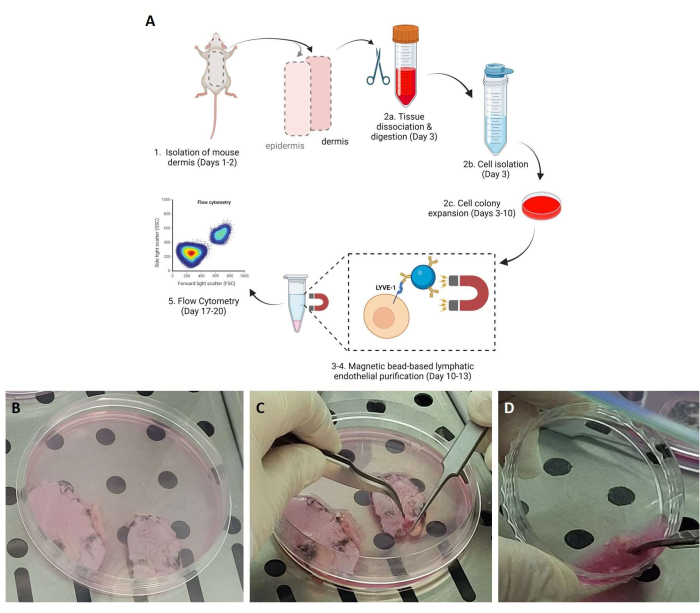
Figure 1: Demonstration of different steps of the tissue and cell isolation process. (A) Graphic scheme of the steps of the protocol. (B) The excised skin is placed in Tissue Culture Plates with Dispase solution for overnight incubation at 4 °C. (C) The detachment of the epidermis from the underlying dermis the following day. (D) Tissue processing of the dermis for the separation and isolation of the tissue cells. Please click here to view a larger version of this figure.
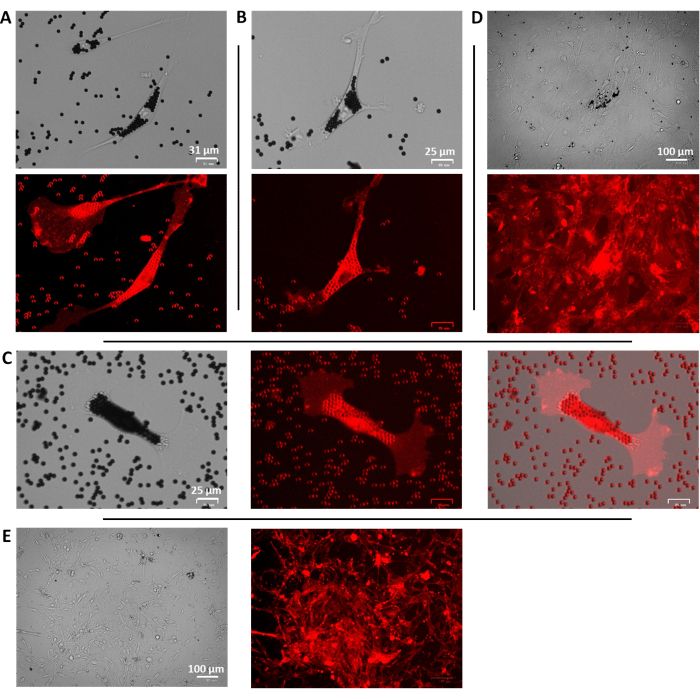
Figure 2: Representative images of the purified LYVE-1+ cells. (A,B) Brightfield (top) and fluorescent (bottom) images of individual LYVE-1+ isolated cells, 24 h after purification, with the beads attached; scale bars = 31 and 25 µm. (C) Brightfield (left), fluorescent (center), and merged (right) images of a representative cell 24 h after purification; scale bar = 25 µm. (D) Brightfield (top) and fluorescent (bottom) images of the isolated cells upon proliferation and with high confluency 7 days after purification; scale bar = 100 µm. (E) Brightfield (left) and fluorescent (center) low magnification images of mixed cell population 5 days after isolation from the dermis; scale bar = 100 µm. Please click here to view a larger version of this figure.
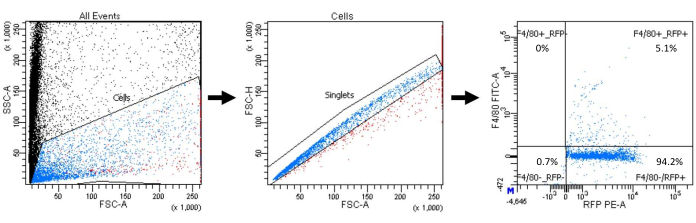
Figure 3: Flow cytometry gating strategy and data. Calculation of the percentage of macrophage population within the selected LYVE-1+ cells (blue: single cells (singlets); red: cell clusters). Abbreviations: FSC-A = forward scatter-peak area; SSC-A = side scatter-peak area; FSC-H = forward scatter-peak height; RFP = red fluorescent protein; FITC = fluorescein isothiocyanate; PE = phycoerythrin. Please click here to view a larger version of this figure.
Discussion
The lymphatic system is an important regulator of the homeostatic function of the body, with the most important functions being the maintenance of fluid plasma, removal of cellular metabolism byproducts and toxic molecules, lipid absorption, and immune cell trafficking1,19. The identification of appropriate markers has provoked a burst in new data in the lymphatics field, revealing novel functions of the lymphatic vasculature, such as their role in the repair and regenerative ability of certain organs2,3. The increasing knowledge about the lymphatic system delineates its role in various pathological conditions, altering the conventional view that was previously established1. The lymphatic system plays an important role in the outcome of many diseases, such as cancer growth and metastasis20, Milroy disease, a form of primary lymphedema21,22, and Kaposiform Lymphangiomatosis, a rare condition characterized by expansion and interconnection of the lymphatic vessels23,24. The above facts highlight the role of the lymphatic system in disease pathology and strengthen the need to further understand the molecular regulation of this system.
As part of this process, the in vitro evaluation of the physiology of the lymphatic endothelial cells can reveal functional deficiencies, characteristic of certain pathological conditions, and can elucidate the molecular mechanisms underlying these conditions. Several protocols for the isolation of lymphatic endothelial cells have been published, most of which use fluorescence-activated cell sorting7,25. The presented method is an efficient method for the isolation of lymphatic endothelial cells without fluorescence-activated cell sorting but is inferior in terms of purity of the lymphatic endothelial cell population from fluorescence-activated cell sorting techniques, which is the major limitation of magnetic bead-based techniques. For this, the method can be useful for crude identification of lymphatic endothelial functions or evaluation of a protein deficiency in conditionally-deficient mice18 but is not recommended for RNA sequencing applications. Fluorescence-activated cell sorting-based protocols offer the advantage of higher purity of the selected cells, since they are selected by co-staining of more than one lymphatic-specific marker, and they are faster as the several days-long step of cell growth prior to magnetic bead purification is omitted. Another significant advantage of the fluorescence-activated cell sorting-based protocols is that magnetic beads are not required, rendering them more cost-efficient. The disadvantage of the magnetic bead-based isolation methods is the decreased yield obtained, but where fluorescence-activated cell sorting equipment is present, it is a preferred purification method.
We previously identified that the contamination of lymphatic endothelial cells isolated from the skin of adult mice with LYVE-1+ immune cells was less than 10%18, with the current example in Figure 3 showing a ~5% contamination of LYVE-1+ macrophages. LYVE-1 is a classic lymphatic marker and has been successfully combined with CD31 and CD34 for magnetic bead-based purification of blood and lymphatic endothelial cells26. The LYVE-1 and CD31 combination provides a lymphatic endothelial cell population with high purity. Alternative magnetic bead-based selection has been reported with the combination of UEA-1 and a D2-40 antibody27. Magnetic bead-based technique for skin lymphatic endothelial cell isolation, via exclusion (negative selection) of the targeting cells and involvement of more purification steps, was successfully performed in mouse embryos28, and if applicable in adult mice, it can definitely serve for isolation of pure lymphatic endothelial populations for relevant downstream applications. The negative selection, however, has the limitation that not all the cells that bind to the beads can be selected, as we have witnessed also non-selected LYVE-1+ positive cells, that are bound with fewer beads and thus not sufficiently bound to the magnet for selection.
A similar magnetic bead-based isolation technique, for the isolation of lymphatic endothelial cells from the lung of adult mice, is based on podoplanin-based selection of the lymphatic endothelial cells14. The structure of the protocols is very similar, with the major difference being the target; however, we have not performed a comparative analysis to identify differences in yield and purity. It is known that different lymphatic subpopulations may vary9,27,29. Under this spectrum, it is important to emphasize that not all dermal lymphatic endothelial cells are LYVE-1+. There are differences in LYVE-1 expression in the same tissue, in inflamed tissues, such as tumors, with LYVE-1 playing a role in CD8+ T cell infiltration29 or in the lymphatic capillaries versus the lymphatic collecting vessels9. This leads to the difference in this method, being specific for lymphatic endothelial cell isolation of a subtype of lymphatic endothelial cells, mostly from the lymphatic capillaries and not the collecting vessels9. Thus, the presented method is applicable for phenomena mainly regulated at the capillaries but also limits the pool of lymphatic endothelial cells that can be isolated.
A limitation of magnetic bead-based methods is the optional evaluation of purity by a flow cytometry experiment. We previously identified that the contamination of lymphatic endothelial cells isolated from the skin of adult mice with LYVE-1+ immune cells was less than 10%18, with the current example in Figure 3 confirming this finding, demonstrating a ~5% contamination of LYVE-1+ macrophages. Flow cytometry experiment with F4/80 staining of the initial isolated cell population (not shown) revealed the presence of ~3% macrophages, which includes also the LYVE-1- macrophages. Thus, the increased percentage in the bead-selected cell population suggests an efficient purification method. Given that the percentage of macrophages in the skin of healthy mice is lower than when tumor or inflammation is present17, this increases the reliability of the method in healthy mice. Of note, there are non-selected cells that have one or more magnetic beads attached (not shown), verifying that for the cells to be bead-selected they must have many beads attached (Figure 2) and that some cells with lower levels of LYVE-1 expression (that have beads are attached) are not selected.
For the successful application of the protocol, it is critical to follow the timing of the incubation of the lymphatic endothelial cells with the LYVE-1 antibody-conjugated magnetic beads, as increased incubation can lead to non-specific binding. Another critical factor is the timing of the mincing of the skin, to ensure the survival of as many cells as possible. Lymphatic endothelial cells are not the dominating cellular population; therefore, after purification, a limited number of cells is expected to be obtained. In terms of the number of mice that can be processed at once, we have processed up to four mice without any noticeable differences in the yield between the first and last mouse. The protocol can be applied in both sexes and different ages, although the age of the adult mice is a determining factor of the replicative potential of the endothelial cells. Similar to human endothelial cells30, increased mouse age leads to increased senescence31,32 and thus, the limited replicative potential of the isolated cells. Higher yield is expected to be provided for young (<3 months of age) mice for the C57BL6 background, which we have used and is the mouse background in this study. Overall, the protocol presents an efficient method for murine dermal lymphatic endothelial cell isolation, which can be used for applications where the absolute purity of the isolated lymphatic endothelial cell population is not prioritized: the purified cells can be used for in vitro experiments or for validation of deficiency of the gene of interest in lymphatic endothelial-specific mutant mice. This can be particularly useful for transgenic mice or experimental animal models, where the fast and crude purification of the lymphatic endothelial cells is the priority and in Institutes and Facilities where fluorescence-activated cell sorter is not readily available.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Hellenic Foundation for Research and Innovation (00376), the National Institutes of Health, the National Cancer Institute (NCI) [Grant R15CA231339], the Texas Tech University Health Sciences Center (TTUHSC) School of Pharmacy Office of the Sciences grant (to C.M.M.), and by the College of Pharmacy, University of Louisiana Monroe start-up funding, the National Institutes of Health (NIH) through the National Institute of General Medical Science [Grant P20 GM103424-21] and the Research Competitiveness Subprogram (RCS) of the Louisiana Board of Regents through the Board of Regents Support Fund (LEQSF(2021-24)-RD-A-23) (to G.M.). The common TTUHSC equipment used was obtained through the Cancer Prevention Research Institute of Texas (CPRIT) Grants RP190524 and RP200572. The funders had no role in the study design, the decision to write, or preparation of the manuscript. The graphical abstract in Figure 1A was created with BioRender.com.
Materials
| 0.2 μm Syringe Filters | Fisher Scientific | 09-719C | |
| 100 mm Tissue Culture Dishes | Fisher Scientific | FB012924 | |
| 60 mm Tissue Culture Dishes | Fisher Scientific | FB012921 | |
| Animal Hair Clipper | Wahl | ||
| Antibiotic-Antimycotic Solution 100x | Fisher Scientific | 15240-062 | |
| Blunt Forceps | Fine Science Tools | 11992-20 | |
| Bovine Serum Albumin | Sigma-Aldrich | A4919 | |
| Cell Strainer 40 μm | Fisher Scientific | 542040 | |
| Cell Strainer 70 μm | Fisher Scientific | 542070 | |
| CO2 Incubator | |||
| Collagen I, High Concentration Rat Tail | Corning | 354249 | dilute in 0.02 M Acetic Acid in H2O |
| Collagenase Type II | Life Technologies | 17101-015 | |
| Dispase | Life Technologies | 17105-041 | |
| DMEM | Fisher Scientific | 11-995-073 | |
| DNAse I Solution (2,500 U/mL) | Thermo Scientific | 90083 | |
| Dynal MPC-L Magnetic Particle Concentrator | Invitrogen | 120-21D | |
| EDTA | Sigma-Aldrich | 3690 | |
| Endothelial Cell Growth Base Medium & Supplement (LEC medium) | R&D Systems | CCM027 | |
| Euthanasia chamber | Euthanex Corporation | ||
| Fine Forceps | Fine Science Tools | 11255-20 | |
| Fine Scissors-Sharp | Fine Science Tools | 14060-10 | |
| FITC anti-mouse F4/80 Antibody | Biolegend | 123107 | |
| Goat Anti-Rabbit IgG Magnetic Beads | New England Biolabs | S1432S | |
| LARC-A E-Z Anesthesia Induction Chamber | Euthanex Corporation | ||
| MagnaBind Goat Anti-Rabbit IgG Beads | Thermo Scientific | 21356 | |
| Paraformaldehyde Solution 4% in PBS | Fisher Scientific | AAJ19943K2 | |
| Phosphate Buffered Saline (PBS) | Fisher Scientific | SH30256FS | |
| Rabbit Anti-Mouse LYVE-1 | ReliaTech GmbH | 103-PA50 | |
| Rotating/Shaking Incubator | |||
| Round-Bottom Polypropylene Tubes | Corning | 352063 | |
| Syringe Filters w 0.2 μm Pores | Fisher Scientific | 09-719C | |
| Trypsin-EDTA | Fisher Scientific | 25-300-120 |
References
- Oliver, G., Kipnis, J., Randolph, G. J., Harvey, N. L. The lymphatic vasculature in the 21(st) century: novel functional roles in homeostasis and disease. Cell. 182 (2), 270-296 (2020).
- Liu, X., et al. Lymphoangiocrine signals promote cardiac growth and repair. Nature. 588 (7839), 705-711 (2020).
- Biswas, L., et al. Lymphatic vessels in bone support regeneration after injury. Cell. 186 (2), 382-397 (2023).
- Tammela, T., Alitalo, K. Lymphangiogenesis: molecular mechanisms and future promise. Cell. 140 (4), 460-476 (2010).
- Ji, R. C. The role of lymphangiogenesis in cardiovascular diseases and heart transplantation. Heart Failure Reviews. 27 (5), 1837-1856 (2022).
- Patnam, M., et al. Lymphangiogenesis guidance mechanisms and therapeutic implications in pathological states of the cornea. Cells. 12 (2), 319 (2023).
- Lokmic, Z., et al. Isolation of human lymphatic endothelial cells by multi-parameter fluorescence-activated cell sorting. Journal of Visualized Experiments: JoVE. (99), e52691 (2015).
- Jackson, D. G. Biology of the lymphatic marker LYVE-1 and applications in research into lymphatic trafficking and lymphangiogenesis. APMIS: Acta Pathologica, Microbiologica, et Immunologica Scandinavica. 112 (7-8), 526-538 (2004).
- Makinen, T., et al. PDZ interaction site in ephrinB2 is required for the remodeling of lymphatic vasculature. Genes and Development. 19 (3), 397-410 (2005).
- Wolters, G. H., Vos-Scheperkeuter, G. H., Lin, H. C., van Schilfgaarde, R. Different roles of class I and class II Clostridium histolyticum collagenase in rat pancreatic islet isolation. Diabetes. 44 (2), 227-233 (1995).
- Kubo, A., Aoki, S., Fujita, H. Whole-mount preparation and microscopic analysis of epidermis. Current Protocols. 2 (7), e464 (2022).
- Thiele, W., Rothley, M., Schmaus, A., Plaumann, D., Sleeman, J. Flow cytometry-based isolation of dermal lymphatic endothelial cells from newborn rats. Lymphology. 47 (4), 177-186 (2014).
- Kazenwadel, J., Michael, M. Z., Harvey, N. L. Prox1 expression is negatively regulated by miR-181 in endothelial cells. Blood. 116 (13), 2395-2401 (2010).
- Lapinski, P. E., King, P. D. Isolation and culture of mouse lymphatic endothelial cells from lung tissue. Methods in Molecular Biology. 2319, 69-75 (2021).
- Choi, E. Y., et al. Del-1, an endogenous leukocyte-endothelial adhesion inhibitor, limits inflammatory cell recruitment. Science. 322 (5904), 1101-1104 (2008).
- Mikelis, C. M., et al. RhoA and ROCK mediate histamine-induced vascular leakage and anaphylactic shock. Nature Communications. 6, 6725 (2015).
- Schledzewski, K., et al. Lymphatic endothelium-specific hyaluronan receptor LYVE-1 is expressed by stabilin-1+, F4/80+, CD11b+ macrophages in malignant tumours and wound healing tissue in vivo and in bone marrow cultures in vitro: implications for the assessment of lymphangiogenesis. Journal of Pathology. 209 (1), 67-77 (2006).
- Akwii, R. G., et al. Angiopoietin-2-induced lymphatic endothelial cell migration drives lymphangiogenesis via the beta1 integrin-RhoA-formin axis. Angiogenesis. 25 (3), 373-396 (2022).
- Petrova, T. V., Koh, G. Y. Biological functions of lymphatic vessels. Science. 369 (6500), eaax4063 (2020).
- Fidler, I. J. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nature Reviews. Cancer. 3 (6), 453-458 (2003).
- Mellor, R. H., et al. Lymphatic dysfunction, not aplasia, underlies Milroy disease. Microcirculation. 17 (4), 281-296 (2010).
- Connell, F., Brice, G., Mortimer, P. Phenotypic characterization of primary lymphedema. Annals of the New York Academy of Sciences. 1131, 140-146 (2008).
- Croteau, S. E., et al. Kaposiform lymphangiomatosis: a distinct aggressive lymphatic anomaly. Journal of Pediatris. 164 (2), 383-388 (2014).
- Ji, Y., Chen, S., Peng, S., Xia, C., Li, L. Kaposiform lymphangiomatosis and kaposiform hemangioendothelioma: similarities and differences. Orphanet Journal of Rare Diseases. 14 (1), 165 (2019).
- Kriehuber, E., et al. Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages. Journal of Experimental Medicine. 194 (6), 797-808 (2001).
- Podgrabinska, S., et al. Molecular characterization of lymphatic endothelial cells. Proceedings of the National Academy of Sciences of the United States of America. 99 (25), 16069-16074 (2002).
- Garrafa, E., et al. Isolation, purification, and heterogeneity of human lymphatic endothelial cells from different tissues. Lymphology. 38 (4), 159-166 (2005).
- Kazenwadel, J., Secker, G. A., Betterman, K. L., Harvey, N. L. In vitro assays using primary embryonic mouse lymphatic endothelial cells uncover key roles for FGFR1 signalling in lymphangiogenesis. PLoS One. 7 (7), e40497 (2012).
- Steele, M. M., et al. T cell egress via lymphatic vessels is tuned by antigen encounter and limits tumor control. Nature Immunology. 24 (4), 664-675 (2023).
- Mammoto, T., et al. Effects of age-dependent changes in cell size on endothelial cell proliferation and senescence through YAP1. Aging. 11 (17), 7051-7069 (2019).
- Regina, C., et al. Vascular ageing and endothelial cell senescence: Molecular mechanisms of physiology and diseases. Mechanisms of Ageing and Development. 159, 14-21 (2016).
- Muhleder, S., Fernandez-Chacon, M., Garcia-Gonzalez, I., Benedito, R. Endothelial sprouting, proliferation, or senescence: tipping the balance from physiology to pathology. Cellular and Molecular Life Sciences: CMLS. 78 (4), 1329-1354 (2021).

