Multi-Photon Time Lapse Imaging to Visualize Development in Real-time: Visualization of Migrating Neural Crest Cells in Zebrafish Embryos
Summary
A combination of the advanced optical techniques of laser scanning microscopy with long wavelength multi-photon fluorescence excitation was implemented to capture high-resolution, three-dimensional, real-time imaging of neural crest migration in Tg(sox10:EGFP) and Tg(foxd3:GFP) zebrafish embryos.
Abstract
Congenital eye and craniofacial anomalies reflect disruptions in the neural crest, a transient population of migratory stem cells that give rise to numerous cell types throughout the body. Understanding the biology of the neural crest has been limited, reflecting a lack of genetically tractable models that can be studied in vivo and in real-time. Zebrafish is a particularly important developmental model for studying migratory cell populations, such as the neural crest. To examine neural crest migration into the developing eye, a combination of the advanced optical techniques of laser scanning microscopy with long wavelength multi-photon fluorescence excitation was implemented to capture high-resolution, three-dimensional, real-time videos of the developing eye in transgenic zebrafish embryos, namely Tg(sox10:EGFP) and Tg(foxd3:GFP), as sox10 and foxd3 have been shown in numerous animal models to regulate early neural crest differentiation and likely represent markers for neural crest cells. Multi-photon time-lapse imaging was used to discern the behavior and migratory patterns of two neural crest cell populations contributing to early eye development. This protocol provides information for generating time-lapse videos during zebrafish neural crest migration, as an example, and can be further applied to visualize the early development of many structures in the zebrafish and other model organisms.
Introduction
Congenital eye diseases can cause childhood blindness and are often due to abnormalities of the cranial neural crest. Neural crest cells are transient stem cells that arise from the neural tube and form numerous tissues throughout the body.1,2,3,4,5 Neural crest cells, derived from the prosencephalon and mesencephalon, give rise to the bone and cartilage of the midface and frontal regions, and the iris, cornea, trabecular meshwork, and sclera in the anterior segment of the eye.4,6,7,8 Neural crest cells from the rhombencephalon form the pharyngeal arches, jaw, and cardiac outflow tract.1,3,4,9,10 Studies have highlighted the contributions of the neural crest to ocular and periocular development, emphasizing the importance of these cells in vertebrate eye development. Indeed, disruption of neural crest cell migration and differentiation lead to craniofacial and ocular anomalies as observed in Axenfeld-Rieger Syndrome and Peters Plus Syndrome.11,12,13,14,15,16,17 Thus, a comprehensive understanding of the migration, proliferation and differentiation of these neural crest cells will provide insight into the complexities underlying congenital eye diseases.
The zebrafish is a powerful model organism for studying ocular development, as the structures of the zebrafish eye are similar to their mammalian counterparts, and many genes are evolutionarily conserved between zebrafish and mammals.18,19,20 In addition, zebrafish embryos are transparent and oviparous, facilitating the visualization of eye development in real-time.
Expanding on previously published work,6,7,20 the migratory pattern of neural crest cells was described using multi-photon fluorescence time-lapse imaging on transgenic zebrafish lines labeled with green fluorescent protein (GFP) under the transcriptional control of the SRY (sex-determining region Y)-box 10 (sox10) or Forkhead Box D3 (foxd3) gene regulatory regions.21,22,23,24. Multi-photon fluorescence time-lapse imaging is a powerful technique that combines the advanced optical techniques of laser scanning microscopy with long wavelength multi-photon fluorescence excitation to capture high-resolution, three-dimensional images of specimens tagged with fluorophores.25,26,27 The use of the multi-photon laser has distinct advantages over standard confocal microscopy, including increased tissue penetration and decreased fluorophore bleaching.
Using this method, two distinct populations of neural crest cells varying in timing of migration and migratory pathways were discriminated, namely foxd3-positive neural crest cells in the periocular mesenchyme and developing eye and sox10-positive neural crest cells in the craniofacial mesenchyme. With this method, an approach to visualize the migration of ocular and craniofacial neural crest migration in zebrafish is introduced, making it easy to observe regulated neural crest migration in real time during development.
This protocol provides information for generating time-lapse videos during early eye development in Tg(sox10:EGFP) and Tg(foxd3:GFP) transgenic zebrafish, as an example. This protocol can be further applied for the high-resolution, three-dimensional, real-time visualization of the early development of any ocular and craniofacial structure derived from neural crest cells in zebrafish. Moreover, this method can further be applied for the visualization of the development of other tissues and organs in zebrafish and other animal models.
Protocol
The protocol described here was performed in accordance with the guidelines for the humane treatment of laboratory animals established by the University of Michigan Committee on the Use and Care of Animals (UCUCA).
1. Embryo Collection for Time-lapse Imaging
- Between 3 and 9 pm, set up male and female adult Tg(sox10:EGFP) or Tg(foxd3:GFP) transgenic zebrafish in a divided breeding tank for pairwise mating.
NOTE: The Tg(sox10:EGFP) and Tg(foxd3:GFP) fish, kind gifts from Thomas Schilling and Mary Halloran, respectively, were crossed into the Casper (roy -/-, nacre -/-) background to decrease auto-fluorescence and interference with pigmentation.- Assemble the breeding tank (slotted inner tank + solid outer tank) and fill it with reverse osmosis (RO) system water.
- Transfer up to 3 females and 3 males into opposing sides of the tank separated by a divider; up to 6 fish can be bred pairwise in a single breeding tank.
- The next morning, remove the divider shortly after the lights turn on in the vivarium.
NOTE: In the present study, the lights are on a 14-hour light (on at 9 am) and 10-hour dark (off at 11 pm) cycle. - Allow undisturbed mating for 20 min or until sufficient numbers of embryos are produced; the embryos will be at the bottom of the breeding tank. When eggs are observed, lift the slotted inner tank out along with the fish and quickly place them (fish and inner tank) into a clean solid outer tank filled with RO water.
- Prepare standard 1X embryo medium by adding the following per 1 L of RO water: 0.287 g (4.9 mM) NaCl, 0.127 g (0.17 mM) KCl, 0.048 g (0.33 mM) CaCl2.2H2O, 0.04 g (0.33 mM) MgSO4 (anhydrous). Stir solution until the salts are completely dissolved.
- Add 100 µL of 0.1% methylene blue as a fungicide. Add 0.25 g of sodium bicarbonate to adjust the pH to 7.75. Store medium at room temperature.
- Collect eggs from the outer tank using an egg-collecting screen, and transfer the eggs to Petri dishes (~50 embryos per dish) containing 30 mL of 1X embryo medium. Incubate the collected eggs at 28.5 °C.
- Embryo preparation
- Tg(sox10:EGFP) embryos.
- For Tg(sox10:EGFP) embryos, at 12 h post-fertilization (hpf), remove the dead eggs and assess the developmental stage of the embryos. Screen for GFP-positive embryos using a dissecting fluorescent stereomicroscope with a standard 460-490 nm band-pass excitation filter. Using forceps, remove the chorions from 3-4 embryos that express GFP, and transfer these embryos to a separate Petri dish containing 30 mL of fresh 1X embryo medium.
- Tg(foxd3:GFP) embryos.
- For Tg(foxd3:GFP) embryos, at 22-24 hpf, remove the dead eggs and assess the developmental stage of the embryos. Screen for positive embryos using a dissecting fluorescent stereomicroscope with a standard 460-490 nm band-pass excitation filter. Using forceps, remove the chorions from 3-4 embryos that express GFP.
- Prepare 1000X Phenylthiourea (PTU) stock solution by dissolving 0.75 g (3%) of PTU in 25 mL of Dimethylsulfoxide (DMSO). Aliquot and store the stock solution at 4 °C. Add 30 µL of 1000X PTU (3%) stock solution to 30 mL of 1X embryo medium to generate 0.003% PTU solution.
- Place the screened and dechorionated GFP-positive embryos in 0.003% PTU solution to inhibit pigmentation. Do not initiate treatment of the embryos with PTU prior to 20 hpf, as early treatment (at <20 hpf) can have adverse effects on neural crest and neuroepithelial development.28
- Prior to conducting the time-lapse experiment, monitor the embryos through live imaging using a standard stereomicroscope at 12-24 h intervals in age-matched comparisons as a control for temperature drift.
- Tg(sox10:EGFP) embryos.
2. Mounting of Embryo for Time-lapse Imaging
- Preparation of time-lapse embryo media
- Prepare standard 0.4% tricaine stock solution by adding 0.004 g of tricaine to 100 mL of RO water. Aliquot (~1 mL) the stock solution into fresh 1.5-mL centrifuge tubes and store at -20 °C.
- Prepare time-lapse embryo medium (50 mL of 1X embryo medium, 0.016% tricaine) by adding 2 mL of 0.4% tricaine solution to 48 mL of 1X embryo medium. If using embryos >22 hpf, then also add 50 µL of 3% PTU stock solution (final concentration.003% PTU) to the embryo medium.
- Preparation of 2% low-melt agarose
- Add 0.4 g of low-melt agarose powder to 20 mL of 1X embryo medium. Heat for 1-2 min or until the solution is clear and all particles are dissolved.
NOTE: Increasing the percentage of the agarose gel decreases the porosity of the matrix, which may physically impede the growth and development of the embryo. Therefore, the agarose solution can be lowered to 1.5% to minimize these effects. However, when the excitation beam is held stationary during laser-scanning microscopy, greater heating can occur, increasing rapidly in a logarithmic relationship with time. In this protocol, heating due to fluorophore absorption is highly localized to the focal region. Thus, in the region of interest, the temperature could increase to a value high enough (≥ 30 °C) to melt agarose solutions made at percentages lower than 1.5, thereby directly exposing the embryo to the laser or enabling conditions in which the embryo floats out of the focal plane. For this reason, the use of agarose solutions lower than 1.5% is not recommended. - Aliquot (~1 mL) the agarose solution into fresh 1.5-mL centrifuge tubes for storage. Store excess agarose solution in liquid form on a heat block (60-70 °C) for 2-3 weeks.
- Add 0.4 g of low-melt agarose powder to 20 mL of 1X embryo medium. Heat for 1-2 min or until the solution is clear and all particles are dissolved.
- Mounting the Embryo
- To set up the open bath chamber, place a small amount of high vacuum grease on the base of the open bath chamber. Place a circular glass coverslip onto the base and screw the top of the open bath chamber onto the base until tight ( Figure 1A, B).
- Pipet (~500-700 µL) of 2% agarose solution (60-70 °C) into the open bath chamber until the base is ~3/4 filled ( Figure 1C). Note that once the agarose solution is in the open bath chamber on the lab bench, the temperature of the solution decreases approximately 1 °C/s.
- Wait 30 s to allow the agarose to cool slightly (~30-40 °C), without completely polymerizing, and subsequently transfer a single embryo to the center of the base ( Figure 1D, E).
- Under a fluorescent stereomicroscope, position the embryo using a 1-10 µL micropipette tip, making sure that the embryo is placed near the bottom of the agarose, as the embryo may float away when covered with embryo media if it is placed too near the surface of the agarose.
NOTE: In the presented videos (Videos 1-3), the embryos are oriented laterally, but depending on the area of interest, the embryos can be oriented ventrally or dorsally. - Monitor and reposition the embryo until the agarose has set. Once the agarose has set, use a transfer pipet to fill the assembled open chamber entirely with time-lapse embryo media ( Figure 1F).
- Place the open bath chamber in the quick exchange platform, which fits onto the stage adapter ( Figure 1G). Place the entire setup (embryo, open bath chamber, quick exchange platform and stage adaptor) onto the stage of the microscope immediately above the condenser ( Figure 1H).
3. Microscope Set-up for Time-lapse Imaging
- Determining laser settings
- Determining laser wavelength to use. Use a wavelength that is twice the excitation wavelength of the fluorophore of interest (e.g. wavelength between 880 and 940 nm for GFP).
NOTE: In the present study, the wavelength setting for GFP was between 880 and 940 nm. The higher the wavelength, the lower the output power of the laser. - Determine laser transmission. A high percent of laser transmission will kill the embryo, use the lowest level of transmission (recommended). For 24 to 48 h time-lapse studies, as presented herein, keep transmission below 5%.
- Determine microscope detection systems and ensure that the correct filters for the fluorophore are in place.
NOTE: For multi-photon microscopes, there are multiple detection systems with various sensitivities for the emitted fluorescence. In general, an internal detection system has less sensitivity than an external detection system. For transgenic lines with high levels of GFP expression, the internal detection system is adequate. For transgenic lines with low levels of GFP expression or with other fluorophores (e.g., red fluorescent protein), an external detection system may be required to maintain the percent of laser transmission at a reasonable level. Regardless the detection system used, the correct filters for the fluorophore must be in place.
- Determining laser wavelength to use. Use a wavelength that is twice the excitation wavelength of the fluorophore of interest (e.g. wavelength between 880 and 940 nm for GFP).
- Adjusting software settings on the multi-photon microscope
- Using the 5X objective, locate the embryo. Manually raise the stage to the highest position, and use the fine focus to position the embryo in the middle of the microscope range.
- Manually lower the stage, and change the 5X objective to the 25X water immersion objective (numerical aperture NA, 0.95). Carefully raise the stage to bring the embryo back into focus.
- In the software, click on the "xyzt" mode for obtaining multiple images at time intervals (t) in the x-y plane over a depth of "z". Use the epifluorescence or brightfield view to find the depth of focus in the area of interest, which will demarcate the Z-stack.
- In the software, click on "begin" button; for these experiments, the lateral edge of the eye was the beginning of the Z-stack. Click on "end" button; the midline of the embryo was the end of the Z-stack.
NOTE: The step size was 0.3-0.6 µm and there was a total of ~200 steps for a z-stack size of 60 to 120 µm).
- In the software, click on "begin" button; for these experiments, the lateral edge of the eye was the beginning of the Z-stack. Click on "end" button; the midline of the embryo was the end of the Z-stack.
- Click on the menu for adjusting acquisition time and imaging frequency. For the present system, ~200 steps requires approximately 5 min for each z-stack acquisition. For adequate recovery of the fluorophore and survival of the embryo, allow for a ratio of at least 1:3 between z-stack acquisition (laser power on) and recovery time (laser power off).
NOTE: For example, z-stacks are acquired every 20 min with 5 min of z-stack acquisition and 15 min of recovery. For this protocol, larger z-stacks can be obtained, but would appropriately increase the time between z-stack acquisitions, resulting in fewer images over the time-lapse course.- With appropriate time for embryo recovery, set the time between z-stacks in the designated window. Set the total length of time for the experiment in the appropriate window.
- Final software, laser, and embryo adjustments.
- Turn on the live image setting to make final adjustments to the laser settings. Adjust laser transmission (see 3.1.2), gain and offset slider bars within the software to optimize the fluorescent image. Also, adjust the orientation of the embryo, as needed, depending on the length of the experiment, anticipated growth of the embryo, etc. Make sure that the area of interest remains within the frame through the duration of the experiment.
- Turn off the epifluorescent light source as it is no longer needed during time-lapse acquisition. Cover the stage with the laser safety box ( Figure 1I). When using the internal detection system, the laser safety box is adequate for protection against background light. Press "start".
NOTE: However, with more sensitive external detection systems, the laser safety box does not block enough background light, and additional covers are required to prevent the disruption of image acquisition.
4. During Time-lapse Acquisition
- Refill the open bath chamber with time-lapse embryo media every 8-12 h (at least 2 times per day) during time-lapse acquisition through the sliding doors on the laser safety box ( Figure 1I).
- Before opening the doors of the laser safety box, ensure that the microscope is not actively acquiring an image.
NOTE: The use of heaters and circulating media systems is not necessary for time-lapse imaging experiments lasting 24 to 48 h. Indeed, the temperatures of both stage and in-line heaters are difficult to control, and during image acquisition the embryo exhibits an adequate development rate at a temperature range from 25-28 °C. Moreover, circulating media systems tend to overflow and potentially damage the equipment. Thus, all embryos are routinely staged post-acquisition.
5. Post-acquisition processing
- In the software, click on the "file" menu and choose "save".
- Open the file in image processing software (see the Table of Materials). Highlight the correct image series. In the software, choose the "Process" menu. Click on 3D Deconvolution and "Apply" to deconvolve each z-stack.
NOTE: The file is large; therefore, this step may take many hours. - In the software, under the "Process" menu, click on "maximum projection." Click on "Apply" to initiate maximum projection to generate 1 image per z-stack. Export each maximum projected file (1 image per z-stack) as a tiff.
- Import individual tiff files into video processing software. Select all tiff files and drag them into the video editor. Adjust length of each image within the video to 0.1s. Export video as mov or mp4 file.
Representative Results
Multi-photon fluorescence time-lapse imaging generated a series of videos that revealed the migration patterns of cranial neural crest cells that give rise to the craniofacial structures and anterior segment of the eye in the Tg(sox10:EGFP) and Tg(foxd3:GFP) zebrafish lines. As an example, sox10-positive neural crest cells between 12 and 30 hpf migrate from the edge of the neural tube into the craniofacial region (Video 1, Figure 2). The cells from the prosencephalon and mesencephalon migrate dorsal and ventral to the developing eye to populate the periocular mesenchyme. In addition, these sox10-positive cells form the frontonasal process. Neural crest cells from the rhombencephalon migrate ventrally and give rise to the pharyngeal arches. Time-lapse imaging of foxd3-positive neural crest cells between 30 and 60 hpf showed that these cells migrated between the optic cup and surface ectoderm and through the ocular fissure (Video 2, Figure 3). These foxd3-positive cells coalesced around the lens, forming the iris stroma.

Figure 1.Setup. A. Each component of the embryo mounting apparatus, including the open bath chamber, quick exchange platform and stage adapter. B. Assembly of the open bath chamber. C. Addition of 2% agarose solution (60-70 °C) to the open bath chamber. D. Placement of the embryo in the open bath chamber under a fluorescent microscope. E. Embryo (at 24 hpf) positioned laterally in the center of the polymerized agarose solution in the open bath chamber. F. Addition of media to the surface of the polymerized agarose solution to completely cover the embryo in the open bath chamber. G. Open bath chamber placed in the quick exchange platform and positioned in the stage adapter. H. Placement of the embryo mounting apparatus on the stage of the multi-photon microscope. I. Laser safety box. The sliding doors (1 and 2) for refilling the open chamber are indicated. Please click here to view a larger version of this figure.
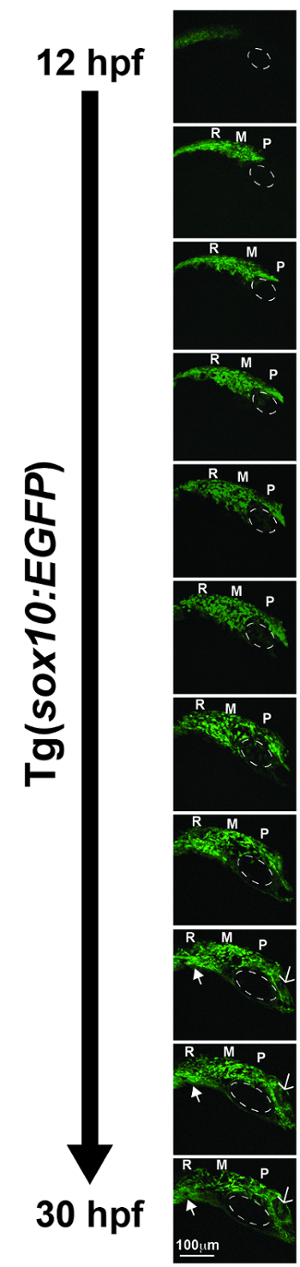
Figure 2. Representative deconvolved and max-projected images from multi-photon time-lapse imaging of a Tg(sox10:EGFP) zebrafish embryo from 12 to 30 hpf. The sox10-positive cells migrated from the prosencephalon (P) and mesencephalon (M) to populate the periocular mesenchyme (the dotted circle denotes the eye) and frontonasal process (open arrow). Sox10-positive cells from the rhombencephalon (R) migrated ventrally to form the pharyngeal arches (closed arrow). Images were obtained every 20 min during the time frame and sewn together to create Video 1. Please click here to view a larger version of this figure.

Figure 3. Representative deconvolved and max-projected images from multi-photon time-lapse imaging of a Tg(foxd3:GFP) zebrafish embryo from 24 to 48 hpf. Foxd3-positive cells entered into the anterior chamber of the eye (asterisk denotes lens) between the surface ectoderm and optic cup, with more cells localized to the dorsal ("D")-posterior ("P") quadrant compared with the anterior ("A") and ventral ("V") quadrants. In addition, foxd3-positive cells migrated adjacent to and through the ocular fissure. Images were obtained every 20 min during the time frame and sewn together to create Video 2. Please click here to view a larger version of this figure.
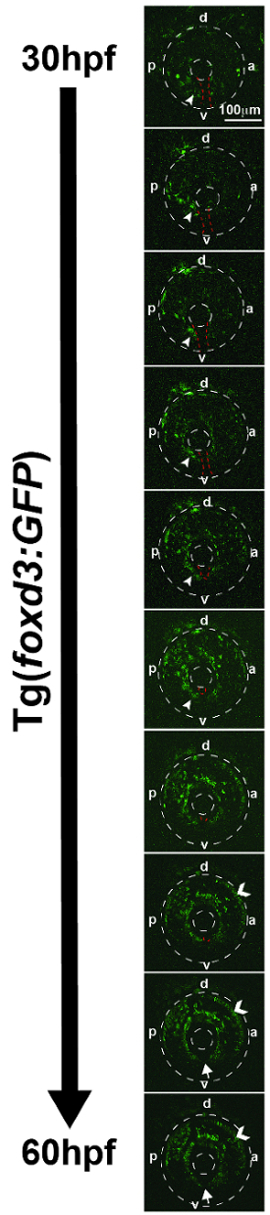
Figure 4. Representative deconvolved and max-projected images from multi-photon time-lapse imaging of a Tg(foxd3:GFP) zebrafish embryo from 30 to 60 hpf. Foxd3-positive cells entered into the anterior chamber of the eye (outer dotted circle denotes peripheral edges of the eye, inner dotted circle denotes lens) between the surface ectoderm and optic cup, with more cells localized to the dorsal ("d")-posterior ("p") quadrant compared with the anterior ("a") and ventral ("v") quadrants. In addition, foxd3-positive cells migrated adjacent to and through the ocular fissure (white arrowhead denotes migrating neural crest, red dashed line demarcates ocular fissure). At 60 hpf, foxd3-positive cells completely encircled the lens (closed arrows), indicating the closure of the fissure. In addition, at 60 hpf, foxd3 was also expressed in photoreceptors (open arrowhead), which was not associated with the expression of this transcription factor in neural crest cells. Images were obtained every 20 min during the time frame and sewn together to create Video 3. Please click here to view a larger version of this figure.
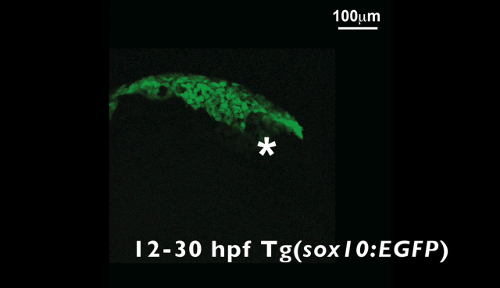
Video 1. Time-lapse video of a Tg(sox10:EGFP) zebrafish embryo from 12 to 30 hpf. The asterisk denotes the eye. Please click here to download this video.
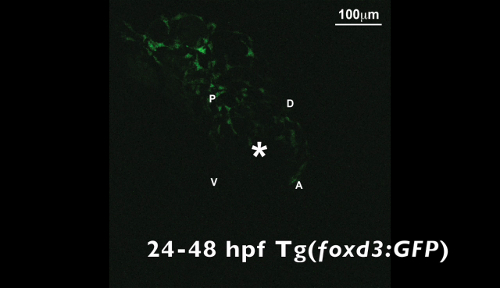
Video 2. Time-lapse video of a Tg(foxd3:GFP) zebrafish embryo from 24 to 48 hpf. The asterisk denotes the lens. The asterisk denotes the ocular fissure. d, dorsal; v, ventral; p, posterior; a, anterior. Please click here to download this video.
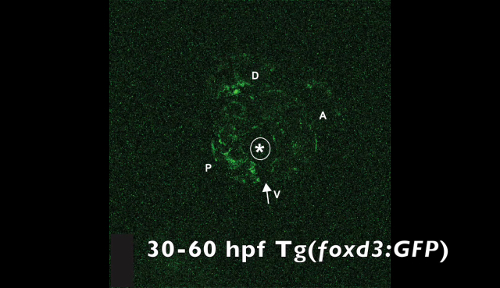
Video 3. Time-lapse video of a Tg(foxd3:GFP) zebrafish embryo from 30 to 60 hpf. The asterisk denotes the lens. The arrow denotes the ocular fissure. d, dorsal; v, ventral; p, posterior; a, anterior. Please click here to download this video.
Discussion
Multi-photon time-lapse imaging enables the in vivo tracking of transient and migratory cell populations. This powerful technique can be used to study embryonic processes in real time, and in the present study, the results of this method enhanced the current knowledge of neural crest cell migration and development. Previous time-lapse imaging studies typically utilize confocal laser scanning microscopy.29,30,31,32 Here, we present the use of a multi-photon technique, which has many advantages over traditional confocal microscopy. The basis of multi-photon microscopy is that two photons of longer infrared wavelengths are used to excite the fluorophore. As a result, there is less scattering and thus decreased background, enabling deeper tissue penetration, achieving imaging depths exceeding 1 mm in biological tissue with more efficient detection than confocal microscopy. These properties also decrease phototoxicity, which is an important consideration with repeated and frequent image acquisition.25,26,27 From these properties, two-photon microscopy can image whole-organ preparations and tissue in small-animal models, (e.g. mice, zebrafish embryos at 12 hpf – as in the case of the present study). Further, these small animal models enable use of genetically encoded fluorescent proteins, which can be localized in the tissue of interest. Thus, the use of multi-photon microscopy can greatly enhance image acquisition for time-lapse experiments.
There are numerous steps in this protocol, which depend on the type of multi-photon laser and detection system being used. Most problems with this protocol arise with the laser settings and acquisition of images based on the detection system used. A working knowledge of the individual system and access to technical support are critical. In addition, previous experience with confocal laser scanning microscopy is helpful for troubleshooting. Moreover, the initial system set up may be time consuming. However, once the system set up is completed, only small adjustments are typically required when using the same transgenic lines.
In the present study, zebrafish embryos between 12 and 60 hpf were used for these experiments. During this age range, the embryos are small, easily embedded in agarose and readily anesthetized with tricaine. However, the embryos can become growth and developmentally delayed depending on the length of the experiment. Therefore, care must be taken to ensure that the embryo is appropriately staged at the end of the experiment.
Multi-photon time-lapse imaging yields high-resolution, three-dimensional, real-time visualization of cell migration, which not only enhances the capability to study in vivo zebrafish embryogenesis, but can also be applied to many other systems. Although the outlined protocol focuses on zebrafish neural crest cell migration, this technique can easily be adapted for other imaging purposes.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
The authors thank Thomas Schilling for kindly gifting the Tg(sox10:eGFP) fish and Mary Halloran for kindly gifting the Tg(foxd3:GFP) fish.
Materials
| Breeding Tanks with Dividers | Aquaneering | ZHCT100 | Crossing Tank Set (1.0-liter) Clear Polycarbonate with Lid and Insert |
| M205 FA Combi-Scope | Leica Microsystems CMS GmbH | Stereofluorescence Microscope – FusionOptics and TripleBeam | |
| Sodium Chloride | Millipore (EMD) | 7760-5KG | Double PE sack. CAS No. 7647-14-5, EC Number 231-598-3 |
| Potassium Chloride | Millipore (EMD) | 1049380500 | Potassium chloride 99.999 Suprapur. CAS No. 7447-40-7, EC Number 231-211-8. |
| Calcium Chloride Dihydrate | Fisher Scientific | C79-500 | Poly bottle; 500 g. CAS No. 10035-04-8 |
| Magnesium Sulfate (Anhydrous) | Millipore (EMD) | MX0075-1 | Poly bottle; 500 g. CAS No. 7487-88-9, EC Number 231-298-2 |
| Methylene Blue | Millipore (EMD) | 284-12 | Glass bottle; 25 g. Powder, Certified Biological Stain |
| Sodium Bicarbonate | Millipore (EMD) | SX0320-1 | Poly bottle; 500 g. Powder, GR ACS. CAS No. 144-55-8, EC Number 205-633-8 |
| N-Phenylthiourea | Sigma | P7629-25G | >98%. CAS Number 103-85-5, EC Number 203-151-2 |
| Dimethylsulfoxide | Sigma | D8418-500ML | Molecular Biology grade. CAS Number 67-68-5, EC Number 200-664-3 |
| Tricaine Methanesulfonate | Western Chemical Inc. | MS222 | Tricaine-S |
| Low-Melt Agarose | ISC Bioexpress | E-3112-25 | GeneMate Sieve GQA Low Melt Agarose, 25 g |
| Open Bath Chamber | Warner Instruments | RC-40HP | High Profile |
| Glass Coverslips | Fisher Scientific | 12-545-102 | Circle cover glass. 25 mm diameter |
| High Vacuum Grease | Fisher Scientific | 14-635-5C | 2.0-lb. tube. DOW CORNING CORPORATION 1658832 |
| Quick Exchange Platform | Warner Instruments | QE-1 | 35 mm |
| Stage Adapter | Warner Instruments | SA-20LZ-AL | 16.5 x 10 cm |
| TC SP5 MP multi-photon microscope | Leica Microsystems CMS GmbH | ||
| Mai Tai DeepSee Ti-Sapphire Laser | SpectraPhysics | ||
| Laser Safety Box | Leica Microsystems CMS GmbH | ||
| Leica Application Suite X (LAS X) Software | Leica Microsystems CMS GmbH | ||
| Photoshop CS 6 Version 13.0 x64 Software | Adobe | ||
| iMovie Version 10.1.4 Software | Apple |
Referenzen
- Barembaum, M., Bronner-Fraser, M. Early steps in neural crest specification. Sem Cell Dev Biol. 16, 642-646 (2005).
- Gage, P. J., Rhoades, W., Prucka, S. K., Hjalt, T. Fate maps of neural crest and mesoderm in the mammalian eye. Invest Ophthalmol Vis Sci. 46 (11), 4200-4208 (2005).
- Minoux, M., Rijli, F. M. Molecular mechanisms of cranial neural crest cell migration and patterning in craniofacial development. Development. 137, 2605-2621 (2010).
- Trainor, P. A. Specification of neural crest cell formation and migration in mouse embryos. Sem Cell Dev Biol. 16, 683-693 (2005).
- Johnston, M. C., Noden, D. M., Hazelton, R. D., Coulombre, J. L., Coulombre, A. Origins of avian ocular and periocular tissues. Exp Eye Res. 29, 27-43 (1979).
- Bohnsack, B. L., Kahana, A. Thyroid hormone and retinoic acid interact to regulate zebrafish craniofacial neural crest development. Dev Biol. 373, 300-309 (2013).
- Chawla, B., Schley, E., Williams, A. L., Bohnsack, B. L. Retinoic acid and pitx2 regulate early neural crest survival and migration in craniofacial and ocular development. Birth Defects Res B Dev Reprod Toxicol. , (2016).
- Trainor, P. A., Tam, P. P. L. Cranial paraxial mesoderm and neural crest cells of the mouse embryo-codistribution in the craniofacial mesenchyme but distinct segregation in branchial arches. Development. 121 (8), 2569-2582 (1995).
- Hong, C. S., Saint-Jeannet, J. P. Sox proteins and neural crest development. Sem Cell Dev Biol. 16, 694-703 (2005).
- Steventon, B., Carona-Fontaine, C., Mayor, R. Genetic network during neural crest induction: from cell specification to cell survival. Sem Cell Dev Biol. 16, 647-654 (2005).
- Dressler, S., et al. Dental and craniofacial anomalies associated wth Axenfeld-Rieger syndrome with PITX2 mutation. Case Report Med. , 621984 (2010).
- Ozeki, H., Shirai, S., Ikeda, K., Ogura, Y. Anomalies associated with Axenfeld-Rieger syndrome. Graefes Arch Clin Exp Ophthalmol. 237 (9), 730-734 (1999).
- Strungaru, M. H., Dinu, I., Walter, M. A. Genotype-phenotype correlations in Axenfeld-Rieger malformation and glaucoma patients with FOXC1 and PITX2 mutations. Invest Ophthalmol Vis Sci. 48, 228-237 (2007).
- Tumer, Z., Bach-Holm, D. Axenfeld-Rieger syndrome and spectrum of Pitx2 and Foxc1 mutations. Eur J Hum Genet. 17, 1527-1539 (2009).
- Schoner, K., et al. Hydrocephalus, agenesis of the corpus callosum, and cleft lip/palate represent frequent associations in fetuses with Peters plus syndrome and B3GALTL mutations. Fetal PPS phenotypes, expanded by Dandy Walker cyst and encephalocele. Prenat Diagn. 33 (1), 75-80 (2013).
- Aliferis, K., et al. A novel nonsense B3GALTL mutation confirms Peters plus syndrome in a patient with multiple malformations and Peters anomaly. Ophthalmic Genet. 31 (4), 205-208 (2010).
- Lesnik Oberstein, ., S, A., et al. Peters Plus syndrome is caused by mutations in B3GALTL, a putative glycosyltransferase. Am J Hum Genet. 79 (3), 562-566 (2006).
- Brittijn, S. A., et al. Zebrafish development and regeneration: new tools for biomedical research. Int J Dev Biol. 53, 835-850 (2009).
- Bohnsack, B. L., Kasprick, D., Kish, P. E., Goldman, D., Kahana, A. A zebrafish model of Axenfeld-Rieger Syndrome reveals that pitx2 regulation by retinoic acid is essential for ocular and craniofacial development. Invest Ophthalmol Vis Sci. 53 (1), 7-22 (2012).
- Williams, A. L., Eason, J., Chawla, B., Bohnsack, B. L. Cyp1b1 regulates ocular fissure closure through a retinoic acid-independent pathway. Invest Ophthalmol Vis Sci. 58 (2), 1084-1097 (2017).
- Curran, K., Raible, D. W., Lister, J. A. Foxd3 controls melanophore specification in the zebrafish neual crest by regulation of Mitf. Dev Biol. 332 (2), 408-417 (2009).
- Dutton, K., Dutton, J. R., Pauliny, A., Kelsh, R. N. A morpholino phenocopy of the colourless mutant. Genesis. 30 (3), 188-189 (2001).
- Dutton, K. A., et al. Zebrafish colourless encodes sox10 and specifies non-ectomesenchymal neural crest fates. Development. 128 (21), 4113-4125 (2001).
- Kucenas, S., Takada, N., Park, H. C., Woodruff, E., Broadie, K., Appel, B. CNS-derived glia ensheath peripheral nerves and mediate motor root development. Nat Neurosci. 11, 143-151 (2008).
- Denk, W., Strickler, J., Webb, W. Two-photon laser scanning fluorescence microscopy. Science. 248 (4951), 73-76 (1990).
- Helmchen, F., Denk, W. Deep tissue two-photon microscopy. Nat Methods. 2 (12), 932-940 (2005).
- Denk, W., Delaney, K. Anatomical and functional imaging of neurons using 2-photon laser scanning microscopy. J Neurosci Methods. 54 (2), 151-162 (1994).
- Bohnsack, B. L., Gallina, D., Kahana, A. Phenothiourea sensitizes zebrafish cranial neural crest and extraocular muscle developmnt to changes in retinoic acid and insulin-like growth factor signaling. PLoS ONE. 6, e22991 (2011).
- Gfrerer, L., Dougherty, M., Laio, E. C. Visualization of craniofacial development in the sox10:kaede transgenic zebrafish line using time-lapse confocal microscopy. J Vis Exp. (79), e50525 (2013).
- Lopez, A. L. R., Garcai, M. D., Dickinson, M. E., Larina, I. V. Live confocal microscopy of the developing mouse embryonic yolk sac vasculature. Methods Mol Biol. 1214, 163-172 (2015).
- McGurk, P. D., Lovely, C. B., Eberhart, J. K. Analyzing craniofacial morphogenesis in zebrafish using 4D confocal microscopy. J Vis Exp. (83), e51190 (2014).
- Nowotschin, S., Ferrer-Vaquer, A., Hadjantonakis, A. K. Imaging mouse development with confocal time-lapse microscopy. Methods Enzymol. 476, 351-377 (2010).

