Modeling Brain Tumors In Vivo Using Electroporation-Based Delivery of Plasmid DNA Representing Patient Mutation Signatures
Summary
Utilizing an immunocompetent, autochthonous tumor model driven by common patient mutations for preclinical testing is critical for immunotherapeutic testing. This protocol describes a method to generate brain tumor mouse models using electroporation-based delivery of plasmid DNA that represent common patient mutations, thus providing an accurate, reproducible, and consistent mouse model.
Abstract
Tumor models are critical for the preclinical testing of brain tumors in terms of exploring new, more efficacious treatments. With significant interest in immunotherapy, it is even more critical to have a consistent, clinically pertinent, immunocompetent mouse model to examine the tumor and immune cell populations in the brain and their response to treatment. While most preclinical models utilize orthotopic transplantation of established tumor cell lines, the modeling system presented here allows for a “personalized” representation of patient-specific tumor mutations in a gradual, yet effective development from DNA constructs inserted into dividing neural precursor cells (NPCs) in vivo. DNA constructs feature the mosaic analysis with the dual-recombinase-mediated cassette exchange (MADR) method, allowing for single-copy, somatic mutagenesis of driver mutations. Using newborn mouse pups between birth and 3 days old, NPCs are targeted by taking advantage of these dividing cells lining the lateral ventricles. Microinjection of DNA plasmids (e.g., MADR-derived, transposons, CRISPR-directed sgRNA) into the ventricles is followed by electroporation using paddles that surround the rostral region of the head. Upon electrical stimulation, the DNA is taken up into the dividing cells, with the potential of integrating into the genome. The use of this method has successfully been demonstrated in developing both pediatric and adult brain tumors, including the most common malignant brain tumor, glioblastoma. This article discusses and demonstrates the different steps of developing a brain tumor model using this technique, including the procedure of anesthetizing young mouse pups, to microinjection of the plasmid mix, followed by electroporation. With this autochthonous, immunocompetent mouse model, researchers will have the ability to expand preclinical modeling approaches, in efforts to improve and examine efficacious cancer treatment.
Introduction
Murine brain tumor models are crucial for understanding the mechanisms of brain tumor formation and treatment. Current models typically include rapidly produced subcutaneous or orthotopic transplantations of commonly used tumor cell lines, based on a limited number of driver mutations or patient-derived xenograft models, using immunodeficient mice that hinder proper immunotherapy studies1,2,3,4. Additionally, these preclinical results can lead to false positives, in that such models can exhibit dramatic, oftentimes curative effects in response to therapy, but this does not translate to the clinic2,5,6,7. Having the ability to rapidly produce genetically engineered preclinical mouse models that are more reflective of patient mutation signatures is imperative for improving the validity of preclinical results.
Electroporation (EP)-based delivery of DNA plasmids to induce both loss of function (LOF) and gain of function (GOF) mutations allows for the generation of such models. We developed a method for an even more precise representation of GOF driver mutations called mosaic analysis with dual-recombinase-mediated cassette exchange, or MADR8. This method allows for the expression of a gene (or genes) of interest in a controlled, locus-specific manner in somatic cells8. In combination with other molecular tools, such as clustered regularly interspaced short palindromic repeats (CRISPR), different patient mutations can be combined to develop mouse brain tumor models. This method has been used for different pediatric brain tumors, including gliomas and ependymomas8, as well as adult brain tumor models, such as glioblastoma (GBM).
While the EP method of tumor modeling is not as common as a transplant, the following demonstrates heretofore the ease and high reproducibility of this modeling system. mTmG mice are used for the insertion of the MADR-plasmid DNA8,9. This system allows for the recombination of loxP and Flp recombinase target (FRT) sites located at the Rosa26 locus for subsequent insertion of the donor DNA plasmid (i.e., GOF gene of interest)8,9. The following protocol demonstrates the straightforwardness of this method after diligent practice, and the ability to develop mouse brain tumor models in an autochthonous, consistent manner.
Protocol
All procedures in this protocol were approved by the Cedars Sinai Medical Center Institutional Animal Care and Use Committee (IACUC). Homozygous mTmG mice were bred with C57BL/6J mice to obtain litters of mixed-sex, heterozygous mTmG mice for use in the following protocol. The animals were obtained from a commercial source (see Table of Materials). Mouse pups were electroporated between postnatal days 0 and 3 (P0-P3).
1. Surgical setup
- Spray down the surgical table with chemical disinfectant (e.g., Vimoba) and wipe off, followed by 70% ethanol, and wipe off again.
- Place the following instruments/materials on the surgical table: pulled glass capillary pipettes (see reference10 on how to pull pipettes), small scissors, small biohazard sharps waste container, microliter pipettor and pipette tips, 2x cut square pieces of paraffin film, electrode gel, electrodes, holder for the microinjector, and DNA plasmid mix (see Table of Materials and Supplementary File 1).
- Place the following attachments onto the equipment on the surgical table or on the floor when indicated: microinjector control panel, holder, and plug attached to the machine, stand for holding the microinjector holder to the side (soldering aid), microinjector foot pedal (on the floor), electrodes attached to the machine, and electrode foot pedal (on the floor) (see Table of Materials).
NOTE: The electrode foot pedal was placed on the right of the microinjector foot pedal as a reminder of the order of use. - Turn on the microinjector and electrode control panels.
- Check the settings on the microinjector are correct for the experiment: the pressure must be between 220-450 hPa, depending on how the plasmid mixture is taken up.
- Check the settings on the electrode panel are correct for the experiment.
NOTE: For the current experiment (and most of our brain tumor models), five pulses of 120 V (50 ms; separated by 950 ms) are used.
2. Pre-surgical preparation
- Prepare the plasmid mix in the microinjector.
- Retrieve one pulled glass capillary pipette and insert it into the microinjector holder. Ensure the tip is screwed on to hold in place.
- Hold the microinjector holder in one hand and the small scissors with the other; have the tip of the pipette over a small sharps biohazard container. With the scissors closed, press down lightly on the lengthened glass capillary pipette. Cut where the bend is seen, approximately 2 mm from the opening. If the cut looks successful, place carefully to the side.
- Place the commercially available plasmid mix tube flat on the surgical table and open. Have the pipette tip box or another item behind the bottom of the tube to keep it stable while drawing up the plasmid mix. Place the glass pipette that is attached to the microinjector line flat on the table and insert gently into the tube of the plasmid mix, making sure the tip is well in the mix near the bottom of the tube.
NOTE: Due to natural gravity suction, the plasmid mix suctioning will begin in the glass pipette before actively drawing in. - With the tip of the glass pipette in the plasmid mix, use the microinjector panel to increase the amount being drawn in. Starting at 0, turn the right knob and dial in the negative direction toward -60-this will bring the plasmid mix into the glass pipette. Only bring in ~1 inch of plasmid mix. Turn the knob back to 0, and then remove the pipette tip from the mix.
NOTE: Do not remove the pipette tip from the mix while in the negative direction, as this will shoot the mix into the microinjector and possibly into the tubing. - Test the amount of mix in one injection by injecting the mix onto one strip of paraffin film. Use the foot pedal and press once to inject the mix onto the paraffin film. Use the pipettor set at 1 µL to take up the mix on the paraffin film to see if it is exactly 1 µL.
NOTE: If it is slightly less than 1 µL, the pressure of the microinjector can be increased. If the pressure is approaching 450 hPa and still less than 1 µL, the glass pipette opening is too small and will need to be trimmed more. It is suggested to place the plasmid mix back into the tube before trimming. Despite this, there will still be a residual mix on the scissors, so it is imperative to thoroughly clean the scissors afterward with DNase to remove any remaining mix. If the injection is more than 1 µL, a new pulled glass pipette will need to be used and trimmed. When the test amount is exactly 1 µL, the glass pipette tip is at the right size. - Repeat step 2.1.4 to draw in additional plasmid mix for the procedure, or about 2-3 inches into the pulled glass pipette. Ensure the mixture does not go too far into the microinjector where one cannot see the end.
NOTE: When drawing in the plasmid mix, it is critical to avoid air bubbles. If bubbles are present, it is necessary to redo this step by removing the plasmid mix from the pulled glass pipette and starting again. Air bubbles present will be detrimental to the following steps when injecting into the ventricle of the pup. - With the plasmid mix ready in the pulled glass pipette, place the readied pipette to the side on the soldering aid stand and out of the way so as not to accidentally touch/bump it.
- Prepare the animals for the experiment.
- Prepare an ice bucket for anesthetizing pups. Add water to the ice bucket to melt and help to cool the anesthetizing holder (e.g., pipette box top).
- Place the box top onto the melted ice to cool.
- Carefully remove the pup(s) from the cage and place them into the cooled box top, which remains on the ice. Place another top loosely over the bottom to help the cooling process.
- After 2-3 min, check on the pups. Separate the pups from each other while remaining in the prone position.
NOTE: The pups will wriggle, but this will slow down as the cold anesthetization kicks in. The box top can be left off for easy viewing of the pups. - To check for proper anesthesia, pinch the tail of the pup and look for a reaction. If there is a reaction, repeat step 2.2.4. If no squeal or little movement is made, the pup is ready for the procedure.
NOTE: Mice should not be kept on ice for extended periods of time as this will affect their ability to recover after the procedure. Only the number of pups that can be both injected and electroporated while still under anesthesia must be put on ice at a time; greater numbers can be done with experience.
3. Microinjection of DNA plasmid mix into the brain ventricle
- Place a pup on the table in the ventral position.
- Pull back slightly on the back of the head to visualize the skull sutures through the skin. Find lambda on the skull. The ventricle/injection site will be midway between lambda and the eye.
NOTE: Lambda is where the sagittal and lambdoid sutures intersect. See reference11 for the identification of lambda on the mouse skull. - Pierce the skin with the glass pipette tip at a perpendicular angle (the pipette is straight up toward to ceiling). Observe a slight concave indentation when pressing on the skull and an initial resistance. Once pierced and the glass pipette tip pokes through the concave indentation, the appropriate injection level is reached.
- Hold the glass pipette tip in place and press the microinjector foot pedal once to inject 1 µL of the plasmid mix.
NOTE: There are two ways to clearly see the injection was successful. First, have another lab member watch the plasmid mix go down in the glass pipette as it is injected. Also, if using fast green or another dye in the plasmid mix, the dye will visibly spread out in the ventricle under the skin once injected and is easily visible when held up to a surgical lamp.
4. Electroporation
- Place a piece of paraffin film (one square) onto the table and squeeze out the electrode gel on top of the film. Cover the electrode paddles in electrode gel by brushing a generous amount onto each paddle. Any excess gel that is dripping off can be swiped off on a paper towel.
NOTE: After the first EP, do not let the gel from each paddle touch the other side. This will transfer the charge and, when applying to the pup's head, a sizzling sound may be heard. This will also happen if the gel on the pup's head from one electrode comes into contact with the gel from another.
CAUTION: Care must be taken to keep the fingers away from the paddles during the electroporation. - Hold the pup's body in one hand (typically the non-dominant hand), keeping the fingers well behind the head.
NOTE: If right-handed, it is easiest to hold the pup with the left hand and electrodes with the right. If left-handed, do the opposite. - Bring the electrodes close to the pup's head to ensure they are in the proper position, with the positive electrode on the outside of the head where the ventricle is being targeted (e.g., if targeting the left ventricle, place the positive on the pup's left side and negative on the pup's right side). While positioning the electrodes, gently slide a finger (typically the thumb) on the dorsal side of the body/neck posterior to the head to help raise the head into position.
- Check the depth of anesthesia by pinching the tail and proceed with electroporation only if the pup is in the appropriate plane of anesthesia. Lightly squeeze the electrode paddles on the rostral part of the mouse head, placing them over the eyes. Press the electrode foot pedal once to start the electroporation. This protocol uses five pulses of 120 V (50 ms, separated by 950 ms); each pulse will be audible.
- With each pulse, rotate the electrode paddles slightly in a counter-clockwise direction so the positive paddle is moving toward the top of the head.
NOTE: With proper electroporation, one will feel/see the mouse pup's body twitch with each pulse.
- With each pulse, rotate the electrode paddles slightly in a counter-clockwise direction so the positive paddle is moving toward the top of the head.
- After the final pulse, remove the paddles and place to the side. Move the pup to a clean paper towel and have another lab member clean the gel off the pup's head and place under a heat lamp on a paper towel "boat".
NOTE: Ensure the heat lamp is not too close to the pups. Make sure to have another lab member help with this step and keep a constant eye on the pups. It often helps to hold the pups in one's hand under the heat lamp to increase the heat for the pup. Gently massaging the abdomen of the pup can also help with recovery. - Return the pups to the home cage once their bodies have a healthy pinkish color and have a squeaking response to a light tail pinch.
NOTE: Before placing them back in the cage, take a small amount of the home cage bedding material and lightly brush the pups with it for the cage scent. Place the pups back in the cage under the bedding material and return to the holding room.
5. Post-surgical steps
- Return the unused plasmid mix from the microinjector glass pipette back to the tube. If there is enough mixture left, this can be used in future procedures. Store at -20 °C.
- Wipe the gel off the electrode paddles with paper towels.
- Return all equipment parts back to their original place.
NOTE: If scissors were used to cut the glass pipette tip after the plasmid mix was taken up, leave the scissors out to thoroughly clean with DNase solution. - Spray down the table with a chemical disinfectant, then wipe off, followed by a spray down of 70% ethanol, then wipe off again.
Representative Results
The protocol described above has been used to successfully develop both pediatric and adult brain tumor mouse models, with the former published in extensive detail in Kim et al.8. With proper technique and careful planning of plasmid design, the success for EP development of tumors is typically 100%. Histology is the quickest and easiest way to check for successful DNA plasmid insertion when a reporter protein is used. This protocol involves steps on how to develop a GBM brain tumor model with 100% penetrance, as confirmed by histological analysis. An MADR donor plasmid expressed both a reporter protein (smTagBFP2-V5) and SpCas9 (Supplementary File 1). Two strong driver LOF tumor suppressor gene single-guide RNAs (sgRNAs) were also included, directed at Nf1 and Trp53 (see reference12 for sgRNA cloning strategy; see Supplementary File 1 for oligonucleotide sequences used in this protocol). Finally, the Cre and Flp recombinase plasmids were added to the plasmid mix for recombination at the Rosa26 locus (Figure 1A; see Supplementary File 1). Mice were moribund by 5 months post-EP, with full tumor growth detected through the reporter protein and histological analysis (Figure 1B).
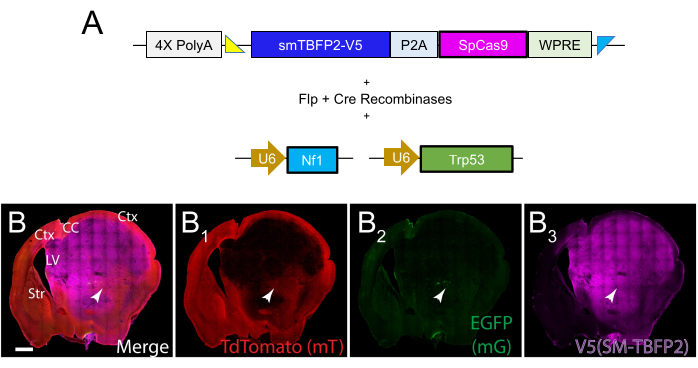
Figure 1: Immunofluorescent staining of successful tumor growth at 5 months post-EP. (A) MADR donor plasmid for spCas9 and the reporter protein TagBFP2-V5. Also included in the plasmid mix was a Cre-Flp recombinase plasmid, along with sgRNAs directed at Nf1 and Trp53. (B) A coronal section taken at 5 months post-EP of a GBM tumor driven by Nf1 and Trp53 LOF. Tumor cells are labeled with the TagBFP-V5 reporter protein linked to spCas9 (B3). Sparse EGFP labeling is also detected (B2) along with all cells that did not express the plasmids labeled with tdTomato (B1). Scale bar = 1,000 µm. Abbreviations: Ctx = cortex; CC = corpus callosum; LV = lateral ventricle; Str = striatum. Please click here to view a larger version of this figure.
Supplementary File 1: Plasmid sequences. Complete sequences of all plasmids and oligonucleotides used in this protocol, including pDonor-SM-TagBFP2-V5-P2A-spCas9-WPRE, pCag-FlpO-2A-Cre, and the oligonucleotides for sgRNA targeting Trp53 and Nf1. Please click here to download this File.
Discussion
Electroporation-based delivery of plasmid DNA allows for the in vivo use of molecular biology, similar to that used in genetically engineered mouse models, but with the speed, localization, and efficiency of viral transduction8,13,14. With the latter, however, comes safety concerns as well as immune responses. We have shown in our modeling system using EP-delivery of plasmid DNA that minimal immune response occurs due to the initial insertion of the glass capillary pipette into the brain ventricle8. Therefore, to improve upon the current tumor modeling systems for immunotherapy, the protocol presented above allows for a more expedient and robust preclinical modeling system for brain tumors.
The first crucial step for successful brain tumor modeling using this method is plasmid design. While not described in this protocol, it has been extensively discussed in Kim et al.8 and Rincon Fernandez Pacheco et al.10 for pediatric brain tumor models, as well as other sources for additional uses15,16. While our lab has successfully electroporated up to six plasmids, there will be an upper limit to how much DNA can be delivered. This upper limit is constrained by plasmid DNA concentration, plasmid sizes, voltage, and the volume of DNA delivered, which is itself limited by the size of the brain ventricle at the age of electroporation. In addition, with multiple DNA plasmids in the mix, it is imperative to have a well-suspended plasmid mix prior to injection. If the mix is not resuspended well, it will be difficult to take up in the narrow glass capillary pipette. Also, with the proper plasmid mix, the addition of fast green dye (10% v/v) allows for clear viewing of the plasmid mix taken up in the brain ventricle during administration (step 3.4). One important caveat to this, however, is that the dye can interfere with antibody staining in the far-red wavelengths (e.g., Cy5) of tissue processed within several weeks after EP8. Although the fast green dye is very helpful when first learning EP, it can be eliminated to avoid false-positive staining. It is worth keeping in mind that, for step 3.4, the delivery of the plasmid mix in the ventricle will not be visible, so it will be crucial to watch the glass pipette for the volume to decrease as it is injected.
For consistency and proper handling of injections, there are several factors to keep in mind: 1) the injection volume. Though it may take several attempts to cut the glass capillary pipette accurately (step 2.1.2), it is crucial that only 1 µL of plasmid mix is injected for each animal. Increases or decreases in the volume between animals will certainly affect the latency of tumor formation. 2) once the injection volume is set, the parameters of the microinjector must not be changed, as this will affect the volume. 3) once the plasmid mix is brought up in the glass capillary pipette, the pipette should be kept out of the way so as to not accidentally poke oneself or others. Using a micropipette stand (soldering aid; see Table of Materials) that clips onto the microinjector holder is useful for this. However, if the plasmid mix is not injected soon after, it will likely clog the tip. Therefore, it is important to do one test microinjection on a piece of parafilm immediately prior to injecting into the brain ventricle to make sure there is no clog blocking the orifice. Finally, similar to every other surgical technique, the execution of the protocol is different for each individual. It is therefore, imperative to have the same technician performing the microinjection and/or EP for all groups of mice in an experiment.
The brain tumor models developed in our lab have focused on gliomas. From a neurobiological perspective, performing this method between postnatal days 0-3 targets the gliogenesis period and the period post-embryonic neurogenesis. Interest in alternative brain tumors may require adjustment in the time point and/or position of electrodes. Several protocols are available for in utero EP that would be pertinent for neural-based tumors15,17,18.
There are a few limitations to this method. The ability to clone and mix and match both LOF and GOF DNA plasmids using CRISPR, transposons, and, more recently, the MADR system allows for endless combinations of patient mutations to be modeled. The techniques of microinjection and EP are relatively simple, though they do take a few practice attempts to improve and become consistent. One limitation of the model presented is the length of time it takes for the GBM tumor to fully develop (5 months). Despite this, we are currently working on additional models using common patient tumor mutations that lead to aggressive tumor growth lasting less than 2 months. In addition to limitations, the equipment itself is a substantial initial investment (around $20,000-$30,000); however, the ability to electroporate tens if not hundreds of mice in one sitting allows for incredible power for a preclinical experiment. With that being said, if there are issues with mouse breeding, this can become the biggest limitation, as it is imperative to have healthy breeders and healthy litters.
It has been several decades since advancements in brain tumor treatments have occurred, with the last greatest improvement of the chemotherapeutic Temozolomide extending survival by only a few months19. Immunotherapy has revolutionized many different cancers, but is yet to make a significant impact on the standard of care in neuro-oncology. Interestingly, currently used mouse brain tumor models have shown great success with immunotherapy, but continuously fail to translate to the clinic and show success with patients. It is therefore imperative to take a step back and re-evaluate the mouse tumor models used. Current models depend on using older cell lines with limited mutations not commonly found in patients while injecting thousands of cells into the brain. With the technique discussed in this protocol, different patient tumor mutation profiles in a mouse model are recapitulated, allowing for an autochthonous, gradual development of tumors within a time frame that would allow for preclinical testing in immunocompetent mice.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
We thank Gi Bum Kim for the immunofluorescent staining and images. We also thank Emily Hatanaka, Naomi Kobritz, and Paul Linesch for helpful advice on the protocol.
Materials
| 0.1-2.5 µL 1-channel pipette | Eppendorf | 3123000012 | |
| 2 µL pipette tips | Fisher Scientific | 02-707-442 | |
| 20 µL pipette tips | Fisher Scientific | 02-707-432 | |
| 2-20 µL 1-channel pipette | Eppendorf | 3123000098 | |
| DNAZap PCR DNA Degradation Solutions | Fisher Scientific | AM9890 | |
| ECM 830 Square Porator Electroporator | BTX | 45-0662 | |
| Electrode Gel | Parker Labs | PLI152CSZ | |
| Fast Green Dye | Sigma-Aldrich | F7258-25G | |
| Helping Hands Soldering Aid | Pro'sKit | 900-015 | |
| Micro Dissecting Scissors, 4.5" Straight Sharp | Roboz | RS-5916 | |
| Mouse Strain: C57BL/6J | The Jackson Laboratory | JAX: 000664 | |
| Mouse Strain: Gt(ROSA)26Sortm4(ACTB-tdTomato,-EGFP)Luo/J | The Jackson Laboratory | JAX: 007676 | |
| Parafilm | Grainger | 16Y894 | |
| Plasmid: pCag-FlpO-2A-Cre EV | Addgene | 129419 | |
| Platinum Tweezertrode, 7 mm Diameter | BTX | 45-0488 | |
| Sharps container, 1-quart | Uline | S-15307 | |
| Standard Glass Capillaries, 4 in, 1 mm OD, 0.58 mm ID | World Precision Instruments | 1B100F-4 | Capillary pipettes need to be pulled – see reference 10 for details. |
| Vertical Micropipette Puller | Sutter Instruments | P-30 | Heat settings: Heat #1 at 880, Heat #2 at 680; pull at 800. See reference 10 for more details on pulling. |
| Vimoba Tablet Solution | Quip Laboratories | VIMTAB | |
| XenoWorks Digital Microinjector | Sutter Instruments | BRE | |
| XenoWorks Micropipette Holder | Sutter Instruments | BR-MH |
Referenzen
- Brabetz, S., et al. A biobank of patient-derived pediatric brain tumor models. Nature Medicine. 24 (11), 1752-1761 (2018).
- Hadad, A. F., et al. Mouse models of glioblastoma for the evaluation of novel therapeutic strategies. Neuro-Oncology Advances. 3 (1), (2021).
- He, C., et al. Patient-derived models recapitulate heterogeneity of molecular signatures and drug response in pediatric high-grade glioma. Nature Communications. 12 (1), 4089 (2021).
- Szatmári, T., et al. Detailed characterization of the mouse glioma 261 tumor model for experimental glioblastoma therapy. Cancer Science. 97 (6), 546-553 (2006).
- Genoud, V., et al. Responsiveness to anti-PD-1 and anti-CTLA-4 immune checkpoint blockade in SB28 and GL261 mouse glioma models. Oncoimmunology. 7 (12), 1501137 (2018).
- Reardon, D. A., et al. Effect of Nivolumab vs Bevacizumab in patients with recurrent glioblastoma: the CheckMate 143 phase 3 randomized clinical trial. JAMA Oncology. 6 (7), 1003-1010 (2020).
- Wainwright, D. A., et al. Durable therapeutic efficacy utilizing combinatorial blockade against IDO, CTLA-4, and PD-L1 in mice with brain tumors. Clinical Cancer Research. 20 (20), 5290-5301 (2014).
- Kim, G. B., et al. Rapid generation of somatic mouse mosaics with locus-specific, stably integrated transgenic elements. Cell. 179 (1), 251-267 (2019).
- Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L., Luo, L. A global double-fluorescent Cre reporter mouse. Genesis. 45 (9), 593-605 (2007).
- Rincon Fernandez Pacheco, D., Sabet, S., Breunig, J. J. Preparation, assembly, and transduction of transgenic elements using mosaic analysis with dual recombinase (MADR). STAR protocols. 1 (3), 100199 (2020).
- White, H. E., Goswami, A., Tucker, A. S. The intertwined evolution and development of sutures and cranial morphology. Frontiers in Cell and Developmental Biology. 9, 653579 (2021).
- Ran, F. A., et al. Genome engineering using the CRISPR-Cas9 system. Nature Protocols. 8 (11), 2281-2308 (2013).
- Breunig, J. J., et al. Ets factors regulate neural stem cell depletion and gliogenesis in Ras pathway glioma. Cell Reports. 12 (2), 258-271 (2015).
- Hambardzumyan, D., Parada, L. F., Holland, E. C., Charest, A. Genetic modeling of gliomas in mice: new tools to tackle old problems. Glia. 59 (8), 1155-1168 (2011).
- Feng, W., et al. CRISPR-mediated loss of function analysis in cerebellar granule cells using in utero electroporation-based gene transfer. Journal of Visualized Experiments. (136), e57311 (2018).
- Zhang, L., et al. Gene knock-in by CRISPR/Cas9 and cell sorting in macrophage and T cell lines. Journal of Visualized Experiments. (177), e62328 (2021).
- Artegiani, B., Lange, C., Calegari, F. Expansion of embryonic and adult neural stem cells in utero electroporation or viral stereotaxic injection. Journal of Visualized Experiments. (68), e4093 (2012).
- Rice, H., Suth, S., Cavanaugh, W., Bai, J., Young-Pearse, T. L. In utero electroporation followed by primary neuronal culture for studying gene function in subset of cortical neurons. Journal of Visualized Experiments. (44), e2103 (2010).
- Zanders, E. D., Svensson, F., Bailey, D. S. Therapy for glioblastoma: is it working. Drug Discovery Today. 24 (5), 1193-1201 (2019).

