Preparation and In Vivo Use of an Activity-based Probe for N-acylethanolamine Acid Amidase
Summary
Here, we describe the preparation and use of an activity-based probe (ARN14686, undec-10-ynyl-N-[(3S)-2-oxoazetidin-3-yl]carbamate) that allows for the detection and quantification of the active form of the proinflammatory enzyme N-acylethanolamine acid amidase (NAAA), both in vitro and ex vivo.
Abstract
Activity-based protein profiling (ABPP) is a method for the identification of an enzyme of interest in a complex proteome through the use of a chemical probe that targets the enzyme's active sites. A reporter tag introduced into the probe allows for the detection of the labeled enzyme by in-gel fluorescence scanning, protein blot, fluorescence microscopy, or liquid chromatography-mass spectrometry. Here, we describe the preparation and use of the compound ARN14686, a click chemistry activity-based probe (CC-ABP) that selectively recognizes the enzyme N-acylethanolamine acid amidase (NAAA). NAAA is a cysteine hydrolase that promotes inflammation by deactivating endogenous peroxisome proliferator-activated receptor (PPAR)-alpha agonists such as palmitoylethanolamide (PEA) and oleoylethanolamide (OEA). NAAA is synthesized as an inactive full-length proenzyme, which is activated by autoproteolysis in the acidic pH of the lysosome. Localization studies have shown that NAAA is predominantly expressed in macrophages and other monocyte-derived cells, as well as in B-lymphocytes. We provide examples of how ARN14686 can be used to detect and quantify active NAAA ex vivo in rodent tissues by protein blot and fluorescence microscopy.
Introduction
Commonly used methods to investigate the expression patterns, interactions, and functions of proteins, including liquid chromatography-mass spectrometry platforms for shotgun analysis1,2, yeast two-hybrid methods3,4, and in vitro assays, are limited in that they are unable to assess the activity of proteins in their native state. Activity-based protein profiling (ABPP) can be used to fill this gap. In this approach, small-molecule probes capable of covalently binding to the active site of an enzyme of interest are conjugated to a reporter group that allows for target detection. Using click chemistry (CC), the reporter can be integrated into the probe or can be introduced after target engagement has occurred5,6. The latter procedure requires the use of probes containing appropriate chemical groups, such as a terminal alkyne or azide, which can be modified with a number of reporter reagents via bio-orthogonal reactions such as the Cu(I)-catalyzed Huisgen [3+2] cycloaddition7-9 or Staudinger ligation10,11.
Recently, we disclosed the compound ARN14686 as the first ABP for the in vitro and in vivo detection of the cysteine hydrolase, NAAA12. NAAA catalyzes the hydrolytic deactivation of saturated and monounsaturated FAEs, including oleoylethanolamide (OEA) and palmitoylethanolamide (PEA), which are endogenous agonists of the anti-inflammatory nuclear receptor PPAR-alpha13-15. NAAA is predominantly expressed in macrophages and other monocyte-derived cells, as well as in B-lymphocytes14,16, suggesting a role in the regulation of the innate immune response. The enzyme is synthesized in the rough endoplasmic reticulum in an inactive form and is activated in acidic compartments of the cell by an autoproteolytic mechanism17. The autoproteolytic cleavage generates a new N-terminal cysteine (C131 in mice and rats, C126 in humans), that is the nucleophile responsible for FAE hydrolysis18,19. Pharmacological inhibition of NAAA activity alters the FAE synthesis/degradation balance in favor of increased cellular levels of FAEs16,20,21. Several β-lactone and β-lactam derivatives have been shown to inhibit NAAA activity with high potency and selectivity16,22-26. These inhibitors act through S-acylation of the catalytic cysteine16,27,28.
The compound ARN14686 was designed based on the chemical structure of the systemically active, serine-derived β-lactam NAAA inhibitor, ARN726 (4-cyclohexylbutyl-N-[(S)-2-oxoazetidin-3-yl]carbamate)16. The 4-butyl-cyclohexyl group of ARN726 was replaced with a C9 saturated aliphatic chain bearing a terminal alkyne tag for subsequent CC conjugation with an azide-bearing reporter tag. We chose to design a two-step ABP to minimally alter the structure of the original scaffold, thus maintaining the affinity of the probe for NAAA. Moreover, avoiding the introduction of bulky tags, such a probe could be more suitable for in vivo treatment than a direct ABP. ARN14686 inhibits NAAA with high potency (hNAAA IC50 = 6 nM, rNAAA IC50 = 13 nM) by forming a covalent adduct with the catalytic cysteine of the enzyme12. Experiments in live rats showed that the probe is selective in capturing NAAA expressed in lungs. Acid ceramidase, another cysteine amidase that shares 33-34% identity with NAAA, was also identified as a low-affinity target when using high probe concentrations (10 µM in vitro, 10 mg/ml intravenous, i.v.)12. We have also used ARN14686 to study the presence of active NAAA in inflamed rat tissues following administration of complete Freund's adjuvant (CFA)29.
Here, we outline a protocol for the preparation of ARN14686 (Figure 1) and its application to the investigation of NAAA activation ex vivo. As an example, we describe an experimental procedure to visualize NAAA in rat paws after CFA administration. In this experiment, proteins are extracted from paw tissue after i.v. injection of the probe, and the ABP-labeled proteome is subjected to CC with biotin-azide. Biotinylated samples are enriched using streptavidin beads, and protein blots are performed. In another application, we describe the localization of active NAAA by fluorescence microscopy in mouse lungs from probe-treated mice. In this case, the tissue is sectioned and sections are subjected to CC for rhodamine addition. A workflow scheme is illustrated in Figure 2.
Protocol
Caution: All chemistry reactions should be carried out in a ventilated fume hood and with the use of a lab coat, gloves, and protective goggles. The reactions should be also carried out in a nitrogen environment.
Ethical statement: Our procedures involving animals are performed in accordance with the Italian regulations on the protection of animals used for experimental and other scientific purposes (D.M. 116192), and European Economic Community regulations (O.J. of E.C. L 358/1 12/18/1986).
NOTE: Synthesis of [(3S)-2-oxoazetidin-3-yl]ammonium acetate is described for large-scale yields (50 g of N-Cbz-L-Serine), but it can be easily scaled down.
1. Synthesis
NOTE: See Figure 1 for the synthesis reaction scheme.
- Preparation of 2-pyridyl Undec-10-ynyl carbonate and Undec-10-ynyl 2-oxopyridine 1-carboxylate
- In a 50 ml round-bottom flask, dissolve 350 mg of undec-10-yn-1-ol in 3.5 ml of dry dichloromethane.
- To the solution in 1.1.1, add 25 mg of 4-dimethylaminopyridine (DMAP) and 530 mg of 2-dipyridylcarbonate (DPC). Stir the mixture at room temperature (RT) for 16 hr.
- Add 20 ml of dichloromethane.
- Transfer the mixture into a separating funnel. Add 15 ml of water, shake it, and allow the two phases to separate.
- Open the stopcock, collect the organic phase (bottom), and then close the stopcock. Add 15 ml of a saturated NaHCO3solution. Shake the separating funnel and let the two phases separate. Repeat this step two more times.
- Dry the organic layer with Na2SO4, filter through cotton into a round-bottom flask (tare first), and evaporate to dryness under reduced pressure (rotary evaporator).
- Weigh the flask and obtain 600 mg of oil that is a mixture of 2-pyridyl undec-10-ynyl carbonate and undec-10-ynyl 2-oxopyridine 1-carboxylate in a ratio of 1.7:1.
1H NMR (400 MHz, DMSO-d6) major component δ = 8.39 (dd, J = 5.0, 2.0, 1H), 7.98 (td, J = 7.9, 2.0, 1H), 7.40 (ddd, J = 7.2, 4.9, 0.9, 1H), 7.30 (d, J = 8.2, 1H), 4.22 (t, J = 6.6, 2H), 2.73 (t, J = 2.4, 1H), 2.18 – 2.11 (m, 2H), 1.76 – 1.62 (m, 2H), 1.50 – 1.23 (m, 12H).
1H NMR (400 MHz, DMSO-d6) minor component δ = 7.74 (dd, J = 7.2, 1.9, 1H), 7.47 (dd, J = 6.7, 2.3, 1H), 6.44 (d, J = 9.4, 1H), 6.30 – 6.25 (m, 1H), 4.35 (t, J = 6.5, 2H), 2.72 (t, J = 2.4, 1H), 2.18 – 2.11 (m, 2H), 1.77 – 1.60 (m, 2H), 1.52 – 1.20 (m, 12H). - Use the oil without any further separation or purification.
- Preparation of [(3S)-2-oxoazetidin-3-yl]ammonium Acetate
- Preparation of benzyl N-[(S)-1-(hydroxymethyl)-2-[(4-methoxyphenyl)amino]-2-oxoethyl]carbamate.
- In a 4-L round-bottom flask, dissolve 141.5 g of p-anisidine in 1.5 L of tetrahydrofuran and 0.5 L of dichloromethane.
- Cool the solution to 0 °C in an ice bath.
- Add 50.0 g of N-Cbz-L-Serine and 43.9 g of N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride.
- Stir the mixture with a magnetic bar at 0 °C for 30 min.
- Remove the solution from the ice bath and stir with a magnetic bar at RT for 16 hr.
- Evaporate the solvents under reduced pressure (rotary evaporator).
- Add 400 ml of 1:1 cyclohexane/ethyl acetate, stir with a spatula, and decant.
- Repeat step 1.2.1.7 two more times.
- Dissolve the gummy residue in 500 ml of ethyl acetate and wash with 400 ml of 0.1 M HCl solution (10 times), with 400 ml of saturated NaHCO3 solution (2 times), and with 400 ml of brine.
- Dry the organic layer with Na2SO4, filter the solution through cotton into a round-bottom flask (tare first), and evaporate it to dryness under reduced pressure (rotary evaporator).
- Weigh the flask and obtain 61.4 g of a white solid.
1H NMR (400 MHz, DMSO-d6) δ = 9.86 (bs, 1H), 7.52 (d, 2H, J = 9.0 Hz), 7.42-7.25 (m, 6H), 6.91-6.85 (m, 2H), 5.06 (d, 1H, J = 12.9 Hz), 5.02 (d, 1H, J = 12.9 Hz), 4.98 (t, 1H, J = 5.6 Hz), 4.24-4.15 (m, 1H), 3.72 (s, 3H), 3.70-3.57 (m, 2H).
- Preparation of Benzyl N-[(S)-1-(4-methoxyphenyl)-2-oxo-azetidin-3-yl]carbamate.
- Dissolve 58.6 g of N-[(S)-1- (hydroxymethyl)-2-[(4-methoxyphenyl)amino]-2-oxo-ethyl]carbamate in 1.6 L of N,N-dimethylformamide.
- Cool the solution to 0 °C with an ice bath.
- Add 50.6 g of 1,1'-sulfonyldiimidazole and stir with a magnetic bar for 30 min.
- Cool the solution to -20 °C with an ice/NaCl bath and add 10.2 g of sodium hydride (60% in mineral oil) portion-wise.
- Stir the mixture at -20 °C for 1 hr, and then quench with 2 ml of methanol and 1 L of water.
- Vacuum filter the precipitate, wash with 200 ml of water, and dry under vacuum.
- Obtain 42.2 g of a white solid.
1H NMR (400 MHz, DMSO-d6) δ = 8.08 (d, 1H, J = 8.5 Hz), 7.42-7.28 (m, 5H), 7.30 (d, 2H, J = 8.9 Hz), 6.95 (d, 2H, J = 8.9 Hz), 5.06 (s, 2H), 4.86 (ddd, 1H, J = 8.5, 5.6, 2.6 Hz), 3.90 (t, 1H, J = 5.6 Hz), 3.73 (s, 3H), 3.55 (dd, 1H, J = 5.6, 2.6 Hz).
- Preparation of Benzyl N-[(S)-2-oxoazetidin-3-yl]carbamate.
- Suspend 9.0 g of benzyl N-[(S)-1-(4-methoxyphenyl)-2-oxoazetidin-3-yl]carbamate in 500 ml of acetonitrile and 400 ml of water.
- Cool the solution to 0 °C in an ice bath.
- Add 45.4 g of ceric ammonium nitrate portion-wise over 45 min and stir with a magnetic bar at 0 °C for 15 min.
- Add 500 ml of saturated NaHCO3 solution cautiously, and then add 500 ml of ethyl acetate.
- Filter the precipitate and wash with 200 ml of ethyl acetate.
- Separate the biphasic solution and wash the aqueous layer with 200 ml of ethyl acetate (3 times).
- Dry the organic layer with Na2SO4, add 5 g of activated charcoal, filter through a pad of diatomaceous silica, and evaporate to dryness under reduced pressure (rotary evaporator).
- Add diethyl ether and stir with a spatula.
- Filter the solid and obtain 4.85 g of an off-white solid.
1H NMR (400 MHz, DMSO-d6) δ = 7.97 (d, 1H, J = 8.7 Hz), 7.94 (bs, 1H), 7.42- 7.30 (m, 5H), 5.05 (s, 2H), 4.67 (ddd, 1H, J = 8.7, 5.4, 2.7 Hz), 3.40 (t, 1H, J = 5.4 Hz,), 3.09 (dd, 1H, J = 5.4, 2.7 Hz).
- Preparation of [(S)-2-oxoazetidin-3-yl]-ammonium Acetate.
- Dissolve 0.93 ml of acetic acid in 245 ml of ethyl acetate. Mark this as "trapping solution."
- Dissolve 3.28 g of benzyl-N-[(R)-2-oxoazetidin-3-yl]carbamate in 298 ml of ethanol.
- Add 14.1 ml of cyclohexadiene and 3.27 g of 10% Palladium on carbon.
- Stir the suspension at RT for 12 hr, and then filter through a short pad of diatomaceous earth. Pour the eluting liquid directly into the trapping solution.
- Evaporate the solvent under reduced pressure (rotary evaporator), keeping the temperature below 35 °C.
- Triturate the obtained solid with tetrahydrofuran and obtain 1.72 g of a white solid.
1H NMR (400 MHz, DMSO-d6) δ = 7.68 (bs, 1H), 3.99 (ddd, 1H, J = 5.2, 2.4, 1.2 Hz), 3.32 (t, 1H, J = 5.2 Hz), 2.79 (dd, 1H, J = 5.2, 2.4 Hz), 1.90 (s, 3H).
- Preparation of benzyl N-[(S)-1-(hydroxymethyl)-2-[(4-methoxyphenyl)amino]-2-oxoethyl]carbamate.
- Preparation of Undec-10-ynyl-N-[(3S)-2-oxoazetidin-3-yl]carbamate
- In a 10 ml round-bottom flask, dissolve 60 mg of [(3S)-2-oxoazetidin-3-yl]ammonium acetate in 2 ml of dry dichloromethane.
- Cool the solution to 0 °C in an ice bath and add 81 µl of N,N-diisopropylethylamine drop-wise.
- Dissolve 350 mg of the crude mixture containing undec-10-ynyl 2-oxopyridine 1-carboxylate in 2 ml of dry dichloromethane, add it to the solution, and stir at RT for 15 hr. Evaporate the solvent to dryness under reduced pressure (rotary evaporator).
- Purify by column chromatography (silica gel) using an automated column chromatography apparatus:
- Absorb the sample onto silica gel, equilibrate the column with cyclohexane, and load the sample into the cartridge.
- Elute with cyclohexane/ethyl acetate from 100:0 to 0:100 and collect peaks on test tubes.
- Evaporate the solvent of the fractions corresponding to the compound to dryness under reduced pressure (rotary evaporator) and obtain 40 mg of a white solid.
2. Preparation of CC Reagents
- Preparation of 5 mM Stock Solution of Tag-azide Molecules
- Dissolve 1.5 mg of Azide-PEG3-Fluor 545 in 0.5 ml of dimethyl sulfoxide (DMSO). Make aliquots in 0.5 ml microcentrifuge tubes and store at -20 °C.
- Dissolve 1.1 mg of Azide-PEG3-Biotin in 0.5 ml of DMSO. Make aliquots in 0.5 ml microcentrifuge tubes and store at -20 °C.
- Preparation of 50 mM stock Solution of Tris(2-carboxyethyl)phosphine (TCEP)
- Dissolve 14.3 mg of TCEP in 1 ml of water, and make it fresh each time.
- Preparation of 50 mM Solution of CuSO4·5H2O
- In a glass vial, dissolve 12.48 mg of CuSO4·5H2O in 1 ml of water. Store at RT for up to one month.
- Preparation of 83.5 mM Stock Solution of Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA)
- In a glass vial, dissolve 8.85 mg of TBTA in 200 µl of DMSO. Store at RT for up to one month.
- Preparation of 1.7 mM Working Solution of TBTA (Immediately Before Use)
- Add 20 µl of 83.5 mM TBTA to a glass vial and dilute with 180 ml of DMSO.
- Add 800 µl of tert-Butanol and vortex.
3. NAAA Expression Analysis in Paw Tissue of CFA-treated Rats
NOTE: Use male Sprague-Dawley rats, weighing 175-200 g, and perform all procedures in accordance with guidelines for ethical use of animals. House rats in ventilated cages on a 12-hr light/dark cycle and give them free access to food and water. For the protocol of CFA treatment, refer to the article published by Bonezzi et al.29 Use animals 7 days after CFA administration.
- Intravenous Administration of ARN14686
- Dissolve ARN14686 in vehicle (15% PEG and 15% Tween saline solution). Calculate the solution concentration according to rat weight (dose 3 mg/kg, injection volume 5 ml/kg).
- Proceed with i.v. injections in three naïve and three CFA-treated rats. Place each rat in an appropriate plastic restraint device. Then, place the rat tail in warm water for 2-4 min to allow vasodilation, and inject the compound i.v. via the tail vein.
- Inject three rats (naïve and CFA-treated) i.v. with vehicle only.
- Paw Collection and Dissection
- Sacrifice the rats by CO2 inhalation 4 hr after probe or vehicle administration and collect their paws by cutting them 0.5 cm above the knee joint using a scalpel.
- Carefully remove the skin by scissor-assisted dissection and dissect out soft tissues by scraping them from the bone. Discard the bones and collect the soft tissues of the paws. Snap freeze samples in liquid nitrogen and store at -80 °C.
- Paw Homogenization and Lysosomal Protein Preparation
NOTE: Avoid using detergents and amine-containing buffers, as they can inhibit the CC reaction.- Combine paw tissue from 3 rats (obtained as described in section 3.2.1) and homogenize them in 4 ml of 320 mM sucrose in phosphate-buffered saline (PBS, pH 7.4) and a protease inhibitor cocktail (see Material/Reagent Table); use a high-performance dispersing instrument (see Material/Reagent Table).
- Centrifuge the tissue homogenate for 20 min at 1,000 x g at 4 °C; save the supernatant.
- Add 2 ml of 320 mM sucrose in PBS and the protease inhibitor cocktail to the tissue pellet and homogenize once more.
- Centrifuge for 20 min at 1,000 x g at 4 °C, and add the supernatant to the one from step 3.3.2.
- Centrifuge the collected supernatants for 30 min at 12,000 x g at 4 °C.
- Weigh the pellet and resuspend in two volumes of PBS (i.e., 200 µl for each 100 mg of pellet). Transfer to a 1.5 ml collection tube and freeze at -80 °C for 1 hr.
- Thaw the samples and freeze again for 1 hr at -80 °C.
- Repeat the freezing/ thawing cycle two more times.
NOTE: This step is intended to solubilize the protein. Freeze overnight if necessary. - Centrifuge for 1 hr at 100,000 x g at 4 °C. Collect the supernatant, which contains soluble lysosomal proteins, and discard the pellet. Store at -80 °C or immediately proceed with protein quantification.
- Quantify the protein content using a BCA protein assay30 commercial kit (see Material/Reagent Table), following the manufacturer's instructions.
- Store the samples at -80 °C until use.
- CC with Azide-PEG3-Biotin
NOTE: The protocol of CC and streptavidin bead enrichment (steps 3.4-3.6) are slightly modified from the protocol published by Speers and Cravatt31.- Prepare 500 µl (1 mg/ml, in PBS) of lysosomal proteins from probe- and vehicle-treated rats (naïve and CFA-treated rats).
- Preclear samples with 40 µl of a 50% slurry of streptavidin agarose (wash three times with 1 ml of PBS before use) for 1 hr at 4 °C. Centrifuge for 4 min at 1,000 x g at 4 °C and take the supernatant.
- Add 11.3 µl of a 5 mM stock of Azide-PEG3-Biotin and vortex.
- Add 11.3 µl of a 50 mM stock of freshly prepared TCEP and vortex.
- Premix 34 µl of a freshly prepared working solution of 1.7 mM TBTA with 11.3 µl of a 50 mM CuSO4·5H2O stock.
- Add 45.3 ml of a premixed TBTA/CuSO4·5H2O solution and vortex.
- Incubate the reaction at 25 °C for 2 hr (a longer incubation time does not affect the reaction). Observe protein precipitation at this step. Mix after the first hour of incubation.
- Removal of Excess CC Reagents
- Centrifuge samples for 4 min at 6,500 x g at 4 °C and remove the supernatant.
- Add 750 µl of cold methanol and resuspend by sonication (5 sec with a probe sonicator).
- Centrifuge the samples for 4 min at 6,500 x g at 4 °C and remove the supernatant using a syringe and needle.
- Repeat step 3.5.2 twice (sonication is not required).
- After the last wash, add 325 µl of sodium dodecyl sulfate (SDS) 2.5% in PBS to the protein pellet and sonicate 3x for 5 sec.
- Heat the samples for 5 min at 65 °C and sonicate again.
- Centrifuge for 5 min at 6,500 x g at RT and save the supernatant.
- Add 1.4 ml of PBS to dilute the SDS concentration to 0.5%. Store at -20 °C or continue with streptavidin enrichment.
- Streptavidin Enrichment
- With PBS, bring the volume of the samples obtained in step 3.5.8 to 4.2 ml. Add 40 µl of a 50% slurry of streptavidin agarose using a cut-end tip (wash three times with 1 ml of PBS before use).
- Incubate for 2 hr at RT with rotation and then centrifuge for 2 min at 1,400 x g.
- Remove the supernatant without drying the beads pellet and use residual supernatant to transfer the beads to a 1 ml spin column (see Material/Reagent Table).
- Wash by gravity with 3x 1 ml of 1% SDS (in PBS), 3x 1 ml of 6 M urea (in PBS), and 4x 1 ml of PBS.
- Use 2x 500 µl of PBS to transfer the beads to a 1.5 ml tube and centrifuge for 2 min at 1,400 x g at RT. Gently aspirate the supernatant with a syringe and needle.
- Elute the resin-bound proteins by adding 25 µl of elution buffer (6 M urea, 2 M thiourea, 2% SDS, and 6 mM biotin, all in PBS) for 15 min at RT followed by 15 min at 95 °C32.
- Protein Blot
- Add 5 µl of 6x Laemmli buffer (for 9 ml: 0.5 ml of 1 M Tris-HCl pH 6.8, 5 ml of 20% SDS, 5 mg Bromophenol blue, 3 ml glycerol, and H2O up to 9 ml) and add 5% β-mercaptoethanol immediately before use. Briefly centrifuge the samples at 2,000 x g to pellet the resin and load 25 µl of the obtained supernatant into a 4-12% polyacrylamide gel.
- Perform gel electrophoresis and protein transfer on a blotting membrane according to the manufacturer's instructions33.
- Saturate the blotting membrane for 1 hr with 10 ml of a blocking buffer (see Material/Reagent Table) containing 0.1% Tween-20. Avoid using milk, as it can increase the background.
- Wash the membrane with 10 ml of 0.05% Tween-20 in PBS and add 10 µl of fluorescent streptavidin (see Material/Reagent Table) dissolved in 10 ml of blocking buffer plus 0.1% Tween-20 for 1 hr at RT.
- Wash 4 times with 0.05% Tween-20 in PBS and once with PBS alone (10 min each).
- Use an image scanner (see Material/Reagent Table). Switch on the instrument and the connected computer. Wait until ready.
- Start the acquisition program and select the fluorescence mode. Select the membrane area and choose the destination folder for saved files.
- Set the following acquisition parameters: 680 nm excitation length, BPFR700 filter, 1,000 V photomultiplier tube (PMT) value (channel 2), and 25 µm pixel size. Acquire the image.
4. Localization of Catalytically Active NAAA in Mouse Lungs by Fluorescence Microscopy
NOTE: Use male 8 to 10 week-old mice and perform all procedures in accordance with the guidelines for the ethical use of animals. House mice in ventilated cages on a 12 hr light/dark cycle and give them free access to food and water.
- Intravenous Administration of ARN14686 in Mice
- Dissolve ARN14686 in vehicle: 15% PEG and 15% Tween-20 saline solution. Calculate the solution concentration according to mouse weight (dose 3 mg/kg, injection volume 5 ml/kg).
- Proceed with the i.v. injection of 3 mice. Place each mouse in an appropriate plastic restraint device. Then, place the mouse tail in warm water for 2-4 min to allow vasodilation and inject the compound i.v. via the tail vein.
- Inject 3 mice i.v. with vehicle only.
- Lung Collection and Slice Preparation
Warning: Handle paraformaldehyde with care in a fume hood, and wear gloves!- Anesthetize mice with chloral hydrate (400 mg/kg). Confirm anesthesia with a toe pinch. Perform a transcardial perfusion as follows:
- Superficially cut the ventral skin with scissors and expose the thoracic and peritoneal membrane surfaces.
- Superficially cut the peritoneal membrane with scissors, just below the xyphoid process, and expose the diaphragm and visceral organs. Be careful not to lacerate any significant vasculature.
- Open the thoracic cavity by cutting the diaphragm from one lateral aspect to the other.
- Insert the 25 G needle connected to the automated syringe pump and infuse 20 ml of 0.9% saline solution followed by 60 ml of 4% PFA in phosphate buffer (0.1 M, pH 7.4).
- Once the perfusion is complete, pull the heart using forceps and carefully dissect it out. Grasp the trachea with forceps and cut completely through it using scissors. Gently tug the trachea upwards and remove the lungs from the rib cage. Dissect the tissue to separate the right lung from the left.
- Postfix the tissue in paraformaldehyde 4% for 1 hr, freeze samples in cold 2-methylbutane, and store them at -80 °C.
- Collect 40 µm sections using a cryostat, mount them immediately on slides (one every fifth), and process them for immunohistochemistry as detailed below.
- Anesthetize mice with chloral hydrate (400 mg/kg). Confirm anesthesia with a toe pinch. Perform a transcardial perfusion as follows:
- Tissue Slices Permeabilization and Blocking
- Wash with PBS (2x for 5 min) and permeabilize with 0.1% Triton X-100 PBS for 15 min at RT.
- Wash with PBS (2x for 5 min) and block with 3% bovine serum albumin (BSA) in PBS for 30 min at RT.
- Wash with PBS (2x for 5 min) and proceed with CC.
- CC with Azide-PEG3-Fluor 545 on Tissue Slices
- Prepare a solution by mixing the CC reagents prepared as described in section 2. For 1 ml solution, add: 2 µl of Azide-PEG3-Fluor 545 (5 mM stock), 20 µl of TCEP (freshly prepared 50 mM stock), 58.8 µl of TBTA (freshly prepared 1.7 mM working solution), 20 µl of CuSO4·5H2O (50 mM stock), and 900 µl of PBS.
- Add around 400 µl of CC mix to the tissue slices, which were prepared according to section 4.2. Pay attention that the chosen volume of CC mix is sufficient to cover the slices. Incubate for 1 hr at RT protected from light.
- Wash with PBS (1x for 5 min), cold methanol (1x for 5 min), a solution of 1% Tween-20 and 0.5 mM EDTA in PBS (3x for 2 min), and PBS (1x for 5 min).
- Air dry, add a drop of antifade mountant with DAPI (see Material/Reagent Table), close with cover slips (avoiding bubble formation), and seal with polish. Store at 4 °C until analysis.
- Image Acquisition
- Use a confocal microscope equipped with 546 nm and 450 nm excitation lasers (see Material/Reagent Table) following the user guide.
- Select a 60X objective lens with NA = 1.40, making sure to preview the slide through the eyepieces and to focus on an area of interest.
- Determine the acquisition parameters according to the sample and equipment features.
Representative Results
ARN14686 was designed based on the scaffold of the NAAA inhibitor ARN726. The 4-butyl-cyclohexyl group of ARN726 was substituted with a C9 saturated aliphatic chain bearing a terminal alkyne tag (Figure 1). The alkyne tag was introduced in order to allow the use of a two-step labeling procedure to add a fluorophore or a biotin molecule via CC. This feature renders ARN14686 a very versatile tool to probe NAAA in vitro and in vivo.
Here, we show two applications of ARN14686, which are representative of the potential of this molecule. Figure 2 is a scheme of the experimental procedure reported here. After i.v. administration of the probe, two different detection methods can be used: i) analysis of active NAAA expression by protein blot and ii) analysis of active NAAA expression and localization within cells by fluorescence microscopy.
The first representative result was recently published by our group29. We analyzed NAAA expression in a rat model of CFA-induced paw inflammation. The probe (3 mg/kg) or vehicle was injected i.v. in naïve and CFA-treated rats. The rats were sacrificed 4 hr later. CC was performed on enriched lysosomal extracts to introduce a biotin-tag onto the probe-labeled proteins. Biotinylated proteins were next enriched using streptavidin beads. The eluted proteins were analyzed by protein blot, showing that levels of active NAAA were markedly increased in the paws of rats treated with CFA relative to those of control rats (Figure 3). The advantage of protein blot analysis is that it enables a detailed examination of the probe-reactive proteome, which is separated by gel electrophoresis. This approach also allows for the unveiling of potential probe off-targets. A no-probe control must be always included in order to exclude endogenous biotinylated proteins, which constitute the experimental background. The arrowhead in Figure 3 indicates such background proteins, which in turn can be used as a loading reference control.
In a second unpublished experiment (Figure 4), we used ARN14686 to probe NAAA for ex vivo detection by fluorescence microscopy. We administered ARN14686 to mice at 3 mg/kg (i.v.) and sacrificed them 2 hr after treatment by transcardial perfusion. Lungs were collected, postfixed, and frozen in cold 2-metylbutane. The CC reaction for fluorophore addition was performed directly on tissue slices of 40 µm thickness, collected with a cryostat. Analysis by fluorescence microscopy showed the presence of catalytically active NAAA in diffused vesicular structures belonging to alveolar macrophages. Compared to the use of a protein-specific antibody, only the active enzyme is observed.
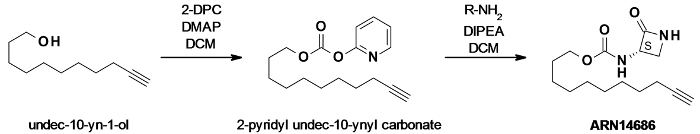
Figure 1: ARN14686 synthesis reaction scheme. Undec-10-yn-2-ol was activated by dipyridylcarbonate (DPC) in the presence of catalytic 4-dimethylaminopyridine (DMAP) to produce a mixed carbonate. This carbonate was then reacted with the amino lactam to obtain the target molecule ARN14686. Please click here to view a larger version of this figure.
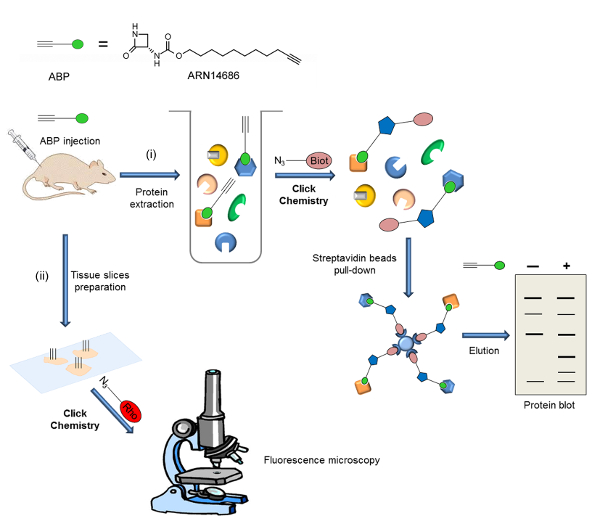
Figure 2: Workflow scheme of the general strategy shown in the present work. The probe is injected into animals (rats or mice) and target expression is analyzed following two different experimental procedures: i) The labeled proteome is extracted and biotin is added by CC. After an enrichment phase of biotinylated proteins on streptavidin beads, probe targets are analyzed by protein blot. ii) Tissue slices are prepared and a fluorophore is added by CC. Probe target localization is analyzed by florescence microscopy. This figure has been adapted from Bonezzi et al.29 Please click here to view a larger version of this figure.
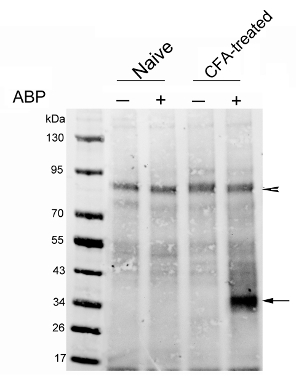
Figure 3: Analysis of NAAA activation in paws of CFA-treated rats. Protein blot analysis of streptavidin-enriched proteins from naïve rats (lanes 1 and 2) or CFA-injected rats 7 days after injection (lanes 3 and 4). Rats received i.v. injections of vehicle or ARN14686 (3 mg/kg). The blotting membrane was probed with a fluorescent streptavidin. The arrow indicates the NAAA band; the arrowhead indicates a biotin-containing band of around 90 kDa, showing that a similar amount of protein was loaded in each lane. This figure has been modified from Bonezzi et al.29 Please click here to view a larger version of this figure.
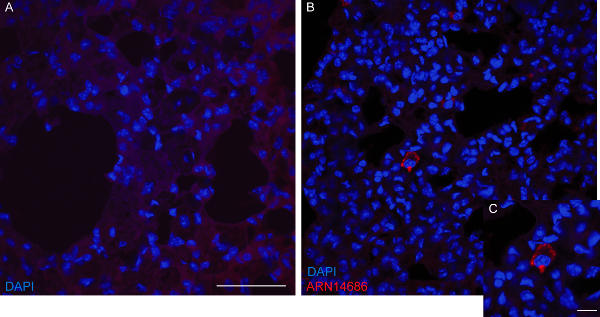
Figure 4: Ex vivo detection of probe-labeled NAAA in mouse lungs by fluorescence microscopy. Representative pictures of lung sections of vehicle- (A) or ARN14686-injected (B) mice after CC with azide-PEG3-Fluor 545 are reported. A positive signal (red cells) was detected in ARN14686-injected mice, while no signal was detected in vehicle-administered mice. A detail of an azide-PEG3-Fluor 545 positive alveolar macrophage is shown in C at higher magnification. Nuclei were marked with DAPI (blue). Scale bar = 50 µm in A and 10 µm in C. Please click here to view a larger version of this figure.
Discussion
Enzyme activity is finely regulated at different levels, including RNA transcription, protein synthesis, protein translocation, post-translational modification, and protein-protein interaction. Often, enzyme expression alone does not account for its activity. ABPP was developed to study the activity of proteins in their native state. Two features are required: a chemical probe that covalently binds to the active site of an enzyme of interest and a reporter tag to detect the probe-labeled enzyme.
Probe design and synthesis are critical points of the procedure. The probe must have adequate affinity and selectivity for its target. Moreover, the presence of a reporter tag must not affect target engagement. This issue is largely overcome by the design of a two-step ABP, in which the reporter tag is introduced after the target has been caught. Tag-free probes are particularly suitable for in vivo studies, in which protein activity can be evaluated in a living cell or organism, with minimal external alteration. The NAAA probe ARN14686 was designed to fulfill the requirements outlined above. The β-lactam reactive warhead was chosen based on prior results obtained with the β-lactam class of NAAA inhibitors16,26. These compounds inhibit NAAA in a potent and selective manner by covalently binding to the catalytic cysteine of the enzyme. Moreover, the compounds were shown to be systemically active16. We introduced a C9 saturated aliphatic chain, taking into account the increased affinity of NAAA for long aliphatic chains. A terminal alkyne was added to allow for two-step labeling.
Another critical step is selecting the dose and the time for in vivo administration. This depends on the stability of the probe in plasma, its target affinity, and its selectivity. The correct dose must be selected to allow for target capture while avoiding engagement of possible off-targets. We found that 3 mg/kg ARN14686 i.v. was optimal to capture NAAA selectively. Higher doses resulted in the capture of the homologous cysteine amidase, acid ceramidase. With regard to treatment length, when analyzing well-perfused organs, like lungs, a short time (2 hr) may be sufficient to allow for the probe to react with the target. For paws, however, we were obliged to double the reaction time.
A possible issue in target analysis by protein blot is due to the presence of naturally biotinylated proteins. These will inevitably be identified together with specific probe targets. We found that introducing a preclearing step with streptavidin beads before performing CC greatly increased the quality of our results. On the other hand, the presence of native biotinylated proteins might be used to control for possible loading artefacts. Finally, with regard to localization studies by fluorescence microscopy, it is very important to be aware of probe selectivity because, unlike protein blots, fluorescence microscopy does not permit the distinction of the target from off-targets. Preliminary selectivity studies should be performed to assess feasibility and to set up optimal experimental conditions.
Limitations of the described technique mainly pertain to the necessity of avoiding experimental conditions that can affect the CC reaction, such as the use of detergents and amine-containing buffers. These aspects must be taken in to account when preparing a cell lysate or a tissue homogenate. Moreover, when a streptavidin enrichment phase is required, the amount of starting material constitutes another issue, because this procedure is only applicable when the protein content is not less than 250 µg. This limit is set due to technical issues, such as working volumes, protein recovery, after CC-induced precipitation, and the amount of streptavidin resin to be used.
Previously, NAAA activity could be only evaluated by performing activity assays, which require the use of an activation buffer for the in vitro activation of the enzyme and for substrate solubilization14. This approach provides information about total NAAA expression, not about the presence of active NAAA. Another possibility is to measure tissue levels of PEA and OEA, but this method represents only an indirect way to evaluate NAAA activity34,35. In addition, FAE levels can be influenced by other factors such as biosynthesis. The chemical probe ARN14686 is the first ABP for NAAA. The protocol described here illustrates a simple procedure for capturing and visualizing the active form of NAAA, both in vitro and in vivo. All sample manipulations are subsequent to probe-target reactions, thus giving reliable information about the in vivo state of the enzyme. Moreover, the use of ARN14686 in fluorescence microscopy represents a unique tool to localize active NAAA. Available antibodies, which recognize the NAAA catalytic subunit, do not discriminate between the NAAA full-length proenzyme and the active enzyme.
Starting from the protocol described here, NAAA expression and activation can be analyzed in different cell lines and animal models of inflammation. Co-localization studies can be performed to better characterize the role of NAAA in physiological and pathological conditions.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
The authors thank the Nikon Imaging Center at Istituto Italiano di Tecnologia, Genova, Italy (NIC@IIT).
Materials
| 1,1’-sulfonyldiimidazole | Sigma Aldrich | 367818 | Harmful |
| 2-dipyridylcarbonate | Fluorochem | 11331 | Harmful |
| 2-Methylbutan | Sigma Aldrich | M32631 | Flamable, toxic,hazardous to the aquatic environment |
| 4-(Dimethylamino)pyridine | Sigma Aldrich | 107700 | Toxic |
| Acetic acid | Sigma Aldrich | 695092 | Flammable, Corrosive |
| Acetonitrile | Sigma Aldrich | 34998 | Flammable, Toxic |
| Activated charcoal | Sigma Aldrich | 161551 | |
| Ammonium chloride | Sigma Aldrich | A9434 | Harmful |
| Azide-PEG3-Biotin | Jena Biosciences | CLK-AZ104P4 | |
| Azide-PEG3-Fluor 545 | Jena Biosciences | CLK-AZ109 | |
| BCA protein assay kit | Thermo Fisher Scientific | 23227 | |
| Bio-spin columns | Biorad | 732-6204 | |
| Biotin | Sigma Aldrich | B4501 | |
| Blocking buffer | Li-Cor Biosciences | 927-40000 | |
| b-mercaptoethanol | Sigma Aldrich | M6250 | Higly toxic |
| Bovin serum albumine (BSA) | Sigma Aldrich | A7030 | |
| Bromophenol blue | Sigma Aldrich | B0126 | |
| Bruker Avance III 400 | Bruker | ||
| Celite | Sigma Aldrich | 419931 | Health hazard |
| Ceric ammonium nitrate | Sigma Aldrich | 22249 | Oxidizing, Harmful |
| Chloral hydrate | Sigma Aldrich | C8383 | Higly toxic |
| CuSO4.5H2O | Sigma Aldrich | 209198 | Toxic |
| Cyclohexadiene | Sigma Aldrich | 125415 | Flammable, Health hazard |
| Cyclohexane | Sigma Aldrich | 34855 | Flammable, Harmful, Health hazard, Environmental hazard |
| Dichloromethane | Sigma Aldrich | 34856 | Harmful, Health hazard |
| Diethyl ether | Sigma Aldrich | 296082 | Flammable, Harmful |
| Dimethyl sulfoxide (DMSO) | Acros Organics | 348441000 | |
| Dimethyl sulfoxide d6 (DMSO-d6) | Sigma Aldrich | 175943 | |
| Ethanol | Sigma Aldrich | 2860 | Flammable, Harmful |
| Ethyl acetate | Sigma Aldrich | 34858 | Flammable, Harmful |
| Glycerol | Sigma Aldrich | G5516 | |
| Irdye 680-LT Streptavidin | Li-Cor Biosciences | 925-68031 | |
| IRDye680-LT Streptavidin | Licor | 925-68031 | Briefly centrifuge before use to precipitate protein complexes |
| Methanol | Sigma Aldrich | 34966 | Highly toxic |
| Methanol | Sigma Aldrich | 34860 | Flammable, Toxic, Health hazard |
| N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride | Sigma Aldrich | E7750 | Harmful, Corrosive |
| N,N-diisopropylethylamine | Sigma Aldrich | D125806 | Flammable, Corrosive, Toxic |
| N,N-dimethylformamide | Sigma Aldrich | 227056 | Flammable, Harmful, Health hazard |
| N-Cbz-L-Serine | Fluorochem | M03053 | Harmful |
| Nikon A1 confocal microscopy | Nikon | Read the user manual | |
| NuPAGE 4-12% Bis-Tris gel | Thermo Fisher Scientific | NP0335BOX | |
| Palladium on carbon | Sigma Aldrich | 330108 | |
| p-anisidine | Sigma Aldrich | A88255 | Toxic, Health hazard, Environmental hazard |
| Paraformaldehyde | sigma Aldrich | 441244 | Toxic, respiratory harmful, corrosive, falmable |
| Poly(ethylene glycol) | Sigma Aldrich | P3265 | |
| ProLong Gold antifade mountant with DAPI | Thermo Fisher Scientific | P36931 | Avoid bubbles formation |
| Protease inhibitor cocktail | Sigma Aldrich | P8340 | |
| Sodium bicarbonate | Sigma Aldrich | S6014 | |
| Sodium dodecyl sulfate (SDS) | Sigma Aldrich | L3771 | Toxic, corrosive, falmmable |
| Sodium hydride | Sigma Aldrich | 452912 | Flammable |
| Sodium sulfate | Sigma Aldrich | 239313 | |
| Starion FLA-9000 immage scanner | FUJIFILM | Read the user manual | |
| Streptavidin agarose | Thermo Fisher Scientific | 20349 | |
| Sucrose | Sigma Aldrich | S7903 | |
| Tert-butanol | Sigma Aldrich | 360538 | Toxic, flammable |
| Tetrahydrofuran | Sigma Aldrich | 186562 | Flammable, Harmful, Health hazard |
| Thiourea | Acros Organics | 424542500 | Toxic, warm at 50 °C to dissolve |
| Tris | Sigma Aldrich | RDD008 | |
| Tris(2-carboxyethyl)phosphine (TCEP) | Sigma Aldrich | C4706 | |
| Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) | Sigma Aldrich | 678937 | |
| Triton-x100 | Sigma Aldrich | X100 | Toxic |
| Tween-20 | Sigma Aldrich | P9416 | |
| Tween-80 | Sigma Aldrich | P1754 | |
| Ultra turrax IKA T18 basic tissue homogenizer | IKA | ||
| Undec-10-yn-1-ol | Fluorochem | 13739 | Harmful |
| Urea | Sigma Aldrich | U5378 | Toxic, warm at 50 °C to dissolve |
Referencias
- Gygi, S. P., Han, D. K., Gingras, A. C., Sonenberg, N., Aebersold, R. Protein analysis by mass spectrometry and sequence database searching: tools for cancer research in the post-genomic era. Electrophoresis. 20, 310-319 (1999).
- Washburn, M. P., Wolters, D., Yates, J. R. Large-scale analysis of the yeast proteome by multidimensional protein identification technology. Nat Biotechnol. 19, 242-247 (2001).
- Zhu, H., Bilgin, M., Snyder, M. Proteomics. Annu Rev Biochem. 72, 783-812 (2003).
- Ito, T., et al. Roles for the two-hybrid system in exploration of the yeast protein interactome. Mol Cell Proteomics. 1, 561-566 (2002).
- Evans, M. J., Cravatt, B. F. Mechanism-based profiling of enzyme families. Chem Rev. 106, 3279-3301 (2006).
- Cravatt, B. F., Wright, A. T., Kozarich, J. W. Activity-based protein profiling: from enzyme chemistry to proteomic chemistry. Annu Rev Biochem. 77, 383-414 (2008).
- Rostovtsev, V. V., Green, L. G., Fokin, V. V., Sharpless, K. B. A stepwise huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew Chem Int Ed Engl. 41, 2596-2599 (2002).
- Meldal, M., Tornoe, C. W. Cu-catalyzed azide-alkyne cycloaddition. Chem Rev. 108, 2952-3015 (2008).
- Speers, A. E., Adam, G. C., Cravatt, B. F. Activity-based protein profiling in vivo using a copper(i)-catalyzed azide-alkyne [3 + 2] cycloaddition. J Am Chem Soc. 125, 4686-4687 (2003).
- Saxon, E., Bertozzi, C. R. Cell surface engineering by a modified Staudinger reaction. Science. 287, 2007-2010 (2000).
- Kohn, M., Breinbauer, R. The Staudinger ligation-a gift to chemical biology. Angew Chem Int Ed Engl. 43, 3106-3116 (2004).
- Romeo, E., et al. Activity-Based Probe for N-Acylethanolamine Acid Amidase. ACS Chem Biol. 10, 2057-2064 (2015).
- Ueda, N., Yamanaka, K., Yamamoto, S. Purification and characterization of an acid amidase selective for N-palmitoylethanolamine, a putative endogenous anti-inflammatory substance. J Biol Chem. 276, 35552-35557 (2001).
- Tsuboi, K., et al. Molecular characterization of N-acylethanolamine-hydrolyzing acid amidase, a novel member of the choloylglycine hydrolase family with structural and functional similarity to acid ceramidase. J Biol Chem. 280, 11082-11092 (2005).
- Tsuboi, K., Takezaki, N., Ueda, N. The N-acylethanolamine-hydrolyzing acid amidase (NAAA). Chem Biodivers. 4, 1914-1925 (2007).
- Ribeiro, A., et al. A Potent Systemically Active N-Acylethanolamine Acid Amidase Inhibitor that Suppresses Inflammation and Human Macrophage Activation. ACS Chem Biol. 10, 1838-1846 (2015).
- Zhao, L. Y., Tsuboi, K., Okamoto, Y., Nagahata, S., Ueda, N. Proteolytic activation and glycosylation of N-acylethanolamine-hydrolyzing acid amidase, a lysosomal enzyme involved in the endocannabinoid metabolism. Biochim Biophys Acta. 1771, 1397-1405 (2007).
- Wang, J., et al. Amino acid residues crucial in pH regulation and proteolytic activation of N-acylethanolamine-hydrolyzing acid amidase. Biochim Biophys Acta. 1781, 710-717 (2008).
- West, J. M., Zvonok, N., Whitten, K. M., Wood, J. T., Makriyannis, A. Mass spectrometric characterization of human N-acylethanolamine-hydrolyzing acid amidase. J Proteome Res. 11, 972-981 (2012).
- Bandiera, T., Ponzano, S., Piomelli, D. Advances in the discovery of N-acylethanolamine acid amidase inhibitors. Pharmacol Res. 86, 11-17 (2014).
- Sasso, O., et al. Antinociceptive effects of the N-acylethanolamine acid amidase inhibitor ARN077 in rodent pain models. Pain. 154, 350-360 (2013).
- Duranti, A., et al. N-(2-oxo-3-oxetanyl)carbamic acid esters as N-acylethanolamine acid amidase inhibitors: synthesis and structure-activity and structure-property relationships. J Med Chem. 55, 4824-4836 (2012).
- Ponzano, S., et al. Synthesis and structure-activity relationship (SAR) of 2-methyl-4-oxo-3-oxetanylcarbamic acid esters, a class of potent N-acylethanolamine acid amidase (NAAA) inhibitors. J Med Chem. 56, 6917-6934 (2013).
- Solorzano, C., et al. Synthesis and structure-activity relationships of N-(2-oxo-3-oxetanyl)amides as N-acylethanolamine-hydrolyzing acid amidase inhibitors. J Med Chem. 53, 5770-5781 (2010).
- Vitale, R., et al. Synthesis, structure-activity, and structure-stability relationships of 2-substituted-N-(4-oxo-3-oxetanyl) N-acylethanolamine acid amidase (NAAA) inhibitors. ChemMedChem 9. 9, 323-336 (2014).
- Fiasella, A., et al. 3-Aminoazetidin-2-one derivatives as N-acylethanolamine acid amidase (NAAA) inhibitors suitable for systemic administration. ChemMedChem 9. 9, 1602-1614 (2014).
- Armirotti, A., et al. beta-Lactones Inhibit N-acylethanolamine Acid Amidase by S-Acylation of the Catalytic N-Terminal Cysteine. ACS Med Chem Lett. 3, 422-426 (2012).
- Nuzzi, A., et al. Potent alpha-amino-beta-lactam carbamic acid ester as NAAA inhibitors. Synthesis and structure-activity relationship (SAR) studies. Eur J Med Chem. 111, 138-159 (2016).
- Bonezzi, F. T., et al. An Important Role for N-Acylethanolamine Acid Amidase in the Complete Freund’s Adjuvant Rat Model of Arthritis. J Pharmacol Exp Ther. 356, 656-663 (2016).
- Smith, P. K., et al. Measurement of protein using bicinchoninic acid. Anal Biochem. 150, 76-85 (1985).
- Speers, A. E., Cravatt, B. F. Activity-Based Protein Profiling (ABPP) and Click Chemistry (CC)-ABPP by MudPIT Mass Spectrometry. Curr Protoc Chem Biol. 1, 29-41 (2009).
- Rybak, J. N., Scheurer, S. B., Neri, D., Elia, G. Purification of biotinylated proteins on streptavidin resin: a protocol for quantitative elution. Proteomics. 4, 2296-2299 (2004).
- Penna, A., Cahalan, M. Western Blotting using the Invitrogen NuPage Novex Bis Tris minigels. J Vis Exp. (264), (2007).
- Giuffrida, A., Piomelli, D. Isotope dilution GC/MS determination of anandamide and other fatty acylethanolamides in rat blood plasma. FEBS Lett. 422, 373-376 (1998).
- Buczynski, M. W., Parsons, L. H. Quantification of brain endocannabinoid levels: methods, interpretations and pitfalls. Br J Pharmacol. 160, 423-442 (2010).

