3D Magnetic Stem Cell Aggregation and Bioreactor Maturation for Cartilage Regeneration
Summary
Chondrogenesis from stem cells requires fine tuning the culture conditions. Here, we present a magnetic approach for condensing cells, an essential step to initiate chondrogenesis. In addition, we show that dynamic maturation in a bioreactor applies mechanical stimulation to the cellular constructs and enhances cartilaginous extracellular matrix production.
Abstract
Cartilage engineering remains a challenge due to the difficulties in creating an in vitro functional implant similar to the native tissue. An approach recently explored for the development of autologous replacements involves the differentiation of stem cells into chondrocytes. To initiate this chondrogenesis, a degree of compaction of the stem cells is required; hence, we demonstrated the feasibility of magnetically condensing cells, both within thick scaffolds and scaffold-free, using miniaturized magnetic field sources as cell attractors. This magnetic approach was also used to guide aggregate fusion and to build scaffold-free, organized, three-dimensional (3D) tissues several millimeters in size. In addition to having an enhanced size, the tissue formed by magnetic-driven fusion presented a significant increase in the expression of collagen II, and a similar trend was observed for aggrecan expression. As the native cartilage was subjected to forces that influenced its 3D structure, dynamic maturation was also performed. A bioreactor that provides mechanical stimuli was used to culture the magnetically seeded scaffolds over a 21-day period. Bioreactor maturation largely improved chondrogenesis into the cellularized scaffolds; the extracellular matrix obtained under these conditions was rich in collagen II and aggrecan. This work outlines the innovative potential of magnetic condensation of labeled stem cells and dynamic maturation in a bioreactor for improved chondrogenic differentiation, both scaffold-free and within polysaccharide scaffolds.
Introduction
Magnetic nanoparticles are already used in the clinic as contrast agents for magnetic resonance imaging (MRI), and their therapeutic applications keep expanding. For example, it has recently been shown that labeled cells can be manipulated in vivo using an external magnetic field and can be directed and/or maintained at a defined site of implantation1,2,3. In regenerative medicine, they can be used to engineer organized tissues in vitro4, including vascular tissue5,6,7, bone8, and cartilage9.
Articular cartilage is immersed in an avascular environment, making repairs of the extracellular matrix components very limited when damages occur. For this reason, research is currently focused on the engineering of hyaline cartilage replacements that can be implanted at the defect site. In order to produce an autologous replacement, some research groups are exploring the use of autologous chondrocytes as a cell source10,11, while others emphasize the capacity of mesenchymal stem cells (MSC) to differentiate into chondrocytes12,13. In previous studies recapitulated here, we selected MSC, as their bone marrow sampling is fairly simple and does not require the sacrifice of healthy chondrocytes, which risk losing their phenotype14.
An early step essential to initiating the chondrogenic differentiation of stem cells is their condensation. Cell aggregates are commonly formed using either centrifugation or micromass culture15; however, these condensation methods neither present the potential to create cell clusters within thick scaffolds nor the potential to control the fusion of aggregates. In this paper, we describe an innovative approach to condensing stem cells using MSC magnetic labeling and magnetic attraction. This technique has been proven to form scaffold-free 3D constructs via the fusion of aggregates with one another to obtain a millimeter-scale cartilaginous tissue9. Magnetic seeding of thick and large scaffolds has also allowed the possibility of increasing the size of the engineered tissue, designing a shape more readily useful for implantation, and diversifying the potential for clinical applications in cartilage repair. Here, we detail the protocol for the magnetic seeding of MSC into porous scaffolds composed of natural polysaccharides, pullulan, and dextran, scaffolds previously used to confine stem cells16,17. Chondrogenic differentiation was finally performed in a bioreactor to ensure continuous nutrient and gas diffusion into the matrix core of the scaffolds seeded with a high density of cells. Besides providing nutrients, chondrogenic growth factors, and gas to the cells, the bioreactor offered mechanical stimulation. Overall, the magnetic technology used to confine stem cells, combined with dynamic maturation in a bioreactor, can markedly improve chondrogenic differentiation.
Protocol
1. Construction of the Magnetic Devices
NOTE: The devices used for cell seeding vary depending on the application (Figure 1). To form aggregates, the number of cells is limited to 2.5×105/aggregate, so the magnetic tips must be very thin (750 µm in diameter). To seed the 1.8 cm2/7 mm-thick scaffolds, the magnets must be larger (3 mm in diameter) and will ensure cell migration through the pores of the scaffold.
- Construction of a device with micro-magnets for aggregate formation (Figure 1A)
- Make micro-holes with a 0.8-mm drill through aluminum plates (3 cm in diameter and 6 mm thick).
- Insert a magnetic tip (750 µm in diameter) into each hole of the plate.
- Place this disk over a permanent neodymium magnet, which ensures magnetization to saturation.
- Construction of a device for scaffold seeding (Figure 1B)
- Cut hard polystyrene into 2.4 cm2 squares.
- Insert 9 small magnets (3 mm in diameter, 6 mm long) at an equal distance over a surface area of 1.6 cm2.
- Place this device over a permanent neodymium magnet.
2. Stem Cell Labeling
NOTE: Stem cells were labeled with 0.1 mM magnetic nanoparticles for 30 min (2.6 ± 0.2 pg iron/cell) to form aggregates, while they were labeled with 0.2 mM magnetic nanoparticles for 30 min (5 ± 0.4 pg iron/cell) to seed scaffolds. These nanoparticle concentrations and incubation times have been used previously and published for MSC and other cells18,19, and it has been determined that nanoparticles impacted neither cell viability nor MSC differentiation capacity. The iron mass incorporated by the stem cells was measured via single-cell magnetophoresis19,20.
- Culture human mesenchymal stem cells (MSC) in complete mesenchymal stem cell growth medium (MSCGM) at 37 °C and 5% CO2 until near to confluence (~90%).
- Prepare the magnetic labeling solution by mixing 0.1 or 0.2 mM maghemite citrate-coated iron oxide (γFe2O3; core: 8 nm diameter) in serum-free McCoy's 5A medium modified at Roswell Park Memorial Institute (RMPI) without glutamine and containing 5 mM sodium citrate.
- Discard the medium, rinse the cells with serum-free RPMI medium without glutamine, and add 10 mL of iron oxide nanoparticle solution per 150-cm2 culture flask, the minimum volume required to cover all the cells.
- Incubate for 30 min at 37 °C and 5% CO2 and then discard the nanoparticle solution. Rinse for 5 min with serum-free RPMI medium without glutamine to internalize the nanoparticles still attached to the plasma membrane.
- Discard the RPMI medium and add 25 mL of complete MSCGM medium per flask. Incubate overnight at 37 °C and 5% CO2.
3. Magnetic Cell Seeding
- Freshly prepare the chondrogenic medium using Dulbecco's Modified Eagle Medium (DMEM) high glucose with L-glutamine by adding 50 µM L-ascorbic acid 2-phosphate, 0.1 µM dexamethasone, 1 mM sodium pyruvate, 0.35 mM L-proline, 1% universal culture supplement containing insulin, human transferrin and selenous acid (ITS-Premix), and 10 ng/mL transforming growth factor-beta 3 (TGF-β3).
- Detach the magnetic cells using 8 mL of 0.05% trypsin-EDTA per 150-cm2 culture flask and centrifuge the dissociated cells at 260 × g for 5 min. Aspirate the medium and count the re-suspended cells.
- Place a glass-bottomed cell culture Petri dish (35 mm) on top of both magnetic devices.
- To magnetically form aggregates, add 3 mL of chondrogenic medium to the Petri dish and gently deposit the smallest volume possible (no more than 8 µL) containing 2.5×105 labeled cells per aggregate (up to 16 aggregates can be deposited). Leave the Petri dish for 20-30 min without moving it, allowing it to form spheroids, and then place the complete device, including the Petri dish containing the 16 aggregates, into the incubator at 37 °C and 5% CO2.
- Form control aggregates following the same protocol and replace the complete medium with chondrogenic medium without TGF-β3.
- To generate the 3D aggregate construct, place 2 aggregates in contact on day 8 to form 8 doublets and initiate the fusion. On day 11, merge 2 doublets to form 4 quadruplets. Finally, fuse the 4 quadruplets on day 15 to obtain the final structure.
- At the same time, form aggregates by centrifuging 2.5×105 labeled stem cells at 260 × g for 5 min in 15-mL tubes with 1.5 mL of chondrogenic medium with or without TGF-β3 (for sample and control, respectively).
- To magnetically seed scaffolds, place each dried scaffold into a Petri dish. Use polysaccharide porous scaffolds made of pullulan/dextran21. For each scaffold, dilute 2×106 labeled stem cells in 350 µL of chondrogenic medium without TGF-β3 and carefully pipette the cells onto the scaffold.
- Incubate for 5 min at 37 °C to allow for full cell penetration within the scaffold and then gently add 3 mL of chondrogenic medium with or without TGF-β3 (for sample or control, respectively) to the Petri dish.
- Incubate the cellularized scaffold on its magnetic device at 37 °C and 5% CO2 for 4 days to allow for cell migration through the scaffold pores and confinement.
- At the same time, seed scaffolds with 2×106 labeled stem cells following the same method and incubate without the magnet to obtain uniformly seeded scaffolds as positive controls.
4. Differentiation into Chondrocytes
NOTE: After 4 days of incubation, remove the magnets and continue the chondrogenic maturation either in a Petri dish (static conditions) or in a bioreactor (dynamic conditions). Negative control samples are matured in static conditions with chondrogenic medium without TGF-β3.
- In static conditions, keep the cellularized scaffolds or aggregates in the same Petri dish. Change the chondrogenic medium twice a week for 21 days.
- In dynamic conditions, prepare the bioreactor.
- Cut silicone tubing at the appropriate length according to the manufacturer's protocol.
- Autoclave all materials: 500-mL culture chamber, tubing, 2-way rotators, and cages.
- Place the bioreactor parts into a sterile microbiological safety station. Connect the tubing to the 2-way rotators and to the culture chamber following the manufacturer's instructions.
- Carefully transfer the cellularized scaffolds into sterilized cages using a sterile spatula. Place 2 scaffolds per cage. When the cages are ready, insert them into the needles of the lid to keep them from moving during further rotation. Fill the culture chamber with chondrogenic medium and close it with the lid containing the cages.
- Turn on the peristaltic pump to fill the tubing with chondrogenic medium and to eliminate air bubbles.
- Place and secure the filled chamber into the motor of the bioreactor and turn on the computer, which controls the rotations both of the arm and of the chamber.
- Apply a rotation speed of 5 rotations per min (rpm) on both the arm and the chamber. Adjust the peristaltic pump at a flow rate of 10 rpm for continuous feeding of the cellularized scaffolds.
5. RNA Extraction and Gene Expression Analysis
NOTE: Prior to RNA extraction, digest the scaffolds with an enzymatic solution.
- Prepare 1 mL of enzymatic solution by adding 100 µL of pullulanase (40 U/mL) and 50 µL of dextranase (60 mg/mL) to 850 mL of serum-free DMEM medium.
- Rinse the scaffolds twice with serum-free DMEM medium, discard the medium, and add 800 µL of the enzymatic solution per scaffold. Incubate for 15-30 min at 37 °C under gentle agitation.
- When the scaffold is completely dissolved, transfer the solution containing the cells to a 1.5-mL tube, centrifuge at 300 × g for 10 min, carefully aspirate the medium, rinse twice with sterile 1× phosphate-buffered saline (PBS), centrifuge at 300 × g for 10 min, and re-suspend the cells in the RNA isolation solution.
- To extract the RNA from the aggregates, place the spheroids in the RNA isolation solution and completely crush them using a homogenizer before performing RNA extraction.
- Isolate the RNA using a kit for total RNA extraction according to the manufacturer's instructions.
- Synthesize complementary DNA from 400 ng of total RNA using reverse transcriptase according to the manufacturer's instructions, using 250-ng random primers, 1 µL of dNTP mix (10 mM each), and 40 U/mL RNase inhibitor; the final volume of reaction is 20 µL. At the end of the reaction, add 80 µL of distilled water to obtain a final volume of 100 µL.
- For quantitative polymerase chain reaction (PCR), use a PCR mix containing a fluorescent reagent to quantify the relative expression of the genes of interest, such as aggrecan (AGC) and collagen II (Col II), with 10× diluted cDNA. Normalize the levels of gene expression with the reference gene Ribosomal Protein, Large, P0 (RPLP0). Perform the calculations with the 2–ΔΔCT formula, where ΔΔCT = ΔCT of differentiated condition – mean ΔCT of control condition, and each ΔCT represents the CT of the gene of interest – the CT of the reference gene (RPLP0).
- Determine statistical measurements as the mean values ± the standard error of the mean (SEM). Perform the analysis with n ≥ 2 independent experiments. Use Student's t-test to analyze the statistical differences between centrifuged pellets and magnetic fusions (* p < 0.05). Determine the significance with a Kruskal-Wallis test (one-way ANOVA nonparametric) to analyze statistical differences between differentiated scaffolds and with the control scaffold (* p < 0.05).
6. Histological Analysis
- Rinse the cellularized scaffolds or aggregates with sterile 1× PBS, fix them in 10% formalin solution for 1 h at room temperature, and rinse with 1× PBS.
- Remove the PBS, embed the samples in optimal cutting temperature compound (OCT), and freeze them in an isopentane bath immersed in liquid nitrogen. Store the sample at -20 °C. Cut the samples with a cryostat to obtain 8-µm cryosections for the aggregates or 12-µm for the cellularized scaffolds.
- Stain the cryosections with 0.5% toluidine blue solution for 2 min, rinse in tap water, dehydrate with 100% ethanol, clarify using toluene, and mount the slides with a mounting medium for light microscopy.
Representative Results
First, aggregates can be individually formed using micro-magnets by depositing 2.5×105 labeled stem cells (Figure 2A). These single aggregates (~0.8 mm in size) can then be fused into larger structures thanks to sequential, magnetically induced fusion. For instance, on day 8 of chondrogenic maturation, aggregates were placed in contact in pairs to form doublets; quadruplets were assembled on day 11 by merging 2 doublets; and finally, on day 15, the 4 quadruplets were fused to form a 3D construct containing 16 of the initially formed aggregates, with a total of 4×106 cells (~4 mm in size) (Figure 2B). Second, the same magnetic attraction technique has been used to form cell aggregates within scaffolds. Scaffolds were seeded over the magnetic device and showed densely condensed cells within the pores of the scaffold, at exact micro-magnet locations (Figure 2C). By contrast, when cells were seeded within a scaffold without magnetic attraction (passive seeding), they were found to be evenly distributed. Cellularized scaffolds were then inserted into cages (Figure 3A) attached to the bioreactor chamber to perform the chondrogenic process under dynamic maturation conditions (Figure 3B). Such a bioreactor improves nutrient and gas exchanges and provides mechanical stimulation by transduction. The rotation speed of both the arm and the chamber was adjusted to 5 rpm, as recommended by the constructor for soft 3D tissue regeneration. A peristaltic pump that provides a continuous supply of medium was set at 10 rpm.
For all conditions of cell organization (fused aggregates and seeding within scaffolds) and tissue maturation (within or without a bioreactor), gene expression was analyzed on day 25. The tissue formed by magnetic fusion showed a significant increase in collagen II expression compared to the pellet obtained by centrifugation (Figure 4A), together with an increased trend of aggrecan expression. For cellularized scaffolds, we obtained an increase in aggrecan and collagen II expression-significant for collagen II-when magnetic seeding was used, as compared to the scaffolds seeded without magnetic forces. In addition, the expression of both genes was much higher (significant for Col II) when magnetic seeding was combined with dynamic differentiation (Figure 4B).
Histological analyses were also performed, for instance using a toluidine blue stain to reveal the glycosaminoglycans (GAG). The sequential magnetic fusion of 16 aggregates exhibited abundant deposition of GAG, as evinced by the blue-purple color (Figure 5A). For the scaffolds, only those magnetically seeded were stained with toluidine blue. GAG content was higher when the scaffolds were differentiated in a bioreactor (Figure 5B) rather than statically (Figure 5C). Taken together, these results demonstrate the potential of magnetic aggregation and magnetic seeding within scaffolds to enhance chondrogenesis. It also indicates that the dynamic maturation conditions within the bioreactor are much more favorable for efficient differentiation.

Figure 1. Construction of the magnetic devices. (A) Example of a magnetic device for aggregate formation: the aluminum plate was drilled (0.8 µm-diameter holes) and tips were inserted in each hole and then placed on a permanent neodymium magnet, which ensured magnetization to saturation. (B) Magnetic device to seed the scaffold: hard polystyrene (24 mm2) with 9 manually made holes was placed on a permanent neodymium magnet, which ensured magnetization to saturation. Small magnets (3 mm in diameter) were then inserted into each hole to form the device. Please click here to view a larger version of this figure.
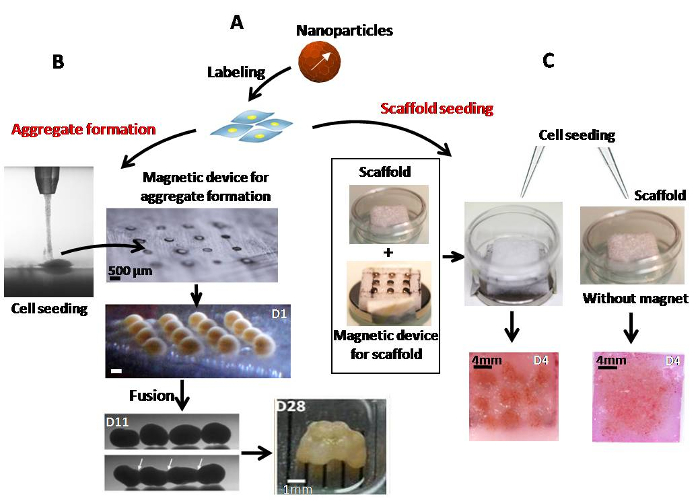
Figure 2. Magnetic labeling of stem cells and seeding. (A) Stem cells were labeled with iron oxide nanoparticles for 30 min at 37 °C. (B) Spheroids were formed from labeled cells attracted by a network of 16 micro-magnets. The aggregates were merged into quadruplets on day 11 by fusion of the doublets formed 3 days before. The quadruplets were then fused on day 15 to construct the final engineered tissue. (C) A scaffold, placed into a glass-bottomed dish, was seeded with or without magnetic forces. On day 4, spots of compacted stem cells were observed in the magnetically seeded scaffold, while the cells appeared uniformly distributed in the scaffold seeded without a magnet. Please click here to view a larger version of this figure.

Figure 3. Dynamic maturation of cellularized scaffolds. (A) After magnetic or passive seeding, cellularized scaffolds were put into cages to avoid disruption. (B) Cages, fixed using the needles of the cap, were placed into the vessel of the bioreactor filled with chondrogenic medium. The bioreactor applied biaxial rotation with an independently controlled speed (1-12 rpm and 1-35 rpm for the arm and the chamber, respectively). A peristaltic pump continuously provided medium. Please click here to view a larger version of this figure.
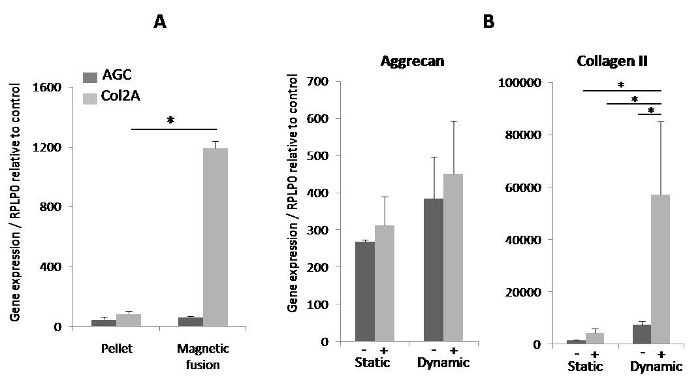
Figure 4. Expression of specific chondrogenic genes on day 25. (A) The magnetically induced fusion of MSC spheroids showed a significant increase in collagen II compared to the pellet formed by centrifugation. *denotes a statistical difference using Student's t-test (p-value < 0.05). (B) The expression of aggrecan and collagen II were clearly increased in scaffolds differentiated with a combination of magnetic seeding and dynamic maturation in a bioreactor. Gene expression was normalized to RPLP0 mRNA and expressed in arbitrary units relative to control (~1 ± SEM). Results are presented as means ± SEM of two to four independent experiments. *denotes a statistical difference using the Kruskal-Wallis test (one-way ANOVA nonparametric test) (p-value < 0.05). (-): seeding without magnet; (+): seeding with magnetic forces. Please click here to view a larger version of this figure.
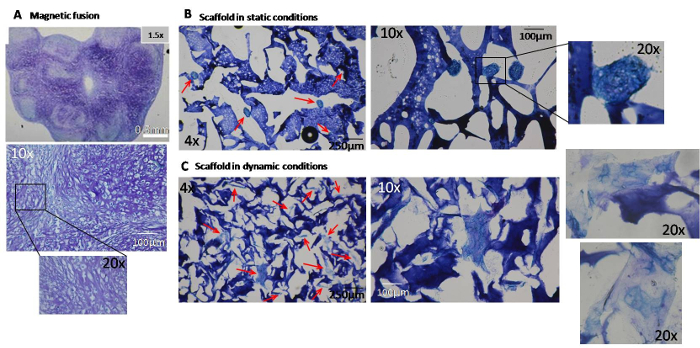
Figure 5. Histological staining of glycosaminoglycans on day 25. Glycosaminoglycan (GAG) deposits are evidenced by blue-purple coloration. (A) A positive toluidine blue stain was observed in the 8-µm cryosections of the final cartilaginous structure obtained from the sequential fusion of 16 aggregates. The 12-µm cryosections of scaffolds magnetically seeded after static (B) or dynamic (C) conditions clearly showed that GAG content was higher in scaffolds differentiated in a bioreactor. Arrows indicate aggregates of differentiated cells. Please click here to view a larger version of this figure.
Discussion
First, because the techniques presented here rely on the internalization of magnetic nanoparticles, one important issue is the outcome of the nanoparticles once they localize within the cells. It is true that iron nanoparticles may trigger potential toxicity or impaired differentiation capacity depending on their size, coating, and time of exposure19,22. However, several studies have shown no impact on cellular physiology when encapsulated iron nanoparticles were used23 in the form of magnetoferritin, a biological magnetic nanoparticle24, or were used with a simple citrate coating and adequate concentrations18. In addition, when iron oxide nanoparticles were used in similar conditions as those described in this paper (with MSC and for chondrogenesis), we recently demonstrated that a rapid and almost total degradation of the nanoparticles occurs within the endosomes in about 10 days upon cellular incorporation and MSC spheroid formation. Interestingly, this massive degradation was associated with the efficient storage of the free iron within the ferritin protein and resulted in a very limited impact on the cellular iron metabolism, boding well for future clinical applications25.
Another critical point is the requirement of cell compaction to initiate chondrogenesis. Usually, the condensation of cells is achieved through centrifugation; however, this method is limited by a low number of cells (not more than 2.5×105 cells). Past this number, the nutrients and gas cannot diffuse to the center of the aggregates, triggering cell necrosis. Here, the magnetic condensation of labeled stem cells into aggregates appears as a significant method to form 3D constructs for cartilage tissue regeneration. Such a magnetic procedure has been used by other authors to build 3D spheroids: by magnetic levitation with a magnet placed on the top of the plate after the dissociation of cells23 or with iron pins to localize the magnetic field26. However, magnetic levitation does not appear to be suited for further stages of aggregate fusion. By contrast, with the magnetic method proposed here, we can control all fusion steps to obtain a step-by-step cartilage tissue construction. In brief, this multi-step process starts with confinement of stem cells into aggregate building blocks and is followed by the fusion of these blocks into a larger structure. The critical steps of this scaffold-free stem cell aggregation procedure are the following: first, one must form each aggregate with as small a volume of cells as possible and second, one must control the fusion steps to avoid the formation of a single, large aggregate. The tissue obtained here was rich in collagen II and aggrecan. It also presented the advantages of having flexible geometry and size and of being scaffold-free.
The magnetic approach was also used to guide stem cells within thick and large scaffolds; another alternative for the design of various shapes and sizes. The critical step here is to seed the scaffolds with an appropriate volume of cells: neither too little to have an overall homogeneous distribution of cells, nor too much to avoid any cell loss. Magnetic forces were previously used to attract and retain cells within scaffolds and to enhance cell seeding27,28. Here, sufficient cell condensation within the pores of the polysaccharide scaffolds led to successful chondrogenesis. The extracellular matrix production was markedly improved when the magnetically cellularized scaffolds were subjected to the transduction/shear stress stimuli provided by the bioreactor thanks to its bi-axial rotation. It has been shown in other studies that multi-axial loading conditions improved the quality of tissues formed from chondrocytes29. This novel bioreactor concept presents a real gain when compared to the existing techniques, where only compressive forces are applied30,31,32.
In conclusion, for chondrogenic differentiation, the use of magnetic confinement of labeled stem cells to form and manipulate aggregates as well as to seed scaffolds allowed for the creation of millimeter-size cartilage cellular constructs. In addition, combining magnetic cell seeding with dynamic differentiation provides a valuable new tool for regenerative medicine applications.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge QuinXell Technologies and CellD, particularly Lothar Grannemann and Dominique Ghozlan for their help with the bioreactor. We thank Catherine Le Visage, who provided us with the pullulan/dextran polysaccharide scaffolds. This work was supported by the European Union (ERC-2014-CoG project MaTissE 648779) and by the AgenceNationalede la Recherche (ANR), France (MagStem project ANR-11 SVSE5).
Materials
| Iron oxide (maghemite) nanoparticules ( γ-Fe2O3) | PHENIX – University Paris 6 | Made and given by C. Ménager | Mean diameter of 8.1 nm and negative surface charge |
| Polysaccharide Pullulan/Dextran scaffolds | LIOAD – University Nantes | Made and given by C. Le Visage | Prepared from a 75:25 mixture of pullulan/dextran in alkaline conditions (10M NaOH). Porosity: 185-205µm; Thickness: 7mm; Surface area: 1.8cm2. |
| TisXell Regeneration System | QuinXell Technologies | QX900-002 | Biaxial bioreactor with 500 mL culture chamber |
| Cage for scaffolds: Histosette II M492 | VWR | 720-0909 | |
| Mesenchymal Stem Cell (MSC) | Lonza | PT-2501 | Three independant batches have been used |
| MSCGM BulletKit medium | Lonza | PT-3001 | For the complete medium, add the provided BulletKit (containing serum, glutamine and antibiotics) to the MSCGM medium |
| DMEM with Glutamax I | Life Technologies | 31966-021 | No sodium pyruvate, no HEPES |
| RPMI medium 1640, no Glutamine | Life Technologies | 31870-025 | No sodium pyruvate, no HEPES |
| PBS w/o CaCl2 w/o MgCl2 | Life Technologies | 14190-094 | |
| 0.05% Trypsin-EDTA (1x) | Life Technologies | 25300-054 | |
| Penicillin (10.000U/mL)/Streptomicin (10.000µg/mL) | Life Technologies | 15140-122 | |
| ITS Premix Universal Culture Supplement (20x) | Corning | 354352 | |
| Sodium pyruvate solution 100mM | Sigma | S8636 | |
| L-Ascorbic Acid 2-phosphate | Sigma | A8960 | Prepare the concentrated solution (25 mM) in distilled water extemporaneously |
| L-Proline | Sigma | P5607 | Prepare the 175 mM stock solution diluted in distilled water and store at 4°C |
| Dexamethasone | Sigma | D4902 | Prepare the 1 mM stock solution diluted in Ethanol 100% and store at -20°C |
| TGF-beta 3 protein 10µg | Interchim | 30R-AT028 | |
| Tri-sodium citrate | VWR | 33615.268 | Prepare the 1M stock solution diluted in distilled water and store at 4°C |
| Pullulanase from Bacillus acidopullulyticus | Sigma | P2986 | |
| Dextranase from Chaetomium erraticum | Sigma | D0443 | |
| NucleoSpin RNA Extraction Kit | Macherey-Nagel | 740955.5 | |
| SuperScript II Reverse Transcriptase | Life Technologies | 18064-014 | |
| Random Primer – Hexamer | Promega | C1181 | 500 µg/mL: Use diluted 1/2 and put 1 µL per sample |
| Recombinant RNAsin ribonuclease inhibitor | Promega | N2511 | 40 U/µL: put 1 µL per sample |
| PCR nucleotide dNTP mix (10mM each) | Roche | 10842321 | |
| SyBr Green PCR Master Mix | Life Technologies | 4368708 | |
| Step One Plus Real-Time PCR System | Life Technologies | 4381792 | |
| Formalin solution 10% neutral buffered | Sigma | HT5012 | |
| OCT solution | VWR | 361603E | |
| Isopentane | Sigma | M32631 | |
| Toluidine blue O | VWR | 1.15930.0025 | |
| Ethanol absolute | VWR | 20821.310 | |
| Toluene | VWR | 1.08323.1000 | |
| Mounting medium Pertex | Histolab | 840 | |
| RPLP0 Primer for qPCR | Eurogentec | 5'-TGCATCAGTAC CCCATTCTATCAT-3' ; 5'-AAGGTGTAATC CGTCTCCACAGA-3' |
|
| Aggrecan Primer for qPCR | Eurogentec | 5'-TCTACCGCTGCGAGGTGAT-3' ; 3'-TGTAATGGAACACGATGCCTTT-5' | |
| Collagen II Primer for qPCR | Eurogentec | 5'-ACTGGATTGACCCCAACCAA-3' ; 3'-TCCATGTTGCAGAAAACCTTCA-5' |
Referencias
- Naumova, A. V., Modo, M., Moore, A., Murry, C. E., Frank, J. A. Clinical imaging in regenerative medicine. Nat Biotechnol. 32 (8), 804-818 (2014).
- Edmundson, M., Thanh, N. T., Song, B. Nanoparticles based stem cell tracking in regenerative medicine. Theranostics. 3 (8), 573-582 (2013).
- Di Corato, R., et al. High-resolution cellular MRI: gadolinium and iron oxide nanoparticles for in-depth dual-cell imaging of engineered tissue constructs. ACS Nano. 7 (9), 7500-7512 (2013).
- Xu, F., et al. Three-dimensional magnetic assembly of microscale hydrogels. Adv Mater. 23 (37), 4254-4260 (2011).
- Kito, T., et al. iPS cell sheets created by a novel magnetite tissue engineering method for reparative angiogenesis. Sci Rep. 3, 1418 (2013).
- Mironov, V., Kasyanov, V., Markwald, R. R. Nanotechnology in vascular tissue engineering: from nanoscaffolding towards rapid vessel biofabrication. Trends Biotechnol. 26 (6), 338-344 (2008).
- Mattix, B. M., et al. Janus magnetic cellular spheroids for vascular tissue engineering. Biomaterials. 35 (3), 949-960 (2014).
- Henstock, J., El Haj, A. Controlled mechanotransduction in therapeutic MSCs: can remotely controlled magnetic nanoparticles regenerate bones?. Regen Med. 10 (4), 377-380 (2015).
- Fayol, D., et al. Use of magnetic forces to promote stem cell aggregation during differentiation, and cartilage tissue modeling. Adv Mater. 25 (18), 2611-2616 (2013).
- Bartlett, W., et al. Autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation for osteochondral defects of the knee: a prospective, randomised study. J Bone Joint Surg Br. 87 (5), 640-645 (2005).
- Batty, L., Dance, S., Bajaj, S., Cole, B. J. Autologous chondrocyte implantation: an overview of technique and outcomes. ANZ J Surg. 81, 18-25 (2011).
- Song, L., Baksh, D., Tuan, R. S. Mesenchymal stem cell-based cartilage tissue engineering: cells, scaffold and biology. Cytotherapy. 6 (6), 596-601 (2004).
- Boeuf, S., Richter, W. Chondrogenesis of mesenchymal stem cells: role of tissue source and inducing factors. Stem Cell Res Ther. 1 (4), 31 (2010).
- Kock, L., van Donkelaar, C. C., Ito, K. Tissue engineering of functional articular cartilage: the current status. Cell Tissue Res. 347 (3), 613-627 (2012).
- Schon, B. S., et al. Validation of a high-throughput microtissue fabrication process for 3D assembly of tissue engineered cartilage constructs. Cell Tissue Res. , (2012).
- Robert, D., et al. Magnetic micro-manipulations to probe the local physical properties of porous scaffolds and to confine stem cells. Biomaterials. 31 (7), 1586-1595 (2010).
- Luciani, N., et al. Successful chondrogenesis within scaffolds, using magnetic stem cell confinement and bioreactor maturation. Acta Biomaterialia. 37, 101-110 (2016).
- Wilhelm, C., Gazeau, F. Universal cell labelling with anionic magnetic nanoparticles. Biomaterials. 29 (22), 3161-3174 (2008).
- Fayol, D., Luciani, N., Lartigue, L., Gazeau, F., Wilhelm, C. Managing magnetic nanoparticle aggregation and cellular uptake: a precondition for efficient stem-cell differentiation and MRI tracking. Adv Healthc Mater. 2 (2), 313-325 (2013).
- Wilhelm, C., Gazeau, F., Bacri, J. C. Magnetophoresis and ferromagnetic resonance of magnetically labeled cells. Eur Biophys J. 31 (2), 118-125 (2002).
- Autissier, A., Le Visage, C., Pouzet, C., Chaubet, F., Letourneur, D. Fabrication of porous polysaccharide-based scaffolds using a combined freeze-drying/cross-linking process. Acta Biomater. 6 (9), 3640-3648 (2010).
- Singh, N., Jenkins, G. J. S., Asadi, R., Doak, S. H. Potential toxicity of superparamagnetic iron oxide nanoparticles (SPION). Nano Reviews. 1, (2010).
- Lee, J. H., Hur, W. Scaffold-free formation of a millimeter-scale multicellular spheroid with an internal cavity from magnetically levitated 3T3 cells that ingested iron oxide-containing microspheres. Biotechnol Bioeng. 111 (5), 1038-1047 (2014).
- Mattix, B., et al. Biological magnetic cellular spheroids as building blocks for tissue engineering. Acta Biomaterialia. 10 (2), 623-629 (2014).
- Mazuel, F., et al. Massive Intracellular Biodegradation of Iron Oxide Nanoparticles Evidenced Magnetically at Single-Endosome and Tissue Levels. ACS Nano. 10 (8), 7627-7638 (2016).
- Kim, J. A., et al. High-throughput generation of spheroids using magnetic nanoparticles for three-dimensional cell culture. Biomaterials. 34 (34), 8555-8563 (2013).
- Shimizu, K., Ito, A., Honda, H. Enhanced cell-seeding into 3D porous scaffolds by use of magnetite nanoparticles. J Biomed Mater Res B Appl Biomater. 77 (2), 265-272 (2006).
- Sensenig, R., Sapir, Y., MacDonald, C., Cohen, S., Polyak, B. Magnetic nanoparticle-based approaches to locally target therapy and enhance tissue regeneration in vivo. Nanomedicine (Lond). 7 (9), 1425-1442 (2012).
- Waldman, S. D., Couto, D. C., Grynpas, M. D., Pilliar, R. M., Kandel, R. A. Multi-axial mechanical stimulation of tissue engineered cartilage: review. Eur Cell Mater. 13, 66-74 (2007).
- Takahashi, I., et al. Compressive force promotes sox9, type II collagen and aggrecan and inhibits IL-1beta expression resulting in chondrogenesis in mouse embryonic limb bud mesenchymal cells. J Cell Sci. 111 (14), 2067-2076 (1998).
- Campbell, J. J., Lee, D. A., Bader, D. L. Dynamic compressive strain influences chondrogenic gene expression in human mesenchymal stem cells. Biorheology. 43, 455-470 (2006).
- Vunjak-Novakovic, G., et al. Bioreactor cultivation conditions modulate the composition and mechanical properties of tissue-engineered cartilage. J Orthop Res. 17 (1), 130-138 (1999).

