Isolation of Stem-like Cells from 3-Dimensional Spheroid Cultures
Summary
Using human primary prostate epithelial cells, we report a novel biomarker-free method of functional characterization of stem-like cells by a spheroid-based label-retention assay. A step-by-step protocol is described for BrdU, CFSE, or Far Red 2D cell labeling; three-dimensional spheroid formation; label-retaining stem-like cell identification by immunocytochemistry; and isolation by FACS.
Abstract
Despite advances in adult stem cell research, identification and isolation of stem cells from tissue specimens remains a major challenge. While resident stem cells are relatively quiescent with niche restraints in adult tissues, they enter the cell cycle in anchor-free three-dimensional (3D) culture and undergo both symmetric and asymmetric cell division, giving rise to both stem and progenitor cells. The latter proliferate rapidly and are the major cell population at various stages of lineage commitment, forming heterogeneous spheroids. Using primary normal human prostate epithelial cells (HPrEC), a spheroid-based, label-retention assay was developed that permits the identification and functional isolation of the spheroid-initiating stem cells at a single cell resolution.
HPrEC or cell lines are two-dimensionally (2D) cultured with BrdU for 10 days to permit its incorporation into the DNA of all dividing cells, including self-renewing stem cells. Wash out commences upon transfer to the 3D culture for 5 days, during which stem cells self-renew through asymmetric division and initiate spheroid formation. While relatively quiescent daughter stem cells retain BrdU-labeled parental DNA, the daughter progenitors rapidly proliferate, losing the BrdU label. BrdU can be substituted with CFSE or Far Red pro-dyes, which permit live stem cell isolation by FACS. Stem cell characteristics are confirmed by in vitro spheroid formation, in vivo tissue regeneration assays, and by documenting their symmetric/asymmetric cell divisions. The isolated label-retaining stem cells can be rigorously interrogated by downstream molecular and biologic studies, including RNA-seq, ChIP-seq, single cell capture, metabolic activity, proteome profiling, immunocytochemistry, organoid formation, and in vivo tissue regeneration. Importantly, this marker-free functional stem cell isolation approach identifies stem-like cells from fresh cancer specimens and cancer cell lines from multiple organs, suggesting wide applicability. It can be used to identify cancer stem-like cell biomarkers, screen pharmaceuticals targeting cancer stem-like cells, and discover novel therapeutic targets in cancers.
Introduction
The human prostate gland contains luminal epithelium with secretory function and basal cells underlying it along with an unusual neuroendocrine cell component. The epithelial cells, in this case, are generated from a rare population of prostate stem cells that are relatively quiescent in vivo and act as a repair system to maintain glandular homeostasis throughout life1. Despite many advances, the identification and functional isolation of prostate stem cells remains a major challenge in the field. Stem cell biomarkers, including cell surface marker-based methodologies combined with flow cytometry are commonly used for stem cell research2,3,4. However, results for enrichment and isolation vary widely as a function of marker combinations and antibody specificity5,6, raising questions about the identity of the isolated cells. Another widely used approach for stem-like cell enrichment is three-dimensional (3D) spheroid culture2,3,4. While resident stem cells are relatively quiescent in vivo with niche restraints, they undergo cell division in 3D matrix culture (both symmetric and asymmetric), generating both stem and progenitor cells that rapidly reproduce toward the lineage commitment7,8. The formed spheroids are a heterogeneous mixture containing both stem cells and progenitor cells at various stages of lineage commitment, including early and late stage progenitor cells. Thus, assays using the whole spheroids are not stem cell exclusive, making the identification of unique stem cell properties inconclusive. Therefore, it is critical to create assays to identify and separate prostate stem cells from their daughter progenitors. Towards this end, the goal of the current protocol is to establish an assay system that allows for the efficient identification and isolation of stem cells from human prostate tissues followed by robust downstream analysis of their biological functions.
Long-term 5-bromo-2′-deoxyuridine (BrdU) label-retention is widely used for in vivo and in vitro lineage tracing of stem cells based on their prolonged doubling time9,10. The current approach for prostate stem cell identification and isolation described herein is based on their relative quiescent characteristic and label-retention properties within a mixed epithelial population. Furthermore, based on the immortal strand DNA hypothesis, only stem cells can undergo asymmetric cell division. The stem cell represents the daughter cell that contains the older parental DNA while the progenitor cell, which is a committed daughter cell, receives the newly synthesized DNA. The unique stem cell property described above is exploited to perform BrdU labeling in parental stem cells in primary cultures and then track their label following BrdU-washout upon transfer to 3D anchor-free spheroid culture. While the majority of primary prostate epithelial cells retain a basal and transit amplifying phenotype in 2D culture, there is also a rare population of multipotent stem cells replenishing and maintaining the epithelial homeostasis as evidenced by formation of spheroids or fully differentiated organoids with corresponding culture media upon transfer to 3D systems3,12. In our current protocol, by using HPrEC prostate spheroids or prostasphere-based BrdU, CFSE, or Far Red retention assays followed by fluorescence activated cell sorting (FACS), we identify label-retaining stem cells in spheroids at a single cell level13.
Importantly, we further confirmed the stem cell characteristics of label-retaining cells within early-stage spheroids compared to the progenitor cells with lineage commitment. These include stem cell asymmetric division, in vitro spheroid formation ability and in vivo tissue regeneration capacity, elevated autophagy activity, augmented ribosome biogenesis and decreased metabolic activity. Subsequently, RNAseq analysis was performed. Differentially expressed genes in label-retaining spheroid cells were observed that may serve as novel biomarkers for human prostate stem cells. This spheroid-based label-retaining approach can apply to cancer specimens to similarly identify a small number of cancer stem-like cells, thus providing translational opportunities to manage the therapeutic resistant populations13. Presented below is the prostasphere-based label-retention assay using human primary prostate epithelial cells (HPrEC) as an example.
Protocol
All cell handling and media preparations should be performed with aseptic technique in a Class II biological safety cabinet (BSC).
1. Culture and maintenance of HPrEC Cells in 2D
- Coat 100 mm culture dishes with 2 mL of 2.5 µg/cm2 fibronectin solution overnight at room temperature (RT). Aspirate the solution and let the culture dishes air dry in the BSC for 45 min. Fibronectin-coated culture dishes can be stored 2–4 weeks at 4 ˚C.
- Add 9 mL of the prostate epithelial cell growth medium (e.g., PrEGM) to a fibronectin-coated 100 mm culture dish and keep the dish warm in a CO2 incubator at 37 ˚C.
- Thaw one vial of frozen HPrEC cells (2 x 105/mL) in a 37 ˚C water bath and resuspend the cells in 10 mL of warm medium.
- Centrifuge the cell suspension at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Thoroughly resuspend the cells in 1 mL of the warm culture medium. Seed cells by pipetting 1 mL of the cell suspension directly into the fibronectin-coated 100 mm culture dish containing 9 mL of the prewarmed medium (total volume of medium and cells is 10 mL). Place culture dishes back into a CO2 incubator at 37 ˚C.
- Replenish the medium every 2 days. To do so, carefully aspirate all media and add 10 mL of fresh warm medium.
- In about 5 days, ensure that the cultures are ~70–80% confluent. Perform the enzymatic digestion by incubating the cells with 3 mL of 0.05% Trypsin/EDTA at 37 ˚C for 5 min.
- Stop the digestion by adding 3 mL of warm PBS containing 10% FBS.
- Collect the cells into a 50 mL centrifuge tube and centrifuge at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Resuspend the cell pellet in 30 mL of warm medium and dispense it equally into three new fibronectin-coated 100 mm culture dishes for the next passage.
NOTE: Primary HPrEC cells can be passaged ~3–5x without altering their basal epithelial cell characteristics. Epithelial phenotype is tested by expressions of basal epithelial cell markers cytokeratin 5 and p63 with immunocytochemistry/immunofluorescent (ICC/IF) assays.
2. Labeling HPrEC with BrdU, CFSE, or Far Red pro-dyes
NOTE: The cells can proceed either to step 2.1 or step 2.2 followed by the transfer to 3D prostasphere culture as described in step 3.1.
- Single labeling of cells with BrdU
- Culture 2 x 105 HPrEC cells in fibronectin-coated 100 mm culture dishes with 10 mL of warm medium containing 1 µM of BrdU. Culture the cells for 10 days (over two passages) to ensure labeling of all cells including prostate stem and progenitor cells.
- After 10 days, carefully aspirate the PrEGM medium containing the BrdU and wash the cells with 5 mL of warm PBS. Repeat 1x.
- Perform the enzymatic digestion using 0.05% trypsin/EDTA as described in steps 1.7–1.8. Centrifuge the cells at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Resuspend the BrdU-labeled pelleted cells (5 x 104) in 500 µL of ice-cold (1:1) basement membrane matrix/medium and transfer to a 3D prostasphere culture (described in step 3.1) for 5 days to permit BrdU washout during the spheroid formation (~6 cell cycles).
- Double labeling of cells with BrdU and CFSE or Far Red pro-dyes
- If co-labeling for live cells is desired, take pre-labeled cells incubated with BrdU for 10 days from step 2.1 and co-label with 5 µM CFSE or Far Red live cell fluorescent pro-dyes for 30 min. Perform the co-labeling at Day 10.
- Carefully aspirate the medium containing BrdU and CFSE or Far Red pro-dyes. Wash the cells with 5 mL of warm PBS. Repeat the wash 1x.
- Perform enzymatic digestion using 0.05% trypsin/EDTA as described in steps 1.7–1.8. Centrifuge the cells at 500 x g for 5 min at RT. Remove and discard the supernatant.
- Resuspend the co-labeled cells (5 x 104) in 500 µL of ice-cold (1:1) basement membrane matrix/medium and transfer to a 3D prostasphere culture (described in section 3.1) for 5 days to permit BrdU and CFSE or Far Red washout during the spheroid formation (~6 cell cycles).
- Perform detection of label-retaining cells in spheres (Day 5 prostaspheres) as described in steps 4.1, 4.2, 4.3, or 5.
NOTE: Carboxyfluorescein diacetate succinimidyl ester (CFSE) and Far Red are long-lasting fluorescent pro-dyes and are well retained within the labeled cells. Intracellular esterase in live cells cleave the acetate groups that activate the green or red fluorescent molecules that are now membrane impermeant.
3. Prostasphere formation in the 3D basement membrane culture system
- Prostasphere culture in the 3D basement membrane matrix system
- Thaw the basement membrane matrix at 4 ˚C overnight and keep on ice before use.
- Gently add 1 mL of ice-cold basement membrane matrix into an equal volume of the ice-cold culture medium. Slowly mix by pipetting up and down, avoiding bubbles.
NOTE: Pre-coat the bottom of 12 well culture plate wells with 100 µL of this solution. This will minimize the number of cells that fall through the matrix and grow as a monolayer. - Resuspend 5 x 104 HPrEC cells in ice-cold (1:1) basement membrane matrix/culture medium mix in a total volume of 500 µL.
- Pipette 500 µL of this cell solution around the bottom rim of each well.
- Swirl the plate to evenly distribute the mixture around the rim of the well. Place the plate in a CO2 incubator at 37 ˚C for 30 min to allow the matrix to solidify.
- Once the matrix has solidified, cover with 1 mL of warm culture medium per well. Do not disturb the basement membrane matrix ring. Carefully aim the pipette tip and dispense the culture medium to the center of the well.
NOTE: The culture medium must be warmed to 37 ˚C when added to the solidified basement membrane matrix. The basement membrane matrix will lose its integrity and dissolve when exposed to cold/cool media. - Replenish medium every 2 days. Carefully aspirate ~500 µL of spent medium and add 500 μL of fresh warm culture medium.
- Monitor the prostasphere formation and growth for 5–7 days using an inverted microscope.
- Passaging of prostaspheres
- Harvest prostaspheres from the matrix by carefully aspirating off as much media as possible. Avoid disturbing or picking up floating pieces of the solidified matrix.
- Add 1 mL of dispase solution (basement membrane matrix:dispase at a 1:2 ratio). Mix thoroughly by pipetting up and down several times.
- Incubate the culture plates in a CO2 incubator at 37 ˚C for 30 min.
- Take images of prostaspheres that are at the bottom of the culture dish for sphere number counting and size measurements.
- Collect the sphere mixture into a 15 mL tube. Centrifuge the suspension at 500 x g for 5 min at RT to pellet the spheres. Aspirate and discard the supernatant.
- Disperse the spheres into single cells by digesting with 500 µL of warm 0.05% trypsin/EDTA and transferring the suspension into a 1.5 mL microcentrifuge tube.
- Incubate the microcentrifuge tube in a CO2 incubator at 37 ˚C for 5 min.
- Stop the trypsin action with 500 µL of warm PBS containing 10% FBS. Pass the digested sphere suspension through a 1 mL syringe with a 26 G needle 5x to dissociate the spheres into single cells.
- Centrifuge the suspension at 500 x g for 5 min to pellet the cells at RT. Aspirate the supernatant and discard.
- Resuspend the cell pellet in 1 mL of warm culture medium and filter through a 40 µm pore size nylon cell strainer.
- Centrifuge the cell suspension at 500 x g for 5 min to pellet the cells at RT. Aspirate and discard the supernatant.
- Resuspend the cell pellet in 1 mL of ice-cold (1:1) basement membrane matrix/culture medium and subculture in a 3D basement membrane matrix for the second passage of the prostasphere.
4. Identification of BrdU-retaining prostate stem cells by immunofluorescent staining
- Outgrow the attached prostaspheres followed by immunofluorescent (IF) staining for prostate stem cell 2D imaging.
- Harvest prostaspheres by dispase digestion as described in steps 3.2.1–3.2.3. Centrifuge the spheres in a 1.5 mL microcentrifuge tube at 500 x g for 5 min. Discard the supernatant.
- Resuspend spheres in 1 mL of warm culture medium.
- Incubate ~50 prostaspheres per well in 8 well chamber slides in 200 µL of culture medium in a 37 ˚C incubator overnight to allow for the attachment and outgrowth of spheres.
- On Day 2, aspirate and discard the culture medium. Wash the outgrown spheres with 200 µL of PBS for 5 min.
- Fix the spheres in 200 µL/well of ice-cold methanol at -20 ˚C for 20 min.
- Wash the spheres with 200 µL/well of PBS for 5 min. Repeat 2x.
- Acid wash spheres with 200 µL/well of 2N HCl for 30 min at RT.
- Wash the spheres with 200 µL/well of PBS for 5 min. Repeat 3x.
- Add 100 µL of blocking solution containing 5% normal goat serum in PBST (PBS with 0.25% Triton X-100) and incubate for 30 min at RT.
- Aspirate off the blocking solution and add 100 µL of PBST containing 2% normal goat serum and primary mouse anti-human BrdU antibody (1:200) to each well and incubate in a humidified box at 4 ˚C overnight.
NOTE: Mouse IgG antibody is used as a negative control. - Aspirate and remove the primary antibody solution. Wash the spheres with 200 µL of PBS for 5 min. Repeat 2x.
- Add 100 µL of PBST containing 2% normal goat serum and secondary goat anti-mouse Alexa Fluor 488 antibody (1: 500) to each well and incubate at RT in the dark for 2 h.
- Aspirate and remove the secondary antibody solution. Wash the spheres with 200 µL of PBS for 5 min. Repeat 3x.
- Mount the slides with ~25-40 µL of aqueous mounting medium containing DAPI.
- Obtain images of the stained spheres/cells with a fluorescent microscope and a color digital camera.
- Whole mount IF staining of prostaspheres for prostate stem cell 3D imaging
NOTE: For whole mount IF staining, prostaspheres are handled using 1.5 mL microcentrifuge tubes.- Harvest the prostaspheres by dispase digestion as described in steps 3.2.1–3.2.3. Centrifuge the spheres in a 1.5 mL microcentrifuge tube at 500 x g for 5 min. Aspirate the supernatant and discard.
- Resuspend and fix the spheres with 1 mL of 4% paraformaldehyde at RT for 20 min. Centrifuge the spheres at 500 x g for 5 min. Aspirate the supernatant and discard.
- Wash the spheres by resuspending the pellet in 1 mL of PBS. Centrifuge the spheres at 500 x g for 5 min. Aspirate the supernatant and discard. Repeat 1x.
- Acid wash the spheres by resuspending the pellet in 1 mL of 2N HCl for 30 min.
- Wash the spheres with 1 mL of PBS for 5 min and centrifuge at 500 x g for 5 min. Aspirate the supernatant and discard. Repeat 3x.
- Resuspend ~15–30 spheres in 100 µL of blocking solution containing 5% normal goat serum in PBST (PBS with 0.25% Triton X-100) and incubate for 30 min at RT.
- To remove the blocking solution, centrifuge the spheres at 500 x g for 5 min. Aspirate and discard the supernatant.
- Resuspend the spheres in 100 µL of PBST containing 2% normal goat serum and primary mouse anti-human BrdU antibody (1:200) and incubate at 4 ˚C overnight.
- Wash the spheres by resuspending the pellet in 1 mL of PBS. Centrifuge the spheres at 500 x g for 5 min. Aspirate the supernatant and discard. Repeat 1x.
- Resuspend the spheres in 100 µL of PBST containing 2% normal goat serum and secondary goat anti-mouse Alexa Fluor 488 antibody (1:1,000) and incubate at RT for 2 h.
- Wash the spheres by resuspending the pellet in 1 mL of PBS. Centrifuge the spheres at 500 x g for 5 min. Aspirate the supernatant and discard. Repeat 1x.
- Resuspend the spheres in 30–50 µL of aqueous mounting medium containing DAPI. To prevent flattening the spheres, dispense them into a well created from a 35 mm uncoated culture dish with a cover glass bottom. Then add a coverslip to the culture dish, covering the well.
- Acquire whole sphere Z-stack images using a transmitted light inverted fluorescent confocal microscope. Convert these Z-stack images into 3D images using an imaging software with freeform drawing capabilities.
NOTE: Mouse IgG antibody is used as a negative control.
- Paired-cell analysis (4 day protocol)
- Culture the BrdU-labeled spheres to Day 5.
- Day 1: Harvest the spheres from a basement membrane matrix with 1 mL of dispase digestion. Disperse into single cells by 500 µL of warm 0.05% trypsin/EDTA in a 1.5 mL microcentrifuge tube as described in steps 3.2.1–3.2.11.
- Resuspend the cells in 1 mL of warm culture medium.
- Plate the dispersed single cells (~300–500 per well) in 8 well chamber slides and culture in 200 µL/well culture medium overnight to allow for attachment.
- Day 2: Change culture medium with 200 µL of 2 µM cytochalasin D in the culture medium and incubate overnight at 37 ˚C to permit one cell division that pauses at late metaphase to anaphase.
- Day 3–4: Aspirate and discard the medium. Fix cells in 200 µL/well of ice-cold methanol at -20 ˚C for 20 min. Follow the immunostaining protocol for BrdU as described in steps 4.1.5–4.1.14.
- Take images of paired cells with a fluorescence microscope. Ensure that the distance between the two nuclei is less than 30 µm. Based on BrdU-retention, identify the paired stem cells as undergoing symmetric or asymmetric cell division.
NOTE: BrdU label-retaining prostate stem cells undergo symmetric division to give rise to two daughter stem cells retaining an equal amount of BrdU, whereas stem cells undergoing asymmetric division give rise to one daughter stem cell retaining all the BrdU and the other daughter progenitor cell which has lost the BrdU label.
5. Isolation of the CFSE label-retaining prostate stem cells by FACS sorting
- Day 5: Harvest the CFSE-labeled day 5 prostaspheres growing in 6 well culture plates in 3D culture by replacing the medium with 2 mL of dispase (basement membrane matrix: dispase at a 1:2 ratio). Pipette up and down several times to mix thoroughly.
- Incubate the plates in a CO2 incubator at 37 ˚C for 30 min to digest the matrix.
- Collect the sphere mixture into a 15 mL centrifuge tube. Centrifuge the spheres at 500 x g for 5 min at RT. Aspirate the supernatant and discard.
- Resuspend the sphere pellet in 500 µL of warm 0.05% trypsin/EDTA, and transfer into a 1.5 mL microcentrifuge tube.
- Incubate the spheres in a CO2 incubator at 37 ˚C for 5 min. Add 500 µL of warm PBS containing 10% FBS.
- Pass the spheres through a 1 mL syringe with a 26 G needle 5x to completely dissociate the spheres into single cells.
- Centrifuge the cells at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Resuspend the cells in 1 mL of warm culture medium containing 1 µg/mL propidium iodide (PI) to stain dead cells. Incubate for 1 min at RT.
- Centrifuge the cells at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Resuspend the cells in 1 mL of warm culture medium. Centrifuge the cells at 500 x g for 5 min at RT. Aspirate and discard the supernatant.
- Resuspend the cells in 500 µL of warm culture medium and collect the cells in 5 mL polystyrene round bottom tubes by filtering them through 35 µm pore size cell strainer snap caps.
- Perform the analysis of trypsin-dispersed CFSE-labeled prostasphere cells with a single-channel FACS analyzer.
- Gate the subpopulations of fractionated CFSEHi, CFSEMed, and CFSELo cells. Use the negative and positive controls for gating.
NOTE: The presence of FACS sorted CFSE label-retaining prostate stem cells (see Hu et al.13) can be confirmed by green fluorescence microscopy and their larger cell size compared to non-retaining cells. This larger cell size is a property of prostate stem cells relative to progenitor cell populations. This may be different with other tissue types.
Representative Results
Primary normal human prostate epithelial cells are placed into fibronectin-coated culture dishes and cell growth is maintained in 2D culture (Figure 1a). Upon transfer into 3D culture with a basement membrane matrix, differentiated epithelial cells slowly die out. Only prostate stem cells can survive in an anchor-free culture and form spheroids in 5 days (Figure 1b).
Dual labeling of prostate epithelial cells in 2D culture followed by spheroid formation in 3D culture indicates the colocalization of BrdU, CFSE, and Far Red in the same label-retaining cells (Figure 2a-i).
Label-retaining cells show stem cell characteristics in Day 5 spheroids. Dual immunostaining shows that label-retaining cells exhibit lower levels of cytokeratin protein KRT14; decreased cell junction protein E-cadherin14; increased stem cell early marker proteins Wnt10B13,15,16 and ALDH1A1; increased autophagy protein LC3, an indicator of autophagy flux activity17; and increased myosin IIB (Figure 3a-f).
A spheroid-based label-retention assay also successfully detects cancer stem-like cells in prostate cancer specimens (Figure 4). This will enable the discovery of true biomarkers for cancer stem-like cells and has the potential to identify novel therapeutic targets for prostate cancer.
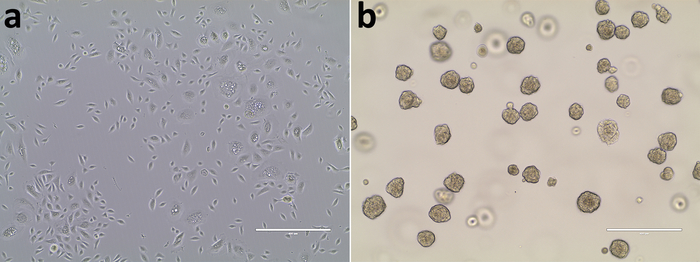
Figure 1: Maintenance of HPrEC in 2D culture and spheroid formation in 3D culture. (a) A 2D primary culture of HPrEC was BrdU-labeled and transferred to 3D culture with prostasphere formation on Day 5 (b). Scale bars = 400 µm. Please click here to view a larger version of this figure.
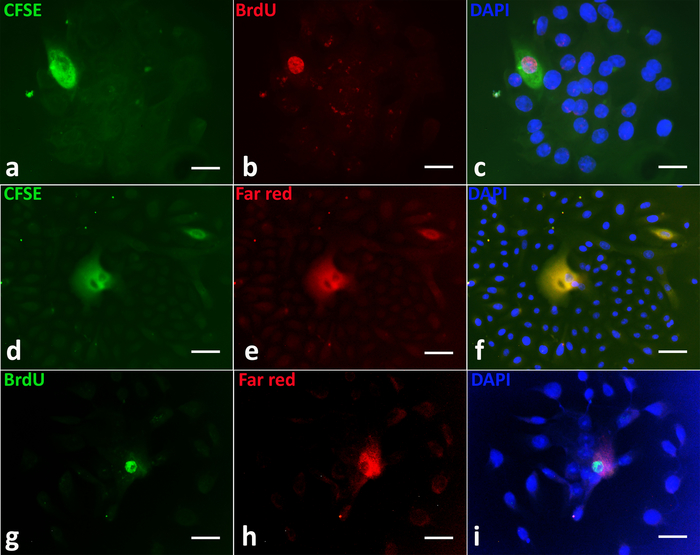
Figure 2: Identification of long-term label-retaining cells in primary prostaspheres. Double labeling of BrdU (red) and CFSE (green); CFSE (green) and Far Red (red); BrdU (green) and Far Red (red) identified the same stem-like cells with retention of parental DNA. Any BrdU, CFSE, or Far Red labels in rapidly dividing progenitor cells (DAPI, blue) were diluted and lost (a-i). Representative images show BrdU/CFSE (a-c) (upper panel), CFSE/Far Red (d-f) (middle panel), and BrdU/Far Red (g-i) (lower panel) co-labeling in single prostasphere (PS) cells. Scale bars = 50 µm. Please click here to view a larger version of this figure.
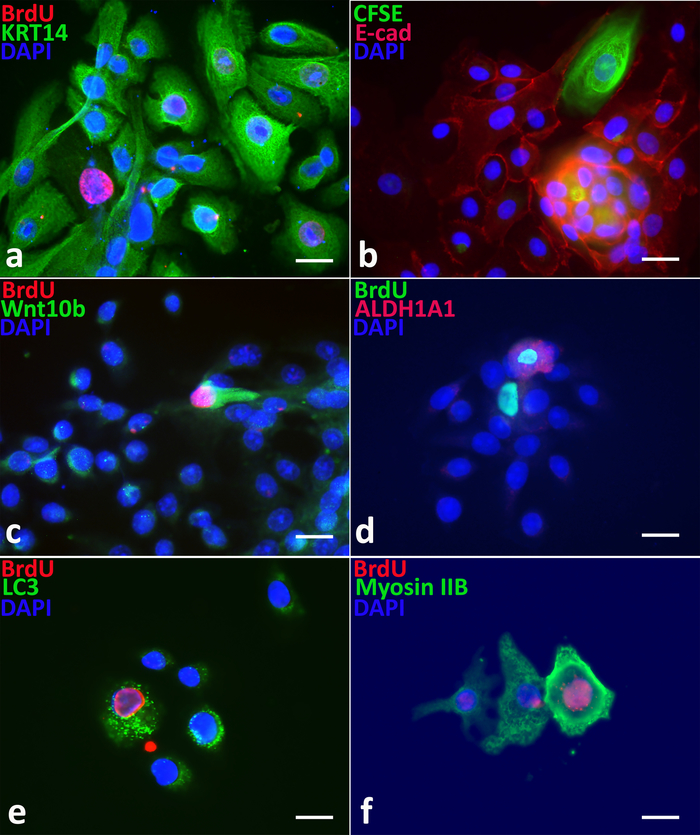
Figure 3: Label-retaining PS cells exhibiting stem cell properties. As compared to non-label-retaining progenitor cells, BrdU or CFSE label-retaining stem cells exhibit (a) lower levels of cytokeratin 14 (KRT 14), (b) decreased levels of E-cadherin, (c) elevated levels of Wnt10B, (d) higher levels of ALDH1A1, (e) increased LC3, and (f) increased myosin IIB proteins. Scale bars = 50 µm. Please click here to view a larger version of this figure.
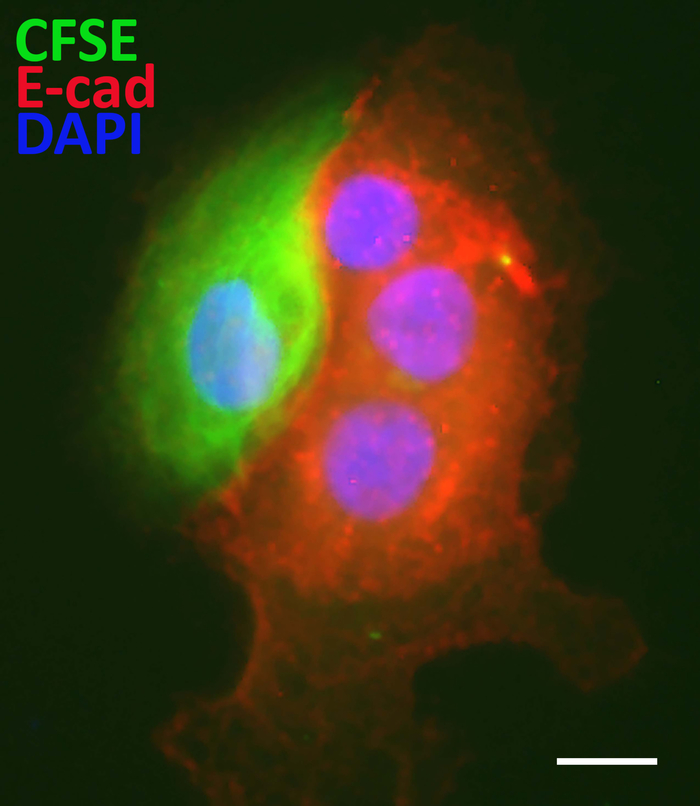
Figure 4: Using the sphere-based label-retaining assay for identification of cancer stem-like cells. CFSE label-retaining cancer stem-like cells in spheroids derived from human prostate cancer specimens exhibited reduced E-cadherin protein relative to the non-labeled progenitor cells. Scale bars = 50 µm. Please click here to view a larger version of this figure.
Discussion
Flow cytometry using multiple stem cell surface markers is a commonly used approach for stem cell research despite lacking both specificity and selectivity1,5,6. While spheroid formation in a 3D culture system is another useful method in enriching the rare stem cell population from primary epithelial cells, including HPrEC, the resulting spheroids are still a heterogeneous mixture of stem and progenitor cells2,3,4. In prostaspheres, compared to the rapidly proliferating progenitor cells, the relatively quiescent character of stem cells allows them to be identified by long-term label-retention assay.
The methods summarized in this paper describe the labeling of HPrEC using BrdU, CFSE, and/or Far Red pro-dyes13. While BrdU, CFSE, and/or Far Red pro-dyes in rapidly proliferating cells are all quickly diluted by each cell division, there is an additional mechanism that accounts for the loss of BrdU known as immortal strand DNA segregation11. This is when the stem cell undergoes asymmetric division, giving rise to one daughter stem cell and a progenitor cell. The daughter stem cell retains all the old parental DNA with BrdU label, while the daughter progenitor cell receives the newly synthesized DNA without the BrdU label. Therefore, a label-retention assay using BrdU allows for clearer and easier identification of label-retaining stem cells by immunofluorescent staining. This is especially useful to confirm stem cell biomarkers by IF double staining using two different antibodies. One major limitation of BrdU labeling is that cells must be fixed for IF staining, thus preventing further functional studies following BrdU labeling and cell fixation.
To solve this issue, two fluorescent pro-dyes for live HPrEC labeling were tested in label-retaining assays. Results indicated that there was a complete overlap of BrdU, CFSE, and Far Red labeled sphere cells. BrdU label could be substituted with CFSE or Far Red in sphere-based label-retaining assays to allow visualization of live cells for imaging and separation by FACS, promoting functional characterization of isolated live stem cells using both in vitro cell culture assays and in vivo xenograft assays13. CFSE or Far Red label-based FACS sorted live stem and progenitor cells could also be used for RNA-seq including single cell RNA-seq, which is very powerful in identifying novel stem cell gene markers and signaling pathways as well as epithelial cell lineage hierarchy.
The novel biomarker-free method of functional characterization of stem cells by a spheroid-based label-retention assay presented here can identify cancer stem-like cells from primary patient specimens and cancer cell lines. Thus, it provides an avenue for discovering novel biomarkers of cancer stem-like cells and developing effective therapeutics targeting cancer13.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This study was supported by grants from the National Cancer Institute R01-CA172220 (GSP, WYH), R01-ES02207 (GSP, WYH). We thank the Flow Cytometry Core at the University of Illinois at Chicago for assistance on cell sorting.
Materials
| 0.05% Trypsin-EDTA | Gibco | 25300-054 | |
| 1 mL tuberculin syringes | Bectin Dickinson | BD 309625 | |
| 1.5 mL microcentrifuge tubes, sterile | |||
| 100 mm culture dishes | Corning/Falcon | 353003 | |
| 12-well culture plate | Corning/Falcon | 353043 | |
| 15 mL centrifuge tubes | Corning/Falcon | 352097 | |
| 22 x 22 mm coverslips, sq | Corning | 284522 | For MatTek 35 mm culture dish |
| 24 x 50 mm coverslips | Corning | 2975245 | |
| 26G x 1.5 inch hypodermic needle | Monoject | 1188826112 | |
| 2N HCl | |||
| 35 mm culture dish with cover glass bottom | MatTek Corp | P35G-0-10-C | Glass bottom No. 0, uncoated,  irradiated irradiated |
| 40 µm pore nylon cell strainer | Corning | 352340 | |
| 5% CO2 culture incubator, 37 °C | Forma | ||
| 50 mL centrifuge tubes | Corning/Falcon | 352098 | |
| 5mL Polystyrene Round-Bottom Tube with strainer snap cap | Corning | 352235 | 35 µm nylon mesh |
| 6-well culture plates | Corning | 353046 | |
| 8-well chamber slides | Millipore Sigma | PEZGS0816 | |
| Aqueous mounting medium containing DAPI | Vector Laboratories | H-1200 | A nuclear fluorescent dye |
| Biological safety cabinet, Level 2 certified | |||
| BrdU (5-bromo-2′-deoxyuridine) | Sigma-Aldrich | B5002 | 1 mM stock solution in DMSO |
| Centrifuge for 1.5 mL microcentrifuge tubes | Eppendorf | ||
| Centrifuge for 15 mL tubes | Beckman Coulter | Allegra 6 | |
| CFSE (carboxyfluorescein succinimidyl ester) | Thermo Fisher Scientific | C34554 | 5 mM stock solution in DMSO |
| cytochalasin D | Thermo Fisher Scientific | PHZ1063 | |
| Dispase 1U/mL | StemCell Technologies | 07923 | |
| FACS CellSorter MoFlo XDP | Beckman Coulter | s | |
| Far-Red pro-dye | Thermo Fisher | C34564 | 5 mM stock solution in DMSO |
| Fetal Bovine Serum (FBS) | |||
| Fibronectin | Sigma-Aldrich | F0895 | For coating 100 mm culture dishes |
| Fluorescent microscope with color digital camera | Carl Zeiss | Axioskop 20 fluorescent microscope; color digital Axiocamera | |
| Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 488 | Thermo Fisher | A-11029 | |
| HPrEC (Primary normal human prostate epithelial cells) | Lifeline Cell Technology | FC-0038 | Pooled from 3 young (19-21yr od) disease-free organ donors; 1 x 105 cells/mL; stored in liquid nitrogen |
| ice bucket and ice | |||
| Inverted microsope with digital camera | |||
| Matrigel, low growth factor, phenol-red free | Corning | 356239 | |
| Methanol | Corning | A452-4 | |
| Mouse anti-BrdU antibody | Cell Signaling | 5292S | |
| Mouse IgG antibody (negative control) | Santa Cruz Biotechnology | sc-2025 | |
| Normal goat serum | Vector Laboratories | S-1000 | |
| Phosphate Buffered Saline (PBS), pH 7.4 | Sigma-Aldrich | P5368-10PAK | |
| Pipettors and tips, various sizes | |||
| PrEGM (ProstaLife Epithelial Cell Growth Medium) | Lifeline Cell Technology | LL-0041 | |
| Propidium Iodide (PI) | R & D Systems | 5135/10 | 10 μg/mL PI in PBS stored at 4 °C in the dark |
| Serological pipets, various sizes | |||
| Software for sphere counting and size measurements | |||
| Software: 3D images using Imaris an imageing software with freeform drawing capabilities | |||
| Triton X-100 | Millipore Sigma | T8787 | |
| Water bath, 37 °C | |||
| z-stack images using a transmitted light inverted fluorescent confocal microscope |
Referencias
- Leong, K. G., Wang, B. E., Johnson, L., Gao, W. Q. Generation of a prostate from a single cell. Nature. 456, 804-808 (2008).
- Xin, L., Lukacs, R. U., Lawson, D. A., Cheng, D., Witte, O. N. Self-renewal and multilineage differentiation in vitro from murine prostate stem cells. Stem Cells. 25, 2760-2769 (2007).
- Hu, W. Y., et al. Estrogen-initiated transformation of prostate epithelium derived from normal human prostate stem-progenitor cells. Endocrinology. 152, 2150-2163 (2011).
- Hu, W. Y., Shi, G. B., Hu, D. P., Nelles, J. L., Prins, G. S. Actions of endocrine disrupting chemicals on human prostate stem/progenitor cells and prostate cancer risk. Molecular and Cellular Endocrinology. 354, 63-73 (2012).
- Collins, A. T., Berry, P. A., Hyde, C., Stower, M. J., Maitland, N. J. Prospective identification of tumorigenic prostate cancer stem cells. Investigación sobre el cáncer. 65, 10946-10951 (2005).
- Vander Griend, D. J., et al. The role of CD133 in normal human prostate stem cells and malignant cancer-initiating cells. Investigación sobre el cáncer. 68, 9703-9711 (2008).
- Wang, J., et al. Symmetrical and asymmetrical division analysis provides evidence for a hierarchy of prostate epithelial cell lineages. Nature Communications. 5, 4758 (2014).
- Neumuller, R. A., Knoblich, J. A. Dividing cellular asymmetry: asymmetric cell division and its implications for stem cells and cancer. Genes and Development. 23, 2675-2699 (2009).
- Cicalese, A., et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell. 138, 1083-1095 (2009).
- Klein, A. M., Simons, B. D. Universal patterns of stem cell fate in cycling adult tissues. Development. 138, 3103-3111 (2011).
- Cairns, J. Mutation selection and the natural history of cancer. Nature. 255, 197-200 (1975).
- Karthaus, W. R., et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell. 159, 163-175 (2014).
- Hu, W. Y., et al. Isolation and functional interrogation of adult human prostate epithelial cells at single cell resolution. Stem Cell Research. 23, 1-12 (2017).
- Bryant, D. M., Stow, J. L. The ins and outs of E-cadherin trafficking. Trends in Cell Biology. 14, 427-434 (2004).
- Clevers, H., Loh, K. M., Nusse, R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science. 346, 1248012 (2014).
- Madueke, I., et al. The role of WNT10B in normal prostate gland development and prostate cancer. The Prostate. 79 (14), 1692-1704 (2019).
- Guan, J. L., et al. Autophagy in stem cells. Autophagy. 9, 830-849 (2013).

