Summary
We developed a lumbar intervertebral disc degeneration mouse model by resection of L3–L5 spinous processes along with supra- and inter-spinous ligaments and detachment of paraspinous muscles.
Abstract
Intervertebral disc degeneration (IDD) is a common pathological change leading to low back pain. Appropriate animal models are desired for understanding the pathological processes and evaluating new drugs. Here, we introduced a surgically induced lumbar spine instability (LSI) mouse model that develops IDD starting from 1 week post operation. In detail, the mouse under anesthesia was operated by low back skin incision, L3–L5 spinous processes exposure, detachment of paraspinous muscles, resection of processes and ligaments, and skin closure. L4–L5 IVDs were chosen for the observation. The LSI model develops lumbar IDD by porosity and hypertrophy in endplates at an early stage, decrease in intervertebral disc volume, shrinkage in nucleus pulposus at an intermediate stage, and bone loss in lumbar vertebrae (L5) at a later stage. The LSI mouse model has the advantages of strong operability, no requirement of special equipment, reproducibility, inexpensive, and relatively short period of IDD development. However, LSI operation is still a trauma that causes inflammation within the first week post operation. Thus, this animal model is suitable for study of lumbar IDD.
Introduction
Intervertebral disc degeneration (IDD) is commonly seen in aging and even young people caused by many factors1. Surgery for patients who suffer from IDD, causing low back pain and impaired movement, is usually performed at a later stage or in severe cases and has potential risks such as nonunion or infection2. Ideal non-operative treatment requires comprehensive understanding of the IDD mechanism. The IDD animal model serves as a crucial tool for studies of IDD mechanism and evaluation of IDD treatment.
Larger animals have been chosen for IDD models such as primates, sheep, goats, dogs, and rabbits due to their similarity with human anatomical structure to a great extent and the strong operability in terms of size of intervertebral discs (IVDs)3,4,5,6,7,8. However, these animal models are time-consuming and cost-intensive9. Mouse IVD is a poor representation of the human IVD based on geometrical measurements of the aspect ratio, nucleus pulposus to disc area ratio, and normalized height10. Despite the difference in size, mouse lumbar IVD segment exhibits mechanical properties similar to human IVD such as compression and torsion stiffness11. In addition, mouse IDD model has the advantage of low cost, relatively short IDD development, and more options for genetically modified animals and antibodies utilized in further mechanistic studies12,13,14,15.
Experimental-induced IDD models vary from the inducers and applications. For example, collagenase-induced extracellular matrix (ECM) degeneration is appropriate for ECM regeneration research16. Genetically modified phenotype are suitable for studying the gene function in the IDD process and in genetic therapies17. Annulus fibrosus incision and smoke models mimic trauma and non-inflammation induced IDD12,18.
Spinal instability (SI) leads to an unstable spine that is not in an optimal state of equilibrium. It can be caused by abnormal movement of a lumbar motion segment due to the weakness of the surrounding supportive tissue such as ligaments and muscles. It is also commonly seen post spinal fusion operation19. SI is considered as the main cause of IDD. Therefore, we aim to develop a SI mice model (focused on lumbar spine) that mimics the human IDD process20,21.
In the protocol, we introduced the procedure of establishing lumbar spinal instability (LSI) mouse model by the resection of lumbar third (L3) to lumbar fifth (L5) spinous processes along with the supraspinous and interspinous ligaments (Figure 1A,B). The animal model develops IDD as early as 1-week post-surgery as shown by hypertrophy and porosity in endplates (EPs). IVD volume starts to decrease 2 weeks post-surgery through 16 weeks along with increased IVD score, which indicates the degree of IDD. We believe the detailed and visualized procedure is useful for researchers to establish the LSI mouse model in their laboratory and apply to IDD research as needed.
Protocol
The investigations described conform to the Guidelines for the Care and Use of Laboratory Animals of the National Institutes of Health and were approved by Shanghai University of Traditional Chinese Medicine Animal Care and Use Committee. All surgical manipulations were performed under deep anesthesia and the animals did not experience pain at any stage during the procedure.
1. Pre-operation preparation
- Instrument sterilization: Steam-sterilize surgical instruments in an autoclave (121 °C for 15 min) prior to the surgery. Pack instruments in a metal container and maintain them until they are used in the surgery.
- Surgery platform setup: Assign a bench area of at least 60 cm x 60 cm for the operation. Clean the surface of area with 75% alcohol and cover with a disposable medical towel. Place a sterile surgical instruments pack, reagents, surgical items onto a disposable medical towel within the upper 1/3 of area. Leave the remaining 2/3 of area clean for surgical operation. Add a hotpad underneath surgical pad for thermal support.
- Animal preparation
- Place the animal (C57BL/6J mice, male, 8-week old) into the induction chamber. Turn on the vaporizer at an induction level of 4% for isoflurane and 4 L/min for oxygen. After the animal is fully anesthetized, maintain the anesthetic with the nose cone and the anesthetic delivery at a level of 1.5% for isoflurane and 0.4 L/min for oxygen during surgery. Monitor the animal for respiration.
- Apply chlortetracycline hydrochloride eye ointment to prevent corneal dryness during the surgery.
- Shave the surgical area on the dorsal surface from the lower thoracic region to the top of the sacral region using a small animal trimmer. Remove the shaved fur with tissue wipes.
- Apply depilatory cream onto the shaved area and leave it there no longer than 3 min. Remove the cream with gauze and flush with 2 mL of 0.9% sterile saline.
- Place a custom-made surgical cylindrical pad (Figure 2A) under the abdomen of the mouse to raise up the lumbar spine and facilitate the surgical operation.
2. Exposure of the lumbar third to lumbar fifth (L3–L5) spinous processes
- Use the index finger to touch the subcutaneous spinous processes of the lumbar vertebrae, which are more outward, and compare with thoracic vertebrae and sacral vertebrae to identify the lumbar region.
- Rinse the skin using 75% alcohol. Perform a 3–4 cm midline skin incision over the lumber region from the mid-thoracic region to the hip using a scalpel blade to expose the fascia.
- Identify the lumbar spine by the morphology of the posterior fascia inserted onto the tips of the spinous processes. In detail, the third lumbar (L3) to the first sacral (S1) fasciae are distinct from other fasciae by their “V” shapes. The last “V” tip connects to the first sacral (S1) fascia and the first “V” tip corresponds to the L3 spinous process (Figure 2B).
- Make the posterior paraspinous muscle incisions along the spinous processes from L3 to L5 on both sides laterally with a scalpel blade (Figure 2C). Control the incision depth towards the facets to reduce hemorrhage.
- Separate the muscle layers using two ophthalmic forceps to expose L3 to L5 spinous processes and supraspinous ligaments.
3. Resection of L3–L5 spinous processes along with the ligaments
- Separate individual spinous processes by cutting off interspinous ligaments using Venus shears (Figure 2D).
- Resect the L3–L5 spinous processes along with the interspinous ligaments with Venus shears (Figure 2E).
- Suture the skin incision with sterile silk braided (suture size 5.0) without reattachment of the paravertebral muscles.
- Apply Chlortetracycline Hydrochloride Eye Ointment to the surgical site.
- Administer Buprenorphine-SR (25 uL per gram of mouse weight) immediately after LSI surgery for analgesia.
- Place the animals in a warm chamber and monitor during recovery from the anesthesia. Monitor food and water intake before returning the animals to the home cage.
- Monitor the animal once daily for the first 3 days after operation. The animal should be able to have a normal appetite and should heal with no sign of pus, hemorrhage, or swelling. They may have minor impairment in locomotion.
- Carry out sham operations only by the detachment of the posterior paravertebral muscles from the L3–L5 vertebrae.
Representative Results
The LSI mouse model is applied in the studies of IDD mechanism, IDD treatment, endplate (EP) degeneration such as sclerosis, and sensory innervation in EP20,21,22,23. The LSI mouse develops IDD and EP degenerative changes, as identified, by decreased IVD volume and height, increased EP volume, and increased IVD and EP scores.
The dissected and fixed lower thoracic and lumbar spine were examined by high-resolution Micro Computed Tomography (μCT) as previously described20,21. The lower thoracic lumbar with ribs were included for the identification of L3–L5 vertebrae (Figure 3A). X-rays of the L3–L5 spine on a lateral view indicate existence and inexistence of spinous processes in Sham and LSI groups (Figure 3B). The results are clearer by 3D-reconstruction of L3–L5 spine on a left anterior oblique view (Figure 3C) and by transverse image of a L3–L5 vertebrae (Figure 3D).
Coronal images of the L4–L5 IVD were used to perform 3D histomorphometric analyses of IVD20 (Figure 4A). IVD volume is defined as the region of interest (ROI) covering the whole invisible space between L4 and L5 vertebrae. Parameter: TV (total tissue volume) was used for 3D structural analysis (Figure 4B). IVD volume significantly increased 1 week post-surgery and started to decrease from 2 weeks to 16 weeks post operation as observed in Figure 4C.
The height of IVD space varied from the anterior to the posterior (Figure 4E,G). LSI had a significant effect on the rear site. Thus, the posterior one-third coronal plane of IVD space was chosen for IVD height measurement (Figure 4D,E). IVD height decreased from 2 weeks to 16 weeks post-surgery (Figure 4F), which was consistent with the findings in IVD volume (Figure 4C).
Coronal images of the L4–L5 IVD space were applied to 3D histomorphometric analyses of both cranial and caudal endplates (Eps) (Figure 5A). Endplate (EP) volume is defined to cover visible bony plate close to the vertebrae (Figure 5A,B)21. The anterior one-fourth coronal plane of five consecutive images of cranial EP were used for 3D reconstruction (Figure 5C), which showed increased cavities within cranial EP in LSI mice (Figure 5D). The results were also indicated by an increased percentage of trabecular separation values which were greater or equal to 0.089 (Figure 5E). Meanwhile, EP volumes significantly increased post-surgery (Figure 5F). Caudal EPs exhibit similar phenotype by LSI (Figure 5G,H), indicating LSI leads to EP hypertrophy and an increase in cavities.
L5 vertebral bodies were reconstructed by drawing the outline of all transverse sections of each L5 vertebral body without accessories and converting all 2D images to a 3D model. The construction and analysis were done with commercial software (e.g., NRecon v1.6 and CTAn v1.9, respectively). The volumes of L5 vertebra slightly increase post-surgery but only have statistic difference between sham group and 16-week LSI group (Figure 6B). A significant decrease in BV/TV was also present 16 weeks post-surgery, indicating that LSI causes vertebral bone loss at a later stage (Figure 6A,C).
LSI induces IVD degeneration and EP degeneration as indicated by increased IVD and EP scores24 (Figure 7A,C). A reduction in intracellular vacuoles of nucleus pulposus cells was accelerated in LSI groups (Figure 7B). Cavities increase in LSI EPs (Figure 7D) accompanied with increased number of osteoclasts as indicated by Trap staining (Figure 7E,F).
The data was shown as mean ± s.d. Statistical significance was determined by a Student’s t-test. The level of significance was defined as p < 0.05. All data analyses were performed using SPSS 15.0.
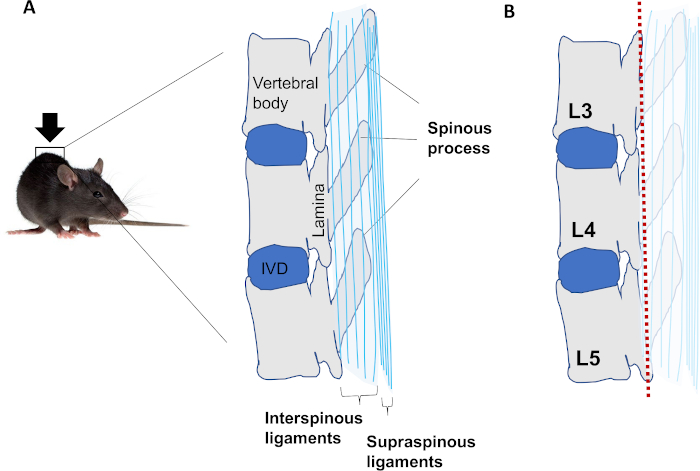
Figure 1: Schematic of LSI mouse model. (A) Anatomy of L3–L5 vertebrae in the lower back area of the mouse. (B) Resection of spinous processes along with interspinous ligaments and supraspinous ligaments (marked pale). A red dotted line indicates a section plane. Please click here to view a larger version of this figure.
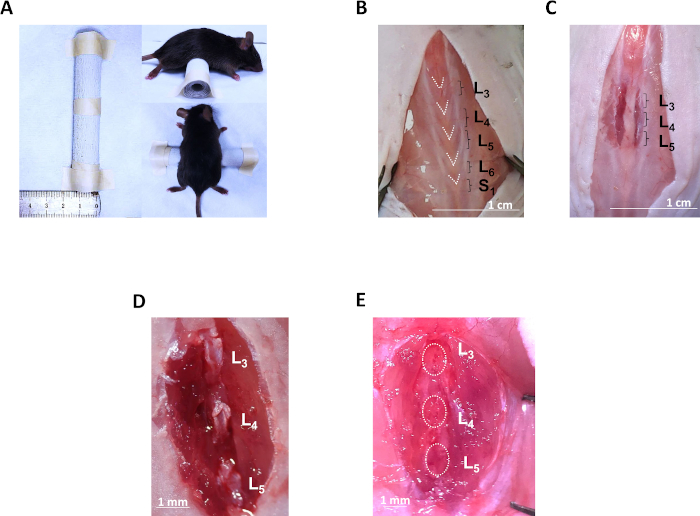
Figure 2: Exposure of L3–L5 spinous processes and LSI operation. (A) A custom-made cylindrical pad is placed under the mouse’s abdomen. (B) Exposure of the lumbar fasciae and identification of L3 to S1 spinous processes by “V” shapes. (C) Lateral paraspinous muscle incisions on both sides of L3 to L5 spinous processes. (D) Exposure of individual spinous processes by cutting off interspinous ligaments. (E) Resection of L3–L5 spinous processes with inter- and supra-spinous ligaments. Please click here to view a larger version of this figure.

Figure 3: LSI identification by μCT. (A) Localization of L3–L5 vertebrae by ribs with thoracic vertebrae in X-rays. (B) X-rays on a lateral view and (C) 3D reconstruction on a left anterior oblique view of L3–L5 vertebrae in Sham and LSI groups. (D) Transverse plane of lumbar vertebra with the resection of the spinous process. (D) has been modified from Bian et al.21. Please click here to view a larger version of this figure.
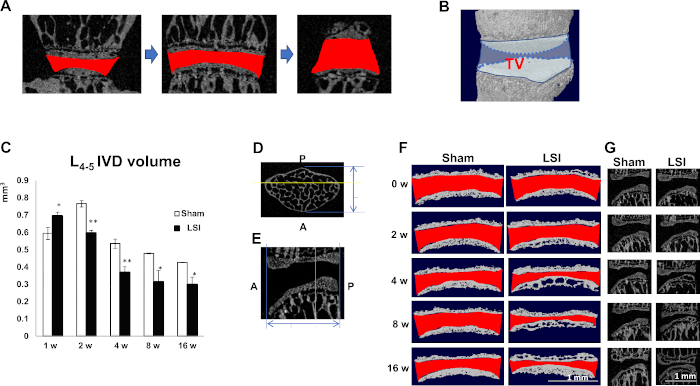
Figure 4: LSI reduces IVD volume. (A) Consecutive images of the invisible space (Red) between L4 and L5 EPs are used for 3D reconstruction. (B) IVD volume is defined by the TV of (A). (C) Quantification of L4–L5 IVD volume at five timepoints post-operation. N = 8 per group. Data is shown as mean ± s.d. *p < 0.05, **p < 0.01 versus Sham. (D) Transverse plane and (E) mid-sagittal plane of lumbar vertebral bodies. Blue double-arrows indicate anteroposterior diameter. Yellow line indicates posterior 1/3 plane. (F) Reconstruction of cranial and caudal EPs using five consecutive images of posterior 1/3 coronal plane of L4–L5. Red indicates IVD space. (G) Mid-sagittal plane of L4–L5. (C) has been modified from Bian et al.20. (F,G) have been modified from Bian et al.21. Please click here to view a larger version of this figure.
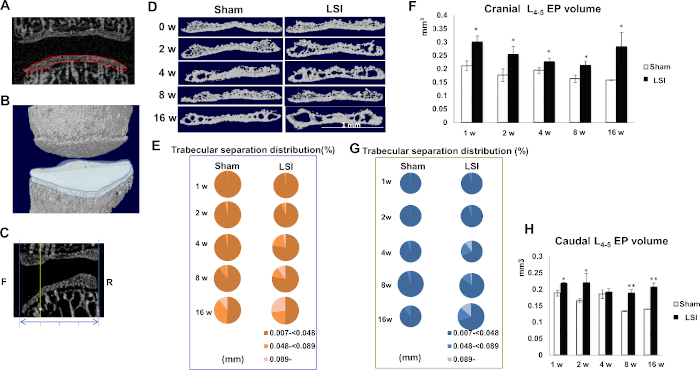
Figure 5: LSI induces EP hypertrophy and porosity. (A) Coronal plane of L4–L5. Red dotted line indicates image of caudal EP used for 3D construction. (B) Reconstruction of caudal L4 and cranial L5. Blue cartoon indicates caudal EP of L4–L5. (C) Mid-sagittal plane of L4–L5. Blue double-arrows indicate anteroposterior diameter. Yellow line indicates anterior 1/4 plane. (D) Reconstruction of cranial EPs by five consecutive images of anterior 1/4 plane of L4–L5. (E,G) Percentage of trabecular separation distribution of cranial (E) and caudal (G) EPs obtained from μCT analyses. (F,H) Quantification of cranial (F) and caudal (H). L4–L5 EP volume in indicated timepoints. N = 8 per group. Data is shown as mean ± s.d.* p < 0.05 versus. Sham. (D–H) have been modified from Bian et al.21. Please click here to view a larger version of this figure.
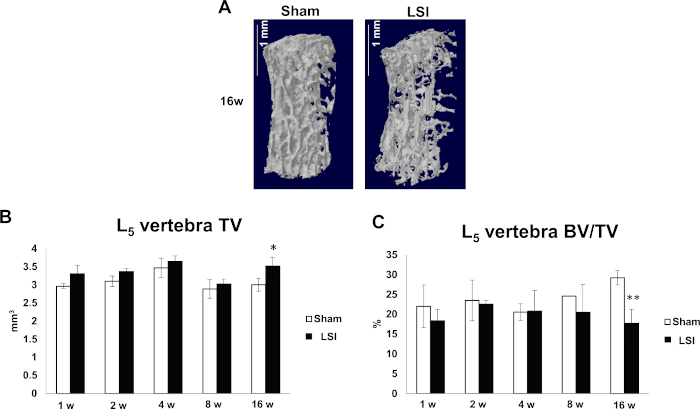
Figure 6: LSI causes vertebral bone loss at late stage. (A) Reconstruction of L5 vertebral bodies in 16-week Sham and LSI groups. (B,C) Quantification of L5 vertebra TV (B) and BV/TV (C). N = 8 per group. Data is shown as mean ± s.d.* p < 0.05, ** p < 0.01 versus. Sham. (B) has been modified from Bian et al.21. Please click here to view a larger version of this figure.
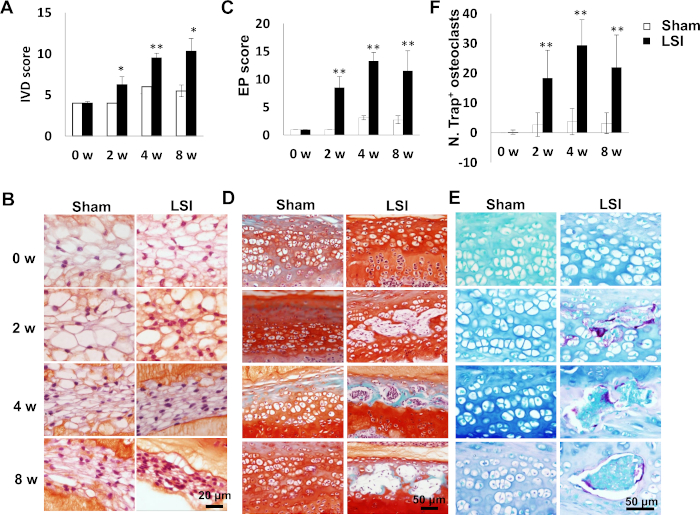
Figure 7: LSI leads to IVD and EP degeneration. (A) IVD score in LSI or sham mice as an indication of IVD degeneration. (B) Representative images of Safranin O staining for NPs in L4–L5 IVD. White indicates vacuoles. Red indicates proteoglycan. (C) EP score in LSI or sham group as an indication of EP degeneration. (D) Representative images of Safranin O-Fast green staining for caudal L4–L5 EPs. Green/blue stains calcified cavities. (E) Representative images of trap staining for caudal L4–L5 EPs. Purple indicates Trap+. N = 6 per group. Data is shown as mean ± s.d.* p < 0.05, ** p < 0.01 versus. Sham. (F) Quantification of Trap+ osteoclasts in (E). (A,B) have been modified from Bian et al.20. (C–F) have been modified from Bian et al.21. Please click here to view a larger version of this figure.
Discussion
We developed the lumbar spine instability mouse model based on the cervical spondylosis mouse model in which the posterior paravertebral muscles from the vertebrae were detached and the spinous processes along with the supraspinous and interspinous ligaments were resected25. We performed a similar operation onto the lumbar spine, which has more prominent spinous processes. The LSI mouse model developed similar IDD in the lumbar spine.
The advantages of the LSI model include strong operability, no requirement of a special equipment, reproducibility, and a relatively short period of IDD development.
Some key points are presented here to help improve the success rate during the operation. These are also the critical steps. First, remove the hair on the lower back portion, as clear as possible, because any shaved hair left in the wound may cause anaphylactic reaction. Second, a cylindrical pad or any other pad is recommended to raise up the lumbar vertebrae. Third, use micro scissors to control the incision depth and hemorrhage. When hematocoelia is noticed during the operation, stop the operation and sacrifice the mouse since the mouse will not survive during or post-surgery. Fourth, reattachment of paraspinous muscle is not recommended because the reattachment may make up the instability. Fifth, a full resection of the whole L3–L5 spinous processes reduces variability in individual model. Sixth, avoid injuring surrounding nerves and blood vessels, otherwise, the mouse may develop non-canonic pathological changes. If the models do not exhibit the typical phenotype as shown in the results, check the above six points.
The success of this LSI model can be assessed by two golden standards including decreased IVD volume measured by small animal MRI or by μCT, and an IVD score based on the histological observation. The LSI model develops IDD as early as 2 weeks post LSI surgery, but develops porosity in endplate as early as 1 week, as observed. It is suitable for study on nucleus pulpous shrinkage, endplate sclerosis, IDD related to osteoclast-induced cytokines, IDD-induced osteoporosis (16 weeks post LSI) etc.
There are some limitations in the LSI model. LSI operation is a relatively great trauma for the mouse. The inflammation is inevitable and usually seen within 7 days post operation. Thus, this model is not suitable for observing early pathological changes of IDD, especially within 7 days caused by mechanical loading changes.
The model can be modified by targeting on different lumbar vertebrae such as L5 only or from L1 to L5. Healthy control is also recommended in addition to sham groups.
In summary, we developed a surgical-induced lumbar IDD mouse model and have the procedure visualized to help others reproduce the animal model and apply it in IDD studies.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81973607) and Essential Drug Research and Development (2019ZX09201004-003-032) from Ministry of Science and Technology of China.
Materials
| Chlortetracycline Hydrochloride Eye Ointment | Shanghai General Pharmaceutical Co., Ltd. | H31021931 | Prevent eye dry, Prevent wound infection |
| C57BL/6J male mice | Tian-jiang Pharmaceuticals Company (Jiangsu, CN) | SCXK2018-0004 | Animal model |
| Disposable medical towel | Henan Huayu Medical Devices Co., Ltd. | 20160090 | Platform for surgical operation |
| Inhalant anesthesia equipment | MIDMARK | Matrx 3000 | Anesthesia |
| Isoflurane | Shenzhen RWD Life Technology Co., Ltd. | 1903715 | Anesthesia |
| Lidocaine hydrochloride | Shandong Hualu Pharmaceutical Co., Ltd. | H37022839 | Pain relief |
| Medical suture needle | Shanghai Pudong Jinhuan Medical Products Co., Ltd. | 20S0401J | Suture skin |
| Ophthalmic forceps | Shanghai Medical Devices (Group) Co., Ltd. Surgical Instruments Factory | JD1050 | Clip the skin |
| Ophthalmic scissors(10cm) | Shanghai Medical Devices (Group) Co., Ltd. Surgical Instruments Factory | Y00030 | Skin incision |
| silk braided | Shanghai Pudong Jinhuan Medical Products Co., Ltd. | 11V0820 | Suture skin |
| Small animal trimmer | Shanghai Feike Electric Co., Ltd. | FC5910 | Hair removal |
| Sterile surgical blades(12#) | Shanghai Pudong Jinhuan Medical Products Co., Ltd. | 35T0707 | Muscle incision |
| Veet hair removal cream | RECKITT BENCKISER (India) Ltd | NA | Hair removal |
| Venus shears | Mingren medical equipment | Length:12.5cm | Clip the muscle and spinous process |
Referencias
- Makino, H., et al. Lumbar disc degeneration progression in young women in their 20’s: a prospective ten-year follow up. Journal of Orthopaedic Science: Official Journal of the Japanese Orthopaedic Association. 22 (4), 635-640 (2017).
- Lee, Y. C., Zotti, M. G. T., Osti, O. L. Operative management of lumbar degenerative disc disease. Asian Spine Journal. 10 (4), 801-819 (2016).
- Wei, F., et al. In vivo experimental intervertebral disc degeneration induced by bleomycin in the rhesus monkey. BMC Musculoskeletal Disorders. 15, 340 (2014).
- Lim, K. Z., et al. Ovine lumbar intervertebral disc degeneration model utilizing a lateral retroperitoneal drill bit injury. Journal of Visualized Experiments: JoVE. (123), e55753 (2017).
- Zhang, Y., et al. Histological features of the degenerating intervertebral disc in a goat disc-injury model. Spine. 36 (19), 1519-1527 (2011).
- Bergknut, N., et al. The dog as an animal model for intervertebral disc degeneration. Spine. 37 (5), 351-358 (2012).
- Kong, M. H., et al. Rabbit Model for in vivo Study of Intervertebral Disc Degeneration and Regeneration. Journal of Korean Neurosurgical Society. 44 (5), 327-333 (2008).
- Gullbrand, S. E., et al. A large animal model that recapitulates the spectrum of human intervertebral disc degeneration. Osteoarthritis and Cartilage. 25 (1), 146-156 (2017).
- Jin, L., Balian, G., Li, X. J. Animal models for disc degeneration-an update. Histology and Histopathology. 33 (6), 543-554 (2018).
- O’Connell, G. D., Vresilovic, E. J., Elliott, D. M. Comparative intervertebral disc anatomy across several animal species. 52nd Annual Meeting of the Orthopaedic Research Society. , (2006).
- Elliott, D. M., Sarver, J. J. Young investigator award winner: validation of the mouse and rat disc as mechanical models of the human lumbar disc. Spine. 29 (7), 713-722 (2004).
- Ohnishi, T., et al. In vivo mouse intervertebral disc degeneration model based on a new histological classification. Plos One. 11 (8), 0160486 (2016).
- Vo, N., et al. Accelerated aging of intervertebral discs in a mouse model of progeria. Journal of Orthopaedic Research. 28 (12), 1600-1607 (2010).
- Oichi, T., et al. A mouse intervertebral disc degeneration model by surgically induced instability. Spine. 43 (10), 557-564 (2018).
- Ohnishi, T., Sudo, H., Tsujimoto, T., Iwasaki, N. Age-related spontaneous lumbar intervertebral disc degeneration in a mouse model. Journal of Orthopaedic Research. 36 (1), 224-232 (2018).
- Stern, W. E., Coulson, W. F. Effects of collagenase upon the intervertebral disc in monkeys. Journal of Neurosurgery. 44 (1), 32-44 (1976).
- Silva, M. J., Holguin, N. LRP5-deficiency in OsxCreERT2 mice models intervertebral disc degeneration by aging and compression. bioRxiv. , (2019).
- Nemoto, Y., et al. Histological changes in intervertebral discs after smoking and cessation: experimental study using a rat passive smoking model. Journal of Orthopaedic Science: Official Journal of the Japanese Orthopaedic Association. 11 (2), 191-197 (2006).
- Mulholland, R. C. The myth of lumbar instability: the importance of abnormal loading as a cause of low back pain. European Spine Journal. 17 (5), 619-625 (2008).
- Bian, Q., et al. Mechanosignaling activation of TGFβ maintains intervertebral disc homeostasis. Bone Research. 5, 17008 (2017).
- Bian, Q., et al. Excessive activation of tgfβ by spinal instability causes vertebral endplate sclerosis. Scientific Reports. 6, 27093 (2016).
- Ni, S., et al. Sensory innervation in porous endplates by Netrin-1 from osteoclasts mediates PGE2-induced spinal hypersensitivity in mice. Nature Communications. 10 (1), 5643 (2019).
- Liu, S., Cheng, Y., Tan, Y., Dong, J., Bian, Q. Ligustrazine prevents intervertebral disc degeneration via suppression of aberrant tgfβ activation in nucleus pulposus cells. BioMed Research International. 2019, 5601734 (2019).
- Boos, N., et al. Classification of age-related changes in lumbar intervertebral discs: 2002 Volvo Award in basic science. Spine. 27 (23), 2631-2644 (2002).
- Miyamoto, S., Yonenobu, K., Ono, K. Experimental cervical spondylosis in the mouse. Spine. 16, 495-500 (1991).

