An Effective Inoculation Method for Phytophthora capsici on Black Pepper Plants
Summary
Pinpricking the basal head of the black pepper plant is a brief and time-saving method to damage it. Here, we provided detailed steps with a video for infecting black pepper plants.
Abstract
Piper nigrum L. (black pepper) is a typical woody vine that is an economically important spice crop across the world. Black pepper production is significantly impacted by root rot disease caused by Phytophthora capsici, which has seriously influenced the industry development as a “choke point” problem. However, the molecular genetic mechanism of resistance in black pepper is unclear, leading to slow progress in the development of new black pepper varieties. An effective inoculation and precise sampling system for Phytophthora capsici on black pepper plants is essential for studying this plant-pathogen interaction. The main aim of this study is to demonstrate a detailed methodology where the basal head of black pepper is inoculated with Phytophthora capsici, while also providing a reference for the inoculation of woody vine plants. The basal head of the black pepper plant was pinpricked to damage it, and mycelial pellets covered the three holes to retain the moisture so the pathogen could infect the plant well. This method provides a better way of solving the instability that is caused by traditional inoculation methods including soil drench or root dipping. It also provides a promising means for studying the mode of action between plants and other soil-borne plant pathogens in agricultural precision breeding.
Introduction
Black pepper (Piper nigrum L.) is a woody climber and one of the most important spice crops. It is known as the “King of Spices”1 and is cultivated in over 40 countries and regions throughout Asia, Africa, and Latin America. Phytophthora root rot is the most devastating disease of black pepper, and is caused by the oomycete Phytophthora capsici. This pathogen also infects cucurbits, eggplants, chili peppers, and tomatoes2,3. With black pepper, an entire crop can sometimes be decimated by this disease. The expansion of pepper planting areas is restricted as a result of the unavailability of resistant varieties, which has significantly hindered the development of the Chinese black pepper industry. An effective inoculation and a precise sampling system for Phytophthora capsici on black pepper plants are essential for studying this plant-pathogen interaction.
The identification and screening of resistance in germplasm resources is the basic requirement for researching the pathogenicity of the pathogen and the breeding and utilization of resistant varieties. A widely used approach is to use a variety of identification methods based on plant species and pathogen groups. Current identification methods include population identification, individual identification, organ identification, tissue identification, cell identification, biochemical identification, and molecular identification, which have been developed in recent years4,5. There has been success in these areas, but there are also many problems. No matter which method is chosen, the basic requirements of plant resistance identification are consistent, including clear objectives, reliable results, and methods that are simple, rapid, and easy to standardize. This principle must also be followed in the identification of black pepper resistance.
In natural field conditions, the identification of disease resistance can be influenced by many environmental factors. Therefore, it was proposed that detached leaves and irrigated roots be used in the laboratory to identify disease resistance. Young leaves from healthy plants were inoculated in vitro in the laboratory, and the diseased leaves area was measured by inoculating the pathogen in order to identify the disease resistance of plants6. However, in vitro leaf inoculation can only be used for general resistance identification and not for molecular interaction studies. Despite this, the disease-resistant status often presents in the irrigated root inoculation, causing uncertainty in the follow-up study of molecular breeding for disease resistance. Therefore, fast and simple indoor detection methods are essential. This study aims to provide a method for resistance identification in the laboratory.
Protocol
1. Preparation of black pepper cutting plants for infection
- Take a five-node cutting, approximately 40 cm long with a diameter of 0.5 cm, from a healthy and vigorously growing orthotropic branch of black pepper using a disinfected pruning knife or secateurs. Prune the lower three nodes of the plagiotropic branches, with the upper two nodes left with approximately 10 leaves intact.
- Prepare rooting substrate containing soil and animal manure (cow dung or sheep dung) at a ratio of 1:1. Autoclave the rooting substrate at 121 °C for 20 min.
- Insert the cuttings into the rooting substrate at an angle of approximately 50°, with the third node just touching the surface of the substrate and the axillary bud on this node above the substrate.
NOTE: The bag used here has the following dimensions: height of 40-60 cm, diameter of 25-30 cm. - Pour 10-20 L of water over the roots of the plant. Place the cuttings in a greenhouse with 90% shade at a temperature of 25-30 °C for rooting and growth.
2. Propagation of Phytophthora capsici (P. capsici)
NOTE: A stock of Phytophthora capsici culture is maintained in the plant protection laboratory of the Spice and Beverage Research Institute, Chinese Academy of Tropical Agricultural Sciences7.
- Brush and clean the potato tubers under running tap water and then cut 200 g of potatoes into cubes of 1 cm3. Place some of the cubes into a beaker containing 800 mL of double-distilled water (ddH2O) and boil for 20 min.
- Filter the broth through double gauze using gravity filtration. Prepare potato dextrose agar (PDA) by adding 20 g of dextrose and 15 g of agar to the filtrate and topping the volume up to 1 L with ddH2O. Autoclave the mixture at 121 °C for 20 min8.
- Pour 20 mL of the sterilized PDA in liquid form into a 9 cm diameter round Petri dish inside a laminar air flow hood. Leave the PDA plates with lids open inside the laminar air flow hood overnight as a means of preventing condensation.
- Use an inoculation loop to pick up mycelia from Phytophthora capsici stock inside a test tube. Place the inoculum with the mycelial side in contact with the PDA in a Petri dish.
3. Infection of black pepper
- Incubation
- Identify an area 5 cm above the substrate surface and near to the roots on the stem for inoculation.
- Core out a disk of mycelia 0.5 cm in diameter at the growing edge of the Phytophthora capsici culture on PDA in a Petri dish using a stopper borer.
- Damage the stem using a syringe needle and make three holes in a triangular pattern at the selected inoculation area. Cover each hole with a mycelial disk. Position the holes close to each other to ensure the wounded area is fully covered with the mycelial disks.
- Cover the mycelial disks with sterilized moistened cotton pads as a means of preventing drying. Tie the pad onto the stem with a polyethylene strip to maintain the position of the inoculating disks.
NOTE: At 8 h after inoculation, the inoculated holes turned black and the lesion extended as time passed. The leaves turned yellow and dropped off, and the inoculated plant died 7-10 days after inoculation. No lesions developed in the control plants. Most of the genes expressed differently following inoculation with Phytophthora capsici in comparison to the control group. Histopathological analysis of the infected tissues demonstrated that Phytophthora capsici colonized in the xylem.
- Sample the plant materials of interest and store at -80 °C in liquid nitrogen for use in subsequent studies.
NOTE: Liquid nitrogen, plastic bags, marker pens, branch shears, and other materials were prepared prior to the experiments. - After the specific plant materials are sampled for use, autoclave all the remaining plant materials, left-over Phytophthora capsici culture and culture medium, and all the instruments and labware used in this inoculation work.
Representative Results
Figure 1 shows the symptoms of black pepper leaves following P. capsici inoculation. Figure 2 shows the symptoms of black pepper stems following P. capsici inoculation. The pathogen infected the black pepper at the basal stem; symptoms including leaf yellowing, wilting appearing, xylem browning, and vessel blacking gradually appearing. Figure 3 shows most of the genes expressed differently after inoculation with Phytophthora capsici compared with the control group. Figure 4 has demonstrated Phytophthora capsici colonized in the xylem by the histopathological analysis of infected tissues.

Figure 1: The symptoms of black pepper leaves following P. capsici inoculation7. CK: control group; Inoculated: after inoculation. Please click here to view a larger version of this figure.

Figure 2: The symptom of black pepper stems following P. capsici inoculation7. Inoculated: after inoculation. Please click here to view a larger version of this figure.
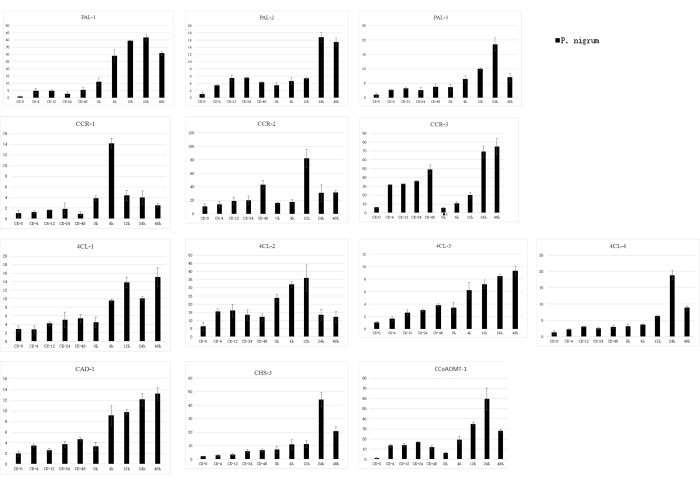
Figure 3: Detailed expression profiles of the genes in the black pepper roots. Error bars in the figures denote the standard error of expression levels from three biological replications. CK-8, CK-12, CK-24, CK-48, 8, 12, 24, and 48 on the x-axis refer to 8, 12, 24, and 48 h on the control and 8, 12, 24, and 48 h, respectively, following inoculation with P. capsici. The y-axis represents the relative expression level compared with ubiquitin. Each column represents the mean value plus SD (standard deviation) from three biological replicates. Please click here to view a larger version of this figure.
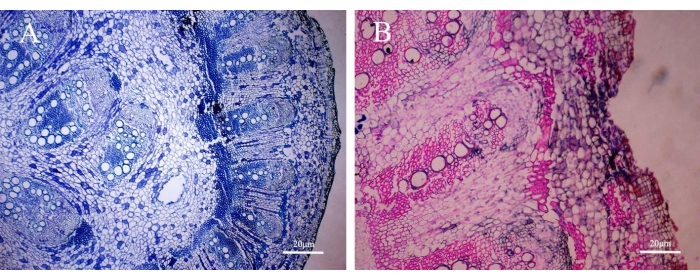
Figure 4: Histopathological analysis of infected tissues. The comparison between toluidine blue O staining alone (left column) and cotton blue and safranin O double-staining (right column) (20X). Please click here to view a larger version of this figure.
Discussion
In this study, the basal head was pinpricked to damage and provide an effective inoculation system in the black pepper plant. Mycelial pellets then covered the three holes to retain moisture and enable the pathogen to infect the plant well. Following inoculation, the leaves turned yellow and dropped off and the inoculated plants died. No lesions developed in the control plants. Most genes expressed differently following inoculation with Phytophthora capsici in comparison to the control group. Fungal diseases are responsible for structural and physiological disorders in a significant number of crops, leading to decreased productivity and economic losses for their producers. Structural studies employing histological techniques on the mode of penetration and colonization of plant tissues by fungi provide a detailed indication of the interactions between the pathogen and the plant tissue. These studies have revealed important aspects to help understand the monocycle of diseases. Histopathological analysis of infected tissues demonstrated Phytophthora capsici colonized in the xylem. This method provides a better means for solving the instability that is caused by traditional inoculation methods including soil drench or root dipping. An effective inoculation and precise sampling system for Phytophthora capsici on black pepper plants are essential for studying this plant-pathogen interaction. It also provides a promising means for studying the mode of action between plants and other soil-borne plant pathogens in agricultural precision breeding.
At the same time, this protocol represents a more efficient way of providing reference for the incubation of woody vine. In previous studies, pathogens were inoculated by root dipping with spore suspensions cultured in V8 medium9. It takes 7 days for the spore suspension to be ready, whereas the use of PDA to culture Phytophthora capsici takes just 5 days. The PDA plate was sealed using permeable surgical tape as a means of avoiding contamination from other bacteria and fungi. The cultures were kept at room temperature. The method used in this study can save more time and be performed more rapidly. Black pepper is a woody vine with many sugars and phenols10, and the zoospores produced by Phytophthora capsici generally occur in soils, making it difficult to infect black pepper vines and cause infection instability in the root11. This protocol provides better results, enabling a strong interaction between vine plants and soil-borne pathogens. The detection of the dynamic process between plants and pathogens is visible and convenient.
The irrigating root method is fast and time-saving, but a problem remains unsolved for black pepper. Phytophthora capsici is a soil-borne pathogen that generally infects plant roots via sporangia and zoospores12. In nature, the sporangia is able to spread via rain and irrigation. Once the zoospores become attached to the plant surface, the germ tubes may quickly develop and penetrate the plant tissue, which results in infection13,14. This can cause uncertainty that choosing hyphae as an infection source will be similar to the spore suspension. The method used in this study starts with pinpricking the basal head of the black pepper plant to damage it. The damaged area is then covered with Phytophthora capsici and moisture is retained, ensuring that the pathogen can infect the plant well. This method is better at solving the instability caused by traditional inoculation methods including soil drench or root dipping. It is also a promising method for studying the mode of action between plants and other soil-borne plant pathogens in agricultural precision breeding.
Acknowledgements
This work was financially supported by the National Key R&D Program of China (2020YFD1001200), the China Agriculture Research System (CARS-11), the specific research fund of The Innovation Platform for Academicians of Hainan Province (YSPTZX202154), the Natural Science Foundation of Hainan Province of China (321RC652), and the Natural Science Foundation of China (No. 31601626).
Materials
| Agar powder | Solarbio | A8190 | |
| Clean bench | Haier | ||
| Dextrose | Xilong Scientific | 15700501 | |
| High temperature sterilizing oven | Zaelway | ||
| Petri dish plates | Biosharp | BS-90-D |
Referencias
- Gordo, S. M., et al. High-throughput sequencing of black pepper root transcriptome. BMC Plant Biology. 12 (1), (2012).
- Leonian, L. H. Stem and fruit blight of Peppers caused by Phytophthora capsici sp. Nov. Phytopathology. 12 (9), 401-408 (1922).
- Ding, X., et al. Priming maize resistance by its neighbors: Activating 1,4-benzoxazine-3-ones synthesis and defense gene expression to alleviate leaf disease. Frontiers in Plant Science. 6, 830 (2015).
- Fonseca, C. E. L., Vianda, D. R., Hansen, J. L., Pell, A. N. Associations among forage quality traits, vigor, and disease resistance in alfalfa. Crop Science. 39 (5), 1271-1276 (1999).
- Altier, N. A., Thies, J. A. Identification of resistance to Pythium seedling disease in Alfalfa using a culture plate method. Plant Disease. 79 (4), 341-345 (1995).
- Pratt, R. G., Rowe, D. E. Evaluation of simplified leaf inoculation procedures for identification of quantitative resistance to Sclerotinia trifoliorum in Alfalfa seedling. Plant Disease. 82 (10), 1161-1164 (1998).
- Hao, C., et al. De novo transcriptome sequencing of black pepper (Piper nigrum L.) and an analysis of genes involved in phenylpropanoid metabolism in response to Phytophthora capsici. BMC Genomics. 17 (1), 1-14 (2016).
- Dong, C., et al. Field inoculation and classification of maize ear rot caused by Fusarium verticillioides. Bio-protocol. 8 (23), 3099 (2018).
- English, J. T., Laday, M., Bakonyi, J., Schoelz, J. E., Érsek, T. Phenotypic and molecular characterization of species hybrids derived from induced fusion of zoospores of Phytophthora capsica and Phytophthora nicotianae. Mycological Research. 103 (8), 1003-1008 (1999).
- Chatterjee, S., et al. Antioxidant activity of some phenolic constituents from green pepper (Piper nigrum L.) and fresh nutmeg mace (Myristica fragrans). Food Chemistry. 101 (2), 515-523 (2007).
- Pfender, W. F. Production of sporangia and release of zoospores by Phytophthora megasperma in soil. Phytopathology. 67 (5), 657-663 (1977).
- Nagila, A., Schutte, B. J., Sanogo, S., Idowu, O. J. Chile pepper sensitivity to mustard seed meal applied after crop emergence. HortScience. 56 (2), 1-7 (2021).
- Lamour, K. H., Stam, R., Jupe, J., Huitema, E. The oomycete broad-host-range pathogen Phytophthora capsica. Molecular Plant Pathology. 13 (4), 329-337 (2012).
- Hardham, A., Gubler, F. Polarity of attachment of zoospores of a root pathogen and pre-alignment of the emerging germ tube. Cell Biology International Reports. 14 (11), 947-956 (1990).

