Retinal Pathophysiological Evaluation in a Rat Model
Summary
Diabetic retinopathy is one of the leading causes of blindness. Histology, blood-retinal barrier breakdown assay, and fluorescence angiography are valuable techniques to understand the pathophysiology of the retina, which could further enhance the efficient drug screening against diabetic retinopathy.
Abstract
A posterior segment eye disease like diabetic retinopathy alters the physiology of the retina. Diabetic retinopathy is characterized by a retinal detachment, breakdown of the blood-retinal barrier (BRB), and retinal angiogenesis. An in vivo rat model is a valuable experimental tool to examine the changes in the structure and function of the retina. We propose three different experimental techniques in the rat model to identify morphological changes of retinal cells, retinal vasculature, and compromised BRB. Retinal histology is used to study the morphology of various retinal cells. Also, quantitative measurement is performed by retinal cell count and thickness measurement of different retinal layers. A BRB breakdown assay is used to determine the leakage of extraocular proteins from the plasma to vitreous tissue due to the breakdown of BRB. Fluorescence angiography is used to study angiogenesis and leakage of blood vessels by visualizing retinal vasculature using FITC-dextran dye.
Introduction
Diabetic retinopathy (DR) is one of the most complex secondary complications of diabetes mellitus. It is also the leading cause of preventable blindness in the working-age population worldwide. In a recent meta-analysis of 32.4 million blind people, 830,000 (2.6%) people were blind due to DR1. The proportion of vision loss attributed to diabetes ranked seventh in 2015 at 1.06% (0.15-2.38) globally2,3.
Diabetic retinopathy is diagnosed by vascular abnormalities in the posterior ocular tissues. Clinically, it is divided into two stages – Non-Proliferative DR (NPDR) and Proliferative DR (PDR), based on the vascularization in the retina. Hyperglycemia is considered the potent regulator of DR as it implicates several pathways involved in neurodegeneration4,5, inflammation6,7, and microvasculature8 in the retina. Multiple metabolic complications induced due to hyperglycemia include the accumulation of advanced glycation end products (AGEs), polyol pathway, hexosamine pathway, and protein kinase-C pathway. These pathways are responsible for cell proliferation (endothelial cells), migration (pericytes), and apoptosis (neural retinal cells, pericytes, and endothelial cells) based on different stages of diabetic retinopathy. These metabolic alterations can lead to physiological changes such as retinal detachment, loss of retinal cells, breakdown of the blood-retinal barrier (BRB), aneurysms, and angiogenesis9.
Streptozotocin (STZ) induced type-1 diabetes is a well-established and well-accepted practice in rats for evaluating diabetes pathogenesis and its complications. Diabetogenic effects of STZ are due to selective destruction of pancreatic islet β-cells10. As a result, the animals will undergo insulin deficiency, hyperglycemia, polydipsia, and polyuria, all of which are characteristic of human type-1 diabetes mellitus11. For severe diabetes induction, STZ is administered at 40-65 mg/kg body weight intravenously or intraperitoneally during adulthood. After approximately 72 h, these animals present blood glucose levels greater than 250 mg/dL10,12.
To understand the physiological alterations of the retina due to neurodegeneration, inflammation, and angiogenesis, different techniques should be optimized in experimental animal models. Structural and functional changes in retinal cells and retinal vessels can be studied by various techniques such as histology, BRB breakdown assay, and fluorescence angiography.
Histology involves the study of the anatomy of cells, tissues, and organs at a microscopic level. It establishes a correlation between the structure and function of cells/tissue. Several steps are performed to visualize and identify the microscopic alterations in tissue structure, thereby comparing healthy and diseased counterparts13. Hence, it is essential to standardize each step of histology meticulously. Various steps involved in retinal histology are fixation of the specimen, trimming the specimen, dehydration, clearing, impregnation with paraffin, paraffin embedding, sectioning, and staining (Hematoxylin and Eosin staining)13,14.
In a healthy retina, the transport of molecules across the retina is controlled by BRB, composed of endothelial cells and pericytes on the inner side, and retinal pigment epithelial cells on the outer side. However, inner BRB endothelial cells and pericytes start degenerating during the diseased condition, and BRB is also compromised15. Due to this BRB breakdown, many low molecular weight molecules leak into vitreous and retinal tissue16. As the disease progresses, many other protein molecules (low and high molecular weight) also leak into vitreous and retinal tissue due to homeostasis disturbance17. It leads to various other complications and ultimately macular edema and blindness. Hence, quantifying the protein levels in the vitreous and comparing healthy and diabetic states measures compromised BRB.
Fluorescence angiography is a technique used to study blood circulation of the retina and choroid using fluorescent dye. It is used to visualize vasculature of the retina and choroid by injecting fluorescein dye via intravenous route or cardiac injection18. Once the dye is injected, it first reaches the retinal arteries, followed by retinal veins. This circulation of dye is usually completed within 5 to 10 min from the injection of dye19. It is an important technique to diagnose various posterior segment ocular diseases, including diabetic retinopathy and choroidal neovascularization20. It helps to detect major and minor vasculature changes in normal and diseased conditions.
Protocol
This protocol follows all the animal care guidelines provided by Institutional Animal Ethics Committee, BITS-Pilani, Hyderabad campus.
1. Retinal histology
- Enucleation and fixation of the eye
- Euthanize a 2 to 3-month-old diabetic Wistar male rat along with the age-matched control (14 to 15 weeks old) using a high dose of pentobarbital (150 mg/kg) injected through the intraperitoneal route. No detectable heartbeat confirms the death within 2-5 min.
- Enucleate the eye by making incisions using a scalpel blade on the nasal and temporal regions of the eye. Then cut along its edges using forceps and micro scissors to remove the eye from the orbital socket.
NOTE: Before sacrificing rats, keep the fixative solutions ready.
CAUTION: Formaldehyde irritates the skin, eye, nose, and respiratory tract. It can also cause cancer as it is a potent skin sensitizer. Wear gloves and handle it under the hood21. - Wash the eye thoroughly with 10 mL of phosphate-buffered saline (PBS) solution in a Petri dish to remove blood. Remove the excess fat surrounding the eye for easy penetration of the fixative solution.
- Immediately place the eye in 5 mL of fixative solution with the help of forceps, and make sure it is not stuck to walls of glass vials. Stir gently for a few seconds and replace the cap with proper labeling. Incubate eye in fixative solution for 24 to 48 h in dark conditions at room temperature.
- Trimming of tissue
- After fixation, remove the eye from the fixative solution, wash with 10 mL of PBS, and place it in a Petri dish containing 10 mL of PBS chilled at 4 °C.
- Using micro forceps, hold the eye with the optic nerve and make a nick at pars plana with the help of micro scissors. Cut through the entire margin of the cornea to separate the anterior cup of an eye. Using forceps, pick the lens gently, discard it, and cut the optic nerve.
- Using curved micro scissors, make a longitudinal section of the posterior eyecup, passing through the optic disc dividing it into two halves. Place it in the base of the cassette and close the lid properly without any disturbance to the tissue. Label the cassettes with the tissue sample name and date.
- Dehydration, clearing, and paraffin impregnation of tissue
- Dehydrate the trimmed tissue by gradual transfer in 80 mL of 50%, 70%, 90%, and 100% ethanol. While transferring the cassettes from one concentration to another, make sure to dab them on clean tissue paper to minimize contamination.
NOTE: Allow the tissue to stand in each transfer for 30 min (twice). The total time consumed for this step is approximately 4 h. - Upon dehydration, transfer the cassettes into 80 mL of xylene for 30 min (twice) to replace the ethanol with xylene.
NOTE: After incubation with xylene, the tissue becomes translucent.
CAUTION: Xylene is an aromatic compound with a benzene ring. It irritates the eye and mucous membrane and may also cause depressions. - Finally, impregnate the tissue with pre-heated paraffin at 60 °C to replace xylene in the tissue. Dab the cassettes several times on tissue paper to minimize the xylene content and place in 200 mL of paraffin (liquid at 60 °C) for 2 h (1 h x 2).
CAUTION: Avoid inhaling melted paraffin, as it produces tiny lipid droplets which may cause respiratory discomfort.
- Dehydrate the trimmed tissue by gradual transfer in 80 mL of 50%, 70%, 90%, and 100% ethanol. While transferring the cassettes from one concentration to another, make sure to dab them on clean tissue paper to minimize contamination.
- Paraffin embedding
- Turn on the paraffin embedding machine at least 1 h before use to melt the paraffin and for the stations to reach desired temperatures. Fill a steel mold with melted paraffin wax by placing it on a hot surface, then remove one cassette at a time from the paraffin container.
- Move the steel mold from a hot to a cold surface (4 °C). At this point, the wax in the mold will start to solidify. Before it solidifies completely, place the tissue in wax with the help of forceps and orient the tissue so that the optic disc portion of the posterior cup is facing the base of the mold.
NOTE: Ensure that the tissue does not move and keep its desired orientation. If not, put the mold back onto the warm work surface until the whole paraffin liquefies, then start again with step 1.4.2. - As the wax in the mold starts solidifying, immediately place the cassette base on top of the mold. Carefully fill the mold with paraffin above the upper edge of the cassette and slowly transfer it to a cold surface (near -20 °C) for rapid solidification. Until it solidifies, repeat the process with other cassettes from step 1.4.2.
- Once all the molds are solidified, separate the steel mold from the cassette. If it is hard, wait a bit longer and try again (do not remove it forcefully). Separation becomes easier upon complete solidification. Now the cassette becomes the base of the paraffin block to be held during sectioning by microtome.
NOTE: This block can be stored at room temperature for further use. At this point, the process can be stopped and continued later (if required).
- Sectioning
- Install a disposable microtome blade in the blade holder. Remove excess paraffin wax around cassettes so that both the upper and lower portions of the blocks remain parallel to the knife. Fit the block on to cassette holder.
- Unlock the hand-wheel to advance it until the surface of the block is in contact with the edge of the knife. Trim the excess paraffin wax on the block until the tissue is visualized on the surface of the block. Set the section thickness to 5 µm and cut a ribbon of five to six sections.
- Using forceps, gently transfer the ribbon onto the surface of the pre-warmed water bath (around 50 °C) to unfold the ribbon. Collect sections on a glass slide coated with Mayer's albumin by holding the slide beneath the section. Gently lift the sections and allow the slides to dry horizontally overnight at 37 °C.
NOTE: Slides can be stored at room temperature in dry boxes for several months. At this step, the process can be stopped and continued later.
- Staining
- Heat the slides to be stained in a hot air oven at 60 °C for 1 h before use for the paraffin to melt, and follow the steps as mentioned in Table 1.
CAUTION: Hematoxylin and Eosin stains are toxic when inhaled or ingested. They have been reported to be carcinogenic.
- Heat the slides to be stained in a hot air oven at 60 °C for 1 h before use for the paraffin to melt, and follow the steps as mentioned in Table 1.
| Reagent | Standing Time | Repetition (Number of times) |
| Xylene | 5 min | 2 |
| 100% Ethanol | 5 min | 2 |
| 90% Ethanol | 5 min | 2 |
| 70% Ethanol | 5 min | 2 |
| 50% Ethanol | 5 min | 2 |
| Water | 5 min | 2 |
| Hematoxylin | 4 min | 1 |
| Water wash | ||
| 1% Acid alcohol in 70% Ethanol | 30 s | 1 |
| Water wash | ||
| Scott's water | 1 min | 1 |
| Water wash | ||
| 50% Ethanol | 1 min | 1 |
| 95% Ethanol | 1 min | 1 |
| 0.25% Eosin | 5 s | 1 |
| Water wash | ||
| Water | 2 min | 1 |
| 95% Ethanol | 1 min | 1 |
| 100% Ethanol | 1 min | 1 |
| Xylene | 5 min | 2 |
| Mountant and coverslip | ||
Table 1. Hematoxylin and Eosin staining procedure
2. Blood-brain barrier breakdown assay
- Blood and vitreous collection
- Anesthetize a 2 to 3-month-old diabetic Wistar male rat along with the age-matched control (14 to 15 weeks) using ketamine (80 mg/kg) and xylazine (8 mg/kg). Confirm the anesthetic state by pedal withdraw reflex (toe pinch). Immediately collect 1 mL of blood by cardiac puncture into an ethylenediaminetetraacetic acid (EDTA) coated tube to separate plasma from whole blood.
- Euthanize the rat using a high dose of pentobarbital (150 mg/kg), injected through the intraperitoneal route. No detectable heartbeat confirms the death within 2-5 min. Enucleate the eye and immediately place it on dry ice.
- Place the eye in a clean Petri dish above dry ice (recommended) and start dissecting the eye as mentioned in step 1.2.2.
- Remove the lens, pull the vitreous carefully from the posterior cup using micro forceps and place it in a homogenization tube containing three to four glass beads (2-4 mm).
NOTE: Dissection on dry ice is recommended as removing the lens and vitreous is easier under freezing conditions.
- Preparation of samples
- Homogenize vitreous humor in a bead homogenizer at medium speed for 10 s.
- Transfer the blood (1 mL) and vitreous samples (10-20 µL) into the microcentrifuge tubes and centrifuge both the samples at 5200 x g for 10 min at 4 °C.
NOTE: In the case of successful homogenization, vitreous has uniform consistency after centrifugation. However, in the case of non-uniform consistency of vitreous, repeat from step 2.2.1 with increased homogenization time. - Collect supernatant from both samples, and dilute vitreous humor and plasma samples in 1:10 and 1:20 ratio, respectively, with PBS.
- Protein quantification and vitreous-plasma protein ratio
- To quantitate protein concentration in vitreous humor and plasma samples, add 5 µL of diluted samples to 250 µL of Bradford reagent, mix well, and read the absorbance at 590 nm within 40 min.
NOTE: If the absorption value of the samples is above 1.0, dilute the samples further and repeat the procedure from step 2.2.3. - Normalize the vitreous protein level to plasma protein level from the same rat and measure the fold difference between healthy vs. diabetic rats.
- To quantitate protein concentration in vitreous humor and plasma samples, add 5 µL of diluted samples to 250 µL of Bradford reagent, mix well, and read the absorbance at 590 nm within 40 min.
3. Fluorescence angiography
- FITC-dextran70000 dye injection
- Anesthetize a 2 to 3-month-old diabetic Wistar male rat along with the age-matched control (14 to 15 weeks) using 3% isoflurane and confirm the pedal withdrawal reflex (toe pinch). Dip the tail in a beaker containing warm water (37-40 °C). Clean the tail with 70% ethanol before injecting the dye. Locate the tail's lateral vein and mark the injection position in the lower portion of the tail.
NOTE: If the insertion of the needle fails, try to insert in another location above the current position (tip to the base of tail). However, it is advised to inject once as repeated pricking may lead to stress development in the rat, which could cause vasoconstriction. - Inject 1 mL of 50 mg/mL FITC-dextran70000 dye solution through the tail vein and allow the dye to circulate for 5 min. Euthanize the rat with a high dose of pentobarbital (150mg/kg), injected through the intraperitoneal route. No detectable heartbeat confirms the death within 2-5 min. Enucleate the eye as mentioned in step 1.1.1.
NOTE: If required, the eye can be fixed in 4% formaldehyde solution, for no more than 30 min.
- Anesthetize a 2 to 3-month-old diabetic Wistar male rat along with the age-matched control (14 to 15 weeks) using 3% isoflurane and confirm the pedal withdrawal reflex (toe pinch). Dip the tail in a beaker containing warm water (37-40 °C). Clean the tail with 70% ethanol before injecting the dye. Locate the tail's lateral vein and mark the injection position in the lower portion of the tail.
- Flat mount preparation
- For preparing flat mounts, keep the dissecting tools ready along with the slides and coverslips. Place the eye in a Petri dish filled with chilled PBS and start dissecting the eye as mentioned in step 1.2.2.
- Now place the tip of a pointed forcep between the sclera and retina. Gently move it all along the rim of the cup, making sure the retina is not attached to the sclera at any point. If there is any obstruction, slowly cut that portion along the rim using curved micro scissors.
- Once it is confirmed that the retina is not attached to the sclera from any side, cut near the optic disc, making a small hole such that the retina completely detaches from the sclera. Slowly push the retina into PBS solution and place it on a clean flat slide with the help of a spatula or forceps.
- Using micro scissors or scalpel blades, make minor cuts in the retinal tissue, dividing it into four quadrants. Make sure that the cuts are at least 2 mm away from the hole left by the optic disc at the center, as shown in Figure 1.
- Remove excess PBS from the sides of tissue using 0.2 mL tips without disturbing the flat mount.
- Place a drop of anti-fading mounting medium (50 µL to 100 µL) on a flat mount, cover with a coverslip, and visualize immediately under a confocal microscope.
NOTE: Maintain minimum light during the entire procedure.
- Visualization of flat mount
- Visualize the flat mount under 10x objective of a confocal microscope. Perform tile scanning to capture the entire flat mount as a single image. The number of tiles depends upon the size of the retinal flat mount. Also, perform Z-stacking to visualize the veins and arteries in different foci (preferred size for Z-stack is 5 µm, depending on which, the number of Z-stack steps varies).
- Use PMT and HyD detector at % gain as 700-900 and 100-150, respectively. Choose emission range between 510-530 nm for FITC-dextran dye.
Representative Results
Retinal histology
In the diabetic retina, retinal cells undergo degeneration. In addition, the thickness of the retinal layers increase due to edema22. The images obtained after Hematoxylin and Eosin staining can be used for cell count and measurement of the thickness of different layers, as shown in Figure 2 using ImageJ.
Blood-retinal barrier breakdown assay
As the BRB is compromised in diabetic rats, leakage becomes prominent, leading to the accumulation of biomolecules from plasma to the retina and vitreous. In diabetic rats, protein leakage from plasma to vitreous is around three folds higher when compared to healthy rats (Figure 3). Protein can be estimated using any kit method such as the Lowry method, Bicinchoninic acid (BCA) method, or Bradford method (mentioned in this paper, section 2.3). It is recommended to freshly dissect the eye, as a delay in processing could lead to strong vitreous adhesion with the retina, making it difficult to separate. Improper separation of vitreous from the retina may lead to false-positive results.
Fluorescence angiography
This technique is used to visualize the retina's leakage and vasculature, including micro and macro vasculature. The selection of the size of FITC-dextran is based on its application for various studies. Low molecular weight FITC-dextran, ranging from 4-6 kDa, is used to visualize leakage in the vasculature. In contrast, high molecular weight FITC-dextran molecules, ranging from 70 kDa to 2 million Da, are used to visualize angiogenesis. Successful injection of dye results in visualizing the entire vasculature of the retinal flat mount, as shown in Figure 4. The obtained images after confocal microscopy can be used to determine the area of vasculature occupied in the entire retina using ImageJ software to compare healthy and diseased groups.
Figure 1. Retinal flat-mount preparation. Please click here to view a larger version of this figure.
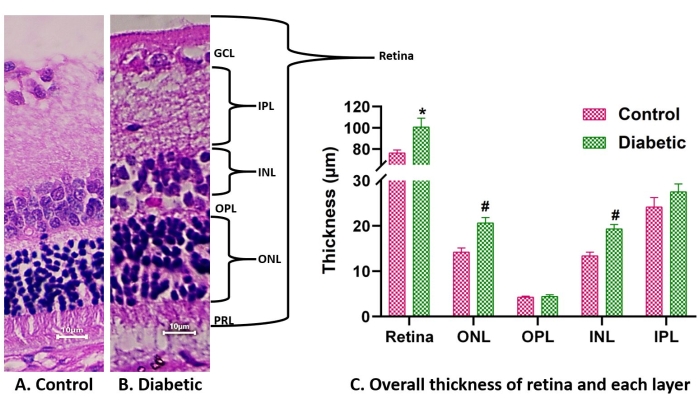
Figure 2. Histology images of healthy vs. diabetic retina. (A) and (B) represent the H&E-stained retinal images of control (A) and diabetic rat (B) under 40x. The overall thickness of the retina and each layer increases in diabetic conditions (C). Abbreviations: PRL = photoreceptor layer, ONL = outer nuclear layer, OPL = outer plexiform layer, INL = inner nuclear layer, IPL = inner plexiform layer, and GCL = Ganglion cell layer. Data are represented as mean ± SD. * represents a significant difference from the control group (P < 0.0001), whereas # represents a significant difference from the control group at P < 0.05, obtained by the ANOVA test. Please click here to view a larger version of this figure.
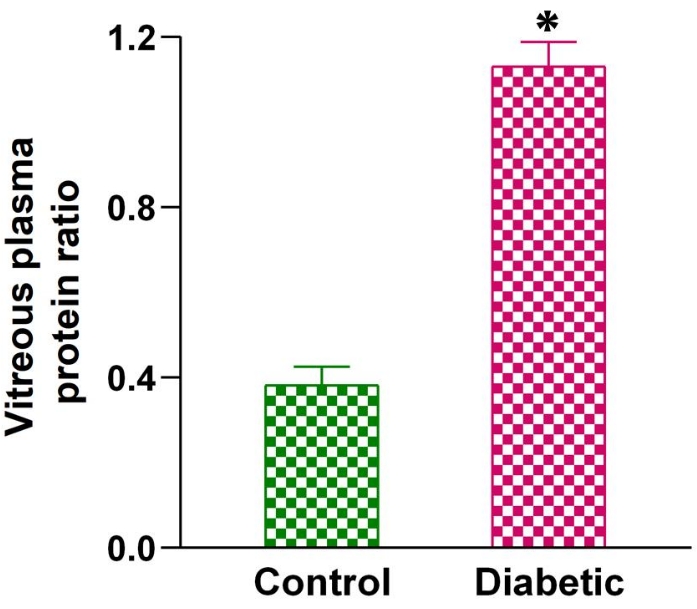
Figure 3. Blood-retinal barrier breakdown in healthy vs. diabetic rats. The graph represents the vitreous plasma protein ratio of healthy vs. diabetic rats. Data are represented as mean ± SEM. * represents a significant difference from the control group (P < 0.01) obtained by unpaired t-test. Please click here to view a larger version of this figure.
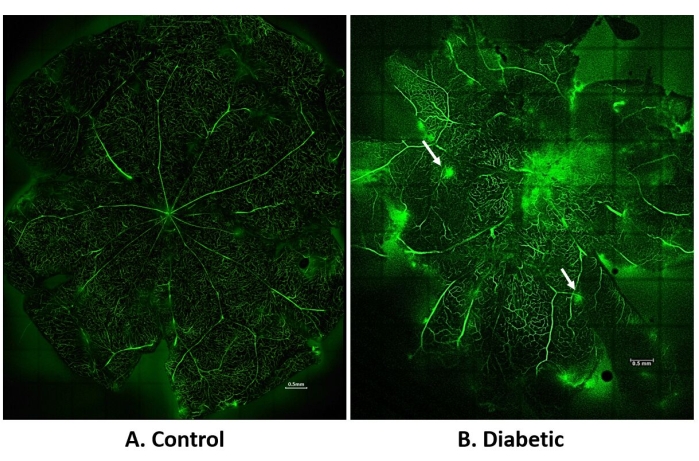
Figure 4. Fluorescence angiography of retinal vasculature. (A) and (B) represent a flat mount of retina visualized by tile scan and stitching the images under 10x. (A) represents control retina, (B) represents diabetic retina. The white arrow indicates leakage. All images were obtained by Z-stack (5 µm thickness). Scale bars in (A) and (B) are 0.5 mm. Please click here to view a larger version of this figure.
Discussion
Histology
Retinal histology is performed to visualize the morphological changes of retinal cells and layers. Various steps, including choice of fixative solution, fixation duration, dehydration, and paraffin impregnation, need to be optimized. The tissue size should not exceed 3 mm, as the fixative penetration becomes slow. The commonly used 4% paraformaldehyde leads to retinal detachment even in the healthy eye due to the relatively high osmolarity of the solution compared to aqueous humor and vitreous humor, which leads to false-positive results23. The high osmolarity of the solution results in volume contraction and leads to shrinkage of tissues. The fixative solution used in this protocol is 1% formaldehyde with 1.25% glutaraldehyde, which has relatively less osmolarity, reduces the tissue distortion, minimizes the retinal detachment from the retinal pigment epithelial layer, and also preserves the structure in its native state. Another choice of fixative solution is acetic acid and ethanol with formaldehyde. Acidic condition of fixative solution can help in rapid fixation of tissue, while ethanol improves the penetration of the fixative solution. However, optimizing the ratio of ethanol and acetic acid is essential as excess concentration may lead to shrinkage or swelling of the tissue, respectively24. Evaluation parameters for successful tissue fixation include detachment of the retina, intact retinal layers, and tissue shrinkage. Another critical factor while performing fixation is the volume of fixative solution, which should be at least 20 to 25 times the size of the eye for proper fixation25. In addition, it is important to swirl the eyeball or tissue whenever transferred from one solution to another, so that it does not stick to the bottom or walls of the container.
Incomplete tissue processing, including dehydration and impregnation with paraffin, can shrink and dry tissue, which can be visualized when depression occurs on the surface of the block. In addition, poor tissue processing will also result in the coloration of sections into white when exposed to water25. If the tissue is not processed correctly, it can be taken back through xylene to remove paraffin, followed by rehydration in downgradient of ethanol, (i.e., 100% ethanol, 90% ethanol, and 70% ethanol). Again, repeat the steps from 1.3 to 1.4 to successfully prepare paraffin blocks.
While preparing paraffin blocks, it is essential to make sure that the tissue is surrounded from all sides by paraffin and no air bubbles are seen. Any mistake in preparing the block will lead to unsuccessful sectioning of tissue. Sometimes the posterior cup gets folded during the processing; at this point, it can be pulled gently apart using forceps (in heated paraffin) but not to the extent that it damages the tissue. Suppose the tissue orientation in steel mold is not as desired while the paraffin starts solidifying; in that case, it can be put back to melted paraffin and tissue to re-orient the tissue to prepare paraffin blocks. Also, while transferring the tissue to the embedding mold, ensure that the paraffin around the tissue does not solidify. It creates a hairline separation between the tissue and embedding medium (paraffin) in the block25. If it happens, melt the paraffin around the tissue by placing it in hot paraffin and placing it in the steel mold again.
During sectioning, the blade must be sharp enough to give unfolded ribbons of tissue. Do not use a particular portion of the blade more than 30 times. If the block does not section properly, assume that the blade is blunt and, therefore, replace it or re-sharpen it. If the sections are not proper, it could also be due to improper paraffin impregnation or the use of old paraffin to impregnate and prepare blocks. The paraffin in the block can be melted down, and fresh paraffin can be used to prepare the block. To avoid the uneven sections, rotate the wheel of the microtome slowly with even turns rather than fast, jerky movements.
While performing Hematoxylin and Eosin staining, optimizing the time of Hematoxylin and Eosin stain is critical as it may lead to under- or over-staining tissues. Improper melting of paraffin or rehydration of the tissue will lead to uneven staining. It can be visualized as the white sections on the slide. Dehydrate the tissue and again melt paraffin in a hot air oven, followed by the steps mentioned in Table 1. Moreover, one of the common errors performed during H&E staining is the use of alcohol as a dehydrant after Eosin stain. Excessive use of alcohol might lead to under-staining of cytoplasm and inclusion bodies which may lead to poorly stained sections25. Use of mountant medium should be done immediately after removing excess xylene, and avoid drying of xylene as it may lead to distortion of the tissue. Also, aqueous-based mountant (such as glycerol in PBS) must be avoided as the tissue is in a dehydrated state. Aqueous-based mountant does not penetrate the tissue and results in hazy images.
Upon optimizing all the steps mentioned above, researchers will be able to perform histology successfully. It takes around three to fice attempts to choose the desired fixative solution and optimize other process parameters.
Blood-retinal barrier breakdown
The most delicate step in this technique is the isolation of the vitreous from the eye. It is best performed on a fresh eye. Use of dry ice is recommended for easy isolation of vitreous, especially if the vitreous remains attached to the retina. Often, lens or retina mix with vitreous can be identified by turbidity of vitreous due to retina. These problems can usually be avoided using freezing conditions. However, if the vitreous tags are beside the lens, they can be separated by cutting at the intersection of the lens and vitreous using micro scissors. If the retina pulls out along with the vitreous, the retina can be removed piece-by-piece using micro forceps. The use of filter centrifugation is also suggested for easy isolation of the vitreous from the lens and retina26. As vitreous is a gel-like structure, it needs homogenization for a homogenous mixture of proteins. Usually, as mentioned in step 2.2.1, a single homogenization cycle is sufficient for proper homogenization, which can be identified by liquefication of vitreous humor upon centrifugation. If the gel nature of the vitreous is observed after centrifugation, repeat homogenization (step 2.2.1). Protein quantification can be performed using any kit method, but should be sensitive to detecting the low protein levels in vitreous up to 2 µg/µL.
Fluorescence angiography
This technique aims to visualize the retina's vascular network, and hence, the dye must reach evenly in the retinal microvessels. Various steps can be optimized to ensure this, such as the injection site and circulation time. A dye can be injected via tail vein injection or cardiac injection. Cardiac injection risks injecting the dye at a site other than the heart if not well trained; therefore, tail vein injection is preferred. However, identifying the lateral vein is easy when dipped in warm water (37-40 °C) and cleaned with 70% ethanol. Ethanol helps dilate the blood vessels, which can help in accessing the location of the vein. It is also essential that the dye is injected into the vein only. Failure to inject dye into the tail vein can be felt by resistance while injecting the dye or bulging the nearby area due to subcutaneous injection. The needle can be removed and re-inserted in another site of the tail. Successful tail vein injection can also be visualized by forced urination of the rat; the color of dye is visible in rat urine within 30 to 40 s.
Also, the circulation time is essential; usually, 5 to 10 min are sufficient to circulate dye in retinal vessels successfully. A flat mount can be prepared in a fresh eye or a fixed eye. Preparing a flat mount in a fresh eye can be challenging, but once mastered, the technique is most preferred as during fixation, the dye starts to leak in fixative solution27. Therefore, fixation should not be prolonged for more than 30 min for the whole eye. In case of difficulty separating fresh retina, it can even be fixed into a fixative solution (4% formaldehyde) after separating the posterior cup from the anterior cup for 15 to 20 min. It is often seen that the dye leaks from the blood vessels into surrounding tissue, even in healthy retina. This could be due to excess fixation; hence, the time needed for fixation should be optimized. It only enhances the easy isolation of the retina and preserves the structure for future use. Moreover, increased circulation time of dye in the animal may also lead to dye leakage from retinal vessels to surrounding tissue, which needs to be optimized.
Size of FITC-dextran is essential as higher molecular weight dye will not enter micro vasculatures. In contrast, lower molecular weight dye will leak from new vessels of the healthy retina28. Therefore, FITC-dextran with a molecular weight of 70 kD is preferred to perform fluorescence angiography, to visualize leakage and vasculature in the retina. However, a significant limitation of this technique is the duration of development of neovascularization in diabetic rats and the invasive procedure of the study, which could be replaced in the future by non-invasive techniques such as electroretinogram, fundus, and other non-invasive optical instruments.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
Authors would like to acknowledge Indian Council of Medical Research (ICMR; ITR-2020-2882) for funding support to Dr. Nirmal J. We would also like to thank University Grant of Commission for providing Junior Research Fellowship to Manisha Malani and Central Analytical Laboratory Facility, BITS-Pilani, Hyderabad campus for providing infrastructural facility.
Materials
| Histology | |||
| Reagents | |||
| Isoflurane | Abbott | Anesthesia agent | |
| Ketamine hydrochloride | Troikaa Pharmaceuticals | Anesthesia agent | |
| Xylazine | Indian Immunologicals Limited | Anesthesia agent | |
| Pentobarbital sodium | Zora Pharma | Euthanesia agent | |
| Fixative solution (1 % formaldehyde, 1.25 % Glutaraldehyde | HiMedia, Avra | MB059, ASG2529 | Prepared in-house |
| Ethanol | Hayman | F204325 | Dehydration |
| Xylene | HiMedia | MB-180 | Clearing of ethanol or paraffin |
| Paraffin wax | HiMedia | GRM10702 | used for embedding tissue |
| Glycerol | HiMedia | TC503 | To prepare albumin coated slides. Glycerol and egg albumin is mixed in 1:1 ratio to coat on slides |
| Hydrochloric acid | Sisco Research laboratories Pvt. Ltd. | 65955 | For preparation of 1 % acid alcohol |
| Acetic acid | HiMedia | AS119 | For preparation of eosin |
| Scotts water | Leica | 3802900 | Bluing reagent |
| Papanicolaou's solution 1b Hematoxylin solution | Sigma | 1.09254.0500 | Staining of nuclei |
| Eosin | HiMedia | GRM115 | Staining of cytoplasm, 0.25 % solution was prepared in-house |
| DPX Mountant media | Sigma | 6522 | Visualization and protection of retinal sections |
| Equipments | |||
| Glassware | Borosil | ||
| Corneal forcep | Stephens Instruments | S5-1200 | Dissection |
| Colibri forcep | Stephens Instruments | S5-1135 | Dissection |
| Curved micro scissor | Stephens Instruments | S7-1311 | Dissection |
| Vannas scissor | Stephens Instruments | S7-1387 | Dissection |
| Iris scissor | Stephens Instruments | S7-1015 | Dissection |
| Cassettes | HiMedia | PW1292 | To hold tissue during histology processing |
| Water bath | GT Sonic | GT Sonic-D9 | Temperature maintenance |
| Paraffin embedding station | Myr | EC 350 | Preparation of paraffin blocks |
| Microtome | Zhengzhou Nanbei Instrument Equipment Co., Ltd. | YD-335A | Sectioning |
| Blades | Leica | Leica 818 | Sectioning |
| Slides | HiMedia | BG005 | Holding paraffin-tissue sections |
| Coverslips | HiMedia | BG014C | To cover tissue after adding mounting media |
| Blood Retinal Barrier breakdown | |||
| Reagents | |||
| Isoflurane | Abbott | B506 | Anesthesia |
| Dry ice | Not applicable | Not applicable | Dissection |
| Bradford reagent | Sigma | B6916 | Protein quantification |
| Equipments | |||
| Corneal forcep | Stephens Instruments | S5-1200 | Dissection |
| Colibri forcep | Stephens Instruments | S5-1135 | Dissection |
| Curved micro scissor | Stephens Instruments | S7-1311 | Dissection |
| Vannas scissor | Stephens Instruments | S7-1387 | Dissection |
| Iris scissor | Stephens Instruments | S7-1015 | Dissection |
| Glassware | Borosil | Not applicable | |
| EDTA coated tubes | J.K Diagnostics | Not applicable | Separate plasma from whole blood |
| Homogenization tubes | MP Biomedicals | SKU: 115076200-CF | Homogenization of vitreous |
| Homogenization caps | MP Biomedicals | SKU: 115063002-CF | Homogenization of vitreous |
| Glass beads | MP Biomedicals | SKU: 116914801 | Homogenization of vitreous |
| Homogeniser | Bertin Instruments | P000673-MLYS0-A | Homogenization of vitreous |
| 96-well plate – Transparent | Grenier | GN655101 | Protein quantification |
| Plate reader | Molecular devices | SpectrMax M4 | Absorbance measurement |
| Centrifuge | REMI | CPR240 Plus | Centrifugation |
| Fluorescence Angiography | |||
| Reagents | |||
| Isoflurane | Abbott | B506 | Anesthesia |
| FITC-dextran 70 kD (FITC, Dextran, Dibutylin dilaurate, DMSO | FITC, Dextran and Dibutylin dilaurate from Sigma; DMSO from HiMedia | FITC-F3651,Dextran-31390,Dibutylin dilaurate -29123, DMSO-TC185 | Prepared in-house |
| Fluoroshied | Sigma | F6182 | Anti-fading mounting medium |
| Equipments | |||
| Corneal forcep | Stephens Instruments | S5-1200 | Dissection |
| Colibri forcep | Stephens Instruments | S5-1135 | Dissection |
| Curved micro scissor | Stephens Instruments | S7-1311 | Dissection |
| Vannas scissor | Stephens Instruments | S7-1387 | Dissection |
| Iris scissor | Stephens Instruments | S7-1015 | Dissection |
| Glassware | Borosil | Not applicable | |
| Slides | HiMedia | BG005 | Flatmount preparation |
| Coverslips | HiMedia | BG014C | To cover tissue after adding mounting media |
| Confocal microscope | Leica | DMi8 | Visualization of flatmount |
Referencias
- Jonas, J. B., Sabanayagam, C. Epidemiology and risk factors for diabetic retinopathy. Diabetic Retinopathy and Cardiovascular Disease. 27, 20-37 (2019).
- Pandova, M. G. . Visual Impairment and Blindness. , (2019).
- Mokdad, A. H., et al. Global, regional, national, and subnational big data to inform health equity research: perspectives from the Global Burden of Disease Study 2017. Ethnicity & Disease. 29, 159-172 (2019).
- Barber, A. J., et al. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. The Journal of Clinical Investigation. 102 (4), 783-791 (1998).
- El-Asrar, A. M. A., Dralands, L., Missotten, L., Al-Jadaan, I. A., Geboes, K. Expression of apoptosis markers in the retinas of human subjects with diabetes. Investigative Ophthalmology & Visual Science. 45 (8), 2760-2766 (2004).
- Schröder, S., Palinski, W., Schmid-Schönbein, G. Activated monocytes and granulocytes, capillary nonperfusion, and neovascularization in diabetic retinopathy. The American Journal of Pathology. 139 (1), 81 (1991).
- Miyamoto, K., et al. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proceedings of the National Academy of Sciences. 96 (19), 10836-10841 (1999).
- Bhanushali, D., et al. Linking retinal microvasculature features with severity of diabetic retinopathy using optical coherence tomography angiography. Investigative Ophthalmology & Visual Science. 57 (9), 519-525 (2016).
- Wang, W., Lo, A. C. Diabetic retinopathy: pathophysiology and treatments. International Journal of Molecular Sciences. 19 (6), 1816 (2018).
- Akbarzadeh, A., et al. Induction of diabetes by streptozotocin in rats. Indian Journal of Clinical Biochemistry. 22 (2), 60-64 (2007).
- Weiss, R. B. Streptozocin: a review of its pharmacology, efficacy, and toxicity. Cancer Treatment Reports. 66 (3), 427-438 (1982).
- Karunanayake, E. H., Hearse, D. J., Mellows, G. The metabolic fate and elimination of streptozotocin. Biochemical Society Transactions. 3 (3), 410-414 (1975).
- Luna, L. G. . Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. , (1968).
- Okunlola, A., et al. Histological studies on the retina and cerebellum of Wistar rats treated with Arteether. Journal of Morphological Sciences. 31 (01), 028-032 (2014).
- Wallow, I., Engerman, R. Permeability and patency of retinal blood vessels in experimental diabetes. Investigative Ophthalmology & Visual Science. 16 (5), 447-461 (1977).
- do Cartmo, A., Ramos, P., Reis, A., Proença, R., Cunha-Vaz, J. Breakdown of the inner and outer blood retinal barrier in streptozotocin-induced diabetes. Experimental Eye Research. 67 (5), 569-575 (1998).
- Shires, T., Faeth, J., Pulido, J. Protein levels in the vitreous of rats with streptozotocin-induced diabetes mellitus. Brain Research Bulletin. 30 (1-2), 85-90 (1993).
- D’amato, R., Wesolowski, E., Smith, L. H. Microscopic visualization of the retina by angiography with high-molecular-weight fluorescein-labeled dextrans in the mouse. Microvascular Research. 46 (2), 135-142 (1993).
- Gupta, D. Fluorescein angiography refresher course: Here’s how to interpret the findings of this useful diagnostic tool. Review of Optometry. 138 (11), 60-65 (2001).
- Edelman, J. L., Castro, M. R. Quantitative image analysis of laser-induced choroidal neovascularization in rat. Experimental Eye Research. 71 (5), 523-533 (2000).
- Szabó, K., et al. Histological evaluation of diabetic neurodegeneration in the retina of Zucker diabetic fatty (ZDF) rats. Scientific Reports. 7 (1), 1-17 (2017).
- Margo, C. E., Lee, A. Fixation of whole eyes: the role of fixative osmolarity in the production of tissue artifact. Graefe’s Archive for Clinical and Experimental Ophthalmology. 233 (6), 366-370 (1995).
- Tokuda, K., et al. Optimization of fixative solution for retinal morphology: a comparison with Davidson’s fixative and other fixation solutions. Japanese Journal of Ophthalmology. 62 (4), 481-490 (2018).
- Luna, L. G. . Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. Third edition. , (1968).
- Skeie, J. M., Tsang, S. H., Mahajan, V. B. Evisceration of mouse vitreous and retina for proteomic analyses. Journal of Visualized Experiments. (50), e2795 (2011).
- D’Amato, R., Wesolowski, E., Smith, L. E. Microscopic visualization of the retina by angiography with high-molecular-weight fluorescein-labeled dextrans in the mouse. Microvascular Research. 46 (2), 135-142 (1993).
- Atkinson, E. G., Jones, S., Ellis, B. A., Dumonde, D. C., Graham, E. Molecular size of retinal vascular leakage determined by FITC-dextran angiography in patients with posterior uveitis. Eye (Lond). 5, 440-446 (1991).

