Technical Refinement of a Bilateral Renal Ischemia-Reperfusion Mouse Model for Acute Kidney Injury Research
Summary
This study established a protocol focusing on the technical refinement of a mouse model of bilateral renal ischemia-reperfusion for acute kidney injury research.
Abstract
Cardiac arrest poses a large public health burden. Acute kidney injury (AKI) is an adverse marker in survivors of cardiac arrest following the return of spontaneous circulation (ROSC) after successful cardiopulmonary resuscitation. Conversely, recovery of kidney function from AKI is a predictor of favorable neurological outcomes and hospital discharge. However, an effective intervention to prevent kidney damage caused by cardiac arrest after ROSC is lacking, suggesting that additional therapeutic strategies are required. Renal hypoperfusion and reperfusion are two pathophysiological mechanisms that cause AKI after cardiac arrest. Animal models of ischemia-reperfusion-induced AKI (IR-AKI) of both kidneys are comparable with patients with AKI following ROSC in a clinical setting. However, IR-AKI of both kidneys is technically challenging to analyze because the model is associated with high mortality and wide variation in kidney damage, which may affect the analysis. Lightweight mice were chosen, placed under general anesthesia with isoflurane, subjected to surgery with a dorsolateral approach, and their body temperature maintained during operation, thereby reducing tissue damage and establishing a reproducible acute renal IR-AKI research protocol.
Introduction
Cardiac arrest occurs more than 80,000 times annually in the United States1,2. The mortality rate of cardiac arrest is extremely high3,4,5,6. AKI is a major risk factor associated with high mortality and poor neurological outcomes in patients with cardiac arrest after ROSC7,8,9,10,11,12,13. Recovery from AKI is a good predictor of favorable neurological outcomes and discharge from the hospital14,15,16. However, effective therapies for IR-AKI are still lacking15,16,17,18,19. Additional therapeutic strategies are required to further improve the clinical outcomes of the disease.
IR-AKI with bilateral renal ischemia approach is one of the animal models used for AKI research20,21,22,23,24,25,26. Renal IR-AKI animal models are less complicated than a whole-body IR injury model for the study of AKI in patients with sudden cardiac arrest following ROSC6,27,28,29,30. This implies that consistent results from a renal IR-AKI animal model are easier to achieve because of the presence of fewer confounding factors in experiments. Moreover, renal IR-AKI protocols commonly involve a unilateral or bilateral renal pedicle occlusion. Conditions in experiments on bilateral renal IR-AKI are comparable to clinical conditions for AKI following ROSC in patients with sudden cardiac arrest after successful cardiopulmonary resuscitation. Although the pathological characteristics of the kidneys in both models reflect the pathological characteristics of human renal IR injury31,32,33, a bilateral renal ischemia approach is more relevant to AKI under human pathological conditions, such as cardiac failure, vasoconstriction, and septic shock35. Bilateral renal IR-AKI animal models are suitable for studies focusing on renal IR injuries in cardiac arrest following ROSC.
Bilateral renal IR-AKI models are associated with technical difficulties, experimental complexity, and long surgery duration23,26,32,33,35,36. To overcome these technical difficulties, the present study established a reliable bilateral IR-AKI research protocol in mice by making some technical modifications. The proposed protocol resulted in fewer surgical complications, less tissue damage, and a lower likelihood of mortality during surgery. Therefore, it can be used to investigate the pathophysiological processes of AKI after ROSC to develop new therapeutic strategies against renal hypoperfusion and reperfusion damage37,38,39.
Protocol
All animal experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals, published by the US National Institutes of Health (NIH publication no. 85-23, revised 1996). The study protocol was approved by and in accordance with the guidelines of the Institutional Animal Care and Use Committee at Fu-Jen Catholic University. See the Table of Materials for details about all materials and instruments used in this protocol.
1. Preparing the mice
- Select 8-week-old C57BL/6 male mice with a weight of 21-23 g.
- House and maintain the mice under a 12 h light and dark cycle at a controlled temperature (21 ± 2 °C) with free access to food, standard mouse food pellets, and tap water.
2. Anesthesia
- Put on a surgical mask and sterile gloves.
- Put the mice under anesthesia with 2% isoflurane mixed with oxygen at 1 L/min in the induction chamber.
- Assess the level of anesthesia by pedal reflex.
NOTE: Pedal reflex is a retraction of the hind paw in response to a firm toe pinch. The anesthetization is complete when the pedal reflex disappears. - Move and place each mouse in prone position on a surgery platform with an electric blanket to maintain their body temperature once the anesthetization is complete. Stabilize the body temperature before surgery and monitor with rectal temperature probes. Apply ophthalmic ointment to both eyes to prevent dryness.
- Tape the paws of the mice to the board.
- Attach a mask to the face of the mice to provide a constant supply of 1% isoflurane and 1 L/min oxygen
- Assess the level of anesthesia by pedal reflex regularly and adjust the anesthetic delivery accordingly during the surgery.
3. Bilateral renal IR-AKI surgery
- Touch the back and find the lumbar spine of the mice manually. Move along the spine cephalically and look for costovertebral angles that are below both sides of the last rib of the mice.
- Apply hair removal lotion to both sides of the costovertebral angle region for approximately 30 s and then remove the fur with saline.
- Disinfect the shaved skin with three rounds of betadine solution and 75% alcohol using cotton balls.
NOTE: Maintaining a sterile field for surgery throughout the procedure is key. Apply a surgical drape and use sterile instruments. - Use fine-tip forceps to gently lift the skin below the left costovertebral angle, and then use scissors to create a 1 cm oblique dorsolateral incision along the skin tension lines from the lumbar midline on the left flank. Transect the muscle wall of the left flank using scissors to visualize the left kidney.
- Repeat the aforementioned surgical procedures to visualize the right kidney. Remove the small amounts of blood produced during the procedure with sterile cotton swabs.
- Push and separate the left kidney carefully from the surrounding tissue with forceps. Identify the renal pedicle after the left kidney is exposed.
NOTE: Be careful not to injure the adrenal gland and the surrounding blood vessels. - Clamp over the left renal pedicle with a microvascular clip for 25 min. Confirm ischemia by a visible change in kidney color from pink to dark red.
- Cover the clamped kidney with sterile saline wet cotton balls to avoid desiccation during left renal pedicle clamping.
- Repeat the aforementioned surgical procedures to clamp the right renal pedicle with a microvascular clip for 25 min.
- Cover the clamped kidney with sterile saline wet cotton balls to avoid desiccation during right renal pedicle clamping.
- Monitor anesthesia depth and humidity of the sterile saline wet cotton balls periodically.
- Open the left microvascular clip to start reperfusion of the left kidney. Confirm reperfusion by a visible color change of the left kidney from dark red to pink.
- Open the right microvascular clip to start reperfusion of the right kidney.
- After the kidney color change is verified, return the kidney to the abdominal cavity.
- Close the abdominal cavity and skin with 6-0 absorbable suture materials.
- Scrub to disinfect the wound with a betadine solution and 75% alcohol using cotton balls.
- Observe the animal carefully until it begins to move freely and feed.
NOTE: Pay close attention to the animals until they have regained sufficient consciousness to maintain sternal recumbency. - Administer carprofen (5 mg/kg in 0.2 mL, administered subcutaneously) for 2-3 days to prevent postsurgical pain.
Representative Results
The quality of the bilateral renal IR-AKI surgery should be assessed before further microscopic or molecular analysis. During surgery, renal ischemia should be confirmed by seeing whether the kidney has changed color from pink to dark red soon after the renal pedicle is clamped with a microvascular clip (Figure 1). After surgery, kidney damage caused by IR-AKI surgery can be further validated with a few microliters of serum through submandibular blood collection for biochemical analysis where the results indicate an increase in the level of blood urea nitrogen and creatinine from baseline (Figure 2).
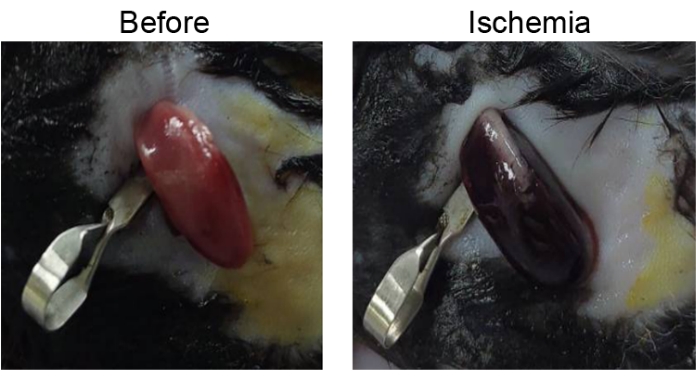
Figure 1: Renal ischemia after renal pedicle clamping. A kidney color change from pink to dark red revealing that the kidney perfusion has become inadequate. Please click here to view a larger version of this figure.
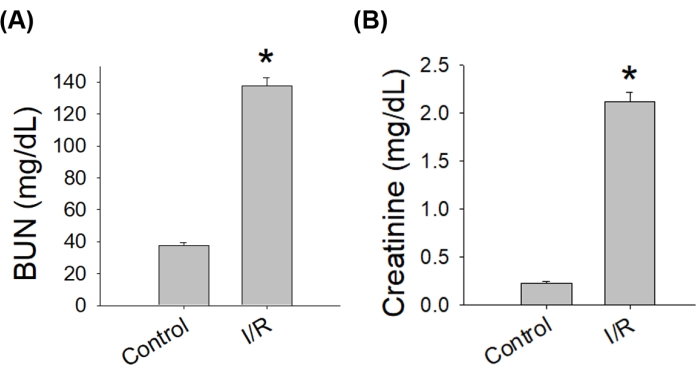
Figure 2: Renal insufficiency after bilateral IR-AKI surgery. Serum levels of blood urea nitrogen and creatinine increased 2 days after renal reperfusion. Abbreviations: IR-AKI = ischemia-reperfusion-induced acute kidney injury; BUN = blood urea nitrogen; I/R = ischemia-reperfusion (n = 4, *p < 0.05 versus control). Please click here to view a larger version of this figure.
Discussion
The proposed bilateral IR-AKI protocol is suitable for investigating the mechanism of hypoperfusion and reperfusion injury of both kidneys. The protocol suggests that lightweight mice, general anesthesia with isoflurane, a dorsolateral approach to the surgery, and body temperature maintenance during the operation mitigate the associated technical difficulties, shorten the duration of the surgery, and increase the consistency of the procedure for acute bilateral renal IR-AKI research.
Technical difficulties influence renal damage severity in bilateral renal IR-AKI surgery33. In addition to mouse strain, sex, age, and heating systems36,40,41,42,43,44, proper placement of the vascular clamp is essential for consistent results. Studies have recommended careful dissection of the surrounding adipose tissue to release the kidney and renal pedicles or arteries23,26,32,35,36. Compared with mice aged 8-20 weeks old typically weighing 25-28 g that have been studied in the literature23,32,35,36, this study used relatively young and light mice (8 weeks old and weighing 21-23 g) to reduce the amount of perirenal adipose tissue, which could expose the kidney and renal pedicles easily without requiring peripheral tissue dissection and proper placement of the vascular clamps. This would reduce procedure-related trauma and the technical complexity, shorten anesthesia and surgery duration, quicken the learning curve for those unfamiliar with the study procedure, and increase the reproducibility of the study.
General anesthesia influences the results of an IR-AKI study. Prolonged anesthesia increases animal loss during surgery33. In the literature, phenobarbital sodium, a long-acting barbiturate that depresses the central nervous system, has been administered subcutaneously for IR-AKI surgery26,33,35. Phenobarbital sets in after 5 min and helps achieve surgical anesthesia in at least 15 min45. Therefore, phenobarbital should be delivered only by skilled surgeons to avoid prolonging anesthesia (>60 mg/kg) and animal loss during surgery33. By contrast, this study's use of isoflurane, which is a nonflammable inhaled anesthetic, induced a rapid onset that achieved surgical anesthesia in 7-10 min and ceased the effect in 15 min after stopping the inhalation46. The delivery of isoflurane, in conjunction with oxygen, is easy for the operator to start, maintain, and stop instantly during surgery and is suggested for renal IR-AKI surgery.
Finally, the method to approaching the renal pedicles may influence the quality of IR-AKI surgery. Some IR-AKI studies have investigated the renal pedicle using midline laparotomy where the abdominal cavity was opened, and the peritoneum and intestines were pushed aside to access the kidney. However, doing so may increase fluid and heat loss, surgery-related trauma, and surgical duration32,35. Therefore, this protocol suggests a dorsolateral approach for IR-AKI research to expose the kidney from the flank and the retroperitoneum to maintain body temperature and to minimize surgery-related injury, subsequently improving the surgical condition and study consistency.
This model has a potential application in studies aiming to identify and characterize markers of bilateral renal damage caused by cardiac arrest following ROSC. However, cytokines released due to surgical damage during the procedure may influence the study results, making them unrelatable to the clinical scenario and limiting the translation of the study results from benchside to bedside.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This model was developed with the financial support from Ministry of Science and Technology, Taiwan (MOST 109-2320-B-030-006-MY3). This manuscript was edited by Wallace Academic Editing.
Materials
| Absorbable Suture, 6-0 | Ethicon | J510G-BX | |
| Betadine solution | Shineteh Istrument | ||
| Carprofen | Sigma | PHR1452 | |
| Cotton balls | Shineteh Istrument | ||
| Graefe Forceps | Fine Science Tools | 11051-10 | |
| Heating pad | Shineteh Istrument | ||
| Isoflurane | Piramal Critical Care Inc. | 26675-46-7 | |
| Moria Vessel Clamp | Fine Science Tools | 18320-11 | |
| Olsen-Hegar needle holder | Fine Science Tools | 12002 – 12 | |
| Saline | Shineteh Istrument | ||
| Scalpel blades | Shinva | s2646 | |
| Small Animal Anesthesia Machine | Sheng-Cing Instruments Co. | STEP AS-01 | |
| Tissue scissors | Fine Science Tools | 14072 – 10 |
Referencias
- Holmberg, M. J., et al. Annual incidence of adult and pediatric in-hospital cardiac arrest in the United States. Circulation: Cardiovascular Quality and Outcomes. 12 (7), 005580 (2019).
- Benjamin, E. J., et al. Heart disease and stroke statistics-2018 update: a report from the American Heart Association. Circulation. 137 (12), 67 (2018).
- Lascarrou, J. B., et al. Targeted temperature management for cardiac arrest with nonshockable rhythm. The New England Journal of Medicine. 381 (24), 2327-2337 (2019).
- Chang, H. C., et al. Factors affecting outcomes in patients with cardiac arrest who receive target temperature management: The multi-center TIMECARD registry. Journal of the Formosan Medical Association. 121 (1), 294-303 (2022).
- Yu, G., et al. Comparison of the survival and neurological outcomes in OHCA based on smoking status: investigation of the existence of the smoker’s paradox. Signa Vitae. 18 (2), 121-129 (2022).
- Chen, Y. C., et al. Major interventions are associated with survival of out of hospital cardiac arrest patients – a population based survey. Signa Vitae. 13 (2), 108-115 (2017).
- Sandroni, C., et al. Acute kidney injury after cardiac arrest: a systematic review and meta-analysis of clinical studies. Minerva Anestesiologica. 82 (9), 989-999 (2016).
- Patyna, S., et al. Acute kidney injury after in-hospital cardiac arrest in a predominant internal medicine and cardiology patient population: incidence, risk factors, and impact on survival. Renal Failure. 43 (1), 1163-1169 (2021).
- Storm, C., et al. Impact of acute kidney injury on neurological outcome and long-term survival after cardiac arrest – A 10 year observational follow up. Journal of Critical Care. 47, 254-259 (2018).
- Geri, G., et al. Acute kidney injury after out-of-hospital cardiac arrest: risk factors and prognosis in a large cohort. Intensive Care Medicine. 41 (7), 1273-1280 (2015).
- Guo, Q. Y., Xu, J., Shi, Q. D. Gasping as a predictor of short- and long-term outcomes in patients with cardiac arrest: a systematic review and meta-analysis. Signa Vitae. 17 (2), 208-213 (2021).
- Chen, P. C., et al. Prognostic factors for adults with cardiac arrest in the emergency department: a retrospective cohort study. Signa Vitae. 18 (3), 56-64 (2022).
- Lee, M. J., et al. Predictors of survival and good neurological outcomes after in-hospital cardiac arrest. Signa Vitae. 17 (2), 67-76 (2021).
- Deakin, C. D., et al. European Resuscitation Council guidelines for resuscitation 2010 section 4. adult advanced life support. Resuscitation. 81 (10), 1305-1352 (2010).
- Cha, K. C., et al. Recovery from acute kidney injury is an independent predictor of survival at 30 days only after out-of-hospital cardiac arrest who were treated by targeted temperature management. Signa Vitae. 17 (2), 119-126 (2021).
- Park, Y. S., et al. Recovery from acute kidney injury as a potent predictor of survival and good neurological outcome at discharge after out-of-hospital cardiac arrest. Critical Care. 23 (1), 256 (2019).
- Mah, K. E., et al. Acute kidney injury after in-hospital cardiac arrest. Resuscitation. 160, 49-58 (2021).
- Pelkey, T. J., et al. Minimal physiologic temperature variations during renal ischemia alter functional and morphologic outcome. Journal of Vascular Surgery. 15 (4), 619-625 (1992).
- Kim, H., et al. Effect of different combinations of initial body temperature and target temperature on neurological outcomes in out-of-hospital cardiac arrest patients treated with targeted temperature management. Signa Vitae. , 1-7 (2022).
- Wyss, J. C., et al. Differential effects of the mitochondria-active tetrapeptide SS-31 (D-Arg-dimethylTyr-Lys-Phe-NH2) and its peptidase-targeted prodrugs in experimental acute kidney injury. Frontiers in Pharmacology. 10, 1209 (2019).
- Wang, Y., Wang, B., Qi, X., Zhang, X., Ren, K. Resveratrol protects against post-contrast acute kidney injury in rabbits with diabetic nephropathy. Frontiers in Pharmacology. 10, 833 (2019).
- Li, S., Yu, L., He, A., Liu, Q. Klotho inhibits unilateral ureteral obstruction-induced endothelial-to-mesenchymal transition via TGF-beta1/Smad2/Snail1 signaling in mice. Frontiers in Pharmacology. 10, 348 (2019).
- Godoy, J. R., Watson, G., Raspante, C., Illanes, O. An effective mouse model of unilateral renal ischemia-reperfusion injury. Journal of Visualized Experiments. (173), e62749 (2021).
- Chen, Q., et al. SIRT1 mediates effects of FGF21 to ameliorate cisplatin-induced acute kidney injury. Frontiers in Pharmacology. 11, 241 (2020).
- Li, H. D., et al. Application of herbal traditional Chinese medicine in the treatment of acute kidney injury. Frontiers in Pharmacology. 10, 376 (2019).
- Grenz, A., et al. Use of a hanging-weight system for isolated renal artery occlusion during ischemic preconditioning in mice. American Journal of Physiology-Renal Physiology. 292, 475-485 (2007).
- Gao, Q., et al. Accumulated epinephrine dose is associated with acute kidney injury following resuscitation in adult cardiac arrest patients. Frontiers in Pharmacology. 13, 806592 (2022).
- Oh, Y. T., et al. Vasoactive-inotropic score as a predictor of in-hospital mortality in out-of-hospital cardiac arrest. Signa Vitae. 15 (2), 40-44 (2019).
- Burne-Taney, M. J., et al. Acute renal failure after whole body ischemia is characterized by inflammation and T cell-mediated injury. American Journal of Physiology-Renal Physiology. 285 (1), 87-94 (2003).
- Adams, J. A., et al. Periodic acceleration (pGz) prior to whole body ischemia reperfusion injury provides early cardioprotective preconditioning. Life Sciences. 86 (19-20), 707-715 (2010).
- Gaut, J. P., Liapis, H. Acute kidney injury pathology and pathophysiology: a retrospective review. Clinical Kidney Journal. 14 (2), 526-536 (2021).
- Hesketh, E. E., et al. Renal ischaemia reperfusion injury: a mouse model of injury and regeneration. Journal of Visualized Experiments. (88), e51816 (2014).
- Wei, Q., Dong, Z. Mouse model of ischemic acute kidney injury: technical notes and tricks. American Journal of Physiology-Renal Physiology. 303 (11), 1487-1494 (2012).
- Wei, Q., Dong, Z. Regulation and pathological role of bid in ischemic acute kidney injury. Renal Failure. 29 (8), 935-940 (2007).
- Grenz, A., et al. Use of a hanging-weight system for isolated renal artery occlusion. Journal of Visualized Experiments. (53), e2549 (2011).
- Skrypnyk, N. I., Harris, R. C., de Caestecker, M. P. Ischemia-reperfusion model of acute kidney injury and post injury fibrosis in mice. Journal of Visualized Experiments. (78), e50495 (2013).
- Han, S. J., Lee, H. T. Mechanisms and therapeutic targets of ischemic acute kidney injury. Kidney Research and Clinical Practice. 38 (4), 427-440 (2019).
- Huang, C. W., et al. A novel caffeic acid derivative prevents renal remodeling after ischemia/reperfusion injury. Biomedicine & Pharmacotherapy. 142, 112028 (2021).
- Spoelstra-de Man, A. M. E., Oudemans-van Straaten, H. M. Acute kidney injury after cardiac arrest: the role of coronary angiography and temperature management. Critical Care. 23 (1), 193 (2019).
- Burne, M. J., Haq, M., Matsuse, H., Mohapatra, S., Rabb, H. Genetic susceptibility to renal ischemia reperfusion injury revealed in a murine model. Transplantation. 69 (5), 1023-1025 (2000).
- Muller, V., et al. Sexual dimorphism in renal ischemia-reperfusion injury in rats: possible role of endothelin. Kidney International. 62 (4), 1364-1371 (2002).
- Schmitt, R., Marlier, A., Cantley, L. G. Zag expression during aging suppresses proliferation after kidney injury. Journal of the American Society of Nephrology. 19 (12), 2375-2383 (2008).
- Oxburgh, L., de Caestecker, M. P. Ischemia-reperfusion injury of the mouse kidney. Methods in Molecular Biology. 886, 363-379 (2012).
- Delbridge, M. S., Shrestha, B. M., Raftery, A. T., El Nahas, A. M., Haylor, J. L. The effect of body temperature in a rat model of renal ischemia-reperfusion injury. Transplantation Proceedings. 39 (10), 2983-2985 (2007).
- IBM Micromedx, I. Phenobarbital sodium. IBM Corporation Available from: https://www-micromedexsolutions-com.autorpa.mmh.org.tw/micromedex2/librarian/CS/53C834/ND_PR/evidencexpert/ND_P/evidencexpert/DUPLICATIONSHIELDSYNC/51EFF0/ND_PG/evidencexpert/ND_B/evidencexpert/ND_AppProduct/evidencexpert/ND_T/evidencexpert/PFActionId/evidencexpert.DoIntegratedSearch?SearchTerm=Phenobarbital+Sodium&fromInterSaltBase=true&UserMdxSearchTerm=%24userMdxSearchTerm&false=null&=null (2022)
- IBM Micromedx, Isoflurane. IBM Corporation Available from: https://www-micromedexsolutions-com.autorpa.mmh.org.tw/micromedex2/librarian/PFDefaultActionId/evidencexpert.DoIntegratedSearch?navitem=headerLogout (2022)

