A Calcium Phosphate-Induced Mouse Abdominal Aortic Aneurysm Model
Summary
This protocol describes a calcium phosphate-induced abdominal aortic aneurysm (AAA) mouse model to study the pathological features and molecular mechanisms of AAAs.
Abstract
An abdominal aortic aneurysm (AAA) is a life-threatening cardiovascular disease that occurs worldwide and is characterized by irreversible dilation of the abdominal aorta. Currently, several chemically induced murine AAA models are used, each simulating a different aspect of the pathogenesis of AAA. The calcium phosphate-induced AAA model is a rapid and cost-effective model compared to the angiotensin II- and elastase-induced AAA models. The application of CaPO4 crystals to the mouse aorta results in elastic fiber degradation, loss of smooth muscle cells, inflammation, and calcium deposition associated with aortic dilation. This article introduces a standard protocol for the CaPO4-induced AAA model. The protocol includes material preparation, the surgical application of the CaPO4 to the adventitia of the infrarenal abdominal aorta, the harvesting of aortas to visualize aortic aneurysms, and histological analyses in mice.
Introduction
An abdominal aortic aneurysm (AAA) is a lethal cardiovascular disease characterized by permanent dilation of the abdominal aorta, with high mortality rates once rupture occurs. AAA is associated with aging, smoking, male gender, hypertension, and hyperlipidemia1. Several pathological processes have been shown to contribute to AAA formation, including extracellular matrix fiber proteolysis, immune cell infiltration, and loss of vascular smooth muscle cells. Currently, the pathological mechanisms of AAA remain elusive, and there are no proven drugs for the treatment of AAA1. Research into human AAA is limited due to the existence of few human aortic samples; thus, several chemical modification-induced animal AAA models have been established and widely adopted, including subcutaneous angiotensin II (AngII) infusion, perivascular or intraluminal elastase incubation, and perivascular calcium phosphate application2. A commonly used mouse model is the application of calcium phosphate (CaPO4) to the adventitia of the infrarenal abdominal aorta, which is cost-effective and does not require genetic modification.
Direct periaortic application of CaCl2 to the carotid artery of rabbits to induce aneurysmal change was initially reported by Gertz et al.3 and was later applied to the abdominal aortas of mice. The model was developed by Yamanouchi et al. to accelerate aortic dilation by using CaPO4 crystals in mice4. Infiltration of CaPO4 into mice aortas recapitulates many pathological features observed in human AAAs, including profound macrophage infiltration, extracellular matrix degradation, and calcium deposition. The risk factors of human AAA, such as hyperlipidemia, also augment CaPO4-induced AAA in mice5. In contrast to AngII perfusion-induced AAA in ApoE-/- or LDLR-/- mice, CaPO4-induced AAA occurs in the infrarenal aortic region, which mimics human AAA. Currently, this method has been widely applied to assess susceptibility to AAA development in genetically modified mice and evaluate the anti-AAA effects of drugs6,7.
Protocol
Animal studies were performed in compliance with the guidelines of the Institutional Animal Care and Use Committee of Peking University Health Science Center and were approved by the Biomedical Ethics Committee of Peking University (LA2015142). All the mice for surgery were anesthetized with isoflurane (1.5%-2%), and anesthesia was carefully monitored to avoid pain or discomfort for the mice.
1. Preparation
- Cut 0.3 cm wide strips of powder-free rubber gloves and gauze.
- Purchase 8-10-week-old C57BL/6J male mice. House the animals in an air-conditioned environment with a 12 h light-dark cycle and free access to food and water.
- Autoclave the gauze, cotton swabs, scissors, and forceps prior to surgery.
- Obtain betadine, 70% ethanol, and antiseptic hand wash.
- Put on a mask, gown, and sterile gloves.
2. Surgical procedure
- Feed chewable carprofen tablets (5 mg/kg dose) to an 8-10-week-old C57BL/6J mouse 2-4 h before surgery. Then, place the mouse in an induction chamber (206 mm x 210 mm x 140 mm) with isoflurane at a flow rate of 1.5%-2%.
- Monitor the mouse for about 5 min until respiration is visibly slowed. Ensure that the mouse has no response to pain stimulation before surgery.
- Apply an ophthalmic ointment to the eyes and provide thermal support with a heating pad or blanket. Confirm the depth of anesthesia with a toe pinch every 15 minutes during the surgical procedure.
- Shave the abdominal hair of the mouse using an electric clipper or hair removal cream. Swab and wipe the shaved area with betadine, followed by 70% ethanol, several times in a circular motion. Change gloves to maintain sterility.
- Use scissors to make a ~1.5 cm incision at the lower abdomen along the midline of the abdomen.
- Use a sterile cotton swab moistened with normal saline to carefully remove the intestine until the infrarenal aorta is visible.
- Dissect the connective tissue and fat from the infrarenal aorta for an approximately 0.5 cm section. Note the small vessels to the dorsal side and avoid tearing them. There is no need to separate the abdominal aorta from the abdominal main vein.
- Pack a piece of the saline-soaked rubber glove strip under the abdominal aorta and abdominal main vein. Use a cotton swab to wipe off excess liquid.
- Pack a piece of gauze soaked with 0.5 M CaCl2 on the adventitia of the infrarenal abdominal vasculature for 10 min. For the sham mouse group, replace the 0.5 M CaCl2 with normal saline.
- Remove the gauze and pack another piece of gauze soaked with PBS solution for 5 min to generate CaPO4 crystals in situ in the adventitia of the aorta.
- Carefully remove the rubber glove strip and gauze. Reset the intestinal tract of the mouse.
- Suture the abdominal incision and skin with a 5-0 suture.
- Place the mouse on a heating pad until the mouse regains consciousness. Provide postsurgical pain recovery housing and analgesia, according to the local animal ethics committee.
- House the mouse for another 14 days. Monitor the mouse closely after the surgery and observe at least 1x every day subsequently. Perform a necropsy immediately if any mice die during this period.
3. Harvesting for imaging of aortas
- 14 days after the surgery, sacrifice the mice using CO2.
- Cut open the mouse thoracic and abdominal cavities ventrally and cut open the right atrium.
- Perfuse the mice with PBS buffer through the left ventricle of the heart to remove blood in the aorta, and then perfuse with 4% paraformaldehyde as previously described8.
- Harvest the aorta under the stereoscope.
- Place harvested aortas in tubes containing 5 mL of 4% paraformaldehyde for 48 h.
- Remove adventitial tissue and fat carefully under the stereoscope and pin the aorta on a black wax plate with insect needles.
- Acquire aortic images.
4. Evaluation of the degradation of the elastic fibers
- Cut the aortic aneurysm tissues into serial cryosections (7 µm thick).
- Analyze the elastic fibers using a commercial elastic van Gieson (EVG) staining kit according to the manufacturer's protocol.
- Grade the elastin degradation. Grade 1: <25% degradation; grade 2: 25% to 50% degradation; grade 3: 50% to 75% degradation; or grade 4: >75% degradation.
Representative Results
14 days after CaPO4 application, the C57BL/6J male mice were euthanized, and their aortas were harvested and cleaned. The morphology of the aortas was imaged to visualize AAA formation. As shown in Figure 1A–B, the application of CaPO4 led to dilation of the infrarenal abdominal aorta. Histologically, CaPO4 resulted in a dramatic degradation of elastic fibers, as illustrated by elastin breaks (Figure 1C).
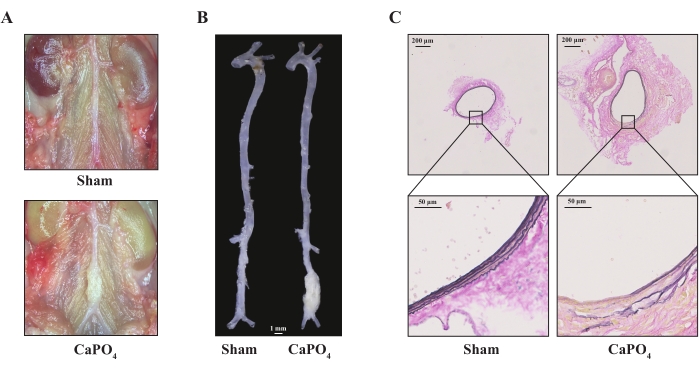
Figure 1: The 8-week-old male C57BL/6J mice were treated with saline (sham) or CaPO4 harvested after 14 days. (A) Representative images of infrarenal abdominal aorta under a stereoscope. (B) Representative morphology images of aortas from mice; scale bar = 1 mm. (C) Representative images of elastic van Gieson staining of aortas. Please click here to view a larger version of this figure.
Discussion
Periaortic application of CaPO4 is a robust approach to induce AAA in mice. Several studies have used the CaPO4 model and consistently reported that this is a rapid and reproducible method to study AAA in mice7,9. This model is considered to recapitulate part of the features of human aortic aneurysm and provide mechanistic insights into AAA pathogenesis, including inflammation and extracellular matrix degradation.
The risk factors of human AAA mainly include aging, male gender, smoking, hyperlipidemia, hypertension, and atherosclerosis10. Although not systematically studied, hyperlipidemia was also found to predispose mice to AAA expansion when using the CaPO4 model. Unlike humans, aging plays a minor role in AAA formation in the mouse CaPO4 model5. Previous large epidemiological studies have shown that diabetes is an independent negative risk factor of human AAA11. While some data suggest metformin, a diabetic drug, causes this effect, alternatively it is of interest that, in the CaPO4 model, hyperglycemia inhibits aortic dilation by the suppression of macrophage activation12.
Currently, several chemically induced murine AAA models have been established, including elastase incubation, CaPO4 incubation, and subcutaneous AngII perfusion2. Generally, the elastase and CaPO4 models have been performed in 8-10-week-old male wild-type mice, and the AngII model has been performed in hyperlipidemia mice (such as ApoE-/- and LDLR-/- mice) or 5-6-month-old mice to induce AAA13. All the three models phenocopy the major pathological characteristics of human AAA, including elastic fiber degradation and immune cell infiltration. Compared with the other two models, CaPO4 application leads to a rapid and more than 1.5-fold expansion of the aorta 7 days after surgery, associated with dramatic elastin degradation and calcium deposition4. The CaPO4 model induces a fusiform dilation at the infrarenal abdominal aorta, which mimics the human AAA condition, whereas AngII perfusion induces both suprarenal AAA and thoracic aortic aneurysm. Considering the cost of elastase, AngII, and the osmotic mini-pump, it is more cost-effective to perform the CaPO4 model. However, it is fair to state that the CaPO4 model cannot induce AAA features such as mural thrombus formation and aortic rupture, and the model is rapid and, thus, less suitable for performing intervention studies with existing AAA. The CaPO4 model is ideal for working with genetically modified mice to assess susceptibility to AAA development.
The mechanism underlying CaPO4-induced AAA formation has not been fully elucidated yet. Previous studies hint that the calcium ion may directly bind to the major arterial structural components, elastin and collagen, facilitating extracellular matrix degradation and decreased vessel wall stability14. CaPO4 crystals have also been identified to trigger significant NOD-like receptor protein 3 (NLRP3) inflammasome activation and smooth muscle cell apoptosis4,15. Besides, microcalcification crystals are capable of inducing mononuclear cells into osteoclast-like cells and promoting matrix metalloproteinase (MMP) production and may, ultimately, result in aortic expansion16.
When performing the CaPO4 model, several issues must be avoided to improve the success rate. One should avoid tearing the dorsal branch blood vessels, avoid the addition of excess CaCl2 solution into the abdominal cavity, and avoid an overdose of anesthesia. Mice with severe bleeding during surgery or with postoperative infection should be excluded from the experiment. As previously reported, when the number of the observations is sufficient, the maximal diameters of the aortas in the CaPO4 model are usually normally distributed, which is different from the AngII model7. Therefore, we recommend a comparison of the maximal diameters of aortas instead of AAA incidence when using the CaPO4 model.
Overall, the CaPO4-induced mice AAA model is a rapid and cost-effective approach to explore the molecular mechanisms and therapeutic strategies of AAA and could be applied in parallel with the other models to fully imitate the features of human AAA.
Acknowledgements
This research was supported by funding from the National Natural Science Foundation of China (NSFC, 81730010, 91839302, 81921001, 31930056, and 91529203) and the National Key R&D Program of China (2019YFA 0801600).
Materials
| CaCl2 | MECKLIN | C805225 | |
| NaCl | Biomed | SH5001-01 | |
| PBS | HARVEYBIO | MB5051 | |
| Small animal ventilator | RWD | H1550501-012 |
Referencias
- Kent, K. C. Abdominal aortic aneurysms. The New England Journal of Medicine. 371, 2101-2108 (2014).
- Patelis, N., et al. Animal models in the research of abdominal aortic aneurysms development. Physiological Research. 66 (6), 899-915 (2017).
- Gertz, S. D., Kurgan, A., Eisenberg, D. Aneurysm of the rabbit common carotid artery induced by periarterial application of calcium-chloride in vivo. Journal of Clinical Investigation. 81 (3), 649-656 (1988).
- Yamanouchi, D., et al. Accelerated aneurysmal dilation associated with apoptosis and inflammation in a newly developed calcium phosphate rodent abdominal aortic aneurysm model. Journal of Vascular Surgery. 56 (2), 455-461 (2012).
- Wang, Y. T., et al. Influence of apolipoprotein E, age and aortic site on calcium phosphate induced abdominal aortic aneurysm in mice. Atherosclerosis. 235 (1), 204-212 (2014).
- Zhao, G., et al. Unspliced xbp1 confers VSMC homeostasis and prevents aortic aneurysm formation via foxo4 interaction. Circulation Research. 121 (12), 1331-1345 (2017).
- Jia, Y., et al. Targeting macrophage TFEB-14-3-3 epsilon interface by naringenin inhibits abdominal aortic aneurysm. Cell Discovery. 8 (1), 21 (2022).
- Gage, G. J., Kipke, D. R., Shain, W. Whole animal perfusion fixation for rodents. Journal of Visualized Experiments. (65), e3564 (2012).
- Yu, B., et al. CYLD deubiquitinates nicotinamide adenine dinucleotide phosphate oxidase 4 contributing to adventitial remodeling. Arteriosclerosis, Thrombosis, and Vascular Biology. 37 (8), 1698-1709 (2017).
- Altobelli, E., Rapacchietta, L., Profeta, V. F., Fagnano, R. Risk factors for abdominal aortic aneurysm in population-based studies: A systematic review and meta-analysis. International Journal of Environmental Research and Public Health. 15 (12), 2805 (2018).
- Theivacumar, N. S., Stephenson, M. A., Mistry, H., Valenti, D. Diabetes mellitus and aortic aneurysm rupture: A favorable association. Vascular and Endovascular Surgery. 48 (1), 45-50 (2014).
- Tanaka, T., Takei, Y., Yamanouchi, D. Hyperglycemia suppresses calcium phosphate-induced aneurysm formation through inhibition of macrophage activation. Journal of the American Heart Association. 5 (3), 003062 (2016).
- Lu, H., et al. Subcutaneous angiotensin II infusion using osmotic pumps induces aortic aneurysms in mice. Journal of Visualized Experiments. (103), e53191 (2015).
- Urry, D. W. Neutral sites for calcium ion binding to elastin and collagen: A charge neutralization theory for calcification and its relationship to atherosclerosis. Proceedings of the National Academy of Sciences of the United States of America. 68 (4), 810-814 (1971).
- Li, Z. Q., et al. Runx2 (runt-related transcription factor 2)-mediated microcalcification is a novel pathological characteristic and potential mediator of abdominal aortic aneurysm. Arteriosclerosis, Thrombosis, and Vascular Biology. 40 (5), 1352-1369 (2020).
- Kelly, M. J., Igari, K., Yamanouchi, D. Osteoclast-like cells in aneurysmal disease exhibit an enhanced proteolytic phenotype. International Journal of Molecular Sciences. 20 (19), 4689 (2019).

