Reconstituting Cytoarchitecture and Function of Human Epithelial Tissues on an Open-Top Organ-Chip
Summary
The present protocol describes the capabilities and the essential culture modalities of the Open-Top Organ-Chip for the successful establishment and maturation of full-thickness organ-on-chip cultures of primary tissues (skin, alveolus, airway, and intestine), providing the opportunity to investigate different functional aspects of the human epithelial/mesenchymal and vascular niche interface in vitro.
Abstract
Nearly all human organs are lined with epithelial tissues, comprising one or multiple layers of tightly connected cells organized into three-dimensional (3D) structures. One of the main functions of epithelia is the formation of barriers that protect the underlining tissues against physical and chemical insults and infectious agents. In addition, epithelia mediate the transport of nutrients, hormones, and other signaling molecules, often creating biochemical gradients that guide cell positioning and compartmentalization within the organ. Owing to their central role in determining organ-structure and function, epithelia are important therapeutic targets for many human diseases that are not always captured by animal models. Besides the obvious species-to-species differences, conducting research studies on barrier function and transport properties of epithelia in animals is further compounded by the difficulty of accessing these tissues in a living system. While two-dimensional (2D) human cell cultures are useful for answering basic scientific questions, they often yield poor in vivo predictions. To overcome these limitations, in the last decade, a plethora of micro-engineered biomimetic platforms, known as organs-on-a-chip, have emerged as a promising alternative to traditional in vitro and animal testing. Here, we describe an Open-Top Organ-Chip (or Open-Top Chip), a platform designed for modeling organ-specific epithelial tissues, including skin, lungs, and the intestines. This chip offers new opportunities for reconstituting the multicellular architecture and function of epithelial tissues, including the capability to recreate a 3D stromal component by incorporating tissue-specific fibroblasts and endothelial cells within a mechanically active system. This Open-Top Chip provides an unprecedented tool for studying epithelial/mesenchymal and vascular interactions at multiple scales of resolution, from single cells to multi-layer tissue constructs, thus allowing molecular dissection of the intercellular crosstalk of epithelialized organs in health and disease.
Introduction
Historically, scientists have relied on preclinical animal testing for drug discovery, but a growing number of these methods have been questioned because of poor correlation with human outcome1. The implementation of the "3Rs" principles to Replace, Reduce, and Refine animal experimentation urges scientists to find new in vitro alternative methods to support preclinical drug and chemical toxicology risk assessment2. However, many in vitro models developed to date lack the biological architecture, cellular complexity, and mechanical environment necessary to recapitulate the dynamic nature of human living organs3,4.
Conventional in vitro preclinical systems typically employ 2D monocultures of human cells grown on a rigid plastic surface. These methods provide a tool for conducting simple mechanistic studies and enable rapid screening of drug candidates. Owing to their relatively low cost and high robustness, 2D models are often paired with automatic high-throughput systems and used for the rapid identification of potential drug candidates during the early stage of the drug development process5,6. However, such 2D models do not provide a translational approach for modeling tissue-level, organ-level, or systemic responses to therapeutic candidates, which is needed for accurate predictions of drug safety and efficacy during the preclinical stage of their development. Flat cell cultures do not recapitulate the native tissue microenvironment, including the complex multicellular interplay, biomechanical properties, and three-dimensional (3D) architecture of human tissues7. Cells growing on a flat surface often do not acquire a mature phenotype and, therefore, cannot respond to pharmacological stimuli as they would in the native tissue. For example, primary human alveolar epithelial cells grown in vitro exhibit a squamous phenotype and lose key phenotypic markers, including surfactant proteins C and B (SP-C and SP-B)8. In addition to insufficient differentiation, primary cells frequently become insensitive to biological stressors in vitro, as certain biochemical pathways associated with tissue inflammation become non-functional9. Such loss of cell function seems to be primarily associated with the use of stiff substrates as well as the lack of soluble factors naturally released by tissue-specific stromal cells such as lung fibroblasts and smooth muscle cells10,11.
Understanding that the lack of chemo-physical and biological complexity limits the physiological behavior of cells in vitro has fostered the development of more sophisticated multicellular models, which have proven to better capture the complexity of human tissues outside the body12,13. Since the creation of the first co-culture models in the early 1970s14, the introduction of synthetic and natural hydrogels has significantly improved the ability to mimic native tissue microenvironments and has become an invaluable tool for driving cellular differentiation, guiding the self-organization of cells into tissue-like structures, and restoration of native tissue functions15,16. For instance, when grown in the appropriate 3D scaffold, human cells can self-arrange into functional structures such as spheroids or organoids, expressing stem cell markers, and are capable of self-renewal17. In contrast, human cells (including stem cells), when grown on traditional 2D substrates, rapidly age and undergo senescence after a few passages18. In addition, hydrogels can be "tailored" to match specific tissue properties such as porosity, pore size, fiber thickness, viscoelasticity, topography, and stiffness or further engineered with tissue-derived cellular components and/or bioactive molecules enabling emulation of the physiological or pathological conditions19,20. Despite their enormous potential for drug testing, 3D hydrogel-based models used in pharmaceutical research do not fully recapitulate the complex cytoarchitecture of the in vivo tissues and lack important hemodynamic and mechanical stimuli normally present in the human body, including hydrostatic pressure, cyclic stretch, and fluid shear21.
Microphysiological systems (MPSs) such as Organs-on-chips (OOCs) have recently emerged as tools that are capable of capturing complex physiological responses in vitro22,23. These models often employ the use of microfluidic platforms, which enable the modelling of the dynamic microenvironment of living organs.
We have combined the principles of 3D tissue bioengineering and mechanobiology to create an Open-Top Chip model of complex human epithelial tissue. This allowed us to closely recapitulate the multicellular and dynamic microenvironment of epithelial tissues. This includes tissue-specific biochemical and biomechanical cues naturally present in living organs but often neglected by traditional in vitro models24. The Open-Top Chip incorporates two compartments: a vascular compartment (Figure 1A) and a stromal compartment (Figure 1B) separated by a porous membrane, allowing for the diffusion of nutrients between the two chambers (Figure 1C). The vascular compartment is exposed to continuous fluid flow to recapitulate physiological shear stress, while the stretchable design of the stromal chamber allows for the modeling of the mechanical strain associated with breathing motions or intestinal peristalsis. The stromal compartment houses the tunable 3D hydrogel scaffold designed to support the physiological growth of tissue-specific fibroblasts. It possesses a removable lid that facilitates the establishment of an air-liquid interface, a condition that allows greater emulation of human physiology of mucosal tissues as well as direct access to the tissue for administrating drugs directly onto the epithelial layer. Supplementary Figure 1 captures some of the key components of the Open-Top Chip design including dimensions and biological compartments (Supplementary Figure 1A–D) as well as the main technical steps described in this protocol (Supplementary Figure 1E).
Perfusion of the Open-Top Chip is achieved with a programmable peristaltic pump (Figure 1D). The peristaltic pump setup allows 12 Open-Top Chips to be perfused simultaneously. Most incubators can house two setups enabling the culture of up to 24 chips per incubator. Mechanical stretching is achieved using a custom-made programmable vacuum pressure regulator (Figure 1E). It consists of an electro-pneumatic vacuum regulator controlled electronically by a digital-to-analog converter. In other words, the electro-pneumatic vacuum regulator generates a sinusoidal vacuum profile with an amplitude and frequency that is determined by the user. Cyclic strain ranging from 0% to 15% is generated by applying negative pressure to the vacuum channel of the Open-Top Chip at an amplitude ranging from 0 to -90 kPa and a frequency of 0.2 Hz. It is a custom-made system equivalent to the commercially available Flexcell Strain Unit previously adopted and described in other papers25. To mimic the mechanical tissue deformation associated, for example, with the breathing motion of the lung or the peristalsis of the intestine, the pneumatic actuator applies sinusoidal vacuum/strain waves whose magnitude and amplitude can be adjusted to match the physiological level of strain and frequency that human cells experience in their native tissue.
Here, we describe an efficient and reproducible method for engineering and culturing organotypic epithelium equivalents on a prototype Open-Top Chip platform. It allows the generation of complex organ models such as skin, alveolus, airway, and colon while integrating a vascular fluid flow and mechanical stretching. We will outline key technical aspects that must be considered while implementing principles of tissue engineering for generating complex epithelial models. We will discuss the advantages and possible limitations of the current design.
An overview of the main steps used to achieve tissue and organ maturation, including flow and stretch parameters, is reported in: Figure 2 for the skin, Figure 3 for the alveolus, Figure 4 for the airway, and Figure 5 for the intestine. Additional information concerning media composition and reagents used for culturing the different organ models are included in the supplementary tables (Supplementary Table 1 for the skin; Supplementary Table 2 for the alveolus; Supplementary Table 3 for the airway, and Supplementary Table 4 for the intestine).
Protocol
Human colonoids were obtained from intestinal resections in accordance with the guidelines of the Institutional Biosafety Committee of the Cincinnati Children's hospital (IBC 2017-2011).
1. Surface activation
- Preparation of activation buffer
- Place the crosslinker and solvent buffer reagents under the biosafety cabinet (BSC) and let them equilibrate at room temperature (RT) for 10 min before use.
- Reconstitute 5 mg of crosslinker in 5 mL of the solvent buffer using a sterile light-impervious container or a transparent 15 mL conical tube wrapped in aluminum foil to protect the crosslinker solution from direct light exposure.
- Vortex the solution for 1 min to remove all clumps, and then pipette 50 µL of the crosslinker solution directly into the bottom channel of the chip and 150 µL into the open-top chamber.
- Remove any excess of crosslinking solution from the surface of the chip using an aspirator. Then pipette additional 50 µL of the crosslinking solution directly into the bottom channel of the chip and 150 µL into the open-top chamber to remove any remaining air bubble.
- Activation with UV crosslinking machine
- Gently remove the lid from the chip under the BSC and store it in a sterile container.
- Transfer the chips containing the crosslinker solution into a Petri dish, close the Petri dish to avoid contamination, and place the dish with the chips under the UV crosslinking machine.
NOTE: Remove the Petri dish lid to maximize the UV exposure. - Set the UV crosslinking machine with a peak wavelength of 365 nm at an intensity of 100 µJ/cm2, and turn on the UV light for 20 min.
NOTE: After 20 min of UV treatment, the crosslinker solution will look darker (brown). - Bring the chips back under the BSC and aspirate the oxidized crosslinker solution. Then, rinse all the chips three times with the solvent buffer and let the chips dry under the BSC for 5-10 min to complete the chemical functionalization of the polydimethylsiloxane (PDMS) surface.
2. Preparation the stroma equivalent
- Preparation of 10x reconstruction buffer (100 mL)
- Dissolve 2.2 g of sodium bicarbonate in 75 mL of 0.067 M NaOH in double-distilled water.
- Add 4.76 g of HEPES and bring the volume to 100 mL using double distilled water.
- Sterile filter the solution under the BSC using a disposable sterile bottle-top filter with a 0.22 µm membrane.
NOTE: The solution is stable for about 6 months when stored at 4 °C.
- Estimation of the pre-gel solution volume
- Multiply the number of chips needed for the experiment by 150 µL (inner volume of the central open-top chamber) to estimate the amount of pre-gel solution required for the experiment:
Volume needed = (Number of chips x 150) µL - Prepare the collagen pre-gel solution on ice by mixing: 1 volume of 10x EMEM containing the cells of choice, 1 volume of 10x reconstruction buffer (see step 2.1), 8 volumes of collagen I solution (10 mg/mL) and 1 µL of 1 N NaOH solution for each mg of collagen I.
NOTE: It is recommended to prepare an extra +15% volume to account for experimental errors; the example described in section 2.2 provides a detailed step-by-step procedure for preparing enough pre-gel solution for 12 chips and an extra 15% volume.
- Multiply the number of chips needed for the experiment by 150 µL (inner volume of the central open-top chamber) to estimate the amount of pre-gel solution required for the experiment:
- Preparation of pre-gel solution for the stroma equivalent (for 12 chips)
- Bring the following solutions under the BSC on ice: 10x EMEM; 10x Reconstruction buffer (see step 2.1); Collagen I solution (10 mg/mL); and Sterile 1 N NaOH solution.
- Culture tissue-specific mesenchymal cells as directed by the providers until 80%-90% confluent, and then dissociate the cells using trypsin or other methods as recommended by the cell provider. Collect the cells in a pellet by centrifugation at 250 x g for 5 min at 24 ˚C.
- Resuspend the cell pellet in 225 µL of ice-cold 10x EMEM, and add 225 µL of ice-cold 10x reconstruction buffer (see step 2.1). Mix the solution by gently pipetting up and down, and then add 1,800 µL of ice-cold collagen I solution.
- Pipette up and down 5-6 times, avoiding bubbles to mix the pre-gel solution while on ice.
- Incorporation of the stroma equivalent on chip (for 12 chips)
- Neutralize the pre-gel solution with 18 µL of 1 N NaOH. Mix gently by pipetting up and down 5-6 times, and then pipette 150 µL of cell-laden hydrogel into the central chamber of the Open-Top Chip avoiding bubbles.
NOTE: If micropatterning is required, please move to the next step (section 3). - Group the chips into separate Petri dishes, including a centrifuge tube cap filled with 2 mL of sterile ddH2O in each Petri dish, and incubate the Petri dishes(s) in the incubator at 37 °C, 5% CO2.
NOTE: After 90 min, the cell-laden hydrogel will be completely polymerized.
- Neutralize the pre-gel solution with 18 µL of 1 N NaOH. Mix gently by pipetting up and down 5-6 times, and then pipette 150 µL of cell-laden hydrogel into the central chamber of the Open-Top Chip avoiding bubbles.
3. Surface micropatterning (optional)
- Perform surface micropatterning of the stromal hydrogel after pipetting the neutralized collagen hydrogel (still in its liquid state) using 3D-printed stamps.
NOTE: The 3D-printed stamps can be obtained in various customizable designs, as previously described elsewhere24. - Pipette 20 µL of the neutralized collagen I pre-gel solution on the patterned surface of a sterile 3D-printed stamp and insert the stamp inside (on top) of the open-top chamber while the stromal hydrogel is still in a liquid form.
- Remove any residue of the hydrogel that may spill from the top of the open-top chamber using an aspirator (or a pipette). Group all chips into separate Petri dishes and include a centrifuge 15 mL conical tube cap filled with 2 mL of sterile ddH2O in each Petri dish.
- Incubate all Petri dishes(s) at 37 °C, 5% CO2 for 90 min, and then bring the chips back under the BSC and gently remove the stamps using precision tweezers to reduce the risk of damaging the hydrogel.
4. Coating the epithelial and vascular surface with tissue-specific ECM proteins
- Coating the vascular microfluidic chamber with extracellular matrix proteins
- Multiply the number of chips needed for the experiment by 20 µL (volume of the vascular channel) to estimate the volume of vascular ECM coating solution required for the experiment:
Volume needed = (Number of chips x 20) µL
NOTE: It is recommended to prepare an extra 15% volume to account for experimental errors. - Prepare the vascular ECM coating solution for all chips (for example, 300 µL per 12 chips) using ice-cold PBS or HBSS.
NOTE: Refer to the specific organ-protocol table in the supplementary materials section (Supplementary Table 1 for the skin; Supplementary Table 2 for the alveolus; Supplementary Table 2 for the airway, and Supplementary Table 4 for the intestine) to identify the specific reagents and recommended ECM composition. - Pipette 20 µL of vascular ECM coating solution into the vascular channel of each chip.
- Multiply the number of chips needed for the experiment by 20 µL (volume of the vascular channel) to estimate the volume of vascular ECM coating solution required for the experiment:
- Coating the apical surface of the stromal equivalent with extracellular matrix proteins
- Multiply the number of chips needed for the experiment by 50 µL (volume of the vascular channel) to estimate the volume of epithelial ECM coating solution required for the experiment:
Volume needed = (Number of chips x 50) µL
NOTE: it is recommended to prepare an extra 15% volume to account for experimental errors. - Prepare enough ECM coating solution for all chips (for example, 750 µL per 12 chips) in ice-cold PBS or HBSS and transfer 50 µL of epithelial ECM coating solution directly on top of the hydrogel surface.
NOTE: Refer to the specific organ-protocol table in the supplementary materials section (Supplementary Table 1 for the skin; Supplementary Table 2 for the alveolus; Supplementary Table 2 for the airway, and Supplementary Table 4 for the intestine) to identify the specific reagents and recommended ECM composition. - Group the chips into separate Petri dishes, including a centrifuge 15 mL conical tube cap filled with 2 mL of sterile ddH2O in each Petri dish, and incubate the Petri dishes(s) in the incubator at 37 °C, 5% CO2 for 2 h before proceeding with epithelial cell seeding.
- Multiply the number of chips needed for the experiment by 50 µL (volume of the vascular channel) to estimate the volume of epithelial ECM coating solution required for the experiment:
5. Seeding epithelial cells on the stromal equivalent
- Epithelial cell culture
- Culture tissue-specific epithelial cells as directed by the providers until 80%-90% confluent.
- Dissociate the cells using proteolytic enzyme procedures as recommended by the cell provider.
NOTE: For best results, harvest epithelial cells at low passage (P1-P2) during the active growth phase when they reach between 70%-90% confluency. - Once dissociated, centrifuge the cells and collect them as a pellet.
- Resuspend the epithelial cells to the appropriate cell/fragment density as indicated in the specific organ protocol table.
NOTE: In this study, cell solution was used at a density of 3 x 106 cells/mL for the skin, 1 x 106 cells/mL for the alveolus, 6 x 106 cells/mL for the airway and 8 x 106 fragments/mL for the intestine.
- Epithelial cell seeding
- Transfer the chips from the incubator into the BSC. Aspirate the coating solution from the vascular channel, and rinse the vascular microfluidic channel three times with 50 µL of fresh endothelial cell culture medium.
- Aspirate the coating solution from the hydrogel surface and rinse the stromal surface three times with 100 µL of fresh epithelial cell culture medium to remove any excess coating solution.
- Aspirate the rising medium and seed the hydrogel surface with 50 µL of the epithelial cell suspension using appropriate cell density, as indicated in the supplementary tables, and then transfer the chips back into the incubator for 2 h (or overnight for colonoids).
- Gently rinse the hydrogel surface with the cell culture medium twice to remove cellular debris. Finally, refresh the medium by autoclaving in sealed autoclavable containers, and connect the chips to the peristaltic pump.
6. Connecting chips to flow
- Preparing the fluidic parts
- Cut 2 inches of biocompatible polypropylene-based thermoplastic elastomer (TPE) transfer tubing (Table of Materials) to prepare the short microfluidic tubing that is required for connecting the chips to the media reservoirs.
- Cut 7.5 inches of biocompatible TPE transfer tubing to produce the long microfluidic tubing.
- Prepare enough 18 G and 19 G metal connectors (Table of Materials).
NOTE: It is recommended to prepare and sterilize the tubing and connectors described in steps 6.1.1 and 6.1.2 at least 1 day before connecting the Open-Top Chips to the peristaltic pumps. - Pierce the lid of each medium reservoirs with a 4-inch hypodermic needle (Table of Materials).
- Medium degassing
- Allow the cell culture medium to equilibrate to room temperature (RT).
- Transfer the volume of medium needed into a conical filtering tube.
- Apply a negative vacuum pressure of -20 PSI to degas the medium (vacuum-driven filtration).
NOTE: If a vacuum is not available, the cell culture medium can be left to equilibrate in the incubator overnight to achieve similar results.
- Prepare the Open-Top Chip for fluid flow
- Bring the chips and the sterile fluidic parts under the BSC and align the lid (top portion) of the Open-Top Chip for sealing the Open-Top Chip prototype before starting fluid flow.
- Pipette 200 µL of the degassed cell culture medium (see organ-specific tables) into the inlet port of both the top and bottom channels of the chip while paying attention to avoid bubbles.
- Pipette 300 µL of the culture medium into the short microfluidic tubing to prime the inner surface of the tubes and connect the short tubing to the inlets of the top and bottom channels of the chip.
- Connect and prime the microfluidic surfaces
- Position the medium reservoirs into the farm rack, connect the hypodermic needle to the bottom inlet of the chips, and finally, accommodate all the chips in the housing carrier(s) inside the incubator.
- Connect the chips to the peristaltic pump, and then inspect all the connectors to make sure that all the chips are properly connected and that there is no visible leakage of cell culture media.
- Use the Purge button on the pump and hold it for about 15 s or until the droplets of the cell culture medium appear at the end of the outflow tubing.
- Use the control system on the pump to set the appropriate organ-chip flow rate (Figure 2, Figure 3, Figure 4, and Figure 5).
7. Maintenance of chips
- Organ-chip maintenance
- Prepare fresh cell culture medium for the epithelium and/or endothelium and perform the degassing steps (as previously described in step 6.2).
- Pause the peristaltic pump, carefully disconnect the chips from the pump, and extract the chip housing carrier(s). Transfer the chips from the incubator to the BSC and remove the volume of media left into the reservoirs.
- Replace the cell culture media to the top and bottom inlet reservoirs with 5 mL of fresh cell culture media and place the chip housing carrier(s) back into the incubator. Connect the chips to the peristaltic pump and restart the flow.
NOTE: It is recommended to use the Purge function to rapidly refresh the medium to the vascular compartment and reduce the risk of air bubbles. - Repeat steps 7.1.1-7.1.3 every other day, as per Figure 2, Figure 3, Figure 4, and Figure 5.
- Establishing air-liquid interface (ALI)
- Pause the peristaltic pump, carefully disconnect the chips from the pump, and extract the chip housing carrier(s). Transfer the chips from the incubator to the biosafety cabinet, and remove the volume of medium left into the top reservoir.
- Gently aspirate all the medium from the top microfluidic channel and clamp the short microfluidic tubing connected to the top inlets using binder clips to reduce media evaporation and maintenance of ALI.
- Place the Open-Top Chip on the housing carrier(s) back into the incubator and reconnect the chips to the peristaltic pump.
NOTE: It is recommended to use the Purge function to rapidly refresh the medium to the vascular compartment and reduce the risk of air bubbles. - Resume the flow by starting the peristaltic pump.
- Stretching (Optional)
- Pause the peristaltic pump and connect the vacuum ports of the chips to the vacuum module using two long microfluidic tubes per chip.
- Use the vacuum module to adjust the stretch setting to the condition recommended for each organ as specified inside the organ protocol tables: Supplementary Table 1 for the skin; Supplementary Table 2 for the alveolus; Supplementary Table 2 for the airway; and Supplementary Table 4 for the intestine.
- Visually inspect the tube connections to make sure all the chips are properly connected and there are no visible droplets of medium dripping.
- Resume the flow by starting the peristaltic pump.
8. Seeding endothelial cells in the vascular compartment
- Prepare cells and chips for vascular cell seeding
- Culture the tissue-specific endothelial cells as directed by the providers until 80%-90% confluent. Dissociate the cells using a proteolytic enzyme procedure (as recommended by the provider) and, finally, resuspend the endothelial cells in a solution of 3 x 106 cells/mL.
NOTE: For best results, harvest the endothelial cells at low passage (P2-P4) during the active growth phase when they reach between 70%-90% confluency. - Pause the peristaltic pump. Transfer the chips from the incubator to the BSC, disconnect the chips from the media reservoirs and any connected tubing, and then group the chips into separate Petri dishes.
- Refresh the cell culture medium of the epithelial compartment with fresh epithelial cell culture medium. Rinse the vascular channel with fresh endothelium cell culture medium twice, and then aspirate the medium from the vascular compartment.
- Culture the tissue-specific endothelial cells as directed by the providers until 80%-90% confluent. Dissociate the cells using a proteolytic enzyme procedure (as recommended by the provider) and, finally, resuspend the endothelial cells in a solution of 3 x 106 cells/mL.
- Endothelial cell seeding
- Seed the bottom (vascular) channel with 25 µL of endothelial cell suspension (3 x 106 cells/mL), flip the chips upside down to allow endothelial cells to attach to the upper surface of the microfluidic chamber.
NOTE: Add 50 µL of the cell suspension per chip (600 µL per 12 chips). - Group the chips into Petri dishes. Place them back into the incubator at 37 °C, 5% CO2, and let the endothelial cells attach for 1 h.
- After 1 h, transfer the chips from the incubator to the BSC. Rinse the vascular channel with endothelial cell culture medium twice to remove cellular debris.
- Repeat steps 8.2.1-8.2.2 to seed the vascular channel once again with endothelial cells and lay the chips flat to facilitate the adhesion of the endothelial cells to the bottom surface of the vascular channel.
- Seed the bottom (vascular) channel with 25 µL of endothelial cell suspension (3 x 106 cells/mL), flip the chips upside down to allow endothelial cells to attach to the upper surface of the microfluidic chamber.
- Reconnect the chip to flow
- Fill up the vascular medium reservoir with the degassed vascular cell culture medium under the BSC.
- Place the chips back inside the chip housing carrier(s) and reconnect the chips to the medium reservoirs on one end to the peristaltic pump on the other end.
NOTE: It is recommended to use the Purge function to rapidly refresh the medium to the vascular compartment and reduce the risk of air bubbles. - Visually inspect the microfluidic connections to make sure all the chips are properly connected and there are no visible droplets of medium dripping. Then, resume fluid flow by starting the peristaltic pump.
9. Common endpoint assays
- Disconnect chips for endpoint assays
- Pause the peristaltic pump, carefully disconnect the chips from the pump, and extract the chip housing carrier(s). Transfer the chip housing carrier(s) from the incubator to the BSC and set the chips free.
- Gently wash the central chamber of the Open-Top Chip with the epithelial cell culture medium and the vascular channel with the endothelium culture medium twice to remove any cellular debris.
- Remove the lid of the Open-Top Chip to access the apical compartment of the chip using tweezers.
- Immunostaining for fluorescence microscopy
- Proceed with conventional sample fixation, permeabilization, and blocking.
NOTE: In this study, the samples were fixed in 200 µL of 4% paraformaldehyde (PFA) for 1 h followed by rinsing with PBS, permeabilization in 0.1% Triton X-100 for 40 min and blocking in 1% bovine serum albumin for 1 h. - Incubate the chips with primary antibodies (Table of Materials) at 4 °C overnight. and then wash both epithelial and endothelial compartments of the chips with 200 µL of PBS twice. Then, proceed with the appropriate secondary antibodies for 2 h.
NOTE: Dilute the primary antibodies and secondary antibodies in PBS + 1% BSA at a dilution of 1:100 (primary antibody) and 1:200 (secondary antibody).
- Proceed with conventional sample fixation, permeabilization, and blocking.
- Immunohistochemistry
- Extract the stromal equivalents from the main chamber of the Open-Top Chip using tweezers, collect them into a 1.5 mL tube filled with 10% neutral buffered formalin and incubate for at least 24 h.
- Transfer the fixed stromal equivalent to the tissue processor and follow the below steps to achieve optimal sample dehydration.
- Submerge the hydrogel in 70% ethanol for 90 min.
- Remove the 70% ethanol and submerge the hydrogel in 80% ethanol for 90 min.
- Remove the 80% ethanol and submerge the hydrogel in 95% ethanol for 90 min.
- Remove the 95% ethanol and submerge the hydrogel in 100% ethanol for 90 min twice.
- Remove the 100% ethanol and submerge the hydrogel in a solution of xylene for 120 min twice.
- Remove the xylene solution and infiltrate the processed stromal equivalents in paraffin wax for 120 min (~2 h) twice.
- Embed the infiltrated stromal equivalents into sectioning paraffin blocks.
- At this point, the stromal equivalents can be sectioned as paraffin blocks using a microtome and processed according to conventional histological techniques26.
Representative Results
Surface micropatterning
Micropatterning of the extracellular matrix (ECM) can be used to replicate the spatial configuration of the intestinal crypt interface. The Open-Top Chip configuration can be modified to integrate micropatterned stamps specifically designed to mimic the natural topography of the colonic epithelium-stroma interface (Figure 6A,B) and the intestinal crypts at micrometer scale (Figure 6C–E). Please note that we used a flat (not patterned) surface for the skin, airway, and alveolus models. The stamp was used in this case to obtain a uniform hydrogel surface to seed epithelial cells. We opted for a design that could mimic the natural architecture of the human intestinal mucosa, consisting of the alternance of positive and negative domes mimicking the intestinal crypts.
Organ-models
We cultured and differentiated four different epithelia (skin, alveolus, airway, and intestine) using the Open-Top Chip prototype to prove this biomimetic platform's versatility. Histological sections of the organ chips confirm the presence of epithelial cells that are phenotypically distinct and representative of: a stratified epithelium in the case of the skin (Figure 7), pseudo-stratified columnar epithelium in the case of the airway (Figure 8 and Supplementary Video 1), simple squamous epithelium in the case of the alveolus (Figure 9), and simple columnar epithelium in the case of the intestine (Figure 10). Skin, airway, and alveolar cells were all obtained from commercially available vendors (as specified in the Table of Materials).
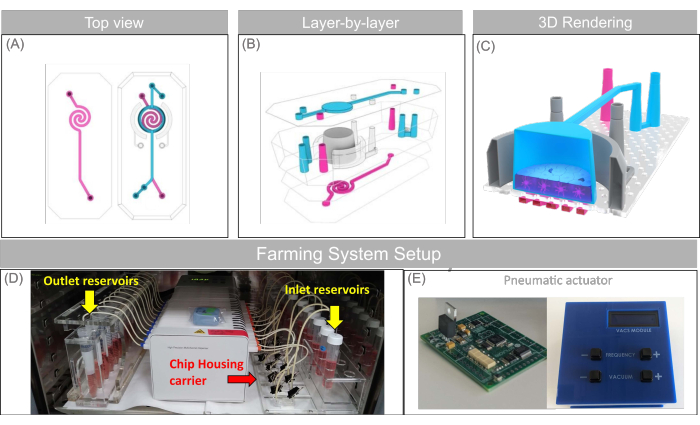
Figure 1: Schematic of the Open-Top Chip and the microfluidic setup used in this study. (A–C) Top-view, layer-by-layer projection, and 3D rendering showing the prototype Open-Top Chip design comprising the removable upper lid with encased micro-fluidic channel (blue), the two semi-lunar vacuum channels alongside the culture chamber (gray), and the bottom spiralized endothelial micro-fluidic channels (magenta). (D) Custom-made chip holder (also named "Farm system") including the chip housing carrier (red arrow), the peristaltic pump, and reservoirs (yellow arrow) arranged in a configuration that fits into a common cell-culture incubator. € Pneumatic actuator, the instrument that controls the negative pressure applied to the vacuum channel of the chips, which is used to generate the cyclic mechanical force cells experience during breathing or peristalsis motion (stretch). This figure has been adapted with permission from Varone et al.24. Please click here to view a larger version of this figure.
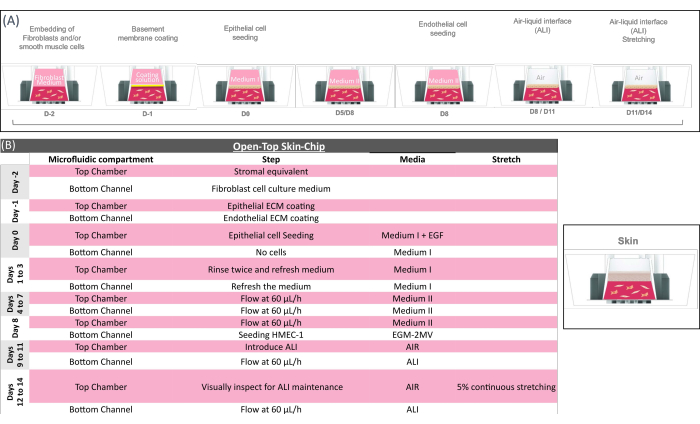
Figure 2: Technical overview of the open-top skin-chip protocol. (A) Schematic showing the sequence of actions for the open-top skin-chip preparation and (B) providing the key biological step of the open-top skin-chip culture. In the initial phase of the chip preparation, mesenchymal cells (fibroblasts) are embedded into the gel and loaded in the Open-Top Chip to form the stromal layer, which is coated for 2-4 h and seeded with epithelial cells. Once the epithelial cells have formed a compact monolayer, they are exposed to air (ALI). The biological system is kept under ALI regime until being sacrificed for analysis on day 14. Mechanical stretching can be applied while the system is under flow and at ALI. The stretching is kept until the tissues are sacrificed for analysis. Additional information on media composition, specific reagents, and cell types can be found in Supplementary Table 1. Please click here to view a larger version of this figure.
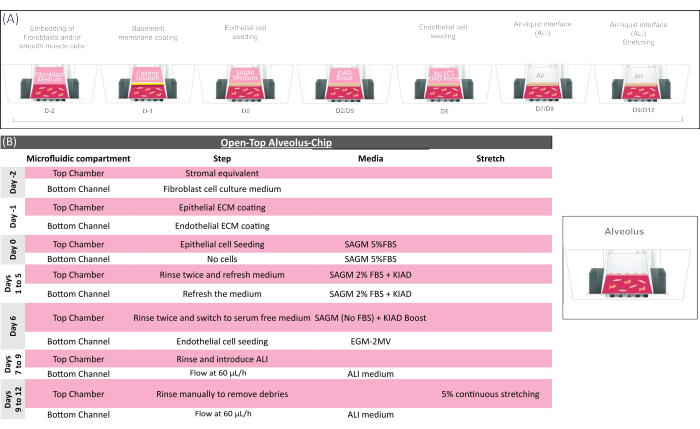
Figure 3: Technical overview of the open-top alveolus-chip protocol. (A) Schematic showing the sequence of actions for the open-top alveolus-chip preparation and (B) providing the key biological step of the open-top alveolus-chip culture. In the initial phase of the chip preparation, mesenchymal cells (fibroblasts) are embedded into the gel and loaded in the Open-Top Chip to form the stromal layer, which is coated for 2-4 h and seeded with airway epithelial cells in a medium supplemented with KIAD (see Supplementary Table 2). EGF-supplemented medium is maintained for ~4 days to support epithelial cell growth. The epithelium is then exposed to air (ALI) for ~10 days to achieve complete tissue maturation. Pulmonary microvascular endothelial cells are seeded on day 14, and the biological system is kept under ALI and flow regime until being sacrificed for analysis on day 21. Please click here to view a larger version of this figure.
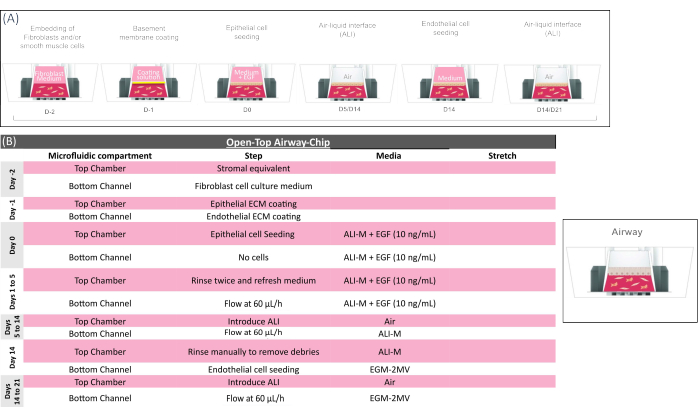
Figure 4: Technical overview of the open-top airway-chip protocol. (A) Schematic showing the sequence of actions for the open-top airway-chip preparation and (B) providing the key biological step of the open-top airway-chip culture. In the initial phase of the chip preparation, mesenchymal cells (fibroblasts and/or smooth muscle cells) are embedded into the gel and loaded in the Open-Top Chip to form the stromal layer, which is coated for 2-4 h and seeded with epithelial cells in medium supplemented with EGF (see Supplementary Table 3). EGF-supplemented medium is maintained for ~4 days to support epithelial cell growth. The epithelium is then exposed to air (ALI) for ~10 days to achieve complete tissue maturation. Pulmonary microvascular endothelial cells are seeded on day 14, and the biological system is kept under ALI and flow regime until being sacrificed for analysis on day 21. Please click here to view a larger version of this figure.
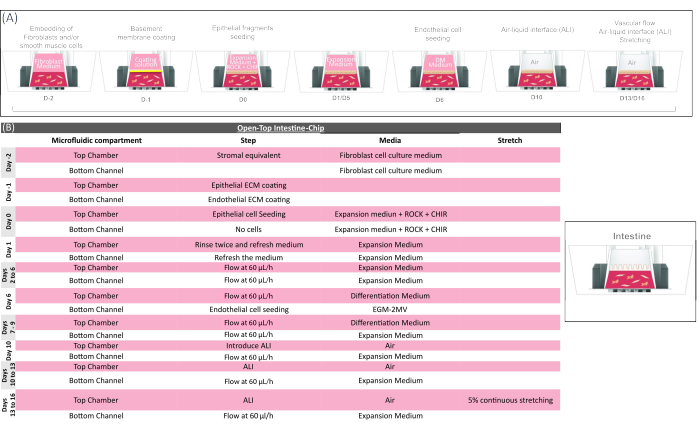
Figure 5: Technical overview of the open-top intestine-chip protocol. (A) Schematic showing the sequence of actions for the open-top intestine-chip preparation and (B) providing the key biological step of the open-top intestine-chip culture. In the initial phase of the chip preparation, mesenchymal cells (colonic fibroblasts) are embedded into the gel and loaded in the Open-Top Chip to form the stromal layer, which is coated for 2-4 h and seeded with fragments of epithelial colonoids obtained from clinical resections. Cell culture medium, including supplements (ROCK and CHIR, see Supplementary Table 4) is required during the seeding step to maintain colonoid cell viability and physiological morphology. Different media are then used to drive the expansion (day 1 to 6) and maturation (day 6 to 9) of the colonic epithelium. Colonic microvascular endothelial cells are seeded on day 6 using an endothelial cell culture medium (EGM2 MV), and then cultured under flow with an epithelial expansion medium for up to 10 more days. The epithelium is exposed to ALI from day 10 to further promote epithelial cell maturation. Mechanical stretching can be applied to the system from day 13 and maintained till day 16, when the organ-model is sacrificed for endpoint analysis. Please click here to view a larger version of this figure.

Figure 6: Micropattern stamping. (A) The lateral and top view of the stamp showing micro-scale texture (500 µm in height and 250 µm width pillar-array) is used to recreate the colonic crypt tissue interface and top and lateral view of the stamp-chip assembly, showing the fitting of the two elements when used to cast the gel surface. (B) Angled lateral view showing the Stamp and Chip interfacing. (C–E) Images of the micropatterned gel surface with and without cells. Scale bars: 200 µm. This figure has been adapted with permission from Varone et al.24. Please click here to view a larger version of this figure.
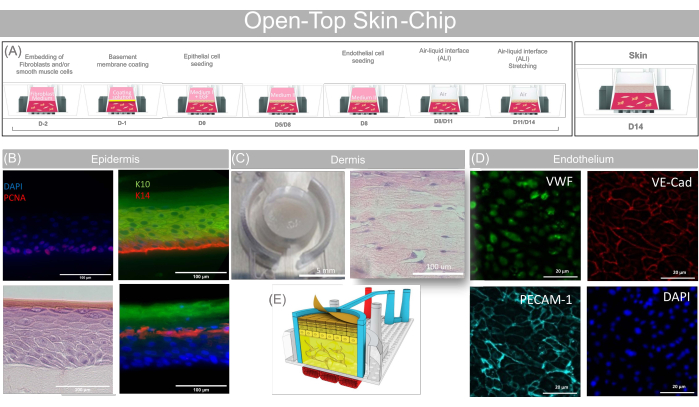
Figure 7: Representative data obtained with the open-top skin-chip. (A) Schematic showing the sequence of action for the open-top skin-chip preparation and providing the key biological steps of the open-top skin-chip culture. (B) PCNA Cytokeratin 14, Cytokeratin10, Involucrin and Fillagrin fluorescence staining and H&E showing mature multilayered stratified epidermis differentiated on-Chip. Scale bars: 100 µm. (C) Top view picture of the Skin-Chip (Scale bars: 5 mm) and H&E cross-section (Scale bars: 100 µm) showing the presence of the fibroblasts inside the dermal layer. (D) PECAM-1, VE-Cadherin and Von Willebrand Fluorescence staining showing differentiation of human microvascular endothelial cells co-cultured in the open-top skin-chip. Scale bars: 20 µm. (E) 3D Cartoon concept rendering of the open-top skin-chip. This figure has been adapted with permission from Varone et al.24. Please click here to view a larger version of this figure.
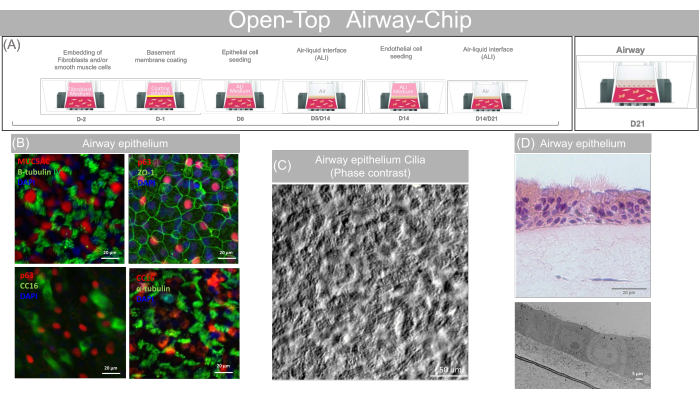
Figure 8: Representative data obtained with the open-top airway-chip. (A) Schematic showing the sequence of action for the open-top airway-chip preparation and providing the key biological step of the Open-Top Airway-Chip culture. (B) MUC5AC (Goblet), α and β-Tubulin (Ciliated cells), Clara Cell protein 16 (Club Cells), p63 (Basal/progenitor cells) and ZO-1 fluorescence staining showing mature airway epithelium. Scale bars: 20 µm. (C) Phase contrast video/image showing the presence of beating cilia. Scale bars: 50 µm. (D) H&E staining (Scale bars: 20 µm) and TEM image (Scale bars: 5 µm) showing mature pseudo-stratified epithelium differentiated on-Chip. Please click here to view a larger version of this figure.
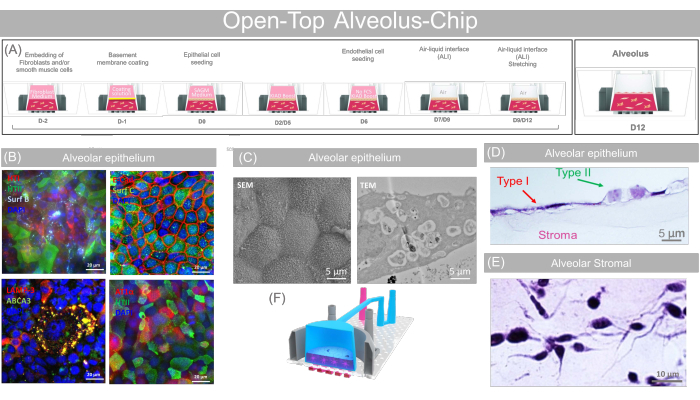
Figure 9: Representative data obtained with the open-top alveolus-chip. (A) Schematic showing the sequence of action for the open-top alveolus-chip preparation and providing the key biological step of the open-top alveolus-chip culture. (B) Type I (HTI-56, AT1-α), Type II (HTII-280, LAMP3, ABCA3, Surfactant (C) and E-Cadherin fluorescence staining showing the presence of mature pneumocytes on-Chip. Scale bars: 20 µm. (C) SEM and TEM image showing the presence of microvilli and lysosomal vesicles evidence of mature alveolar phenotype. Scale bars: 5 µm. (D) H&E cross-section (Scale bars: 5 µm) confirming the presence of flat, squamous cells consistent with Type I phenotype and cuboidal, cobblestone-like cells coherent with Type II phenotype, and (E) showing the presence of the fibroblasts inside the dermal layer (Scale bars: 10 µm). (F) 3D cartoon concept rendering of the open-top alveolus-chip. This figure has been adapted with permission from Varone et al.24. Please click here to view a larger version of this figure.
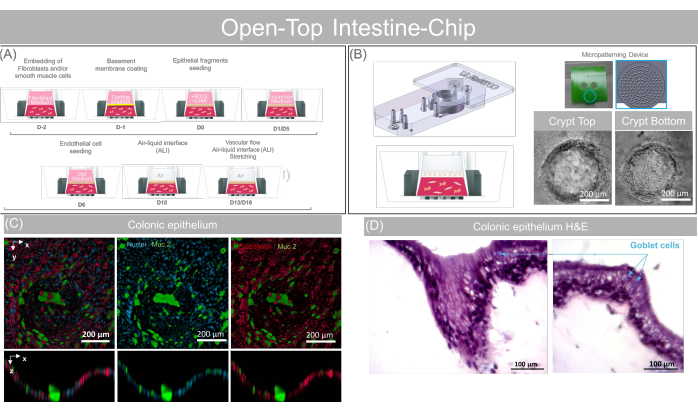
Figure 10: Representative data obtained with the open-top intestine-chip. (A) Schematic showing the sequence of action for the Open-Top Intestine-Chip preparation and providing the key biological step of the Open-Top Intestine-Chip culture. (B) Angled lateral view showing the Stamp and Chip assembly during the gel casting phase, the cartoon concept of the micropatterned gel and phase contrast images of a crypt-like structure micropatterned on the gel surface and seeded with colonoids at two different heights. Scale bars: 200 µm. (C) Mucin 2 and E-Cadherin fluorescence staining showing the presence of enterocytes and mature goblet cells on-Chip. Scale bars: 200 µm. (D) H&E cross-section of a crypt-like structure showing the presence of the fibroblasts inside the dermal layer and confirming the presence of a simple columnar epithelium. Scale bars: 100 µm. Please click here to view a larger version of this figure.
Supplementary Video 1: Phase contrast video showing beating cilia. Scale bars: 100 µm. Please click here to download this File.
Supplementary Figure 1: Open-top chip assembly. (A) Schematic showing the three-dimensional rendering of the Open-Top Chip assembly and the cartoon rendering of the open-top skin-chip with the different biological compartments highlighted and including epithelial (blue), dermal (yellow), and vascular (red). (B) The assembled microfluidic platform has a 35 mm x 17 mm format, a tissue culture area of 0.32 cm2, a bottom-spiraled microfluidic channel, and a chamber lid with a microfluidic channel. (C) The platform comprises a chamber with a 5-degree angled wall, which has a diameter of 6 mm at the level of the membrane and 5.7 mm at the top of the PDMS chamber wall and a height of 4 mm and width. The porous membrane is 50 µm thick and the pores are 7 µm in diameter. (D) The bottom spiral-shaped microfluidic channel has cross-section dimensions of 400 µm (height) x 600 µm (width). (E) An overview of the experiment timeline and the steps required for preparing the Open-Top Organ-Chip. Please click here to download this File.
Supplementary Table 1: Skin. The table provides a summary of the key daily steps and the stretching and flow parameters used during the three phases of the Open-Top Skin-Chip culture (growth, proliferation, and differentiation). The table also provides the list of materials, the medium formulations, and instructions on how to prepare the media necessary for this protocol. The composition of the three media is optimized for the different phases of the protocol. Specifically, Medium I is optimized for the seeding and early keratinocyte culture phase. Medium II is optimized for proliferation and early differentiation (formation of a stratified epithelium). The ALI medium is optimized for maintaining keratinocytes at an air-liquid interface until a fully differentiated epidermis is produced. Please click here to download this File.
Supplementary Table 2: Alveolus. The table provides a summary of the key daily steps and the stretching and flow parameters used during the three phases of the open-top alveolus-chip culture (growth, proliferation, and differentiation). The table also provides the list of materials, the medium formulations, and instructions on how to prepare the media necessary for this protocol. The composition of the two media is optimized for the different phases of the protocol. Please note that the addition of supplements (KIAD) to the cell culture medium is critical to achieve optimal differentiation of the pneumocytes. Please click here to download this File.
Supplementary Table 3: Airway. The table provides a summary of the key daily steps and the stretching and flow parameters used during the three phases of the open-top airway-chip culture (growth, proliferation, and differentiation). The table also provides the list of materials, the medium formulation, and instructions on how to prepare the medium necessary for this protocol. The composition of the medium is optimized for maintaining airway cells at the air-liquid interface, which, in turn, induces terminal differentiation and stimulates the production of mucus. Please click here to download this File.
Supplementary Table 4: Intestine. The table provides a summary of the key daily steps and the stretching and flow parameters used during the three phases of the open-top intestine-chip culture (growth, proliferation, and differentiation). The table also provides the list of materials, the medium formulations, and instructions on how to prepare the media necessary for this protocol. The compositions of both the media are optimized for the different phases of the protocol. Specifically, the expansion medium supplemented with the CHIR and ROCK inhibitors is optimized for the seeding and early phase of culture because it promotes the survival and growth of the colonic organoid fragment as a monolayer. The expansion medium is optimized for the proliferation and early differentiation of epithelium monolayer. The differentiation medium is optimized for terminal differentiation of the epithelium monolayer before exposure to the air-liquid interface. Please click here to download this File.
Discussion
The Open-Top Chip represents an enabling platform for investigating the complex cellular interplay occurring between endothelium, stroma, and epithelium in a controlled microenvironment, in real time. This technology offers critical advantages over conventional organotypic and organoid cultures, such as the integration of physical and biochemical cues that are relevant to reconstitute the human tissue microenvironment, including fluidic shear (flow), cyclic stretching, and reconstruction of the epithelial surface topography achieved via micropatterning. Human cells growing within this platform act in synergy to recapitulate tissue-specific functions that can be analyzed via conventional techniques, including immune histochemistry and biochemical assays using the outflow fluids (or effluents) from the top and/or bottom compartments. The current design allows easy access to the epithelial layer, where cells grow in direct contact with tissue-specific fibroblasts and other stromal cells, mimicking the multicellular architecture of epithelial tissues. Notably, the Open-Top Chip prototype offers a viable solution to common challenges associated with the use of other organ-on-chip platforms. While it enables the incorporation of several different cell types within a stromal compartment leading to the creation of very complex 3D tissues, it also allows for easy extraction of the established tissue constructs from the chip device for downstream analysis, including conventional H&E staining.
The current design presents a few limitations. For instance, the elastic membrane interposed between the vascular microchannel and the stromal compartment (hydrogel) of the prototype Open-Top Chip is considerably thicker (≈ 50 µm versus ≈ 1 µm) than the interstitial space separating endothelial tissues from the stroma in the native human organs. Although the elastic membrane does not represent a physical barrier to the diffusion of large molecules, such as hormones and other paracrine factors that mediate the intercellular crosstalk between tissues, it may limit the direct, cell-cell interactions and the migration of cells from the vascular to the stromal compartment. Finally, the prototype Open-Top Chip is made of PDMS, which is known to absorb a vast number of hydrophobic compounds. This limitation is shared by many PDMS-based platforms that can be a serious obstacle in applications intended for testing the pharmacodynamics and pharmacokinetics of small therapeutic compounds27.
One of the main challenges of integrating 3D hydrogels into a microfluidic device such as the Open-Top Chip, is that the PDMS does not provide an optimal substrate for the binding of human proteins or cells. Chemical functionalization of the PDMS surface is therefore one critical step in this protocol that is required to ensure proper ECM coating and hydrogel adhesion to the gel chamber of the Open-Top Chip model. To achieve optimal results, the crosslinking agent in the ER1 solution must always be protected from direct exposure to light. An important indicator of the reactive status of the crosslinker is its color. The crosslinker in the ER1, in fact, undergoes a color transition from brilliant orange to dark brown when it oxidizes. The color transition can be used as an indicator after the UV activation step to check whether the solution has effectively reacted with the PDMS surface. During the preparation of the crosslinker solution, the color transition should be monitored to ensure that an accidental exposure to direct light does not photo-bleach the chemical compound in the solution. To protect the crosslinker solution and avoid any unwanted photobleaching, we suggest wrapping aluminum foil, approximately 15 cm x 15 cm, around a 15 mL conical tube or using an amber tube available on the market. The use of the ER1 allows for a simple and effective method for achieving rapid functionalization of the PDMS surfaces; however, there are other molecules that can be used in place of ER1. For example, the chemical crosslinkers 3-aminopropyl-trimethoxysilane (APTMES) is not as photosensitive as the ER1 and it can be used to achieve similar results with a few additional steps as we have previously described elsewhere28,29. Independently of the molecule of choice, one of the main constraints in working with a chemical crosslinker is the presence of leftover residues, which can induce cytotoxicity. Following the activation reaction, it is important to rinse the microfluidic surfaces with abundant volumes of washing solution.
Because air bubbles tend to form and grow within the hydrophobic interfaces of the chips, tubing, and connectors, it is important to assess whether the microfluidic path is free from air bubbles before starting the fluid flow. Bubbles can indeed disrupt the flow and even kill the cells when chips are connected to flow. Priming of the microfluidic components before starting fluid flow will reduce the risk of generating air bubbles and help achieve optimal and reproducible results. If the priming cycle described in this protocol was not sufficient, rinsing the microfluidic tubes with ethanol (5 min), and then with HBSS (20 min) will further mitigate the risk of generating air bubbles inside the fluidic connectors.
Despite its limitations, PDMS possesses a rare combination of chemical properties that has enabled the fabrication of highly biocompatible, transparent, and elastic microfluidic devices. All these properties have made PDMS the most broadly applied material for the construction of the Organ-on-Chip models over the past decade. Recent advancements in the fields of material science and chemical engineering30,31 suggest that the PDMS component of this platform could be replaced in the near future with new synthetic polymers or biomaterials. If successfully achieved, it could enable recapitulation of tissue-specific properties, including porosity, ECM composition of the interstitial space providing closer mimicry to human native tissues, and more advanced models for pharmacology studies. We anticipate that the future evolution of the Open-Top Chip design will enable the modeling of human tissues and organs with an unprecedented level of detail.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
None
Materials
| 10x EMEM | Lonza | 12-684F | Medium; Stroma |
| 18 Gauge needle | MicroGroup | 316H18RW | Tube stainless steel 316 welded, 18RW Full Hard |
| 19 Gauge needle | MicroGroup | 316H19RW | Tube stainless steel 316 welded, 19RW Full Hard |
| 2-Stop PharMed BPT | Cole-Palmer | EW-95723-12 | Tube, 0.25 mm, 12/pack |
| 70% ethanol and wipes | - | - | For surface sterilization |
| 8-Bromoadenosine 3′,5′-cyclic monophosphate sodium salt (8-Br-cAMP) | Sigma | B7880 | Medium supplement |
| A-83-01 | Tocris | 2939 | |
| Adenine | Sigma | A9795 | |
| Advanced DMEM/F12 | Thermo | 12634010 | |
| Airway Epithelial Cells | Lifeline Cell Technology | FC-0016 | |
| Aluminum foil | - | - | - |
| Alveolar cells | Cell Biologics | H6621 | |
| Anti-ABCA3 | ABCAM | ab24751 | Mouse monoclonal antibody [3C9] |
| Anti-Aquaporin5 Alexa Fluor 647 | ABCAM | ab215225 | Rabbit monoclonal antibody [EPR3747] |
| Anti-Aquaporin5 | ABCAM | ab92320 | Rabbit monoclonal antibody [EPR3747] |
| Anti-beta IV Tubulin | ABCAM | ab11315 | Mouse monoclonal antibody [ONS.1A6] |
| Anti-CD31 (PECAM-1) | ABCAM | ab9498 | Mouse monoclonal [JC/70A] antibody |
| Anti-CK5 | ABCAM | ab75869 | Rabbit recombinant monoclonal [AY1E6] |
| Anti-Cytokeratin 10 | ThermoFisher | MA5-13705 | Mouse monoclonal antibody (DE-K10) |
| Anti-Cytokeratin 14 | ABCAM | ab7800 | Mouse monoclonal antibody |
| Anti-E-Cadherin | ABCAM | ab1416 | Mouse monoclonal antibody |
| Anti-Filaggrin | ThermoFisher | PA5-79267 | Rabbit polyclonal antibody |
| Anti-HTI-56 | Terrace Biotech | TB-29AHT1-56 | Mouse monoclonal antibody (IgG1) |
| Anti-HTII-280 | Terrace Biotech | TB-27AHT2-280 | Mouse monoclonal antibody (IgM) |
| Anti-Involucrin | ThermoFisher | MA5-11803 | Mouse monoclonal antibody (SY5) |
| Anti-Isoforms TA p63-α, -β, -γ | Biolengend | 618902 | Rabbit polyclonal antibody |
| Anti-Ki67 | ABCAM | ab8191 | Mouse monoclonal antibody [B126.1] |
| Anti-LAMP3 | ABCAM | ab111090 | Rabbit polyclonal antibody |
| Anti-Mature SP-B | Seven Hill | WRAB-48604 | Rabbit polyclonal antibody |
| Anti-MUC5AC | ThermoFisher | PA5-34612 | Rabbit polyclonal antibody |
| Anti-Mucin-2 | SantaCruz Biotechnology | sc-7314 | Mouse monoclonal antibody (IgG1) |
| Anti-p63 | Dako | GA662 | Mouse monoclonal antibody p63 Protein (Dako Omnis) Clone DAK-p63 |
| Anti-PCNA | ThermoFisher | PA5-32541 | Rabbit polyclonal antibody |
| Anti-Podoplanin (AT-1α) | ABCAM | ab128994 | Rabbit polyclonal antibody |
| Anti-Pro + Mature Surfactant Protein B | ABCAM | ab40876 | Rabbit polyclonal antibody |
| Anti-Surfactant C | Seven Hill | WRAB-9337 | Rabbit polyclonal antibody |
| Anti-Uteroglobin/SCGB1A1 | Hycult Biotech | HM2178 | Mouse monoclonal antibody [AY1E6] |
| Anti-VE-cadherin | ABCAM | ab33168 | Rabbit polyclonal antibody |
| Anti-ZO-1 | ThermoFisher | 33-9100 | Mouse monoclonal antibody [1A12] |
| Ascorbic acid | Sigma | A4544 | |
| Aspirating pipettes | Corning / Falcon | 357558 | 2 mL, polystyrene, individually wrapped |
| Aspirating tips | - | - | Sterile (autoclaved) |
| B27 | Thermo | 17504044 | |
| Blocker BSA (10X) in PBS solution | ThermoFisher | 37525 | Blocker agent |
| Calcium Chloride | Sigma | C7902 | |
| CHIR 99021 | Tocris | 4423 | |
| Collagen I | Advanced Biomatrix | 5133 | 10 mg/mL (Stroma) |
| Collagen I | Advanced BioMatrix | 5005 | 3 mg/mL (Vascular ECM) |
| Collagen IV | Sigma | C5533 | |
| Collagen-IV | Sigma | C5533-5MG | Collagen from human placenta, 5 mg powder, reconstitute to 1 mg/mL |
| Colonic Fibroblasts | Cell Biologics | H6231 | |
| Colonic microvascular endothelial cells | Cell Biologics | H6203 | |
| Conical tubes | - | - | 15 mL and 50 mL polypropylene, sterile |
| Crosslinker (ER-1) | Emulate | 10461 | 5 mg powder |
| DAPI (4',6-Diamidino-2-Phenylindole, Dilactate) | ThermoFisher | D3571 | DNA probe |
| Dermal fibroblasts | ATCC | PCS-201-010 | |
| Dermal microvascular endothelial cells | ATCC | CRL-3243 | |
| Dexamethasone | Sigma | D4902 | |
| DMEM | ThermoFisher | 11054020 | |
| DMEM/F-12 | GIBCO | 11320082 | |
| DMEM/F-12, GlutaMAX | GIBCO | 10565-018 | Basal medium for ALI medium |
| Donkey Anti-Mouse IgG H&L (Alexa Fluor 488) | ABCAM | ab150105 | Donkey Anti-Mouse secondary antibody |
| Donkey Anti-Mouse IgG H&L (Alexa Fluor 568) | ABCAM | ab175472 | Donkey Anti-Mouse secondary antibody |
| Donkey Anti-Mouse IgG H&L (Alexa Fluor 647) | ABCAM | ab150107 | Donkey Anti-Mouse secondary antibody |
| Donkey Anti-Rabbit IgG H&L (Alexa Fluor 488) | ABCAM | ab150073 | Donkey Anti-Mouse secondary antibody |
| Donkey Anti-Rabbit IgG H&L (Alexa Fluor 568) | ABCAM | ab175470 | Donkey Anti-Mouse secondary antibody |
| Donkey Anti-Rabbit IgG H&L (Alexa Fluor 647) | ABCAM | ab150075 | Donkey Anti-Mouse secondary antibody |
| Dulbecco’s PBS (DPBS-/-) (without Ca2+, Mg2+) | Corning | 21-031-CV | 1x |
| Epidermal Growth Factor (EGF) human, recombinant in E. coli | PromoCell | C-60170 | Medium supplement |
| F-12 Ham’s | Invitrogen | 21700-108 | For vascular ECM |
| FibriCol | Advanced BioMatrix | 5133-20ML | Collagen-I solution (10 mg/mL) |
| Fibronectin | Corning | 356008 | |
| Fibronectin, Human, Natural, | Corning | 47743-654 | human plasma fibronectin |
| Fine-tip precision tweezers | Aven | 18056USA | Technik Style 5B-SA Precision Stainless Steel Tweezers |
| Glutamax | Invitrogen | 21700-108 | |
| Glutamax | Invitrogen | 35050061 | |
| Goat Anti-Mouse IgG H&L (Alexa Fluor 594) | ABCAM | ab150080 | Goat Anti-Mouse secondary antibody |
| Goat Anti-Mouse IgG H&L (Alexa Fluor 647) | ABCAM | ab150115 | Goat Anti-Mouse secondary antibody |
| Goat Anti-Mouse IgG H&L (FITC) | ABCAM | ab6785 | Goat Anti-Mouse secondary antibody |
| Goat Anti-Mouse IgG1 Alexa Fluor 568 | ThermoFisher | A-21124 | Goat Anti-Mouse IgG1 secondary antibody |
| Goat Anti-Mouse IgM Alexa Fluor 488 | ThermoFisher | A-21042 | Goat Anti-Mouse IgM secondary antibody |
| Handheld vacuum aspirator | Corning | 4930 | - |
| Heat Inactivated HyClone FetalClone II Serum (FCS) | GE Healthcare Life Sciences | SH30066.03 | |
| Hemocytometer | - | - | - |
| Heparin sodium salt from porcine intestinal mucosa | Sigma | H3149 | |
| HEPES | Thermo | 15630080 | |
| Human [Leu15] – Gastrin | Sigma | G9145 | |
| Human colonoids | Obtained from clinical resections | Obtained from clinical resections | |
| Human EGF Recombinant Protein | Thermo | PHG0311L | |
| human epithelial growth factor | Thermo | PHG0311 | |
| HyClone FetalClone II Serum (U.S.) | GE Healthcare | SH30066.02HI | Sterile FBS heat-inactivated |
| Hydrocortisone 21-hemisuccinate sodium salt | Sigma | H4881 | |
| Hydrocortisone | PromoCell | C-64420 | Medium supplement |
| Ice bucket | - | - | - |
| Ismatec IPC-N | Cole-Palmer | EW-78000-41 | Low-Speed Digital Peristaltic Pump; q24-Channel (1 per 12 Chips) |
| ITES | BioWhittaker | 17-839Z | |
| Keratinocyte Growth Factor (KGF), also known as Basic Fibroblast Growth Factor 7 (FGF-7), human, recombinant in HEK | PromoCell | C-63821 | |
| Keratinocytes | ATCC | PCS-200-010 | |
| Laminin | Biolamina | CT521-0501 | |
| Laminin, 521 CTG (CT521) | Biolamina | CT521-0501 | human recombinant laminin 521 |
| Lung Fibroblast | Cell Biologics | H6013 | |
| Lung Fibroblast | Lifeline Cell Technology | FC-0049 | |
| Lung microvascular endothelial cells | Lonza | CC-2527 | |
| Lung smooth muscle cells | Lifeline Cell Technology | FC-0046 | |
| Manual counter | - | - | - |
| Masterflex (TPE) Transfer Tubing | Cole-Palmer | FV-96880-02 | PharMed BPT, 1/32" ID x 5/32" OD |
| Medium 199, no phenol red | Thermo | 11043023 | |
| Microcentrifuge tube | - | - | 1.5 mL, sterile |
| Microscope (with camera) | - | - | For bright-field imaging |
| N2 | Sigma | 17502001 | |
| N-acetyl cysteine | Sigma | A5099 | |
| Noggin (HEK293T conditioned medium) | Sigma | N17001 | |
| Normal Goat Serum | ThermoFisher | 50062Z | Blocking solution |
| O-phosphosrylethanolamine | Sigma | P0503 | |
| Paraformaldehyde (4% wt/vol) | EMS | 15710 | Fixing agent |
| Penicillin Streptomycin | GIBCO | 15140122 | |
| Penicillin-streptomycin | Sigma | P4333 | 10,000 U/mL; 10 mg/mL |
| Pipette tips | - | - | P20, P200, and P1000 sterile, low adhesion |
| Pipette | Gilson | F167380 | P20, P200, and P1000 |
| PluriQ Serum Replacement (or alternatively KO Serum replacement) | AMSBIO (or Thermo) | N/A (or C1910828010) | |
| Poly-L-Lysine coated microscope glass slides | Sigma | P0425 | Glass slides |
| Primocin | InvivoGen | ant-pm-1 | |
| Progesterone | Sigma | P8783 | |
| ProLong Gold | ThermoFisher | P36931 | Antifade Mountant with DAPI |
| Retinoic Acid | Sigma | R2625 | |
| ROCK inhibitor (Y27632) | Tocris | TB1254-GMP/10 | |
| R-spondin (HEK293T conditioned medium) | Sigma | SCC111 | |
| SAGM SingleQuots supplements | Lonza | CC-4124 | |
| SAGMTM Small Airway Epithelial Cell Growth medium BulletKitTM | Lonza | CC-4124 | Medium supplements |
| SB2001190 | Tocris | 1264/10 | |
| Serological pipettes | - | - | 2 mL, 5 mL, 10 mL, and 25 mL low endotoxin, sterile |
| Small Airway Epithelial Cell Growth medium (SAGM) | Lonza | CC-4124 | |
| Solvent Buffer (ER-2) | Emulate | 10462 | 25 mL bottle |
| Steriflip-HV | Millipore | SE1M003M00 | Sterile filtering conical tube |
| Sterilin 100 mm Square Petri Dishes | Thermo | 103 | Sterile, 1 per 6 chips |
| T25 flasks | - | - | - |
| T75 flasks | - | - | - |
| Tri-iodothyronine | Sigma | T5516 | |
| Triton X-100 (0.3% (vol/vol) | Sigma | T8787 | Permeabilization agent |
| Trypan blue | Sigma | 93595 | 0.4% solution |
| TrypEE solution | Sigma | 12604013 | Cell detaching solution |
| TWEEN-20 | Sigma | P2287 | Permeabilization agent |
| UV Light Oven (peak frequency 365nm, intensity of 100 µJ/cm2) | VWR | 21474-598 | UVP, Long Range UV, 365 nm 60Hz Model CL-1000L |
| Vacuum set-up | - | - | Minimum pressure: -70 kPa |
| Vascular Endothelial Growth Factor 165 (VEGF-165) human, recombinant in E. coli | PromoCell | C-64420 | |
| VEGF-165 | PromoCell | C-64420 | Medium supplement |
| Von Willebrand Factor conjugated FITC | ABCAM | ab8822 | Sheep polyclonal antibody |
| Water bath (or beads) | - | - | Set to 37 °C |
| Wnt3A (L-Wnt3A conditioned medium) | ATCC | CRL-2647 |
Referencias
- Van Norman, G. A. Limitations of animal studies for predicting toxicity in clinical trials: Is it time to rethink our current approach. JACC: Basic to Translational Science. 4 (7), 845-854 (2019).
- Wange, R. L., Brown, P. C., Davis-Bruno, K. L. Implementation of the principles of the 3Rs of animal testing at CDER: Past, present and future. Regulatory Toxicology and Pharmacology. 123, 104953 (2021).
- Mosig, A. S. Organ-on-chip models: New opportunities for biomedical research. Future Science OA. 3 (2), (2017).
- Alépée, N., et al. State-of-the-art of 3D cultures (organs-on-a-chip) in safety testing and pathophysiology. Altex. 31 (4), 441-477 (2014).
- MacArron, R., et al. Impact of high-throughput screening in biomedical research. Nature Reviews Drug Discovery. 10 (3), 188-195 (2011).
- Hughes, J. P., Rees, S. S., Kalindjian, S. B., Philpott, K. L. Principles of early drug discovery. British Journal of Pharmacology. 162 (6), 1239-1249 (2011).
- Kitaeva, K. V., Rutland, C. S., Rizvanov, A. A., Solovyeva, V. V. Cell culture based in vitro test systems for anticancer drug screening. Frontiers in Bioengineering and Biotechnology. 8, 322 (2020).
- Mao, P., et al. Human alveolar epithelial type II cells in primary culture. Physiological Reports. 3 (2), 12288 (2015).
- Zaitseva, M., Vollenhoven, B. J., Rogers, P. A. W. In vitro culture significantly alters gene expression profiles and reduces differences between myometrial and fibroid smooth muscle cells. Molecular Human Reproduction. 12 (3), 187-207 (2006).
- Singh, A., Brito, I., Lammerding, J. Beyond tissue stiffness and bioadhesivity: Advanced biomaterials to model tumor microenvironments and drug resistance. Trends in Cancer. 4 (4), 281-291 (2018).
- Nawroth, J. C., et al. Stem cell-based Lung-on-Chips: The best of both worlds. Advanced Drug Delivery Reviews. 140, 12-32 (2019).
- Jensen, C., Teng, Y. Is it time to start transitioning from 2d to 3d cell culture. Frontiers in Molecular Biosciences. 7, 33 (2020).
- Kapałczyńska, M., et al. 2D and 3D cell cultures – a comparison of different types of cancer cell cultures. Archives of Medical Science. 14 (4), 910-919 (2018).
- Sutherland, R. M., Inch, W. R., McCredie, J. A., Kruuv, J. A multi-component radiation survival curve using an in vitro tumour model. International Journal of Radiation Biology. 18 (5), 491-495 (1970).
- Chandra, P., Lee, S. J. Synthetic extracellular microenvironment for modulating stem cell behaviors. Biomarker Insights. 10, 105-116 (2015).
- Nicolas, J., et al. 3D extracellular matrix mimics: Fundamental concepts and role of materials chemistry to influence stem cell fate. Biomacromolecules. 21 (6), 1968-1994 (2020).
- Brassard, J. A., Lutolf, M. P. Engineering stem cell self-organization to build better organoids. Cell Stem Cell. 24 (6), 860-876 (2019).
- Lutolf, M. P., Gilbert, P. M., Blau, H. M. Designing materials to direct stem-cell fate. Nature. 462 (7272), 433-441 (2009).
- Mantha, S., et al. Smart hydrogels in tissue engineering and regenerative medicine. Materials. 12 (20), 3323 (2019).
- Langhans, S. A. Three-dimensional in vitro cell culture models in drug discovery and drug repositioning. Frontiers in Pharmacology. 9, 6 (2018).
- Li, H., et al. Biomechanical cues as master regulators of hematopoietic stem cell fate. Cellular and Molecular Life Sciences. 78 (16), 5881-5902 (2021).
- Donoghue, L., Nguyen, K. T., Graham, C., Sethu, P. Tissue chips and microphysiological systems for disease modeling and drug testing. Micromachines. 12 (2), 139 (2021).
- Ma, C., Peng, Y., Li, H., Chen, W. Organ-on-a-chip: A new paradigm for drug development. Trends in Pharmacological Sciences. 42 (2), 119-133 (2021).
- Varone, A., et al. A novel organ-chip system emulates three-dimensional architecture of the human epithelia and the mechanical forces acting on it. Biomaterials. 275, 120957 (2021).
- Hassell, B. A., et al. Human organ chip models recapitulate orthotopic lung cancer growth, therapeutic responses, and tumor dormancy in vitro. Cell Reports. 21 (2), 508-516 (2017).
- Sadeghipour, A., Babaheidarian, P. Making formalin-fixed, paraffin embedded blocks. Biobanking. 1897, 253-268 (2019).
- Grant, J., et al. Simulating drug concentrations in PDMS microfluidic organ chips. Lab on a Chip. 21 (18), 3509-3519 (2021).
- Barrile, R., et al. Organ-on-chip recapitulates thrombosis Induced by an anti-CD154 monoclonal antibody: Translational potential of advanced microengineered systems. Clinical Pharmacology & Therapeutics. 104 (6), 1240-1248 (2018).
- Jain, A., et al. Primary human lung alveolus-on-a-chip model of intravascular thrombosis for assessment of therapeutics. Clinical Pharmacology & Therapeutics. 103 (2), 332-340 (2018).
- Campbell, S. B., Wu, Q., Yazbeck, J., Liu, C., Okhovatian, S., Radisic, M. Beyond polydimethylsiloxane: Alternative materials for fabrication of organ-on-a-chip devices and microphysiological systems. ACS Biomaterials Science and Engineering. 7 (7), 2880-2899 (2021).
- Pun, S., Haney, L. C., Barrile, R. Modelling human physiology on-chip: Historical perspectives and future directions. Micromachines. 12 (10), 1250 (2021).

