Percutaneous Endoscopic Unilateral-Approach Bilateral Decompression for Lumbar Spinal Stenosis
Summary
The present protocol describes the steps and key points of lumbar endoscopic unilateral laminotomy for bilateral decompression for the treatment of degenerative lumbar spinal stenosis.
Abstract
Lumbar spinal stenosis (LSS) involves the narrowing of the spinal canal due to degenerative changes in the vertebral joints, intervertebral discs, and ligaments. LSS encompasses central canal stenosis (CCS), lateral recess stenosis (LRS), and intervertebral foramen stenosis (IFS). The utilization of lumbar endoscopic unilateral laminotomy for bilateral decompression (LE-ULBD) has gained popularity in the treatment of CCS and LRS. This popularity is attributed to the rapid development of endoscopic instruments and the progress of endoscopic philosophy.
In this technical report, a detailed introduction to the steps and key points of LE-ULBD is provided. Simultaneously, a retrospective review of 132 consecutive patients who underwent LE-ULBD for central canal and/or lateral recess stenosis was conducted. The outcomes after more than two years of follow-up were assessed using the visual analogue score (VAS), Oswestry Disability Index (ODI), Japanese Orthopaedic Association (JOA) scores, and the modified MacNab criteria to evaluate surgical efficacy. All 132 patients underwent LE-ULBD successfully. Among them, 119 patients were rated as “excellent,” while 13 patients were rated as “good” based on the modified MacNab criteria during the last follow-up. Incidental dural tears occurred in four cases, but there were no post-operative epidural hematomas or infections. The experience demonstrates that LE-ULBD is a less invasive, effective, and safe approach. It can be considered as an alternative option for treating patients with lumbar central canal stenosis and/or lateral recess stenosis.
Introduction
Degenerative lumbar spinal stenosis (DLSS) can result from alterations in bony, discal, capsular, or ligamentary anatomical structures. Clinically, LSS presents with a range of symptoms, including radiating sciatic pain in the legs, neurogenic claudication during ambulation, and sensory disturbances, all of which significantly affect patients' quality of life1,2,3. An initial conservative management period of two months is the recommended therapeutic approach for LSS. If conservative measures prove ineffective, transitioning to surgical decompression therapy becomes the subsequent recommendation.
Historically, open laminectomy has been the traditional surgical approach for addressing spinal stenosis. Peer-reviewed studies have confirmed its safety and cost-effectiveness, with outcomes showing notable superiority compared to non-surgical interventions4,5,6,7,8. However, the complexities involved in achieving comprehensive bony decompression of neural components can introduce the risk of segmental spinal instability. This may lead to symptom recurrence or a subsequent need for arthrodesis9,10. Distinctively, Ghogawala et al.11 documented that approximately 34% of patients, after undergoing traditional laminectomy for lumbar spinal stenosis with concomitant stable spinal spondylolisthesis, required revision surgery within four years post-operatively.
Over the past five decades, a discernible shift toward minimally invasive spinal surgical procedures has become evident. The overarching objective has been to reduce approach-related tissue disruption and subsequently mitigate post-operative discomfort and disability. This transition aligns seamlessly with the advent and progression of percutaneous endoscopic spine surgery.
Synchronized with advancements in endoscopic technology and a deepened understanding of endoscopic anatomical intricacies, contemporary endoscopic spine surgeons are now equipped to address a spectrum of LSS presentations safely and effectively. Specifically, lumbar endoscopic unilateral laminotomy for bilateral decompression (LE-ULBD) has gained traction as a favored modality for LSS management12,13,14,15,16,17(Figure 1). Paramount benefits of LE-ULBD include minimal incision requirements, avoidance of soft tissue disruption and muscular denervation, along with improved visualization.
Between May 2017 and May 2021, our institution employed the LE-ULBD technique to manage 132 patients with LSS. Relevant technical details and outcomes over a two-year follow-up period are elucidated herein. Based on these findings, LE-ULBD emerges as a minimally invasive, effective, and secure modality, positioning it as a viable alternative therapeutic strategy for patients diagnosed with lumbar central canal stenosis and/or lateral recess stenosis.
Protocol
This study was conducted in strict accordance with the protocols established by the Institutional Review Boards of Zhongshan Hospital and Minhang Hospital, both affiliated with Fudan University (approval numbers: 2021-042 and 2021-037-01X, respectively). All participating patients provided informed written consent. Exclusion criteria were rigorously applied: patients with foraminal stenosis, multi-level stenosis, significant instability, a medical history of previous lumbar spine surgical interventions, or those presenting with degenerative spondylolisthesis of grade 2 or higher were considered ineligible for the study.
1. Patient preparation
- Perform the surgery under general anesthesia following approved protocols.
- Place the patient in a prone position on a radiopermeable operating table, and flex appropriately (Figure 2).
- Select the responsible lumbar segment causing clinical symptoms as the operative segment.
NOTE: For the convenience of description, the most common L4-5 stenosis was selected as an example to describe the surgical technique in this report. Other segments are similar to L4-5. - Generally, select the more symptomatic side as the side of the surgical approach. In this report, the left side was selected as the surgical side for introduction.
- Determine the skin incision and working channel docking point on the anteroposterior view of fluoroscopy. Select the lateral edge of the left lumbar 4-5 interlaminar window as the docking target, and select its vertical projection on the body surface as the entry point of the skin.
- After the patient is routinely sterilized and draped, make a 10 mm stab wound in the skin deep to the fascia layer. Introduce a pencil-like rod with a 2 mm diameter to touch the bone (left L4-5 articular) under fluoroscopic guidance.
2. Insertion of endoscope
- Gradually expand the vertebral muscle and fascia using the soft tissue extender (see Table of Materials). Then, insert the 10 mm work sleeve with an oblique mouth. Verify the position of the working sleeve with fluoroscopy (Figure 3).
- Introduce the endoscopic surgical system (15° viewing angle, outer diameter 10 mm, working channel diameter of 6 mm, and working length 125 mm), and perform all the subsequent steps under constant irrigation with endoscopic visualization.
3. Surgical procedure
- Under high-definition endoscopic visualization, dissect the soft tissue off the bone, and perform hemostasis using the radiofrequency probe (see Table of Materials). Confirm the presence of important anatomical structures, including the lower half of the left L4 lamina, left L4-5 facet joint, the upper 1/3 of the left L5 lamina, the base of the spinous process of L4 and L5, and the proliferative ligamentum flavum within the lumbar 4-5 interlaminar window1.
- Initiate ipsilateral bony decompression, starting from the L4 lamina. Remove the lower part of the L4 lamina, the medial part of the L4-5 articular process, and the upper edge of the L5 lamina using a 3.5 mm endoscopic diamond bur, trephine, and Kerrison Rongeur (see Table of Materials).
- Utilize the 5 mm diameter endoscopic trephine (see Table of Materials) to effectively remove the L4 inferior articular process, which is surrounded by joint capsule and ligament. Separate and remove the superficial layer of the ligamentum flavum from the inner layer with a rongeur.
NOTE: Retain the inner layer of the ligamentum flavum in place, as it is tightly attached to the inner surface of the lamina and the medial border of the L5 superior articular process. - The goal of lamina decompression in this step is to expose the cephalad and caudal free margins of the deep layer of the ligamentum flavum. Cut the medial border of the L5 superior articular process to free the lateral margin of the ligamentum flavum.
- Utilize the 5 mm diameter endoscopic trephine (see Table of Materials) to effectively remove the L4 inferior articular process, which is surrounded by joint capsule and ligament. Separate and remove the superficial layer of the ligamentum flavum from the inner layer with a rongeur.
- Proceed with contralateral bony decompression, starting from the base of the spinous process of L4. Shave the base of the spinous process to create sufficient space, facilitating the insertion of the working cannula (working channel diameter of 6 mm, and working length 125 mm) towards the contralateral side.
- Polish the contralateral lower part of the L4 lamina from the inner surface to expose the cephalad free margin of the ligamentum flavum.
- Similarly, shave the base of the L5 spinous process and remove the upper edge of the L5 lamina to expose the caudal free margin of the ligamentum flavum. Detach the contralateral lateral margin of the ligamentum flavum from the contralateral L5 superior articular process.
- Remove the medial part of the L5 superior articular process using a diamond bur and/or Kerrison Rongeur.
- After completing bony decompression and releasing ligamentum flavum attachments, remove the ligamentum flavum either in an en bloc manner or in a piecemeal manner.
- Remove the inner layer of the ligamentum flavum at the end of the operation. This step helps protect nerve elements during osteotomy and reduces vision blurring caused by epidural hemorrhage.
- Explore both lateral recesses to ensure thorough decompression of bilateral traversing nerves. If necessary, perform further decompression of the ligamentum flavum and its attached bony structures using endoscopic Kerrison Rongeur.
- Conclude the procedure when the dura and bilateral traversing nerve roots are free. Perform careful hemostasis using endoscopic radiofrequency bipolar. Close the skin incision with one or two stitches (approximately 1 cm).
4. Post-operative care and follow-up
- After lying down for 8 h post-surgery, ask the patients to walk, but ensure that they wear support around their waist.
- Provide proper guidance to the patients on exercises to aid in their recovery. Carefully monitor and address any swelling or pain that may occur after the operation.
- After 7 days of the surgery, encourage them to gradually resume their usual routines. Conduct regular check-ins to assess their healing progress.
Representative Results
Outcome evaluation
Surgical results were assessed using the Visual Analog Scale (VAS) scores for leg and back pain, Japanese Orthopaedic Association (JOA) Scores, Oswestry Disability Index (ODI) scores, and the modified MacNab standard12,15,17. These indicators were measured pre-operatively and at two days, six months, 12 months, and 24 months post-surgery.
Clinical outcome
All 132 patients who underwent surgery successfully completed the 24-month follow-up. There is a statistically significant difference in scores before and after the operation (P < 0.001). According to the modified MacNab criteria17, all 132 patients exhibited satisfactory results at the final follow-up. Figure 4 and Figure 5 illustrate a clinical example of LE-ULBD performed. Changes in VAS scores, JOA scores, and ODI scores over time are presented in Table 1 and Figure 6. Figure 7 shows the clinical results based on the modified MacNab criteria.
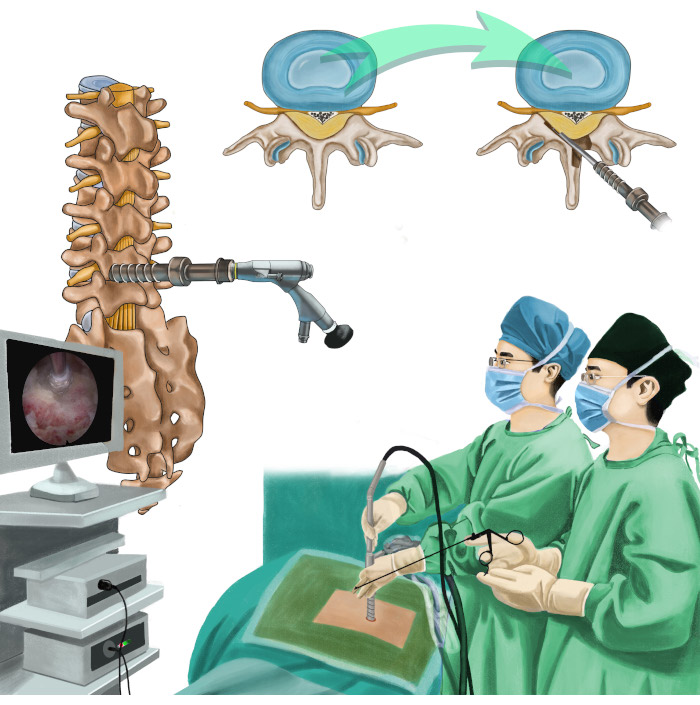
Figure 1: Overview of the protocol. Illustration depicting percutaneous endoscopic unilateral-approach bilateral decompression for lumbar spinal stenosis. Please click here to view a larger version of this figure.
Figure 2: Patient positioning. The patient is positioned in a prone posture on a radiolucent operating table and appropriately flexed. Please click here to view a larger version of this figure.
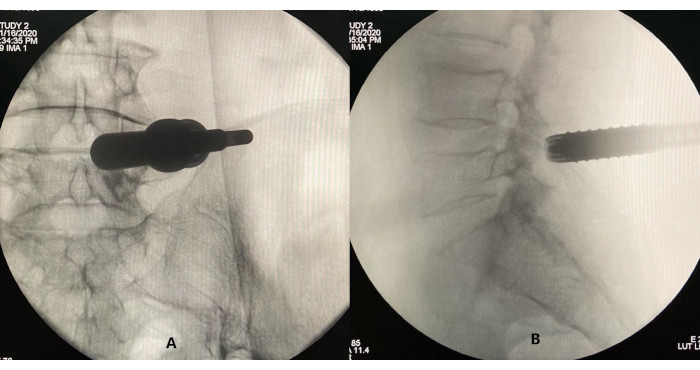
Figure 3: Confirming the correct placement of the working cannula through fluoroscopy. (A) Lateral view and (B) anteroposterior view. Please click here to view a larger version of this figure.
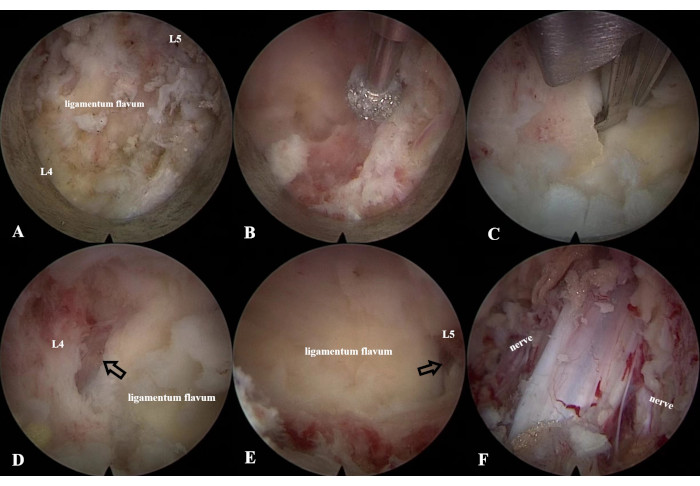
Figure 4: LE-ULBD procedure. (A) Identification of the anatomical structures of the left L4 lamina, L5 lamina, and ligamentum flavum using (B) a diamond bur and (C) an endoscopic Kerrison Rongeur to enlarge the interlaminar window. (D,E) Identification of the cranial and caudal ends of the ligamentum flavum. (F) Decompression of the dural sac and bilateral traversing nerve roots. Please click here to view a larger version of this figure.
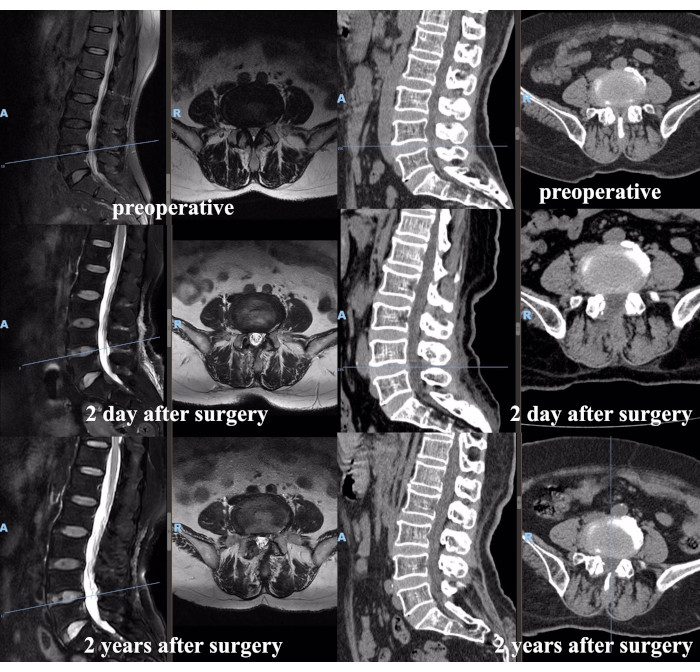
Figure 5: Pre- and post-operative MRI and CT scans of a representative patient. Please click here to view a larger version of this figure.
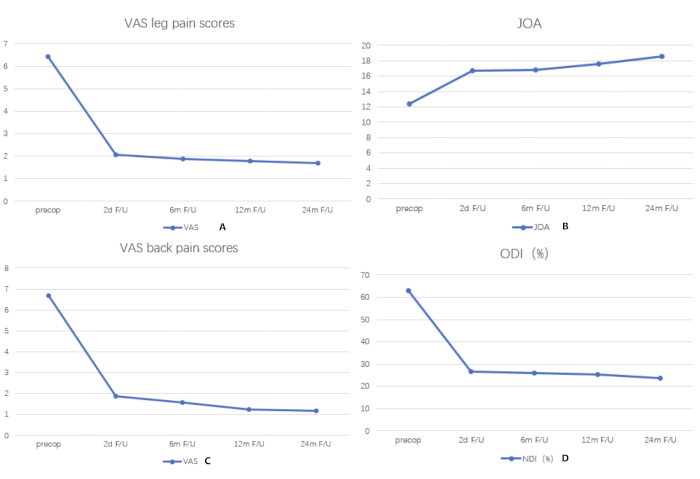
Figure 6: Changes in VAS, JOA, and ODI scores over time. Significant improvements in leg and back VAS scores (A,C), JOA scores (B), and ODI (D) during the follow-up period. The x-axes depict time, and the y-axes depict the scores. Please click here to view a larger version of this figure.
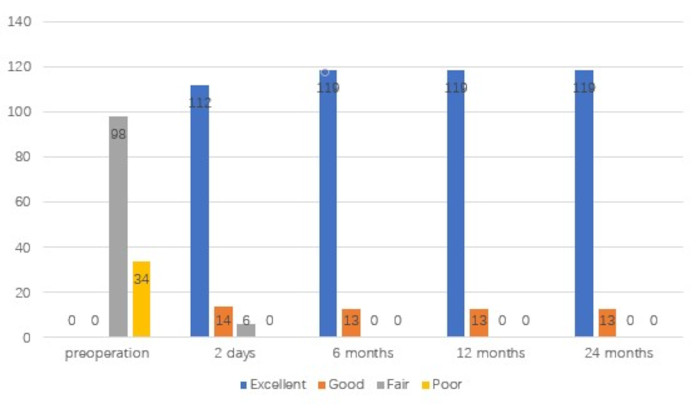
Figure 7: Clinical results assessed according to the modified MacNab criteria. The x-axes depict time, and the y-axes depict the scores. Please click here to view a larger version of this figure.
| Time | VAS leg pain scores | VAS back pain scores | JOA | ODI |
| scores | (%) | |||
| Pre-operative | 6.41 ± 1.33 | 6.68 ± 1.43 | 12.44 ± 1.87 | 62.94 ± 9.52 |
| post-operative | ||||
| 2 d | 2.06 ± 1.12 | 1.89 ± 0.83 | 16.69 ± 1.60 | 26.56 ± 6.93 |
| 6 m | 1.88 ± 1.16 | 1.58 ± 0.88 | 16.81 ± 1.85 | 25.89 ± 6.74 |
| 12 m | 1.78 ± 0.90 | 1.26 ± 0.90 | 17.62 ± 2.91 | 25.44 ± 7.29 |
| 24 m | 1.70 ± 1.10 | 1.17 ± 1.02 | 18.53 ± 2.80 | 23.55 ± 7.46 |
| F value | 789.446 | 1130.254 | 287.64 | 2243.436 |
| P value | P < 0.001 | P < 0.001 | P < 0.001 | P < 0.001 |
| data as (x̄̄ ± s) |
Table 1: Pre- and post-operative scores for 132 patients.
Discussion
With the progressive evolution of percutaneous endoscopic spine surgery and clinicians' profound comprehension of endoscopic procedures, the therapeutic indications for endoscopic lumbar interventions have expanded to encompass all manifestations of LSS, branching out from merely addressing lumbar disc herniation. The spectrum of endoscopic management for LSS includes stenosis decompression, contralateral decompression, and fusion facilitated by endoscopic visualization. An increasing volume of research underscores that LE-ULBD yields outcomes analogous to those obtained via micro-lumbar decompression laminectomy. The LE-ULBD technique is endowed with several merits, including high-definition intraoperative visualization, minimized soft tissue trauma, and an expedited post-operative recovery trajectory12,13,14,15,16,17.
LE-ULBD is optimally tailored for addressing central canal stenosis, lateral recess stenosis, or an amalgamation of both presentations. For those presenting with grade 1 spondylolisthesis1, LE-ULBD can be judiciously considered as a surgical intervention. For cases indicative of intervertebral foramen stenosis, transforaminal endoscopic decompression is typically the method of choice.
The procedure consists of the following key steps. First, bone decompression must be performed before the complete removal of the ligamentum flavum. Drilling above the ligamentum flavum can protect it from inadvertent durotomy. Second, the endpoint of lamina decompression is the cephalad and caudal aspects of the ligamentum flavum being exposed. Third, the endpoint for cutting the superior articular process is the lateral margin of the ligamentum flavum being exposed. In this step, the resection of the articular process should not exceed 1/2 because this may cause lumbar instability. Fourth, exploring the medial edge of the pedicle with a Kerrison rongeur is an important indicator of the decompression of the traversing nerve root. Of course, the best endpoint for nerve decompression is the lateral margin of the nerve root from the emitting site to the intervertebral foramen presented on the screen. Fifth, to obtain enough space to insert the working sleeve into the opposite side, we ground the base of the spinous process. Lastly, the endpoint is reached when the dura and bilateral traversing nerve roots are free.
Within our cohort, dural tears were observed in four patients, although none exhibited related symptoms. Upon detecting a dural tear, we promptly used a gelatin sponge to seal the compromised region of the dural sac and aimed to conclude the surgical intervention within the next 30 min. No post-operative infections occurred. Over time, Visual Analog Scale (VAS) scores for the 132 patients consistently declined, while Japanese Orthopedic Association (JOA) scores showed an ascending trend, and the Oswestry Disability Index (ODI) likewise decreased. At the 24-month post-operative point, the therapeutic efficacy, assessed using the modified MacNab criterion, was rated as "excellent" for 119 patients and "good" for the remaining13.
However, we must acknowledge the inherent limitations of our study: (1) This investigation was retrospective and limited by a small sample size. (2) Since the focus was exclusively on patients with single-level stenosis, the outcomes may not apply to cases involving multi-level stenosis. (3) Implementing this specific surgical technique requires meticulous preoperative planning and a deep familiarity with LE-ULBD operations.
The field of endoscopic spinal surgery is expanding, moving beyond its initial focus on lumbar disc degeneration to encompass a wider range of spinal pathologies, from the craniovertebral junction to the sacral vertebrae. Looking ahead, we anticipate the integration of cutting-edge technologies, such as robotics and artificial intelligence-enhanced navigation, which are poised to bring transformative innovations to the realm of endoscopic spine surgery.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| Kerrison Rongeur | Joimax GmbH, Karlsruhe, Germany | ||
| The endoscopic high-speed diamond burr | NSK-Nakanishi International, Co., Ltd., Osaka, Japan | Primado P200-RA330 | |
| The endoscopic surgical system Delta | Joimax GmbH, Karlsruhe, Germany | ||
| The radiofrequency probe | Elliquence LLC, Baldwin, New York | Trigger-FlexR Bipolar System | |
| Trephine | Joimax GmbH, Karlsruhe, Germany |
Referencias
- Benini, A. Lumbar spinal stenosis. An overview 50 years following initial description. Der Orthopade. 22 (4), 257-266 (1993).
- Cinotti, G., Postacchini, F., Fassari, F., Urso, S. Predisposing factors in degenerative spondylolisthesis: a radiographic and CT study. International Orthopaedics. 21, 337-342 (1997).
- Torudom, Y., Dilokhuttakarn, T. Two portal percutaneous endoscopic decompression for lumbar spinal stenosis: preliminary study. Asian Spine Journal. 10 (2), 335 (2016).
- Atlas, S. J., Keller, R. B., Robson, D., Deyo, R. A., Singer, D. E. Surgical and nonsurgical management of lumbar spinal stenosis: four-year outcomes from the maine lumbar spine study. Spine. 25 (5), 556-562 (2000).
- Atlas, S. J., Keller, R. B., Wu, Y. A., Deyo, R. A., Singer, D. E. Long-term outcomes of surgical and nonsurgical management of lumbar spinal stenosis: 8 to 10 year results from the maine lumbar spine study. Spine. 30 (8), 936-943 (2005).
- Malmivaara, A., et al. Lumbar spinal research group. surgical or nonoperative treatment for lumbar spinal stenosis?: a randomized controlled trial. Spine. 32 (1), 1-8 (2007).
- Parker, S. L., et al. Cost-effectiveness of multilevel hemilaminectomy for lumbar stenosis-associated radiculopathy. The Spine Journal. 11 (8), 705-711 (2011).
- Parker, S. L., et al. Two-year comprehensive medical management of degenerative lumbar spine disease (lumbar spondylolisthesis, stenosis, or disc herniation): a value analysis of cost, pain, disability, and quality of life. Journal of Neurosurgery: Spine. 21 (2), 143-149 (2014).
- Fox, M. W., Onofrio, B. M., Hanssen, A. D. Clinical outcomes and radiological instability following decompressive lumbar laminectomy for degenerative spinal stenosis: a comparison of patients undergoing concomitant arthrodesis versus decompression alone. Journal of Neurosurgery. 85 (5), 793-802 (1996).
- Jönsson, B., Annertz, M., Sjöberg, C., Strömqvist, B. A prospective and consecutive study of surgically treated lumbar spinal stenosis: part II: five-year follow-up by an independent observer. Spine. 22 (24), 2938-2944 (1997).
- Ghogawala, Z., et al. Laminectomy plus fusion versus laminectomy alone for lumbar spondylolisthesis. New England Journal of Medicine. 374 (15), 1424-1434 (2016).
- Komp, M., et al. Bilateral spinal decompression of lumbar central stenosis with the full-endoscopic interlaminar versus microsurgical laminotomy technique: a prospective, randomized, controlled study. Pain Physician. 18 (1), 61 (2015).
- Ito, F., et al. Step-by-step sublaminar approach with a newly-designed spinal endoscope for unilateral-approach bilateral decompression in spinal stenosis. Neurospine. 16 (1), 41 (2019).
- Lee, C. W., Yoon, K. J., Jun, J. H. Percutaneous endoscopic laminotomy with flavectomy by uniportal, unilateral approach for the lumbar canal or lateral recess stenosis. World Neurosurgery. 113, e129-e137 (2018).
- McGrath, L. B., White-Dzuro, G. A., Hofstetter, C. P. Comparison of clinical outcomes following minimally invasive or lumbar endoscopic unilateral laminotomy for bilateral decompression. Journal of Neurosurgery: Spine. 30 (4), 491-499 (2019).
- Pairuchvej, S., Muljadi, J. A., Ho, J. C., Arirachakaran, A., Kongtharvonskul, J. Full-endoscopic (bi-portal or uni-portal) versus microscopic lumbar decompression laminectomy in patients with spinal stenosis: systematic review and meta-analysis. European Journal of Orthopaedic Surgery & Traumatology. 30, 595-611 (2020).
- Kim, H. S., Wu, P. H., Jang, I. T. Lumbar endoscopic unilateral laminotomy for bilateral decompression outside-in approach: a proctorship guideline with 12 steps of effectiveness and safety. Neurospine. 17 (Suppl 1), S99 (2020).

.
