Generation of Centromere-Associated Protein-E CENP-E-/- Knockout Cell Lines using the CRISPR/Cas9 System
Summary
This article reports the construction of centromere-associated protein-E (CENP-E) knockout cells using the CRISPR/Cas9 system and three phenotype-based screening strategies. We have utilized the CENP-E knockout cell line to establish a novel approach to validate the specificity and toxicity of the CENP-E inhibitors, which is useful for drug development and biological research.
Abstract
The CRISPR (clustered regularly interspaced short palindromic repeats)/Cas9 system has emerged as a powerful tool for precise and efficient gene editing in a variety of organisms. Centromere-associated protein-E (CENP-E) is a plus-end-directed kinesin required for kinetochore-microtubule capture, chromosome alignment, and spindle assembly checkpoint. Although cellular functions of the CENP-E proteins have been well studied, it has been difficult to study the direct functions of CENP-E proteins using traditional protocols because CENP-E ablation usually leads to spindle assembly checkpoint activation, cell cycle arrest, and cell death. In this study, we have completely knocked out the CENP-E gene in human HeLa cells and successfully generated the CENP-E-/- HeLa cells using the CRISPR/Cas9 system.
Three optimized phenotype-based screening strategies were established, including cell colony screening, chromosome alignment phenotypes, and the fluorescent intensities of CENP-E proteins, which effectively improve the screening efficiency and experimental success rate of the CENP-E knockout cells. Importantly, CENP-E deletion results in chromosome misalignment, the abnormal location of the BUB1 mitotic checkpoint serine/threonine kinase B (BubR1) proteins, and mitotic defects. Furthermore, we have utilized the CENP-E knockout HeLa cell model to develop an identification method for CENP-E-specific inhibitors.
In this study, a useful approach to validate the specificity and toxicity of CENP-E inhibitors has been established. Moreover, this paper presents the protocols of CENP-E gene editing using the CRISPR/Cas9 system, which could be a powerful tool to investigate the mechanisms of CENP-E in cell division. Moreover, the CENP-E knockout cell line would contribute to the discovery and validation of CENP-E inhibitors, which have important implications for antitumor drug development, studies of cell division mechanisms in cell biology, and clinical applications.
Introduction
Engineered genome editing mediates the targeted modifications of genes in a variety of cells and organisms. In eukaryotes, site-specific mutagenesis can be introduced by the applications of sequence-specific nucleases that stimulate homologous recombination of target DNA1. In recent years, several genome editing technologies, including zinc finger nucleases (ZFNs)2,3, transcription activator-like effector nucleases (TALENs)4,5, and homing meganucleases6,7, have been engineered to cleave genomes at specific sites, but these approaches require complex protein engineering and redundant experimental procedures. Studies have shown that the type II prokaryotic clustered regularly interspaced short palindromic repeats (CRISPR)/Cas system is an efficient gene editing technology, which specifically mediates RNA-guided, site-specific DNA cleavage in a wide variety of cells and species8,9,10,11. The CRISPR/Cas9 gene knockout technology has revolutionized the fields of basic biology, biotechnology, and medicine12.
Bacteria and most archaea have evolved an RNA-based adaptive immune system that uses CRISPR and Cas proteins to identify and destroy viruses and plasmids13. Streptococcus pyogenes Cas9 (SpCas9) endonuclease contains the RuvC-like Holliday junction resolvase (RuvC) and His-Asn-His (HNH) domain, which can efficiently mediate sequence-specific, double-stranded breaks (DSBs) by providing a synthetic single-guide RNA (sgRNA) containing CRISPR RNAs (crRNA) and trans-activating crRNA (tracrRNA)14,15,16. DSBs can be repaired through the indel-forming non-homologous end joining (NHEJ) or homology-directed repair (HDR) pathway, which introduces multiple mutations, including insertions, deletions, or scar-less single nucleotide substitutions, in mammalian cells1,8. Both the error-prone NHEJ and the high-fidelity HDR pathway can be used to mediate gene knockout through insertions or deletions, which can cause frameshift mutations and premature stop codons10.
Kinesin-7 CENP-E is required for kinetochore-microtubule attachment and chromosome alignment during cell division17,18,19. Antibody microinjection20,21, siRNA depletion22,23, chemical inhibition24,25,26, and genetic deletion27,28,29 of CENP-E leads to chromosome misalignment, the activation of spindle assembly checkpoint and mitotic defects, which results in aneuploidy and chromosomal instability19,30. In mice, CENP-E deletion results in abnormal development and embryonic lethality at the very early stages of development27,29,31. Genetic deletion of CENP-E usually leads to chromosome misalignment and cell death26,27,29, which is an obstacle in studying the functions and mechanisms of the CENP-E proteins.
A recent study has established a conditional CENP-E knockout cell line using an Auxin-inducible CRISPR/Cas9 gene-editing method32, which enables rapid degradation of CENP-E proteins in a relatively short time33. However, to date, stable CENP-E knockout cell lines have not been established, which is an unresolved technical challenge in CENP-E biology. Considering genetic robustness34, genetic compensation responses35,36,37, and complex intracellular environments, as the direct consequences of complete deletion of CENP-E may be complex and unpredictable, it is important to establish CENP-E knockout cell lines for the investigation of mechanisms of chromosome alignment, spindle assembly checkpoint, and downstream signaling pathways.
The discovery and applications of CENP-E inhibitors are important for cancer treatment. To date, seven types of CENP-E inhibitors have been found and synthesized, including GSK923295 and its derivatives24,25, PF-277138,39, imidazo[1,2-a]pyridine scaffold derivatives40,41, compound-A42,43, syntelin44,45, UA6278446, and benzo[d]pyrrolo[2,1-b]thiazole derivatives47. Among these inhibitors, GSK923295 is an allosteric and efficient CENP-E inhibitor that binds to the motor domain of CENP-E and inhibits CENP-E microtubule-stimulated ATPase activity with a Ki of 3.2 ± 0.2 nM24,25. However, compared with the inhibitory effects of GSK923295 on cultured cancer cells, the therapeutic effects of GSK923295 in clinical cancer patients are not ideal48,49, which also raised concerns about the specificity of GSK923295 for CENP-E. Moreover, the specificity and side effects of other CENP-E inhibitors on the CENP-E proteins are key issues in cancer research.
In this study, we have completely knocked out the CENP-E gene in HeLa cells using the CRISPR/Cas9 system. Three optimized phenotype-based screening strategies have been established, including cell colony screening, chromosome alignment phenotypes, and the fluorescent intensities of CENP-E proteins, to improve the screening efficiency and success rate of CENP-E gene editing. Furthermore, CENP-E knockout cell lines can be used to test the specificity of candidate compounds for CENP-E.
Protocol
1. Construction of the CRISPR/Cas9 gene knockout vectors
- Select the target genomic DNA sequence on the human CENP-E gene (GenBank Accession No. NM_001286734.2) and design the sgRNA using an online CRISPR design tool (http://crispor.tefor.net/).
- Input a single genomic sequence, select the genome of "Homo sapiens-human-UCSC Dec 2013 (hg38 analysis set) + single nucleotide polymorphisms (SNPs): dbSNP148", and select the protospacer adjacent motif "20 bp-NGG-spCas9". Choose two guide sequences, including sgRNA-1, 5'-CGGCCGCACTCGCACGCAGA-3' and sgRNA-2 5'-TTCTTTAGAGACGCGGGCTC-3', according to the specificity scores50, the predicted efficiency10, and the minimal off-targets.
- Order and synthesize the single-stranded DNA oligonucleotides (ssODNs). Dilute the ssODNs in ddH2O to a final concentration of 100 µM. To phosphorylate and anneal the sgRNA oligos, add 1 µL of forward oligo, 1 µL of reverse oligo, 0.5 µL of T4 polynucleotide kinase, 1 µL of T4 polynucleotide kinase buffer, and 6.5 µL of RNase-free water in a polymerase chain reaction (PCR) tube, and incubate the tube at 37 °C for 30 min, 95 °C for 5 min; then, ramp down to 25 °C at 5 °C min-1.
- Digest the pX458 plasmid using the Bbs
 restriction enzyme. Add 1 µg of the pX458 plasmid, 2 µL of 10x buffer G, 1 µL of Bbs
restriction enzyme. Add 1 µg of the pX458 plasmid, 2 µL of 10x buffer G, 1 µL of Bbs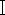 , and add RNase-free ddH2O to 20 µL in a 1.5 mL centrifuge tube. Incubate the tube at 37 °C for 2 h. Then, purify the linear plasmid using the column DNA gel extraction kit according to the manufacturer's protocols.
, and add RNase-free ddH2O to 20 µL in a 1.5 mL centrifuge tube. Incubate the tube at 37 °C for 2 h. Then, purify the linear plasmid using the column DNA gel extraction kit according to the manufacturer's protocols. - Ligate the sgRNA oligos into the pX458 plasmid (pSpCas9(BB)-2A-green fluorescent protein (GFP) plasmid, Addgene ID. 48138). Add 1 µL of the annealed oligonucleotides, 25 ng of the linear plasmid, 1 µL of 10x T4 DNA ligation buffer, 0.5 µL of T4 DNA ligase (350 U/µL) and make up to 10 µL with ddH2O in a 1.5 mL centrifuge tube. Incubate the ligation solution at 16 °C for 2 h.
- Incubate 10 µL of the constructed plasmid with the competent DH5α cells on ice for 30 min, heat shock at 42 °C for 45 s, and immediately place them on ice for 5 min. Add 500 µL of Luria-Bertani (LB) medium and seed the cells on an LB plate containing ampicillin (100 µg/mL). Incubate it at 37 °C for 16 h and screen the resistant clones.
- Select 5-10 single colonies with a sterilized pipette tip and transfer them to a 1 mL culture of LB medium with ampicillin (100 µg/mL). Incubate the culture at 37 °C and shake it at 180 rpm for 12-16 h.
- Isolate the plasmid DNA using a plasmid extraction kit according to the manufacturer's protocols. Validate the plasmid DNA of each colony by Sanger sequencing from the U6 promoter site using the U6-Fwd primer 5'-GAGGGCCTATTTCCCATGATTCC-3'.
- Extract and purify the plasmids using the endo-free plasmid DNA kit according to the manufacturer's protocols.
2. Transfection, isolation, and screening of the CENP-E knockout HeLa cells
- Culture HeLa cells in Dulbecco Modified Eagle's Medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin in a humidified incubator at 37 °C with 5% CO2.
- Incubate the HeLa cells using 0.25% trypsin/ethylene diamine tetraacetic acid (EDTA) at 37 °C for 2-3 min, dissociate the cells by pipetting gently, and then, seed the cells in new plates at a dilution ratio of 1:4 for cell passage.
- Seed the cells in a 12-well plate, and culture to 60-70% confluency before transfection. Transfect the pX458-sgRNA plasmids into HeLa cells using the referenced reagents according to the manufacturer's protocols (see the Table of Materials).
- Gently mix 1 µg of validated pX458-sgRNA plasmid and 50 µL of reduced serum medium in tube A. Then, gently mix 2 µL of the transfection reagent and 50 µL of reduced serum medium in tube B. Incubate tubes A and B separately at room temperature for 5 min.
- Mix both tube A and tube B gently and incubate at room temperature for 5 min. Add the mixture to a 12-well plate, incubate the cells for 6 h, and replace the medium with a fresh DMEM medium.
- Examine HeLa cells after 24 h or 48 h using a fluorescence microscope for transfection efficiency. After transfection for 48 h, completely dissociate the transfected HeLa cells using 0.25% trypsin/EDTA at 37 °C for 3-5 min, count the number of cells using a Neubauer chamber or an automated cell counter, and plate the cells into three separate 96-well plates for each transfected population according to serial dilution methods10.
- Return the cells to a humidified incubator and then culture them for another 1-2 weeks. Change the culture medium once every 3 days. For the phenotype-based screening and validation of CENP-E knockout, dissociate and expand the cells in 24-well or 12-well plates for 5-7 days.
- Screen the mutant cells with smaller cell colony diameters using an inverted microscope equipped with 10x and 20x objective lenses.
NOTE: CENP-E mutations usually result in a significant increase in the number of dividing cells in cell colonies, which can be one of the key indicators for screening the CENP-E mutant cells. - Harvest the cells for DNA extraction using the column animal genomic DNA extraction kit according to the manufacturer's protocols. Set up the PCR reactions as follows: 0.25 µL of Taq polymerase, 5 µL of 10x buffer (Mg2+ plus), 4 µL of dNTP mixtures, 500 ng of DNA template, 1 µL of forward oligo, 1 µL of reverse oligo, and adjust ddH2O to 50 µL. Use the following PCR program settings for gene amplification: 98 °C, 10 s; 98 °C, 10 s, 55 °C, 30 s, 72 °C, 60 s for 33 cycles; 72 °C, 10 min, and then hold at 4 °C.
NOTE: The specific primers for cloning of the target locus are listed as follows: CENP-E target F1, 5'-GAGGGTCCTGGCCATTTTCCTG-3'; CENP-E target R1, 5'-AGATCTCCGATCCTCCCCTGTC-3'; CENP-E target F2, 5'-TGGTAACTGCATTTTGGTGTTCTAC-3'; CENP-E target R2, 5'-CCTGTTGCAACGTGAGGGAAG-3'. - Ligate the target DNA into the pMD18-T vector according to the manufacturer's protocols. Transfect the ligated plasmid into competent DH5α cells and culture for selection of clones.
- Perform Sanger sequencing to determine the types of CENP-E knockout. Isolate the plasmid DNA using a plasmid extraction kit according to the manufacturer's protocols. Carry out Sanger sequencing of the plasmid DNA of each colony using the M13 forward and reverse primers. M13F primer, 5'-TGTAAAACGACGGCCAGT-3'; M13R primer, 5'-CAGGAAACAGCTATGACC-3'.
- Seed the wild-type and CENP-E mutant HeLa cells on 12 mm glass coverslips in a 24-well plate, respectively. Remove the complete DMEM medium and fix the cells in 4% paraformaldehyde/phosphate-buffered saline (PBS) fixative solution at room temperature for 10 min.
- Stain the nuclei with the 4',6-diamidino-2-phenylindole (DAPI) solution at room temperature for 5 min. Screen and validate the CENP-E mutant cells based on the phenotype of chromosome alignment using a fluorescence microscope equipped with a Plan Fluor 40x/ Numerical Aperture (NA) 0.75 objective.
- Screen and validate the CENP-E knockout cells using immunofluorescence staining and analysis (see section 3) of CENP-E proteins.
3. Immunofluorescence staining and high-resolution confocal microscopy
- Fix the cells with 4% paraformaldehyde/PBS fixative solution at room temperature for 10 min and immerse for 2 x 5 min in 1x PBS.
NOTE: Ensure that the cells are in healthy condition and cells are collected and fixed gently to avoid metaphase cell detachment. - Permeabilize the cells with 0.25% Triton X-100/PBS at room temperature for 10 min and immerse them in 1x PBS for 2 x 5 min.
- Block the cells with 1% BSA/PBST (0.1% Tween 20 in PBS) at room temperature for 1 h. Dilute the primary antibodies with 1% BSA/PBST and incubate the samples at 4 °C for 12 h.
- Discard the primary antibody solutions, rinse the cells 3 x 5 min with 1x PBS, and incubate the cells with diluted secondary antibodies at room temperature for 2 h.
- Discard the secondary antibodies and rinse the cells in 1x PBS for 3 x 5 min.
- Stain the nuclei with DAPI at room temperature for 5 min. Mount the slides with the mounting medium. Seal the slides with nail polish.
- Observe and record the fluorescence images using a scanning confocal microscope equipped with a 63x/NA 1.40 objective.
4. Chromosome preparation and karyotype analysis
- Culture the wild-type and CENP-E-/- HeLa cells in complete DMEM medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin for 24 h and then, incubate the wild-type and CENP-E-/- HeLa cells with 300 nM colchicine for 5 h.
- Incubate the cells with 0.25% trypsin/EDTA at 37 °C for 3 min and collect the cells into 1.5 mL centrifuge tubes.
- Centrifuge the cells at 1,000 × g for 5 min at room temperature, discard the supernatants, add 1.2 mL of 0.075 mol/L KCl solution, and incubate at 37 °C for 20 min.
- Centrifuge the cells at 1,000 × g for 5 min at room temperature, discard the supernatants, add 0.2 mL of the fixative solution (methanol: glacial acetic acid = 3:1) for prefixation, and mix gently for 1 min.
- Centrifuge the cells at 1,000 × g for 5 min at room temperature, collect the pellets, add 1.5 mL of the fixative solution at room temperature for 10 min, and then centrifuge at 1,000 × g for 5 min at room temperature.
- Discard the supernatants, add 0.6 mL of the fixative solutions, and prepare a cell suspension. Add 3-5 drops of the cell suspensions from a height of 35-40 cm on the ice slides.
NOTE: Keep the height for releasing the cell suspensions onto the slides at 35-40 cm; otherwise, the chromosomes may be too dispersed or not separated from each other. - Dry the slides immediately with an alcohol lamp, stain the samples with 10% Giemsa staining solution for 7 min, rinse the slides with running water for 2 min, and observe the samples using a light microscope equipped with a Plan Fluor 40x/NA 0.75 objective.
5. Cell colony formation assay
- Prepare the cell suspensions in the 6-well plates at a cell density of 1,000-2,000 cells/well. Gently shake the plates to evenly distribute the cells.
- Place the 6-well plates into a CO2 incubator and incubate for 2-3 weeks at 37 °C with 5% CO2. Change culture medium once every 5 days. Record the images using an inverted microscope equipped with 10x and 20x objective lenses. Harvest the cells when colonies are visible (hundreds of cells within a clone).
- To study the effects of GSK923295, culture the cells in the 6-well plates for 24 h, and add 2 mL of GSK923295 at the final concentrations of 10, 25, 50, 100, and 200 nM.
NOTE: Do not move the culture dish at the early stage of clone formation to avoid cell shedding, which can form new clones and affect the screening of transfected cells. - Discard the culture medium and fix the cells with 1% PFA at room temperature for 10 min. Stain the colonies with 0.1% crystal violet staining solution at room temperature for 15 min. Rinse the samples with 1x PBS three times. Record the images of cell colonies and quantify the diameters of each colony using ImageJ software. Choose the Straight line selection tool, click Analyze, choose Measure, and record the length.
- Rinse the colonies with 1 mL of 10% acetic acid solution for 5 min. Transfer 200 µL of the supernatant solutions in a 96-well plate, and measure absorbance values using a microplate spectrophotometer at A600 nm.
6. Cell viability assay
- Seed the cells in the 24-well plates and culture the cells for 48 h to 80-90% density.
- Incubate the cells with 100 µL of 0.25% trypsin/EDTA at 37 °C for 3 min.
- Mix 400 µL of PBS with the cells with a pipette gun. Transfer 100 µL of the cell suspensions into the 96-well plate.
- Add 20 µL of MTS solution in each well with a final concentration of 317 µg/mL and mix gently according to the manufacturer's protocols. Incubate the samples at 37 °C for 1-3 h in a CO2 incubator.
- Record the absorbance values of each well at the absorbance peak of A490 nm using a microplate spectrophotometer. Use a reference wavelength of A630 nm to subtract the background contributed by cell debris and nonspecific absorbance (Cell viability = A490nm– A630 nm).
Representative Results
The CENP-E-/- HeLa cells were successfully generated using the CRISPR/Cas9 system (Figure 1). The timeline and critical experimental steps of this method are shown in Figure 1. First, we designed and synthesized the CENP-E-specific sgRNAs, annealed and ligated the sgRNAs into the pX458 plasmid, transfected the plasmid into HeLa cells, and cultured them for 48 h. The transfected cells were dissociated and seeded in a 96-well plate using serial dilutions (Figure 1A). Single colonies were selected and screened using three phenotype-based strategies. The mutation types and the CENP-E knockout cells were validated using PCR, Sanger sequencing, and immunofluorescence assays (Figure 1A). Exon 1 and exon 13 of the CENP-E gene were selected on chromosome 4 as the targeting sites (Figure 1B). The CRISPR/Cas9 system can induce DSBs proximal to the protospacer adjacent motif (PAM) sequences using the RuvC and HNH endonuclease domains, which trigger the NHEJ pathways and result in gene editing-small insertion, deletion, and substitution (Figure 1C). Due to these error-prone repair mechanisms, we obtained four gene knockout HeLa cell populations. The transfection efficiency of pX458-sgRNA was detected using an immunofluorescence assay (Figure 1D). We also extracted the genomic DNA of the control, target 1, and target 2 HeLa cells (Figure 1E), amplified the DNA of the target locus, and ligated them into pMD18-T for DNA sequencing and validation of the mutation types (Figure 1F). In summary, approximately 100 clones were screened and four stable CENP-E knockout HeLa cells were successfully generated, including the insertion of + 1 bp (position 28 in Clone 1 and Clone 2), the deletion of -7 bp (position 1102-1108 in Clone 3), and a deletion of -1 bp (position 1102 bp in Clone 4) (CENP-E coding sequence, GenBank Accession No. NM_001286734.2) (Figure 1G,H). These mutations both resulted in frameshift mutations and premature stops, which resulted in the complete deletion of the CENP-E gene in Clone 1/2 and two CENP-E mutants in Clone 3/4 HeLa cells.
To improve the efficiency and experimental success of the CENP-E knockout experiments, three phenotype-based screen strategies were developed. These were based on cell colony screening using phase contrast microscopy (Figure 2A), on chromosome alignment using DAPI staining (Figure 2B), and on the fluorescence intensities of the CENP-E proteins using the immunofluorescence assay (Figure 2C). The critical steps of the CRISPR/Cas9 system included the guide RNA cloning, transfection of pX458-sgRNA into cell lines, incubation for 48 h, cell dilution into a 96-well plate, cell harvest, screening, and validation using three phenotype-based strategies (Figure 2D). Immunofluorescence staining of the control and CENP-E-/- groups showed that the fluorescence intensities of the CENP-E proteins were completely knocked out in the CENP-E-/- Clone 1 cells and significantly decreased in CENP-E mutant Clone 3 cells (Figure 2E), which further demonstrates the effectiveness of the CRISPR/Cas9 mediated gene knockout of the CENP-E gene in HeLa cells. The ratios of metaphase cells with chromosome misalignment increased from 0.30 ± 0.21% in the control group to 4.15 ± 0.57% in the Clone 1 HeLa cells and to 5.85 ± 0.80% in the Clone 3 HeLa cells (Figure 2F). Meanwhile, the ratios of metaphase cells increased from 3.49 ± 0.47% in the control to 5.86 ± 0.57% CENP-E-/- Clone 1 cells and to 6.70 ± 0.77% in CENP-E mutant Clone 3 cells (Figure 2G). In addition, the ratio of interphase cells slightly increased in the Clone 1 and Clone 3 groups after CENP-E deletion compared with the control group (Figure 2H). Both the rates of prophase, anaphase, and telophase cells were slightly decreased after CENP-E deletion, whereas the rates of metaphase cells were increased after CENP-E deletion (Figure 2I). To further investigate the phenotypes of CENP-E deletion, we performed immunofluorescence staining of BubR1, a key protein of spindle assembly checkpoint, and found that the localization of BubR1 was abnormal and kinetochore accumulation of BubR1 was significantly influenced in the absence of CENP-E (Figure 2J–L).
To better understand the cell growth and proliferation capacity of the CENP-E knockout HeLa cells, colony formation assays and crystal violet staining were performed for four CENP-E-/- HeLa cells, including Clone 1, Clone 2, Clone 3, and Clone 4 cells (Figure 3A,B). CENP-E deletion results in a reduced clone formation ability, smaller colony diameter, and decreased cell numbers in each clone (Figure 3A–D), which suggests that CENP-E is essential for clone formation and cell proliferation. Furthermore, we utilized the 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt (MTS) method and performed cell viability assays, and found that compared with 1.05 ± 0.00 in the control group, cell viability values were decreased to 0.66 ± 0.01 and 0.79 ± 0.00 in the CENP-E-/- HeLa cells (Figure 3E). To further investigate the roles of CENP-E in chromosome stability, we performed chromosome preparation, the Giemsa staining, and karyotype analysis, and found a decrease in chromosome numbers from 67.09 ± 0.50 in the wild-type HeLa cells to 56.08 ± 0.6 – 61.68 ± 0.83 in the CENP-E-/- HeLa cells (Figure 3F,G). Together, these results indicate that CENP-E deletion significantly inhibits cell proliferation, colony formation, and cell viability, which suggests CENP-E is essential for cell growth, proliferation, and chromosomal stability.
In this study, the CENP-E knockout cell line has been developed as a valuable tool for the identification and validation of CENP-E-specific inhibitors. A useful approach to validate the specificity and toxicity of CENP-E inhibitors has been established (Figure 4). Using GSK92329524,25 as an example, we treated wild-type HeLa cells and CENP-E-/- Clone 1 HeLa cell lines with a series of concentrations of GSK923295 and further explored the effects of CENP-E inhibitors on these two HeLa cells using the colony formation assays and the immunofluorescence assays (Figure 4). The single cells inoculated in the 6-well plates were subjected to colony formation assays. Cells were treated with different concentrations of GSK923295 and cultured for 2-3 weeks and then harvested for further analysis (Figure 4A–D). After crystal violet staining, the colony diameters of wild-type HeLa cells and CENP-E-/- cells were measured using the ImageJ software. In wild-type HeLa cells, colony formation was significantly decreased after the treatment of the increasing concentrations of GSK923295. In addition, the diameters of colonies became smaller after CENP-E inhibition in the wild-type HeLa cells (Figure 4B–D). Notably, CENP-E knockout Clone 1 HeLa cells showed no significant decrease in clone diameters in the presence of different concentrations of GSK923295, which suggests that GSK923295 is a specific inhibitor with no significant off-target effects and no obvious toxic side effects at the concentration range of 10 nM to 10 µM.
Subsequently, the chromosomal alignment phenotype in the wild-type HeLa cells and the CENP-E-/- cells was examined using the immunofluorescence assay (Figure 4E). Quantitative analysis has shown that the ratio of metaphase cells significantly increased after the treatment of 50 nM, 100 nM, 400 nM, 2 µM, and 10 µM GSK923295 (Figure 4E,F). In addition, compared with the significant increase in misaligned chromosomes in wild-type HeLa cells, the CENP-E-/- Clone 1 HeLa cells were not affected by different concentrations of GSK923295. Taken together, this CENP-E-/- Clone 1 cell line is a specific type of GSK923295-resistant cell line, which does not respond to the CENP-E inhibitors due to CENP-E deletion and adaption, genetic redundancy34, and potential genetic compensation effects35,36,37. These findings demonstrate that the CENP-E-/- Clone 1 HeLa cell is a powerful and useful tool to validate the specificity, toxicity, and side effects of CENP-E inhibitors.
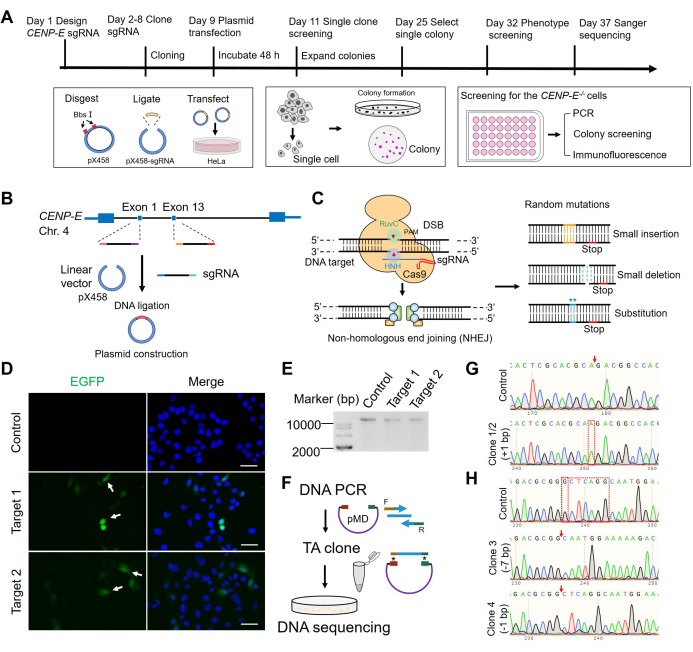
Figure 1: The construction of CENP-E-/- HeLa cells using the CRISPR/Cas9 system. (A) Protocol timeline for CRISPR/Cas9 vector construction, transfection, cell clone formation and selection, and functional validations. Timeline for the construction of the CENP-E-/- cell lines, including sgRNA design, construction of pX458-sgRNA plasmid, and the screening of mutant cells. (B) A schematic model of the cloning of sgRNA into the linearized pX458 vector. Exon 1 and exon 13 of the CENP-E gene in chromosome 4 were selected as the target sites. (C) Mechanisms of the CRISPR/Cas9 system for gene editing. DSB followed by non-homologous end joining generates three random mutations, including insertion, deletion, and substitution. (D) Representative fluorescence images of HeLa cells transfected with the pX458-sgRNA1 (Target 1) and pX458-sgRNA2 (Target 2). Scale bars = 50 µm. (E) Representative agarose gel electrophoresis of genomic DNA in the control, target 1, and target 2. (F–H) Sanger sequencing results of the target regions in the control, Clone 1, and Clone 3 groups. PCR amplification was carried out using the specific forward and reverse primers for the target locus in isolated Clone 1 and Clone 3 cells. The DNA fragments were cloned into pMD18-T vectors and transfected into DH5α E. coli for clone formation and Sanger sequencing. Representative sequencing results are shown. In Clone 1 HeLa cells, there was an insertion of + 1 bp at 28 bp. In Clone 2 HeLa cells, there was also an insertion of + 1 bp at 28 bp. In Clone 3, there was a deletion of -7 bp at 1,102-1,108 bp. In Clone 4, there was a deletion of -1 bp at 1,102 bp. These two mutations both result in a frameshift mutation and a premature stop in the CENP-E proteins. Please click here to view a larger version of this figure.
Figure 2: The construction and phenotype-based screening strategy of CRISPR/Cas9-mediated CENP-E knockout HeLa cell lines. (A) The screening strategy is based on cell colonies. Compared with the control group, the CENP-E knockout cells showed a significant increase in dividing cells. Scale bar = 50 µm. (B) The screening strategy is based on chromosome alignment. Compared with the control group, the CENP-E knockout cells showed an increase in chromosome misalignment. Several chromosomes were located proximal to the spindle pole after CENP-E deletion. DAPI, blue. Scale bar = 10 µm. (C) The screening strategy is based on fluorescence intensities of CENP-E proteins. CENP-E, green. Scale bar = 10 µm. (D) The construction of the CRISPR/Cas9 system. The CENP-E-specific sgRNA was ligated into the pX458 plasmid and sequenced for validation. The pX458-sgRNA plasmid was transfected into HeLa cells using a transfection reagent and cultured for 48 h. Cells were lysed using 0.25% trypsin/EDTA, seeded in the 96-well plate, and cultured for the formation of cell colonies. (E) Representative immunofluorescence images of CENP-E and α-tubulin in the control and CENP-E-/- groups. DAPI, blue; CENP-E, green; α-tubulin, red. Scale bar = 10 µm. (F) The ratios of metaphase cells with misalignment chromosomes in the control group and CENP-E-/- groups. Control, 0.30 ± 0.21%, n = 9; Clone 1, 4.15 ± 0.57%, n = 9; Clone 3, 5.85 ± 0.80%, n = 9. (G) The ratios of metaphase cells in the control and CENP-E-/- groups. Control, 3.49 ± 0.47%, n = 9; Clone 1, 5.86 ± 0.57%, n = 9; Clone 2, 6.70 ± 0.77%, n = 9. (H) The ratios of interphase cells in the control and CENP-E-/- groups. Control, 70.08 ± 2.71%, n = 7; Clone 1, 86.77 ± 0.91%, n = 7; Clone 3, 87.67 ± 0.91%, n = 7. (I) Mitotic index analysis of the control and CENP-E-/- groups, including the ratios of the prophase, metaphase, anaphase, and telophase cells. (J) Representative immunofluorescence images of BubR1 and CENP-B in the control and CENP-E-/- groups. DAPI, blue; BubR1, green; CENP-B, red. Scale bar = 10 µm. (K) Line-scan analysis of BubR1 and CENP-B in the control and CENP-E-/- groups. The X-axis indicates the relative distance. The Y-axis indicates the fluorescence intensity. (L) Relative fluorescence intensities of BubR1 (fluorescence intensities of BubR1 at the kinetochores/fluorescence intensities of BubR1 in the cytoplasm) in the control and CENP-E-/- groups. Control, 1.44 ± 0.10, n = 16; Clone 1, 0.98 ± 0.07, n = 18. For all graphs, mean ± SEM was shown. In Figure 2F–I, ANOVA Dunnett's multiple comparisons tests. ns, p > 0.05; **, p < 0.01; ***, p < 0.001; ****, p < 0.0001. In Figure 2L, the unpaired Student's t-test. ***, p < 0.001. Please click here to view a larger version of this figure.
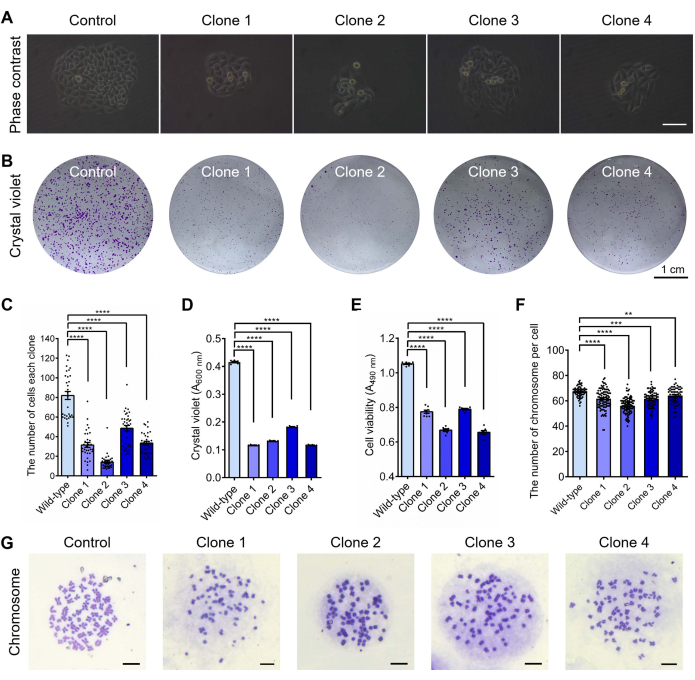
Figure 3: Cell growth assays of CENP-E knockout HeLa cells, including cell colony formation, cell viability assay, and karyotype analysis. (A) Representative images of the cell colony in the control and CENP-E-/- groups. Scale bar = 50 µm. (B) Representative crystal violet staining of cell colony in the control and CENP-E-/- groups. Scale bar = 1 cm. (C) The number of cells per clone in the wild-type (Control) and CENP-E-/- groups. Wild-type, 81.97 ± 4.09, n = 32; Clone 1, 31.52 ± 2.51, n = 33; Clone 2, 13.97 ± 1.31, n = 33. Clone 3, 48.48 ± 3.01, n = 33; Clone 4, 33.39 ± 1.87, n = 33. (D) Relative cell viability of the cell colony was measured by crystal violet absorbance at A600 nm. Wild-type, 0.42 ± 0.00, n = 6; Clone 1, 0.12 ± 0.00, n = 6; Clone 2, 0.13 ± 0.00, n = 6. Clone 3, 0.18 ± 0.00, n = 6; Clone 4, 0.12 ± 0.00, n = 6. (E) The cell viability of the wild-type and CENP-E-/- groups using the MTS and cell viability assay. Wild-type, 1.05 ± 0.00, n = 8; Clone 1, 0.77 ± 0.01, n = 8; Clone 2, 0.67 ± 0.01, n = 7. Clone 3, 0.79 ± 0.00, n = 7; Clone 4, 0.66 ± 0.01, n = 7. (F) The chromosome numbers per cell in the wild-type and CENP-E-/- groups. Wild-type, 67.09 ± 0.50, n = 66; Clone 1, 61.68 ± 0.83, n = 105; Clone 2, 56.08 ± 0.6, n = 106. Clone 3, 61.28 ± 0.63, n = 90; Clone 4, 64.18 ± 0.83, n = 61. (G) Representative images of the chromosome spread in the control group and CENP-E-/- group. Scale bar = 10 µm. For all graphs, ANOVA Dunnett's multiple comparisons tests. Error bars, mean ± SEM. **, p < 0.01; ***, p < 0.001; ****, p < 0.0001. Please click here to view a larger version of this figure.
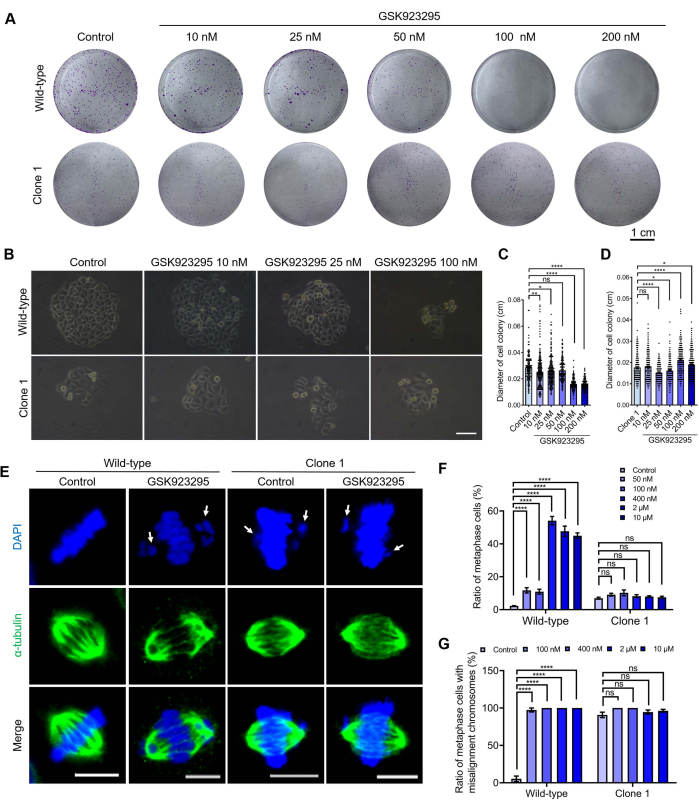
Figure 4: Using CENP-E knockout HeLa cells to validate the specificity and toxicity of the CENP-E inhibitors. (A) Representative crystal violet staining of the control and CENP-E-/- groups after the treatment of different concentrations of GSK923295. Scale bar = 1 cm. (B) Representative images of the cell colony in the control and CENP-E-/- groups after treatment with different concentrations of GSK923295. Scale bar = 50 µm. (C) The diameters of cell colonies in the control group after the treatment of different concentrations of GSK923295. Control, 28.68 ± 0.85 µm, n = 149; 10 nM, 25.40 ± 0.86 µm, n = 223; 25 nM, 26.07 ± 0.68 µm, n = 255; 50 nM, 26.41 ± 0.60 µm, n = 180; 100 nM, 16.02 ± 0.32 µm, n = 185; 200 nM, 15.61 ± 0.29 µm, n = 185. (D) The diameters of cell colonies in the CENP-E-/- groups after the treatment of different concentrations of GSK923295. Clone 1, 17.64 ± 0.40 µm, n = 230; 10 nM, 17.98 ± 0.39 µm, n = 230; 25 nM, 15.03 ± 0.26 µm, n = 230; 50 nM, 16.14 ± 0.32 µm, n = 230; 100 nM, 21.13 ± 0.42 µm, n = 230; 200 nM, 19.00 ± 0.40 cm, n = 230. (E) Representative immunofluorescence images of α-tubulin in the control and CENP-E-/- groups after treatment with different concentrations of GSK923295, DAPI, blue. α-tubulin, green. Scale bar = 10 µm. (F) The ratios of metaphase cells in the control and CENP-E-/- group after treatment with different concentrations of GSK923295. (G) The ratios of metaphase cells with misalignment chromosomes in the control and CENP-E-/- groups after treatment with different concentrations of GSK923295. For all graphs, ANOVA Dunnett's multiple comparisons tests. Error bars, mean ± SEM. ns, p > 0.05; *, p < 0.05; **, p < 0.01; ****, p < 0.0001. Please click here to view a larger version of this figure.
Discussion
Kinesin-7 CENP-E is a key regulator in chromosome alignment and spindle assembly checkpoint during cell division17,19,20. Genetic deletion of CENP-E usually results in the activation of spindle assembly checkpoint, cell cycle arrest, and cell death27,29,51,52. Thus, the construction of stable CENP-E knockout cell lines is an important unresolved issue in CENP-E biology. In this study, the CENP-E knockout HeLa cells have been successfully generated using the CRISPR/Cas9 system. This study has established three optimized phenotype-based screening strategies – cell colony screening, chromosome alignment phenotypes, and the fluorescent intensities of CENP-E proteins – which significantly improve the efficiency and success rate of the CENP-E knockout cells.
There are several crucial steps in this study. First, designing optimized sgRNA is essential to minimize off-target effects. Second, the transfection of the CRISPR plasmids into cultured cells through lipid-based reagents, electroporation, or lentivirus transfection can improve transfection efficiency and gene knockout cell ratios. In addition, the applications of fluorescence-activated cell sorting53,54,55 and puromycin selection56,57 would accelerate the screen speed and shorten the experimental period. Third, the validation of gene knockout phenotypes can be fulfilled by PCR, western blot, immunofluorescence assays, and genomic DNA sequencing. Fourth, the functional analysis of CENP-E knockout phenotypes can be studied using a variety of assays, including cell proliferation assays, immunofluorescence, cell cycle analyses, and high-resolution imaging.
Compared with traditional genome editing technologies, such as ZFNs and TALENs, the CRISPR/Cas9 system exhibits several advantages, including simplicity, high efficiency, ease of customization, versatility, and multiplex capability10. Moreover, the CRISPR/Cas9 system can be scaled up for high-throughput screening and large-scale engineering projects58,59. The CRISPR/Cas9 system has also been adapted as an efficient gene editing technology that enables one-step mutations in multiple genes60, which accelerates the studies of functionally redundant genes, interacting genes, and polygenic genetic diseases. In addition, the CRISPR/Cas9 system can be improved in its efficiency and versatility8. The high-fidelity HDR pathway, an alternative major DNA repair pathway, can be used to mediate the precise gene editing at the target locus in dividing cells in the future10. Moreover, co-transfection of multiple sgRNA can result in the large deletion and complete knockout of CENP-E in cells, which improves the stability and reliability of CENP-E knockout cells.
A key limitation of the CRISPR/Cas9 system is that SpCas9 is prone to off-target mutagenesis, which occasionally targets the unintended sites in the genome and leads to off-target effects. This can be minimized by improved guide RNA design, off-target prediction algorithms, and whole-genome sequencing10,59. Several approaches, including the use of alternative Cas12a or Cas13 proteins53, the use of base editors61, and the delivery of Cas9 proteins and guide RNA12, have been developed to reduce off-target effects. The second limitation is that the widely used Streptococcus pyogenes Cas9 (SpCas9) requires a requisite 5'-NGG PAM for target loci recognition, which limits the selection of the target sites of SpCas9 in genomes. The development of engineered SpCas9 variants that recognize the relaxed or diverse PAMs could increase the target range of Cas9 in the CRISPR/Cas9 genome engineering toolbox62,63,64. In addition, in several cases, the CRISPR/Cas9 system leads to mosaicism and results in different edits in cell populations65. Some animals may stimulate an immune response to the Cas9 proteins, which limits its applications in certain species12,66. Despite these limitations, the CRISPR/Cas9 system remains a powerful gene editing technology for genome editing and studying gene functions in basic science, biotechnology, gene therapy, and medicine.
The interaction modes and molecular mechanisms of CENP-E and its inhibitors are important research fields in cell biology and cancer research. Site-directed mutagenesis and molecular biology studies have revealed that GSK923295 interacts with Ile182 and Thr183 and with the CENP-E proteins, being sandwiched between helices α2 and α3 and near loop 525. In this study, the CENP-E knockout HeLa cell line has been established as a valuable tool for the validation of the specificity and toxicity of CENP-E inhibitors. To date, the binding sites and mechanisms of most CENP-E inhibitors are not well understood. The combinations of molecular mass spectrometry imaging67, nuclear magnetic resonance spectroscopy68, optical tweezers69, molecular structure modifications, X-ray crystallography70, and cryo-electron microscopy71,72 would contribute to the elucidation of the interaction sites and mechanisms of CENP-E inhibition. In the future, homology-directed repair-mediated target modification of CENP-E at specific sites can generate the CENP-E gene editing cells, which would help to reveal the specific binding sites and mechanisms of CENP-E-specific inhibitors.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
We thank all members of the Cytoskeleton Laboratory at Fujian Medical University for helpful discussions. We thank Jun-Jin Lin, Zhi-Hong Huang, Ling Lin, Li-Li Pang, Lin-Ying Zhou, Xi Lin, and Min-Xia Wu at Public Technology Service Center, Fujian Medical University for their technical assistance. We thank Si-Yi Zheng, Ying Lin, and Qi Ke at the Experimental Teaching Center of Basic Medical Sciences at Fujian Medical University for their support. This study was supported by the following grants: National Natural Science Foundation of China (grant number 82001608 and 82101678), Natural Science Foundation of Fujian Province, China (grant number 2019J05071), the Joint Funds for the Innovation of Science and Technology, the Fujian Province, China (grant number 2021Y9160), and Fujian Medical University high-level talents scientific research start-up funding project (grant number XRCZX2017025).
Materials
| 0.25% Trypsin-EDTA | Gibco | 25200056 | |
| 1.5 mL centrifuge tube | Axygen | MCT-150-C | |
| 24-well plate | Corning | 3524 | |
| 4S Gelred, 10,000x in water | Sangon Biotech (Shanghai) | A616697 | |
| 50 mL centrifuge tube | Corning | 430828 | |
| 6 cm Petri dish | Corning | 430166 | |
| 95% ethanol | Sinopharm Chemical Reagent | 10009164 | |
| 96-well plate | Corning | 3599 | |
| Acetic acid | Sinopharm Chemical Reagent | 10000218 | Dissolve in H2O to prepare a 10% working solution. |
| Agarose | Sangon Biotech (Shanghai) | A620014 | |
| Alexa Fluor 488-labeled Goat Anti-Mouse IgG(H+L) | Beyotime | A0428 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:500 dilution. |
| Alexa Fluor 488-labeled Goat Anti-Rabbit IgG(H+L) | Beyotime | A0423 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:500 dilution. |
| Alexa Fluor 555-labeled Donkey Anti-Mouse IgG(H+L) | Beyotime | A0460 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:500 dilution. |
| Anhydrous ethanol | Sinopharm Chemical Reagent | 100092690 | |
| Anti-BubR1 rabbit monoclonal antibody | Abcam | ab254326 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:100 dilution |
| Anti-CENP-B mouse monoclonal antibody | Santa Cruz Biotechnology | sc-376392 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:50 dilution. |
| Anti-CENP-E rabbit monoclonal antibody | Abcam | ab133583 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:100 dilution. |
| Anti-fade mounting medium | Beyotime | P0131 | Slowing down the quenching of fluorescent signals. |
| Anti-α-tubulin mouse monoclonal antibody | Abcam | ab7291 | For immunofluorescence. Dissolve in 1% BSA/PBST. 1:100 dilution. |
| Biotek Epoch Microplate Spectrophotometer | Biotek Instruments | Biotek Epoch | |
| Bovine Serum Albumin (BSA) | Sinopharm Chemical Reagent | 69003435 | |
| BpiI (BbsI) | Thermo Fisher Scientific | ER1011 | |
| CellTiter 96 aqueous one solution cell proliferation assay | Promega | G3580 | |
| Centrifuge | Eppendorf | 5424BK745380 | |
| Colchicine | Sinopharm Chemical Reagent | 61001563 | |
| Confocal scanning microscope | Leica | Leica TCS SP8 | |
| Coverslip | CITOTEST | 80344-1220 | |
| DAPI | Beyotime | C1006 | |
| DH5α competent cells | Sangon Biotech (Shanghai) | B528413 | |
| DL2000 DNA marker | TaKaRa | 3427A | |
| Dulbecco's Modified Eagle Medium (DMEM) | Gibco | C11995500BT | |
Endo-free plasmid mini kit  |
Omega | D6950 | |
| Ezup Column Animal Genomic DNA Purification Kit | Sangon Biotech (Shanghai) | B518251 | |
| Fetal bovine serum | Zhejiang Tianhang Biotechnology | 11011-8611 | |
| Gentian violet | Sinopharm Chemical Reagent | 71019944 | Dissolve in PBS to prepare 0.1% gentian violet/PBS. |
| Giemsa staining solution | Sinopharm Chemical Reagent | 71020260 | |
| GraphPad Prism version 8.0 software | GraphPad | www.graphpad.com | Statistical analysis. |
| GSK923295 | MedChemExpress | HY-10299 | |
| HeLa cell line | ATCC | CCL-2 | |
| Humidified incubator | Heal Force | HF90/HF240 | |
| Image J software | National Institutes of Health | https://imagej.nih.gov/ij/ | Image processing and analysis. |
| Inverted microscope | Nanjing Jiangnan Novel Optics | XD-202 | |
| LB agar powder | Sangon Biotech (Shanghai) | A507003 | |
| Lipo6000 transfection reagent | Beyotime | C0526 | |
| Nikon Ti-S2 microscope | Nikon | Ti-S2 | |
| Opti-MEM reduced serum medium | Gibco | 31985070 | |
| Paraformaldehyde | Sinopharm Chemical Reagent | 80096618 | Dissolve in PBS to prepare 4% paraformaldehyde/PBS. |
| Penicillin-streptomycin solution | HyClone | SV30010 | |
| SanPrep column DNA gel extraction kit | Sangon Biotech (Shanghai) | B518131 | |
| SanPrep column plasmid mini-preps kit | Sangon Biotech (Shanghai) | B518191 | |
| T4 DNA ligase | TaKaRa | 2011A | |
| T4 polynucleotide kinase | TaKaRa | 2021A | |
| TaKaRa Ex Taq | TaKaRa | RR001A | |
| Triton X-100 | Sinopharm Chemical Reagent | 30188928 | Dissolve in PBS to prepare 0.25% Triton X-100/PBS. |
| Tween 20 | Sinopharm Chemical Reagent | 30189328 | Dissolve in PBS to prepare 0.1% Tween 20/PBS. |
Referencias
- Jiang, W., Bikard, D., Cox, D., Zhang, F., Marraffini, L. A. RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nature Biotechnology. 31 (3), 233-239 (2013).
- Bibikova, M., Beumer, K., Trautman, J. K., Carroll, D. Enhancing gene targeting with designed zinc finger nucleases. Science. 300 (5620), 764 (2003).
- Urnov, F. D., Rebar, E. J., Holmes, M. C., Zhang, H. S., Gregory, P. D. Genome editing with engineered zinc finger nucleases. Nature Reviews Genetics. 11 (9), 636-646 (2010).
- Joung, J. K., Sander, J. D. TALENs: a widely applicable technology for targeted genome editing. Nature Reviews Molecular Cell Biology. 14 (1), 49-55 (2013).
- Wood, A. J., et al. Targeted genome editing across species using ZFNs and TALENs. Science. 333 (6040), 307 (2011).
- Stoddard, B. L. Homing endonuclease structure and function. Quarterly Reviews of Biophysics. 38 (1), 49-95 (2005).
- Pâques, F., Duchateau, P. Meganucleases and DNA double-strand break-induced recombination: perspectives for gene therapy. Current Gene Therapy. 7 (1), 49-66 (2007).
- Cong, L., et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 339 (6121), 819-823 (2013).
- Shmakov, S., et al. Discovery and functional characterization of diverse class 2 CRISPR-Cas systems. Molecular Cell. 60 (3), 385-397 (2015).
- Ran, F. A., et al. Genome engineering using the CRISPR-Cas9 system. Nature Protocols. 8 (11), 2281-2308 (2013).
- Jia, N., Patel, D. J. Structure-based functional mechanisms and biotechnology applications of anti-CRISPR proteins. Nature Reviews Molecular Cell Biology. 22 (8), 563-579 (2021).
- Hsu, P. D., Lander, E. S., Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 157 (6), 1262-1278 (2014).
- Horvath, P., Barrangou, R. CRISPR/Cas, the immune system of bacteria and archaea. Science. 327 (5962), 167-170 (2010).
- Gasiunas, G., Barrangou, R., Horvath, P., Siksnys, V. Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proceedings of the National Academy of Sciences of the United States of America. 109 (39), E2579-E2586 (2012).
- Jinek, M., et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 337 (6096), 816-821 (2012).
- Jackson, S. A., et al. CRISPR-Cas: Adapting to change. Science. 356 (6333), eaal5056 (2017).
- Wood, K. W., Sakowicz, R., Goldstein, L. S., Cleveland, D. W. CENP-E is a plus end-directed kinetochore motor required for metaphase chromosome alignment. Cell. 91 (3), 357-366 (1997).
- Craske, B., Welburn, J. P. I. Leaving no-one behind: how CENP-E facilitates chromosome alignment. Essays in Biochemistry. 64 (2), 313-324 (2020).
- Yu, K. W., Zhong, N., Xiao, Y., She, Z. Y. Mechanisms of kinesin-7 CENP-E in kinetochore-microtubule capture and chromosome alignment during cell division. Biology of the Cell. 111 (6), 143-160 (2019).
- Schaar, B. T., Chan, G. K., Maddox, P., Salmon, E. D., Yen, T. J. CENP-E function at kinetochores is essential for chromosome alignment. Journal of Cell Biology. 139 (6), 1373-1382 (1997).
- McEwen, B. F., et al. CENP-E is essential for reliable bioriented spindle attachment, but chromosome alignment can be achieved via redundant mechanisms in mammalian cells. Molecular Biology of the Cell. 12 (9), 2776-2789 (2001).
- Yao, X., Abrieu, A., Zheng, Y., Sullivan, K. F., Cleveland, D. W. CENP-E forms a link between attachment of spindle microtubules to kinetochores and the mitotic checkpoint. Nature Cell Biology. 2 (8), 484-491 (2000).
- Kim, Y., Heuser, J. E., Waterman, C. M., Cleveland, D. W. CENP-E combines a slow, processive motor and a flexible coiled coil to produce an essential motile kinetochore tether. Journal of Cell Biology. 181 (3), 411-419 (2008).
- Qian, X., et al. Discovery of the first potent and selective inhibitor of Centromere-Associated Protein E: GSK923295. ACS Medicinal Chemistry Letters. 1 (1), 30-34 (2010).
- Wood, K. W., et al. Antitumor activity of an allosteric inhibitor of centromere-associated protein-E. Proceedings of the National Academy of Sciences of the United States of America. 107 (13), 5839-5844 (2010).
- Pisa, R., Phua, D. Y. Z., Kapoor, T. M. Distinct mechanisms of resistance to a CENP-E inhibitor emerge in near-haploid and diploid cancer cells. Cell Chemical Biology. 27 (7), 850.e6-857.e6 (2020).
- Putkey, F. R., et al. Unstable kinetochore-microtubule capture and chromosomal instability following deletion of CENP-E. Developmental Cell. 3 (3), 351-365 (2002).
- Weaver, B. A., Silk, A. D., Montagna, C., Verdier-Pinard, P., Cleveland, D. W. Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell. 11 (1), 25-36 (2007).
- Silk, A. D., et al. Chromosome missegregation rate predicts whether aneuploidy will promote or suppress tumors. Proceedings of the National Academy of Sciences of the United States of America. 110 (44), E4134-E4141 (2013).
- Guo, Y., Kim, C., Ahmad, S., Zhang, J., Mao, Y. CENP-E-dependent BubR1 autophosphorylation enhances chromosome alignment and the mitotic checkpoint. Journal of Cell Biology. 198 (2), 205-217 (2012).
- Weaver, B. A., Cleveland, D. W. Aneuploidy: instigator and inhibitor of tumorigenesis. Investigación sobre el cáncer. 67 (21), 10103-10105 (2007).
- Nishimura, K., Fukagawa, T. An efficient method to generate conditional knockout cell lines for essential genes by combination of auxin-inducible degron tag and CRISPR/Cas9. Chromosome Research. 25 (3-4), 253-260 (2017).
- Owa, M., Dynlacht, B. A non-canonical function for Centromere-associated protein-E controls centrosome integrity and orientation of cell division. Communications Biology. 4 (1), 358 (2021).
- Barabási, A. L., Oltvai, Z. N. Network biology: understanding the cell’s functional organization. Nature Reviews Genetics. 5 (2), 101-113 (2004).
- Rossi, A., et al. Genetic compensation induced by deleterious mutations but not gene knockdowns. Nature. 524 (7564), 230-233 (2015).
- Ma, Z., et al. PTC-bearing mRNA elicits a genetic compensation response via Upf3a and COMPASS components. Nature. 568 (7751), 259-263 (2019).
- El-Brolosy, M. A., et al. Genetic compensation triggered by mutant mRNA degradation. Nature. 568 (7751), 193-197 (2019).
- Kung, P. P., et al. Chemogenetic evaluation of the mitotic kinesin CENP-E reveals a critical role in triple-negative breast cancer. Molecular Cancer Therapeutics. 13 (8), 2104-2115 (2014).
- Kim, J. H., et al. Development of a novel HAC-based "gain of signal" quantitative assay for measuring chromosome instability (CIN) in cancer cells. Oncotarget. 7 (12), 14841-14856 (2016).
- Hirayama, T., et al. Synthetic studies of centromere-associated protein-E (CENP-E) inhibitors: 1. Exploration of fused bicyclic core scaffolds using electrostatic potential map. Bioorganic & Medicinal Chemistry. 21 (17), 5488-5502 (2013).
- Hirayama, T., et al. Synthetic studies on Centromere-associated protein-E (CENP-E) inhibitors: 2. Application of electrostatic potential map (EPM) and structure-based modeling to Imidazo[1,2-a]pyridine derivatives as anti-tumor agents. Journal of Medicinal Chemistry. 58 (20), 8036-8053 (2015).
- Ohashi, A., et al. A novel time-dependent CENP-E inhibitor with potent antitumor activity. PLoS One. 10 (12), e0144675 (2015).
- Ohashi, A., et al. Aneuploidy generates proteotoxic stress and DNA damage concurrently with p53-mediated post-mitotic apoptosis in SAC-impaired cells. Nature Communications. 6, 7668 (2015).
- Ding, X., et al. Probing CENP-E function in chromosome dynamics using small molecule inhibitor syntelin. Cell Research. 20 (12), 1386-1389 (2010).
- Liu, X., et al. Phase separation drives decision making in cell division. Journal of Biological Chemistry. 295 (39), 13419-13431 (2020).
- Henderson, M. C., et al. UA62784, a novel inhibitor of centromere protein E kinesin-like protein. Molecular Cancer Therapeutics. 8 (1), 36-44 (2009).
- Yamane, M., et al. Identification of benzo[d]pyrrolo[2,1-b]thiazole derivatives as CENP-E inhibitors. Biochemical and biophysical research communications. 519 (3), 505-511 (2019).
- Lock, R. B., et al. Initial testing of the CENP-E inhibitor GSK923295A by the pediatric preclinical testing program. Pediatric Blood & Cancer. 58 (6), 916-923 (2012).
- Chung, V., et al. First-time-in-human study of GSK923295, a novel antimitotic inhibitor of centromere-associated protein E (CENP-E), in patients with refractory cancer. Cancer Chemotherapy and Pharmacology. 69 (3), 733-741 (2012).
- Hsu, P. D., et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nature biotechnology. 31 (9), 827-832 (2013).
- Weaver, B. A., Cleveland, D. W. Decoding the links between mitosis, cancer, and chemotherapy: The mitotic checkpoint, adaptation, and cell death. Cancer Cell. 8 (1), 7-12 (2005).
- Manchado, E., Guillamot, M., Malumbres, M. Killing cells by targeting mitosis. Cell Death and Differentiation. 19 (3), 369-377 (2012).
- Dang, Y., et al. Optimizing sgRNA structure to improve CRISPR-Cas9 knockout efficiency. Genome Biology. 16, 280 (2015).
- Ratz, M., Testa, I., Hell, S. W., Jakobs, S. CRISPR/Cas9-mediated endogenous protein tagging for RESOLFT super-resolution microscopy of living human cells. Scientific Reports. 5, 9592 (2015).
- Koch, B., et al. Generation and validation of homozygous fluorescent knock-in cells using CRISPR-Cas9 genome editing. Nature Protocols. 13 (6), 1465-1487 (2018).
- Yan, Q., et al. Multiplex CRISPR/Cas9-based genome engineering enhanced by Drosha-mediated sgRNA-shRNA structure. Scientific Reports. 6, 38970 (2016).
- Cheng, Y., et al. CRISPR/Cas9-mediated chicken TBK1 gene knockout and its essential role in STING-mediated IFN-β induction in chicken cells. Frontiers in Immunology. 9, 3010 (2019).
- Zhou, Y., et al. High-throughput screening of a CRISPR/Cas9 library for functional genomics in human cells. Nature. 509 (7501), 487-491 (2014).
- Zeng, W., Guo, L., Xu, S., Chen, J., Zhou, J. High-throughput screening technology in industrial biotechnology. Trends in Biotechnology. 38 (8), 888-906 (2020).
- Tsai, S. Q., et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nature Biotechnology. 33 (2), 187-197 (2015).
- Kim, D., et al. Genome-wide analysis reveals specificities of Cpf1 endonucleases in human cells. Nature Biotechnology. 34 (8), 863-868 (2016).
- Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A., Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature. 533 (7603), 420-424 (2016).
- Nishimasu, H., et al. Engineered CRISPR-Cas9 nuclease with expanded targeting space. Science. 361 (6408), 1259-1262 (2018).
- Wang, H., et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell. 153 (4), 910-918 (2013).
- Ablain, J., Durand, E. M., Yang, S., Zhou, Y., Zon, L. I. A CRISPR/Cas9 vector system for tissue-specific gene disruption in zebrafish. Developmental Cell. 32 (6), 756-764 (2015).
- Pickar-Oliver, A., Gersbach, C. A. The next generation of CRISPR-Cas technologies and applications. Nature Reviews Molecular Cell Biology. 20 (8), 490-507 (2019).
- Nalawansha, D. A., Gomes, I. D., Wambua, M. K., Pflum, M. K. H. HDAC inhibitor-induced mitotic arrest is mediated by Eg5/KIF11 acetylation. Cell Chemical Biology. 24 (4), 481.e5-492.e5 (2017).
- Kavalapure, R. S., et al. Design, synthesis, and molecular docking study of some 2-((7-chloroquinolin-4-yl) amino) benzohydrazide Schiff bases as potential Eg5 inhibitory agents. Bioorganic Chemistry. 116, 105381 (2021).
- Calligaris, D., Lafitte, D. Chemical inhibitors: the challenge of finding the right target. Chemistry & Biology. 18 (5), 555-557 (2011).
- Łomzik, M., et al. Metal-dependent cytotoxic and kinesin spindle protein inhibitory activity of Ru, Os, Rh, and Ir half-sandwich complexes of Ispinesib-derived ligands. Inorganic Chemistry. 59 (20), 14879-14890 (2020).
- Ferro, L. S., et al. Structural and functional insight into regulation of kinesin-1 by microtubule-associated protein MAP7. Science (New York, N.Y.). 375 (6578), 326-331 (2022).
- Atherton, J., et al. The mechanism of kinesin inhibition by kinesin-binding protein. eLife. 9, e61481 (2020).

