Modeling Age-Associated Neurodegenerative Diseases in Caenorhabditis elegans
Summary
Here, we introduce and describe widely accessible methodologies utilizing some versatile nematode models, including hyperactivated ion channel-induced necrosis and protein aggregate-induced neurotoxicity, to monitor and dissect the cellular and molecular underpinnings of age-associated neurodegenerative diseases.
Abstract
Battling human neurodegenerative pathologies and managing their pervasive socioeconomic impact is becoming a global priority. Notwithstanding their detrimental effects on the human life quality and the healthcare system, the majority of human neurodegenerative disorders still remain incurable and non-preventable. Therefore, the development of novel therapeutic interventions against such maladies is becoming a pressing urgency. Age-associated deterioration of neuronal circuits and function is evolutionarily conserved in organisms as diverse as the lowly worm Caenorhabditis elegans and humans, signifying similarities in the underlying cellular and molecular mechanisms. C. elegans is a highly malleable genetic model, which offers a well-characterized nervous system, body transparency and a diverse repertoire of genetic and imaging techniques to assess neuronal activity and quality control during ageing. Here, we introduce and describe methodologies utilizing some versatile nematode models, including hyperactivated ion channel-induced necrosis (e.g., deg-3(d) and mec-4(d)) and protein aggregate (e.g., α-syunclein and poly-glutamate)-induced neurotoxicity, to monitor and dissect the cellular and molecular underpinnings of age-related neuronal breakdown. A combination of these animal neurodegeneration models, together with genetic and pharmacological screens for cell death modulators will lead to an unprecedented understanding of age-related breakdown of neuronal function and will provide critical insights with broad relevance to human health and quality of life.
Introduction
Over the last two decades, C. elegans has been widely used as a model organism to investigate the molecular mechanisms of necrotic cell death. C. elegans offers an exceptionally well-characterized and mapped nervous system, transparent body structure and a diverse repertoire of genetic and imaging methods to monitor in vivo cellular function and survival throughout ageing. Thus, several C. elegans genetic models of neurodegeneration have been already developed to assess neuronal viability. In particular, well-described and used nematode models include the hyperactive ion channel-induced necrosis1,2,3 and cell death triggered by increased protein aggregation4,5,6,7,8,9,10 and heat stroke11,12, among others.
Short-term exposure to sub-lethal temperatures conferred resistance against necrotic cell death, triggered by a subsequent heat stress both in nematodes and mammalian neurons11. Interestingly, daily preconditioning of nematodes at a mild elevated temperature protects against neurodegeneration, which is inflicted by diverse stimuli, such as ionic imbalance (e.g., mec-4(u231) and/or deg-3(u662)) and protein aggregation (e.g., α-synuclein and polyQ40)11,13.
Here, we describe versatile methodologies using C. elegans to monitor and evaluate age-dependent neurodegeneration in well-established models of human diseases, such as excitotoxicity-triggered cell death, Parkinson’s and Huntington’s disease. Moreover, we underline the neuroprotective role of heat preconditioning in several models of neurodegeneration. A combination of these techniques together with genetic and/or pharmacological screens will result in the identification and characterization of novel cell death modulators, with potential therapeutic interest.
Protocol
1. Necrotic cell death-induced by hyperactive ion channels
NOTE: Gain-of-function mutations in the gene family of degenerins, including mec-4 and deg-3 among others, results in the generation of hyperactive ion channels triggering necrotic cell death of six touch receptor neurons required for mechanosensation in worms3. Necrosis induced by the aberrant stimulation of degenerins displays several mechanistic and morphological similarities to excitotoxicity in mammals. The maintenance of energy metabolism and calcium homeostasis has a crucial role on neuronal survival during necrosis11. The following strains can be used to monitor necrotic cell death triggered by hyperactive ion channels, mec-4(u231)X and deg-3(u662)V.
- Maintenance, synchronization and preparation of mutant worms for examination of necrotic cell death
- Day 1: Pick L4 larvae of mec-4(u231) or deg-3(u662) mutant nematodes onto Nematode Growth Media (NGM; Table 1) plates seeded with Escherichia coli (OP50) using a dissecting stereomicroscope.
- Place 10 L4 nematodes per seeded NGM plate and grow them at the standard temperature of 20 °C.
- Day 5: Wash the plates with 1 mL of M9 buffer (Table 1) and collect the animals in a 1.5 mL tube.
- Centrifuge at 10,000 x g for 30 s and remove the supernatant.
- Add 0.5 mL of bleaching solution (7 mL of H2O, 1 mL of 5 N NaOH and 2 mL of bleach).
NOTE: Bleaching solution gradually lose its efficiency; hence it should be prepared daily. - Vortex and monitor periodically until the worms have dissolved.
NOTE: Avoid bleaching for periods longer than 5 min because the embryos’ viability will be affected. - Centrifuge at 10,000 x g for 30 s and remove the supernatant.
- Wash twice the pellet (eggs) with 1 mL of M9 buffer.
- After washing, resuspend the eggs in 200 µL of M9 buffer and incubate them for 25 min at 34 °C in a water bath.
- Maintain a separate group of control sample (eggs) at 20 °C.
- Pipette 100 µL of control or heat shock-treated eggs and place them on unseeded NGM plates. Each plate contains at least 100-200 eggs.
- Incubated the eggs at 20 °C until hatching.
- Mounting L1 larvae for examination using differential interference contrast (DIC; Nomarsky) microscopy
- Prepare 2% agarose pads.
- Use 1 mL of M9 buffer to wash the plates and collect L1 larvae nematodes in 1.5 mL tubes.
- Centrifuge at 10,000 x g for 30 s.
- Remove the supernatant and keep the pellet (nematodes).
- Add 100 µL of 20 mM M9/levamisole buffer to anesthetize the nematodes.
NOTE: Avoid sodium azide as an anesthetic. Sodium azide triggers mitochondrial damage and oxidative stress leading eventually to cell death induction - Pipette 10 µL of M9/levamisole buffer containing L1 worms and mount them on 2% agarose pads (Table 1).
- Gently place a coverslip on the top of the sample.
NOTE: Seal the coverslip with nail polish to preserve humidity throughout the process of imaging. - Observe worms using differential interference contrast (DIC; Nomarsky) microscopy.
- Score neurodegeneration of the six touch receptor neurons by counting cells with the characteristic vacuolated appearance per nematode.
2. Protein aggregate-induced neurodegeneration
NOTE: The following strains can be used to investigated protein aggregates-induced neurotoxicity: (A) overexpression of human α-synuclein in dopaminergic neurons, UA44: Is[baIn1; pdat-1α-syn, pdat-1GFP] and (B) overexpression of human polyglutamine protein (PolyQ) pan-neuronally, AM101: rmsIs110[prgef-1Q40::YFP]6,10.
- Maintenance, synchronization and preparation of transgenic nematodes for neurodegeneration assay
- Use a dissecting stereomicroscope to monitor C. elegans development and growth.
- Day 1: Synchronize the population of transgenic nematodes by picking and transferring 15-20 L4 larvae of each strain on freshly OP50-seeded NGM plates.
NOTE: Use at least three plates of each strain per condition. - Incubate and let the nematodes to grow at the standard temperature of 20 °C.
- Day 2: Perform daily preconditioning for 30 minutes by transferring the plates in an incubator set at 34 °C. Then, return the preconditioned nematodes back at the standard temperature of 20 °C.
NOTE: Different genetic backgrounds might be sensitive to high temperature for long periods exposure. - (A) If investigating overexpression of human α-synuclein in dopaminergic neurons, UA44: Is[baIn1; pdat-1α-syn, pdat-1GFP]: on Day 9, monitor 7-days-old transgenic nematodes for dopaminergic neuronal cell death.
- (B) If investigating overexpression of human polyglutamine protein (PolyQ) pan-neuronally, AM101: rmsIs110[prgef-1Q40::YFP]: on Day 5, measure neuronal PolyQ aggregates in the head region of 4-days-old transgenic animals expressing Q40::YFP.
- Mounting the samples for microscopic examination
- Prepare 2% agarose pads (Table 1).
- Add 10 µL of 20 mM M9/levamisole buffer drop at the center of the agarose pad.
- Pick the respective transgenic nematodes and transfer them in a M9/levamisole drop.
NOTE: Place 20-30 nematodes per drop. - Place gently a coverslip on the top of the sample.
NOTE: Seal the coverslip with nail polish to preserve humidity throughout the process of imaging. - Proceed to microscopic examination of the respective samples.
- Acquisition process and data analysis of transgenic nematodes co-expressing α-synuclein and cytosolic GFP in dopaminergic neurons
- Use an epifluorescence microscope combined with a camera (e.g., EVOS FL Auto 2).
- Detect and capture z-stack images of the head region at 20x magnification.
- Save and collect the maximum intensity projection images.
- Proceed to the analysis of the acquired images.
- Examine the transgenic worms for neurodegeneration by scoring the following cellular characteristics, (i) loss of fluorescence from neurons expressing GFP under the promoter of dat-1, (ii) neurons showing soma and/or axonal blebbing, outgrowths or dendritic loss.
- Import and analyze the data by using a software package (e.g., Excel).
3. Acquisition process and data analysis of transgenic nematodes expressing pan-neuronally PolyQ40 fused with YFP
- Use an epifluorescence microscope combined with a camera (e.g., EVOS FL Auto 2).
- Detect and capture z-stack images of the head region at 20x magnification.
- Save and collect the maximum intensity projection images.
- Proceed to the analysis of the acquired images by using Fiji software.
- Open images with Fiji program14.
- Select Split Channels command via the Image and Color drop-down menu.
NOTE: Keep the green channel image. - Use the freehand selection tool to manually set the fluorescent region of interest (ROI; e.g., head).
- Add the respective ROI in ROI Manager via Analyze and Tools drop-down menu.
- Subtract the background to 50% by selecting Process and Subtract Background.
- Set up and apply threshold values via the menu command Image | Adjust | Threshold. Keep and set the same threshold values throughout image analysis of the entire experiment.
- Select the respective ROI from the ROI Manager.
- Analyze the number of protein aggregates by using the menu command Analyze and Analyze Particles.
- Repeat steps 3.5-3.12 for each acquired image.
- Copy the display values from the separate Results window.
- Paste/Import and analyze the results by using a software package.
4. Report statistical analysis
- Use at least 30 nematodes for each experimental condition. Perform three biological replicates.
- Use statistical analysis software to either conduct student t-test (comparison between two groups) or ANOVA (comparison among multiple groups) for statistical analysis with p < 0.05 as significant.
Representative Results
Necrotic cell death-induced by hyperactive ion channels
Using the procedures presented here, mec-4(u231) and deg-3u662) mutant embryos were either incubated for 25 min at 34 °C or kept at the standard temperature of 20 °C. Upon hatching, the number of neuronal cell corpses was determined at the L1 larval stage of both groups. Necrotic cell death is diminished in nematodes that hatched from heat shock preconditioned eggs (Figure 1A-1B).
Protein aggregate-induced neurodegeneration
Transgenic nematodes overexpressing (A) human α-synuclein and cytoplasmic GFP in dopaminergic neurons and (B) human polyglutamine protein (PolyQ) fused with YFP pan-neuronally, were exposed daily for 30 min at 34 °C. Heat shock preconditioning promotes neuroprotection against α-synuclein-induced cell death in 7-day-old adult hermaphrodites (Figure 2A) and decreases Q40::YFP protein aggregates in the head region of 4-day-old adults (Figure 2B and Figure 3).
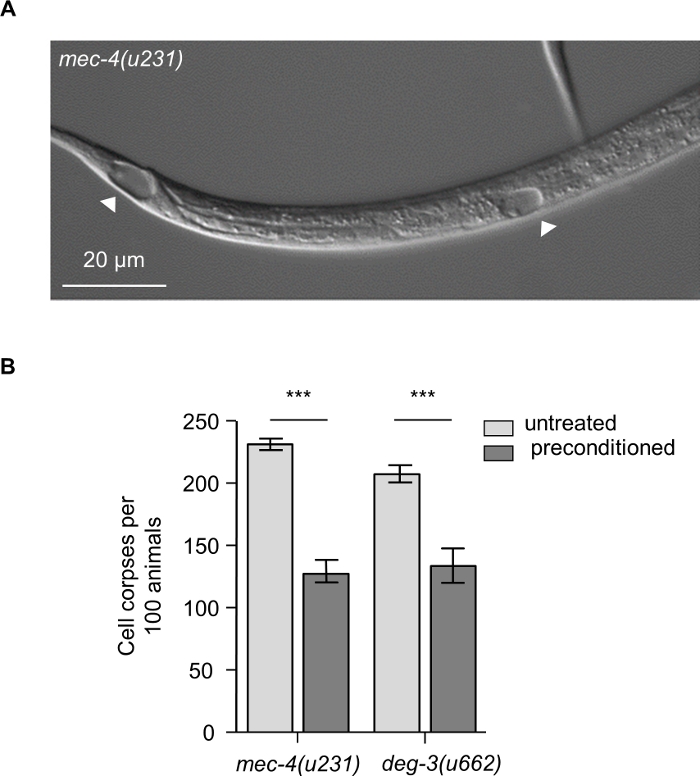
Figure 1: Hyperactive ion channel-induced necrosis. (A) Representative DIC microscopy image of mec-4(u231) L1 larva, with arrowheads indicating characteristic necrotic vacuoles. At early stage of neurodegeneration swollen nuclei are displayed within cells. Image was acquired by using a 40x objection lens. Scale bar, 20 μm. (B) Number of neuronal cell corpses at the L1 larval stage of development, per 100 animals carrying the neurotoxic mec-4(u231) or deg-3(u662) alleles. Necrotic cell death is suppressed in L1 larvae hatched from preconditioned eggs compared to untreated counterparts (n= 100 animals per genotype and assay; Data represent mean ±S.E.M., ***P< 0.001 for untreated versus preconditioned; t-test) Please click here to view a larger version of this figure.
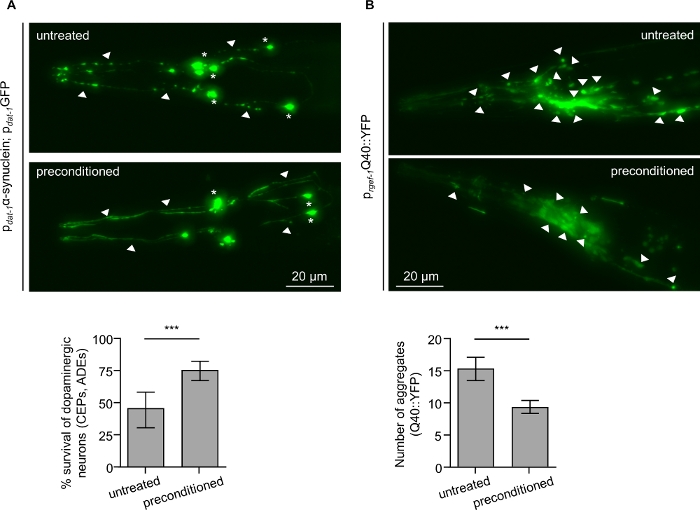
Figure 2: Daily preconditioning at 34 °C confers neuroprotection against α-synuclein and decreases polyQ aggregates in C. elegans. (Α) Survival of anterior dopaminergic neurons (CEPs and ADEs) in untreated and preconditioned nematodes co-expressing cytoplasmic GFP together with the human α-synuclein. Preconditioned nematodes display enhanced neuroprotection compared to untreated. Remnants of neuronal cell bodies (asterisks) and axonal beading (arrowheads) are seen in untreated nematodes (top panel). Both soma (asterisks) and neuronal processes (arrowheads) are preserved upon preconditioning. Images acquired by using a 20x objection lens and depict maximum intensity z-projection. Acquisition details: Bright, 0.15. Scale bar, 20 μm. Worms were scored for neuronal survival of the anterior dopaminergic neurons on the seventh day of adulthood. (B) Neuronal polyQ aggregates detected on the fourth day of adulthood in the head region of untreated and preconditioned transgenic nematodes. Preconditioned transgenic worms present less neuronal Q40::YFP aggregates compared to untreated. Representative images of the head region are shown with arrowheads indicating polyQ protein aggregates in neuronal cells. Images acquired by using a 20x objection lens and depict maximum intensity z-projection. Acquisition details: Bright, 0.0175. Scale bar, 20 μm. 30-35 animals were quantified per condition in each of three independent experiments. Data represent mean ±S.E.M., ***P < 0.05, unpaired t-test. Please click here to view a larger version of this figure.
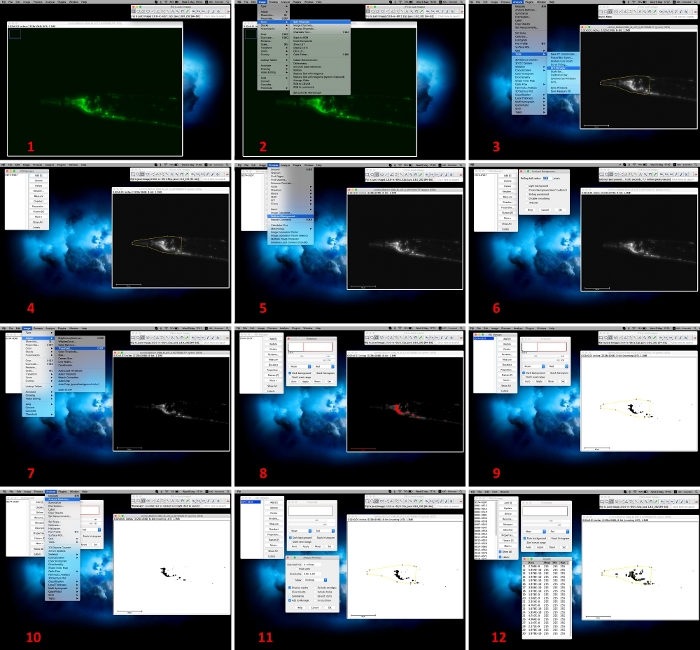
Figure 3: Image analysis by using Fiji software. 1. Open an acquired image with Fiji software; 2. Select “Split channel” command via the “Image” and “Color” drop down menu to convert the image; 3, 4. Keep “green channel” image and by using the “freehand selection” tool, enwrap the fluorescent region of interest (ROI; e.g., head). Add the respective ROI in “ROI Manager” via “Analyze” and “Tools” drop-down menu; 5, 6. Subtract the background to 50% by selecting “Process” and “Subtract Background”; 7, 8. Set up and apply threshold values via the menu command “Image”, “Adjust” and “Threshold”; 9. Select the respective ROI from the “ROI Manager”; 10-12. Analyze the number of protein aggregates by using the menu command “Analyze” and “Analyze Particles”. Please click here to view a larger version of this figure.
| Reagent | Recipe | |
| 2% agarose pads | 1. Weigh 0.5 g of agarose in a cylindrical glass beaker. | |
| 2. Add 25 mL of M9 buffer. | ||
| 3. Heat in a microwave until close to boiling. Take out, stir with a pipette tip and boil again. Repeat until the agarose is dissolved. | ||
| 4. Place an empty microscope slide on the bench. | ||
| 5. Put a drop (~ 50 µL) of fresh 2% agarose solution in the middle of the slide. | ||
| 6. Take a second microscope slide and place it on top of the agarose drop. Gently press down to flatten the drop. | ||
| 7. Let the agarose harden for 30 seconds and remove gently the top microscope slide. | ||
| 8. Immediately proceed with the sample preparation, since the agarose pads will start drying within approximately 5 minutes. | ||
| Tip: Leave the top microscope slide as a cover to preserve the humidity longer (~ 1 hour). Thus, several agarose pads can be prepared and used swiftly during the experiments. | ||
| M9 buffer | 1. Dissolve 3 g of KH2PO4, 6 g of Na2HPO4, 5 g of NaCl in 1 L of distilled water and autoclave. | |
| 2. Let cool and add 1 mL of 1 M MgSO4 (sterile). | ||
| 3. Store M9 buffer at 4 °C. | ||
| Nematode growth medium (NGM) agar plates | 1. Mix 3 g of NaCl, 2.5 g of bactopeptone, 0.2 g of streptomycin, 17 g of agar and add 900 mL of distilled water. Autoclave. | |
| 2. Let cool to 55-60 °C. | ||
| 3. Add 1 mL of cholesterol stock solution, 1 mL of 1 M CaCl2, 1 mL of 1 M MgSO4, 1 mL of nystatin stock solution, 25 mL of sterile 1 M phosphate buffer, pH 6.0, and distilled sterile water up to 1 L. | ||
| 4. Pipette 10 mL of medium per Petri dish and leave to solidify. | ||
| 5. Store the plates at 4 °C until used. | ||
Table 1: Recommended recipes for reagents used. All the reagents recipes used in the presented protocol are outlined here.
Discussion
Here, we introduce and describe widely accessible methodologies for growth, synchronization and microscopic examination of some versatile C. elegans models investigating age-dependent neurodegeneration. Particularly, we assess and dissect the cellular and molecular underpinnings of age-related neuronal breakdown by using hyperactivated ion channel-induced necrosis and protein aggregate-induced neurotoxicity1,2,3,4,5,7,9,10,11.
Although the described procedures for in vivo cell death assessment are straightforward and can be easily performed in any laboratory, there are some critical steps that should be taken into consideration. Caloric restriction and starvation are known to induce multiple stress pathways, such as autophagy, that might interfere with neurodegeneration or protein aggregates accumulation13,15,16. Thus, well-fed and non-starved nematodes should be used. Heat shock preconditioning confers neuroprotection against several neurodegenerative stimuli and is used as an established cell death modulator11,13. However, some mutants are susceptible to high temperature exposure for long periods. Thus, the appropriate developmental stage, age and duration of heat shock preconditioning should be experimentally determined each time, when animals of different genetic backgrounds, that might be sensitive to high temperatures, are used. A gradual increase of nematode intestinal autofluorescence is observed during aging. Thus, neuronal cell bodies and processes close to the intestinal area should be avoided during the imaging process of AM101 strain. Focus on neuronal cells, which are located in the head and/or tail region to bypass intestine-derived autofluorescence. Use M9 buffer instead of water to generate M9/levamisome buffer and 2% agarose pads. M9 buffer ensure a favorable osmotic environment protecting the nematodes from drying out throughout the microscopic visualization and analysis.
The described methodologies underscore that the combination of nematodes models of neurodegeneration, together with genetic and pharmacological screens for cell death modulators could lead to an unprecedented understanding of age-associated impairment of neuronal circuits and boost the development of novel therapeutic interventions against neurodegenerative disorders promoting human health and quality of life.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
We thank Chaniotakis M. and Kounakis K. for video recording and editing. K.P. is funded by a grant from the Hellenic Foundation for Research and Innovation (HFRI) and the General Secretariat for Research and Technology (GSRT). N.T. is funded by grants from the European Research Council (ERC – GA695190 – MANNA), the European Commission Framework Programmes, and the Greek Ministry of Education.
Materials
| Agar | Sigma-Aldrich | 5040 | |
| Agarose | Biozym | 8,40,004 | |
| AM101: rmsIs110[prgef-1Q40::YFP] | Caenorhabditis Genetics Center (CGC) | ||
| Calcium chloride dehydrate (CaCl2∙2H2O) | Sigma-Aldrich | C5080 | |
| Cholesterol | SERVA Electrophoresis | 17101.01 | |
| deg-3(u662)V or deg-3(d) | Caenorhabditis Genetics Center (CGC) | Maintain animals at 20 °C | |
| DIC microscope (Nomarsky) | Zeiss | Axio Vert A1 | |
| Dissecting stereomicroscope | Nikon Corporation | SMZ645 | |
| Epifluorescence microscope | Thermo Fisher Scientific | EVOS Cell Imaging Systems | |
| Escherichia coli OP50 strain | Caenorhabditis Genetics Center (CGC) | ||
| Greiner Petri dishes (60 mm x 15 mm) | Sigma-Aldrich | P5237 | |
| image analysis software | Fiji | https://fiji.sc | |
| KH2PO4 | EMD Millipore | 1,37,010 | |
| K2HPO4 | EMD Millipore | 1,04,873 | |
| Magnesium sulfate (MgSO4) | Sigma-Aldrich | M7506 | |
| mec-4(u231)X or mec-4(d) | Caenorhabditis Genetics Center (CGC) | Maintain animals at 20 °C | |
| Microscope slides (75 mm x 25 mm x 1 mm) | Marienfeld, Lauda-Koenigshofen | 10 006 12 | |
| Microscope cover glass (18 mm x 18 mm) | Marienfeld, Lauda-Koenigshofen | 01 010 30 | |
| Microsoft Office 2011 Excel software package | Microsoft Corporation, Redmond, USA | ||
| Na2HPO4 | EMD Millipore | 1,06,586 | |
| Nematode growth medium (NGM) agar plates | |||
| Nystatin stock solution | Sigma-Aldrich | N3503 | |
| Peptone | BD, Bacto | 211677 | |
| Phosphate buffer | |||
| Sodium chloride (NaCl) | EMD Millipore | 1,06,40,41,000 | |
| Standard equipment for preparing agar plates (autoclave, Petri dishes, etc.) | |||
| Standard equipment for maintaining worms (platinum wire pick, incubators, etc.) | |||
| statistical analysis software | GraphPad Software Inc., San Diego, USA | GraphPad Prism software package | |
| Streptomycin | Sigma-Aldrich | S6501 | |
| Tetramisole hydrochloride | Sigma-Aldrich | L9756 | |
| UA44: Is[baIn1; pdat-1α-syn, pdat-1GFP] | Upon request: G. Caldwell (University of Alabama, Tuscaloosa AL) |
Referencias
- Nikoletopoulou, V., Tavernarakis, N. Necrotic cell death in Caenorhabditis elegans. Methods in Enzymology. 545, 127-155 (2014).
- Syntichaki, P., Tavernarakis, N. The biochemistry of neuronal necrosis: rogue biology. Nature Reviews Neuroscience. 4 (8), 672-684 (2003).
- Syntichaki, P., Tavernarakis, N. Genetic models of mechanotransduction: the nematode Caenorhabditis elegans. Physiological Reviews. 84 (4), 1097-1153 (2004).
- Berkowitz, L. A., et al. Application of a C. elegans dopamine neuron degeneration assay for the validation of potential Parkinson’s disease genes. Journal of Visualized Experiments. (17), (2008).
- Brignull, H. R., Moore, F. E., Tang, S. J., Morimoto, R. I. Polyglutamine proteins at the pathogenic threshold display neuron-specific aggregation in a pan-neuronal Caenorhabditis elegans model. Journal of Neuroscience. 26 (29), 7597-7606 (2006).
- Gidalevitz, T., Ben-Zvi, A., Ho, K. H., Brignull, H. R., Morimoto, R. I. Progressive disruption of cellular protein folding in models of polyglutamine diseases. Science. 311 (5766), 1471-1474 (2006).
- Gitler, A. D., et al. The Parkinson’s disease protein alpha-synuclein disrupts cellular Rab homeostasis. Proceedings of the National Academy of Sciences of the United States of America. 105 (1), 145-150 (2008).
- Klaips, C. L., Jayaraj, G. G., Hartl, F. U. Pathways of cellular proteostasis in aging and disease. Journal of Cell Biology. 217 (1), 51-63 (2018).
- Labbadia, J., Morimoto, R. I. The biology of proteostasis in aging and disease. Annual Review of Biochemistry. 84, 435-464 (2015).
- Tucci, M. L., Harrington, A. J., Caldwell, G. A., Caldwell, K. A. Modeling dopamine neuron degeneration in Caenorhabditis elegans. Methods in Molecular Biology. 793, 129-148 (2011).
- Kourtis, N., Nikoletopoulou, V., Tavernarakis, N. Small heat-shock proteins protect from heat-stroke-associated neurodegeneration. Nature. 490 (7419), 213-218 (2012).
- Kourtis, N., Tavernarakis, N. Small heat shock proteins and neurodegeneration: recent developments. BioMolecular Concepts. 9 (1), 94-102 (2018).
- Kumsta, C., Chang, J. T., Schmalz, J., Hansen, M. Hormetic heat stress and HSF-1 induce autophagy to improve survival and proteostasis in C. elegans. Nature Communications. 8, 14337 (2017).
- Schindelin, J., et al. Fiji: an open-source platform for biological-image analysis. Nature Methods. 9 (7), 676-682 (2012).
- Palikaras, K., Lionaki, E., Tavernarakis, N. Coordination of mitophagy and mitochondrial biogenesis during ageing in C. elegans. Nature. 521 (7553), 525-528 (2015).
- Samara, C., Syntichaki, P., Tavernarakis, N. Autophagy is required for necrotic cell death in Caenorhabditis elegans. Cell Death and Differentiation. 15 (1), 105-112 (2008).

