A Mouse Model of the Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy Procedure Aided by Microscopy
Summary
Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy (ALPPS) has been shown to result in remarkable liver regeneration after several weeks post first-stage surgery. Animal models that have undergone ALPPS have been developed to explore potential liver regeneration ability and achieve favorable clinical outcomes.
Abstract
Hepatectomy is widely regarded as the primary treatment for hepatic malignancies; yet, postoperative liver failure remains a major cause of perioperative mortality, severely impacting patient outcomes. In a robust hepatic environment, the future liver remnant (FLR) must exceed 25%, and in cases of cirrhosis, this requirement increases to over 40%. The inadequacy of FLR is currently a major obstacle in the progression of hepatic surgery.
Traditional methods to enhance FLR hypertrophy mainly focus on portal vein embolization (PVE), but its effectiveness is considerably limited. In recent years, there have been numerous reports on a novel biphasic hepatectomy method involving hepatic partitioning and portal vein ligation, known as associating liver partition and portal vein ligation for staged hepatectomy (ALPPS). ALPPS surpasses PVE in efficiently and considerably inducing FLR hypertrophy. However, the detailed mechanisms driving ALPPS-facilitated hepatic regeneration are not fully understood. Thus, replicating ALPPS in animal models is crucial to thoroughly investigate the molecular mechanisms of hepatic regeneration, offering valuable theoretical and practical insights.
Introduction
The liver harbors formidable regenerative potential, swiftly proliferating and restoring metabolic demands within a mere 3 months following resection for diverse hepatic ailments1. However, the imperative to ascertain the completeness of tumor margins necessitates the inevitability of expansive hepatic excision. Thus, ensuring an ample volume of the participatory hepatic milieu, known as the future liver remnant (FLR), assumes paramount importance2. ALPPS has been a breakthrough technique in hepatic surgery over the past few decades, particularly tailored for patients with inadequate residual hepatic volume subsequent to tumor resection, heralded as one of the most auspicious breakthroughs in the realm of hepatic oncological surgery3.
Remarkable progress has been made in developing ALPPS animal models. An ideal model typically requires independent blood inflow (portal vein and hepatic artery) and outflow (hepatic vein) in the preserved hepatic lobe and a clear separation between the preserved and the to-be-resected hepatic lobes to prevent collateral circulation4. Although ALPPS stimulates rapid hepatic regeneration in the remaining liver tissue, the specific mechanisms of this process are still unclear.
Currently, ALPPS models are categorized into three types: large animal models (e.g., swine and sheep), medium-sized models (e.g., rabbits and rodents), and small models (e.g., mice)5. The use of mice, with their quick breeding, cost-effectiveness, and ease of genetic modification, is particularly effective for in-depth studies of hepatic regeneration mechanisms6. Moreover, the liver structure of mice, especially their middle hepatic vein, closely resembles that of humans, making them highly suitable for ALPPS model development.
It is important to note that most hepatocellular carcinoma patients in clinical practice have underlying liver conditions, unlike the healthy liver models typically used in studies7. Thus, using mice preconditioned with hepatic fibrosis or viral infections can more accurately simulate the surgical responses and postoperative liver regeneration seen in patients with various liver diseases8. This approach could reveal new therapeutic targets of clinical relevance.
So far, a few renowned research groups, such as those at the University of Zurich and the University of Tokyo, have successfully developed murine ALPPS models9,10. Creating a standardized murine model under controlled conditions could advance our understanding of the rapid liver regeneration observed after ALPPS procedures.
Protocol
All experiments in this protocol were approved by the Veterinary Authorities of the Jiangxi Provincial People's Hospital (number 70/2022). Furthermore, all experimental steps were performed in strict compliance with the Institutional Animal Care and Use Committee.
1. Start of surgery
NOTE: Male C57BL/6 mice weighing 20-22 g were housed in a well-ventilated cage, maintained in a standard pathogen-free environment with a 12 h/12 h light/dark cycle. The animals were provided ad libitum access to food and water at a controlled ambient temperature of 22 ± 1 °C. The surgery was performed in a dedicated microsurgical room using a surgical microscope (Figure 1). The mice in the sham group were only operated by laparotomy, but no operation was performed on the liver. In the PVL group, part of the portal vein branches was ligated, and in the ALPPS group, the middle liver lobe was transected on the basis of PVL.
- Subject the mice to a 12 h preoperative fasting period.
- Arrange sterile surgical instruments, including microforceps, scissors, abdominal retractors, and pre-prepared 8-0 portal vein ligature sutures, on a side table. Soak intraoperative materials comprising sterile gauze and cotton balls in 37 °C saline solution.
- For induction, expose the animals to 5% v/v isoflurane for 30 s, followed by maintenance anesthesia using 3 vol% isoflurane until achieving a deep anesthetic state. Examine the toe pinch reflex to ascertain the level of anesthesia. Subcutaneously inject medetomidine at a dose of 5 mg/kg during the induction phase.
- Apply ointment to the animals' eyes and inject 1 mL of lactated Ringer's solution mixed with 5% glucose subcutaneously, dividing the dose equally between the lower abdomen on each side.
- Meticulously shave the abdominal region using small animal hair clippers, encompassing a 2 cm lateral area from the xiphoid process to the genital region.
- Thoroughly disinfect the shaved area by employing a sponge soaked in povidone-iodine solution, repeating the process 3x.
- Make a precise midline incision on the skin using a surgical scalpel, subsequently utilizing surgical scissors to delicately open the abdominal cavity.
- Employ the abdominal retractor to expand the incision, facilitating the gentle traction of the duodenum and a segment of the small intestine using sterile, moist cotton swabs. Cover these structures with the pre-prepared sterile gauze, simultaneously exposing the hepatic segment of the portal vein.
2. Portal vein ligation (PVL)
NOTE: Ensure that all actions are gentle throughout the whole procedure. Use moistened cotton balls when manipulating liver tissue.
- Peel off the peritoneum and intestinal tract and make sure the portal vein is fully exposed.
- In the course of exposing the main portal vein, proceed by encircling its branches using slow, deliberate forward-pushing and spreading movements.
- Sequentially identify the various branches of the portal vein in the following order: (1) the right posterior branch, (2) the combined left lateral and left median branches, and (3) the caudate branch (Figure 2A).
3. Dissection of the right posterior branch of portal vein branches
- Upon traversing the hepatic hilum via the main portal vein, observe a conspicuous branch of the right posterior lobe that has become visible.
- Use moistened cotton balls as a fulling agent and separate the liver's middle lobe and the right posterior lobe to avoid potential injury.
- Be sure to dissect along the Glisson's capsule (the peritoneum that extends over the surface of the liver) and pull a 1.5 cm silk to tie around portal vein branches.
- Ligate the right posterior branch of the portal vein with 8-0 silk suture. A reliable indicator of successful portal vein ligation is visible pallor of the liver.
4. Dissection of the left lateral and left medial branches of the portal vein
- The left lateral lobe (LLL) and left medial lobe (LML) branches share a common main portal vein. Be sure to dissect any potential interstice between the LLL and the caudate lobe.
NOTE: Ligation of the LLL and LML branches of the portal vein is the most difficult in this surgical model because the microforceps must traverse the space around the portal vein and avoid liver injury. - Introduce the microforceps through the entrance in step 4.1 and move the hand slowly, as even the slightest resistance hints at the potential proximity to the liver or blood vessels.
- When resistance is felt, adjust the microforceps slightly to move forward and through the surface of Glisson's capsule of the right medial lobe (RML) (Figure 3C).
- Employ the prepared 8-0 silk suture to tie around the portal vein of the LLL and LML branches. Look for a striking ischemic demarcation within the liver middle lobe, while the left lobe assumes pallor, upon ligation of the portal vein branches.
5. Dissection of the caudate branch of the portal vein branches
- Dissect the caudate lobe and use a cotton swab to fill the potential space between the main portal vein and the caudate lobe.
- Peel off the peritoneum of the caudate lobes to visualize the portal vein branches of the lobe.
- Use Stitch-up suture rather than tie-around to ligate the caudate branch, ensuring that the needle path adequately wraps the caudate branch of the portal vein.
NOTE: The difficulty of ligating the caudate branch of the portal vein is because the caudate lobe portal vein encapsulates the liver tissue and is close to the main portal vein. A needle penetration depth of 1 mm is recommended.
6. Liver transection and cholecystectomy
- Observe a distinct demarcation line in the middle liver lobe and perform the transection along this line (Figure 2B).
- Use an electrocautery pen to create a 0.5 mm precut line along the demarcation line.
- Use micro forceps and an electrocautery pen to cut liver tissue, applying cotton swabs to stop bleeding.
NOTE: The difficulty in the transection is to master the depth. The goal is to split the liver as close to the vena cava as possible. Intraoperative bleeding is difficult to avoid and requires repetitive practice to master the depth of transection. Hot burning, cotton swabs, and gelatin sponges can be used to assist in hemostasis. - Remove the gallbladder last as this will provide a good pulling point during the whole operation.
- Employ a cotton swab to apply controlled pressure for hemostasis, and if hemorrhage persists, the inferior vena cava is injured and the effect of suture repair will be poor; euthanize the animal without hesitation.
7. Final steps of surgery
- Ensure meticulous placement of the small bowel within the abdominal cavity prior to closure.
- Securely close the peritoneum and abdominal wall using 6-0 silk sutures and the skin with 4-0 silk sutures.
- Disinfect the skin surface again to ensure optimal cleanliness.
- Subcutaneously administer 1 mL of saline solution and meloxicam 4 mg/kg to alleviate postoperative pain and address any supplementary intraoperative fluid loss.
- Place the mice on a thermostatic pad to facilitate natural awakening, and monitor the blood supply to their extremities. Subsequently, house them individually in cages for continued observation as they regain consciousness.
- Subcutaneously inject medetomidine at a dose of 4 mg/kg upon recovery, continuing for 24 h postoperatively.
8. Intraoperative measurement of liver regeneration and liver function
- Euthanize the mice and collect liver as well as blood samples at intervals of 1, 2, 3, and 7 days post surgery.
- Perform HE staining and liver function test and examine serum inflammatory markers, thereby providing additional validation for the impact of the model construction.
- Document the daily variations in the body weight and FLR/BW (body weight) ratio of the mice over a span of 1 week following the surgical procedure.
Representative Results
In contrast to the PVL (protocol steps 2,3,4, and 5), the mice that were subjected to the ALPPS (steps 2,3,4,5, and 6) surgical intervention demonstrated a heightened proclivity towards cellular proliferation. Figure 2 delineates the ischemic demarcation of the middle liver lobe following portal vein ligation, along with the visual field during liver transection, which represents a key distinction between ALPPS and PVL. Notably, the levels of inflammatory mediators, including (Tumor Necrosis Factor-α (TNF-α), interleukins, and hepatic growth factor (HGF), within the hepatic tissue, surpass those observed in the PVL group. This is seen in Figure 4C where the ALPPS group had higher IL-6 levels than the PVL group, suggesting that inflammation may be involved in rapid liver regeneration. To further substantiate these findings, a comparative analysis of the liver-to-body weight ratio and the expression levels of ki67, a widely recognized marker of cellular proliferation, within the hepatic tissue, two days post-operatively, has been done. Figure 4A,B and Figure 5 show that the liver in the ALPPS group demonstrates enhanced proliferative capacity.
Figure 1: Surgical instruments and consumables. (A) 1: Topical antibiotics for incisions; 2: 6-0, 8-0 Surgical sutures; 3: hemostatic gelatin sponges. (B) 1: Scissors and 2: needle holders are employed for the incision and suturing of the abdominal cavity. (C) Microscopic procedures necessitate the use of 1: scissors, 2: forceps, 3, 4: dissecting forceps, 5: abdominal retractors. Please click here to view a larger version of this figure.
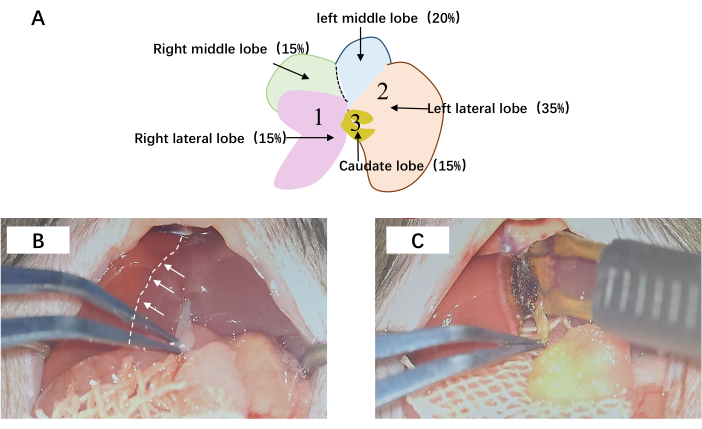
Figure 2: The hepatic surgical procedure. (A) Portal vein ligation and transection. The sequential ligation of 1, 2, and 3 is meticulously executed, concomitantly with the meticulous division of the hepatic parenchyma within the middle lobe. (B) The demarcation line of the middle lobe is visually depicted by the presence of a white dashed line. (C) Subsequent to microscopic examination, the liver is meticulously dissected along the delineated ischemic boundary. Abbreviations: RLL = right lateral lobe; LML = left middle lobe; LLL = left lateral lobe; RML = right median lobe. Please click here to view a larger version of this figure.
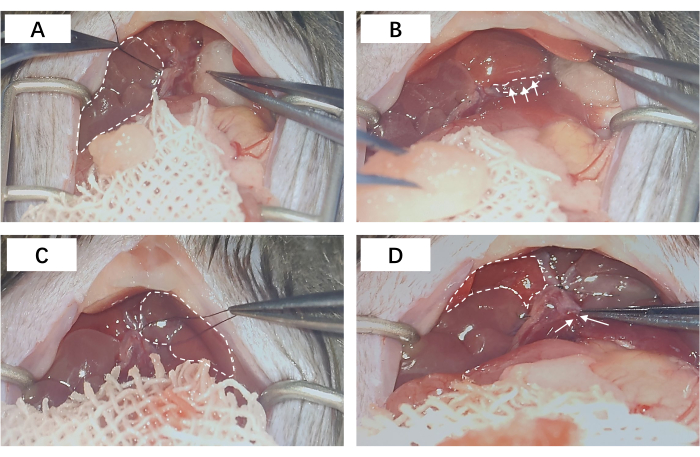
Figure 3: Portal vein ligation. (A) The portal vein ligation of the right lateral lobe; the dashed area corresponds to the ligated RLL. (B) The portal vein ligation of the left middle lobe and the left lateral lobe; the dashed line area represents the entrance for ligation of the portal vein branches. (C) The dashed area corresponds to the ligated LML and LLL. (D) The portal vein ligation of the caudate lobe; the dashed region delineates the physiologically perfused right middle lobe, whereas the arrows demarcate the portal vein branch of the caudate lobe, discreetly ensconced within the hepatic parenchyma. Abbreviations: RLL = right lateral lobe; LML = left middle lobe; LLL = left lateral lobe. Please click here to view a larger version of this figure.
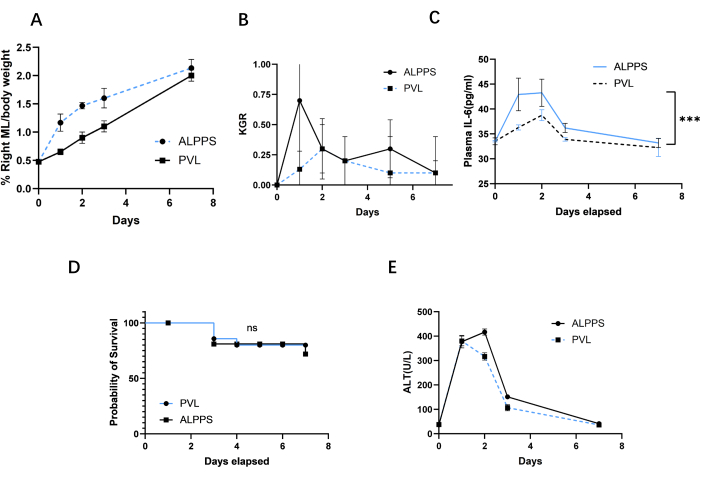
Figure 4: Comparison of future liver remnants, kinetic growth ratio, survival rate, and plasma ALT during the first week between ALPPS and PVL. (A) FLR was assessed in the PVL group and PVL+T group post surgery. (B) KGR was assessed as the gain of LMR/bodyweight ratio per day. KGR in the ALPPS group increased rapidly within 2 days after surgery, then gradually decreased, but was still higher than that in the PVL group. (C) Plasma IL-6 between PVL and ALPPS. (D) Survival curve after PVL and ALPPS. (E) Plasma AST between PVL and ALPPS. Abbreviations: FLR = future liver remnants; PVL = portal vein ligation; PVL + T = portal vein ligation + transection; KGR = kinetic growth ratio; LMR = left ; ALPPS = associating liver partition and portal vein ligation for staged hepatectomy; ALT = Alanine aminotransferase. Please click here to view a larger version of this figure.
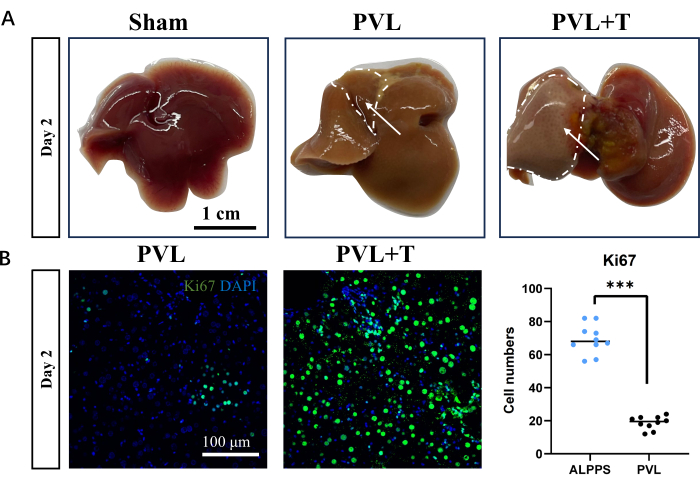
Figure 5: Markers and morphology of liver regeneration following portal vein ligation or combined transection. (A) Hepatic morphology was assessed in the sham surgery group, PVL group, and PVL+T group 2 days post surgery. The delineated area signifies the regenerated hepatic lobes; scale bar = 1 cm. (B) Fluorescence staining unveiled heterogeneous levels of cellular proliferation across distinct surgical cohorts of the liver; scale bar = 100 µm, ***P < 0.001. Please click here to view a larger version of this figure.
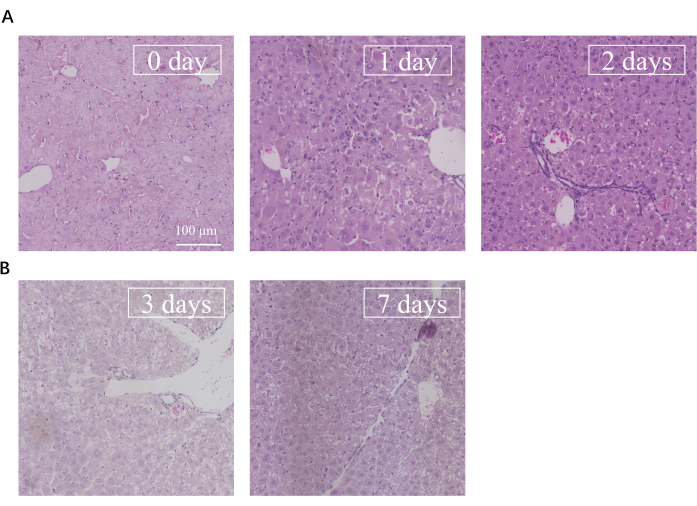
Figure 6: HE images from portal vein to central vein after ALPPS. (A) The liver of ALPPS HE staining from 0 day to 2 days. (B) HE staining from 3 days to 7 days. Scale bar = 100 µm. Abbreviations: HE = hematoxylin and eosin; ALPPS = associating liver partition and portal vein ligation for staged hepatectomy. Please click here to view a larger version of this figure.
Discussion
This study presents a protocol for the initial phase of ALPPS surgery in mice, involving partial ligation of the portal vein and division of the median liver lobe. This method closely mimics the human ALPPS procedure, with the mouse liver's unique lobular structure and dual portal venous supply to the median lobe ensuring distinct blood flow post-surgery11. It is noteworthy that the ALPPS procedure comprises two distinct surgical stages. In this study, we exclusively present the initial stage involving portal vein branch ligation and liver transection, denoted as PVL+T. Notably, the second stage, involving partial liver lobectomy, is not executed in our study. To maintain uniformity in our description, we consider ALPPS equivalent to PVL+T. While previous research has largely focused on rat models, the University of Zurich's team uniquely reported on a mouse-specific ALPPS protocol12. However, a standardized operational procedure for mice remains elusive. Compared to rats, mice offer easier genetic manipulation and broader research opportunities in liver regeneration, making a mouse-specific ALPPS model highly valuable for understanding liver regeneration under clinical conditions
The key steps in this protocol include simplifying the ALPPS procedure as PVL combined with T and emphasizing the anatomy of the mouse portal vein for beginners. Each mouse liver lobe has a separate portal vein, resembling clinical liver resection strategies (Figure 3A). Critical aspects are the careful ligation of portal vein branches to prevent bleeding and ensure postoperative survival, as well as identifying gaps around these branches for easier ligation. Sterile cotton balls aid in protecting the liver during the procedure, and gentle handling is crucial due to the liver's delicate texture. When ligating the left lateral and median lobes, finding the correct gap is essential, as is a suturing ligation method for the caudate lobe's portal vein branches.
The two distinct surgical procedures, portal vein ligation (PVL) and PVL with transection (PVL+T), yield markedly different growth kinetics and future liver remnant (FLR) outcomes. PVL elicits a moderate volume increase within 2 days, while PVL+T exhibits a substantially larger right median lobe (RML) enlargement, as illustrated in Figure 4A and Figure 5A. This observation can be corroborated through assessments of the FLR/body weight (BW) ratio and the ratio of Ki67-positive staining, as illustrated in Figure 4B,D. In terms of FLR/BW ratio or IHC staining (e.g., Ki67/MCM2/Ccnd1), the expected increase of FLR/BW ratio is ~1.3-1.5 after 2 days of ALPPS described in protocol step 1. The positive staining of Ki67 is 15-20 positive cells per 100 hepatocytes.
The protocol adapts the rat ALPPS procedure, aiming to enhance anatomical understanding, refine surgical techniques, and improve postoperative outcomes in mice. However, it may not fully simulate liver regeneration in mice under varied physiological conditions, as clinical ALPPS patients often have conditions such as hepatitis, cirrhosis, or liver cancer. Future research should include diverse liver or systemic disease backgrounds for more accurate liver status assessment post-ALPPS13.
To assess the risk of liver failure across different groups, we quantified postsurgery transaminase levels in the liver and compared their respective survival curves (Figure 4D,E). Remarkably, ALPPS demonstrated the capacity for rapid liver regeneration and correction of metabolic disorders arising from liver insufficiency. Additionally, considering the substantial regional heterogeneity of the liver, we conducted HE staining on hepatocytes spanning from the portal vein to the central vein at various postoperative time points. This analysis revealed that the liver undergoes apoptosis and necrosis 2-3 days after surgery, followed by a swift return of liver cells to the preoperative state at ~7 days (Figure 6). Notably, no significant differences were observed among distinct liver regions, suggesting that delving into liver heterogeneity may provide further insights into the intricate relationship between liver regeneration and hepatic diversity.
Moreover, certain details require careful consideration during the preoperative preparation. Regarding preoperative fasting, it is crucial to consider that the abdominal cavity needs to be accessed for surgery. A distended bowel can pose challenges in exposing the portal vein during the procedure, escalating the risk of inadvertent bowel injury. An intriguing aspect pertains to the learning curve, influenced by factors such as anatomical knowledge, manual dexterity, and surgical aptitude. Attaining proficiency in micromanipulation, with approximately 100 h of experience, involves repetitive manipulation in ~30 mice.
Liver transection stands out as a pivotal step in the ALPPS surgical model. Choosing the appropriate degree of transection is crucial to prevent collateral damage to the inferior vena cava, a topic that will be elaborated upon in the revised manuscript. For hemostasis, electrocoagulation and cotton swab compression must be used, systematically transecting along the ischemic line from the surface to the deep layers. Notably, there was no incidence of excessive bleeding in the liver section following meticulous layer-by-layer hemostasis.
In conclusion, our adaptation of the rat ALPPS protocol for mice has improved survival rates and reduced intraoperative bleeding. Yet, questions remain about the drivers of liver regeneration in this context. Is it altered hemodynamics due to portal vein ligation or an inflammatory response from liver transection? The accidental discovery of ALPPS in liver cancer patients and its development as a surgical method to enhance liver volume still leaves the liver regeneration mystery unresolved. Our research, incorporating CRISPR gene editing and single-cell sequencing14,15, aims to unravel the interplay between inflammatory factors, hepatocytes, immune cells, and other cells in liver regeneration, contributing to novel therapeutic strategies for post resection liver regeneration and preventing liver failure.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
None
Materials
| 5% glucose injection (500 mL) | Shapuaisi pharma (http://www.zjspas.com/) | H20003666 | Efficient, cheap,Easy to access |
| anaesthesia machine | RWD (www.rwdls.com) | R500 | low price and valuable quality. It is suitable for operations with beginners |
| C57 BL/6 | The Jackson Lab | 22349-2023 | Stability of strains |
| isoflurane | KCSW (kcsw.szqisoubao.com) | H19980141 | durable, cost-effective |
| meloxicam | Boehringer-Ingelheim | H20020217 | Durable and efficient |
| microforceps | maydeal | 60018920 | Durable and efficient |
| microinstrument | CH microsurgical instrument factory | HC-A804-1 | durable, cost-effective |
| sodium lactate ringer | Shapuaisi pharma (http://www.zjspas.com/) | H20193277 | Efficient, cheap, easy to access |
| suture line | CH microsurgical instrument factory | 6-0/8-0 | high performance-price ratio |
| topical antibodies | chenxin pharmacology (www.cisen-pharma.com) | H20020217 | Effectively avoid incision infection |
Referencias
- Michalopoulos, G. K., Bhushan, B. Liver regeneration: Biological and pathological mechanisms and implications. Nat Rev Gastroenterol Hepatol. 18 (1), 40-55 (2021).
- Chapelle, T., et al. Estimation of the future remnant liver function is a better tool to pr edict post-hepatectomy liver failure than platelet-based liver scores. Eur J Surg Oncol. 43 (12), 2277-2284 (2017).
- Isfordink, C. J., et al. Portal vein ligation versus portal vein embolization for induction of hypertrophy of the future liver remnant: A systematic review and meta-analysis. Surg Oncol. 26 (3), 257-267 (2017).
- Linecker, M., et al. Risk adjustment in alpps is associated with a dramatic decrease in ear ly mortality and morbidity. Ann Surg. 266 (5), 779-786 (2017).
- Darnis, B., et al. Subtotal hepatectomy in swine for studying small-for-size syndrome and portal inflow modulation: Is it reliable. HPB (Oxford). 17 (10), 881-888 (2015).
- Casellas, J. Inbred mouse strains and genetic stability: A review. Animal. 5 (1), 1-7 (2011).
- Zhang, J., et al. Evolution of associating liver partition and portal vein ligation for staged hepatectomy from 2012 to 2021: A bibliometric analysis. Review. Int J Surg. 103, 106648 (2022).
- Parola, M., Pinzani, M. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical iss ues. Mol Aspects Med. 65, 37-55 (2019).
- Kikuchi, Y., et al. Impact of associating liver partition and portal vein occlusion for st aged hepatectomy on tumor growth in a mouse model of liver metastasis. Eur J Surg Oncol. 44 (1), 130-138 (2018).
- Schlegel, A., et al. Alpps: From human to mice highlighting accelerated and novel mechanism s of liver regeneration. Ann Surg. 260 (5), 839-846 (2014).
- Dili, A., Lebrun, V., Bertrand, C., Leclercq, I. A. Associating liver partition and portal vein ligation for staged hepate ctomy: Establishment of an animal model with insufficient liver remnan t. Lab Invest. 99 (5), 698-707 (2019).
- Schadde, E., Hertl, M., Breitenstein, S., Beck-Schimmer, B., Schläpfer, M. Rat model of the associating liver partition and portal vein ligation for staged hepatectomy (alpps) procedure. J Vis Exp. (126), e55895 (2017).
- Allaire, M., et al. New frontiers in liver resection for hepatocellular carcinoma. JHEP Rep. 2 (4), 100134 (2020).
- Memi, F., Ntokou, A., Papangeli, I. Crispr/cas9 gene-editing: Research technologies, clinical applications and ethical considerations. Semin Perinatol. 42 (8), 487-500 (2018).
- Saviano, A., Henderson, N. C., Baumert, T. F. Single-cell genomics and spatial transcriptomics: Discovery of novel cell states and cellular interactions in liver physiology and disease biology. J Hepatol. 73 (5), 1219-1230 (2020).

.
