Optical Mapping of Langendorff-perfused Rat Hearts
Summary
This article describes a high temporal and spatial resolution technique to optically image action potential movement on the surface of Langendorff-perfused rat hearts using a potentiometric dye (di-8-ANEPPS).
Abstract
Protocol
Part 1: Prepare solutions and the isolated perfused heart system
- On the morning of the experiment, 4.0 L of Krebs-Henseleit solution is prepared as previously described[5, 6].
- 11 mMol/L 2,3-butanedione monoxime (BDM) is dissolved in 1.0 L of Krebs-Henseleit solution decanted from the perfusate prepared in step 1.1.
- An additional 150 mL of Krebs-Henseleit is removed from the perfusate prepared in step 1.1 and mixed with 5 Mol/L di-8-ANEPPS (diluted from a 10 mMol/L stock dissolved in dimethyl sulfoxide (DMSO)).
- These 3 solutions are transferred to water-jacketed glass reservoirs (Radnoti) where they are pre-warmed to 41.0° C and oxygenated with a submerged bubbler (Radnoti) using 0.2 μm-filtered 95% O2; 5% CO2 gas. Solutions from the 3 stock reservoirs are pumped to the wall-mounted Langendorff system with MasterFlex™ L/S peristaltic pumps and low-absorption silicone tubing (Cole-Parmer).
- Prior to assembly,the Langendorff system glassware (Radnoti) is meticulously washed with E-TOXA-CLEAN reagent (Sigma-Aldrich) and thoroughly rinsed with 0.1 M/L HCl, 100% ethanol, and distilled water.
- The non-recirculating Langendorff apparatus was constructed to deliver 70 mm Hg constant-pressure from 3 separate oxygenated reservoirs, which are separated from the heart with water-jacketed heating coils and bubble traps (Radnoti)[7]. The 3 independent perfusion lines converge above the heart at two 3-way stopcocks, allowing us to precisely control delivery of the solutions prepared in steps 1.1 to 1.3. Water-jacketed glassware is connected in series with MasterFlex PharMed BPT tubing (Cole-Parmer) and warmed to 39° C with distilled water using two E100 circulators (Lauda). Heat loss through the platinum-cured silicone tubing (Cole-Parmer) that connects the pressure-head reservoirs to the heating coils and bubble traps resulted in a 37° C perfusate reaching the heart.
Part 2: Harvest the rat heart and set-up Langendorff perfusion
- To induce deep general anesthesia in 200-250 g Lewis rats, 100 mg/kg Ketamine and 10 mg/kg Xylazine is injected into the intraperitoneal cavity. To this mixture, we add 500 U/kg Heparin to prevent blood coagulation and myocardial ischemia during the explantation procedure.
- For easy access to the heart and great vessels, the anterior chest wall is removed. After that, surrounding tissue is carefully dissected and the pericardial sack opened.
- Following identification of the inferior vena cava, this vessel is ligated with 5-0 silk (Ethicon) and the entire heart-lung block is explanted. Tissue is immediately placed in ice-cold Krebs-Henseleit solution contained in a 50 mL beaker on ice.
- The ascending aorta is quickly identified and dissected from the surrounding tissue. An appropriately-sized cannula (Harvard Apparatus) is inserted into the aorta, taking care to avoid interrupting obligatory perfusion of the coronary arteries by inserting the cannula too far into the aortic root. The cannula is secured to the ascending aorta with 5-0 silk (Ethicon).
- The rat heart is then placed on the Langendorff apparatus without introducing air bubbles into the cannula. Retrograde coronary vascular perfusion is now established with the warm, oxygenated Krebs-Henseleit solution from the pressure-head described in step 1.6.
- Extra tissue, including the lungs, is now removed and the heart is perfused for 20 minutes to permit recovery of function and stabilize the rhythm. During this time, a very thin thermocouple temperature probe (Cole-Parmer) is introduced into the left ventricular cavity and sutured in place with 5-0 Prolene suture (Ethicon). The probe is connected to a thermo-controller (Digi-Sense) to ensure the temperature of the heart is maintained at 37° C by adjusting the settings of the water circulatory pumps. Motion from perfusate dripping from the cardiac apex is minimized by placing a piece of gauze in the effluent receptacle.
Part 3: Load heart with potentiometric dye and acquire electrographic and optical signals
- Di-8-ANEPPS (Invitrogen) is loaded into the heart by switching to the perfusion line containing the Krebs-Henseleit mixed with the fluorescent dye using a stopcock. In addition, an 18G cannula is placed into the left and/or right atrium and additional 50 mL of dye solution is slowly administered into each of these chambers because they are not sufficiently perfused with dye introduced through the coronary arteries.
- During the loading procedure, 3 ECG leads (Harvard Apparatus) are gently placed on the surface of the heart that is not facing the optics used for mapping. Electrode No 1 is positioned on the posterior apical portion of the left ventricle, No 2 on the left atrium, and No 3 as a reference electrode on the aortic root (Figure 1). The atrial and ventricular electrographic signals are subsequently amplified, digitized, and displayed alongside optical signals using the software (RedShirt Imaging) (Figure 2). An oscilloscope (Tektronix model TDS 1002) is also used to visualize the surface ECG s in real-time and to assure adequate pacing.
- ECG amplifier (Hugo Sachs Elektronik) settings:
High Pass Filter: 0.1 Hz
Low Pass Filter: 150 Hz - The CMOS camera (RedShirt Imaging) and macroscope is positioned using the X-Y-Z adjustments so that the surface of the heart is in focus and centered in the acquisition frame. The camera and optics are mounted on a vibration isolation table (Minus K Technology) to minimize resonant frequencies. At the same time, a coaxial pacing electrode (Harvard Apparatus) controlled with an isolated, S48 electrical-stimulation unit (Grass), is placed on the right atrium and the heart is paced at 300 beats per minute (Figure 1).
- Electrical stimulation (Grass) settings:
Rate: 5 pulses per second
Delay: 0.2 ms
Duration: 2 ms
Volts: 6-12 V
Mode: Repeat
Pulse: Single - For optical recordings which lack motion artifacts from contraction, the heart needs to be electromechanically uncoupled. We do this by again switching perfusion lines to Krebs-Henseleit solution that contains 11 mMol/L BDM. Between acquisitions, the heart is perfused with unadulterated Krebs-Henseleit to help preserve viability of the preparation.
- The recording parameters are set using the software (RedShirt Imaging) with the following acquisition settings:
Configuration: 2,000 Hz; 128×128 pixel array
Frame Interval: 0.5 ms
Camera Amplifier Gain: 5x
On-Chip Gain: 8x-12Me-well
Shutter: 500 ms delay
Number of Frames: 4000
Duration: 2000 ms - All room and equipment lights are turned off or shielded to eliminate background noise during recording. The LED light illuminates the heart only during the optical recording to reduce photo-bleaching and dye toxicity. The light source shutter is controlled with a 5 V pulse delivered through the control panel by way of a D-to-A board in the computer (RedShirt Imaging).
Part 4: Analyze acquisition information using RedShirt Imaging Software
- Following acquisition, data is processed using different filter settings. We generally use the default settings except when adjusting the Band Stop/Pass filter, which is set with the left boundary at 44.0 and the right boundary at 98.0. Afterward the recorded information is processed and a movie is generated (RedShirt Imaging).
- Data from one acquisition corresponds to the local electrical activation at 16,384 sites on the heart surface over a period of 2 seconds. The software allows these local signals to be directly compared with one another and with the atrial and ventricular electrographic recordings. Data is then visualized by mapping local electrical activation to color and rendering this information as an animation showing the spatiotemporal electrical activation on the cardiac surface. To create such an animation, we use the software to:
- temporally and/or spatially filter the data
- select a start and end time for the animation
- map optical signals to color based on resting light intensity of each pixel
- overlay the resulting color data with a picture of the heart
- generate the animation
Part 5: Representative results
If the perfused heart preparation was motionless during recording, the optical signals show one distinct peak for every pixel involved in a change of the emission intensity of di-8-ANEPPS. The corresponding movies (Figures 3 and 4) demonstrate an excitation wave front propagating across the epicardial surface of the heart as well as the simultaneously acquired electrographic recordings (Figure 2).

Figure 1. A photograph of a Langendorff-perfused heart preparation depicting the positions of the pacing electrode on the right atrium and the ECG leads as described in step 3.2.
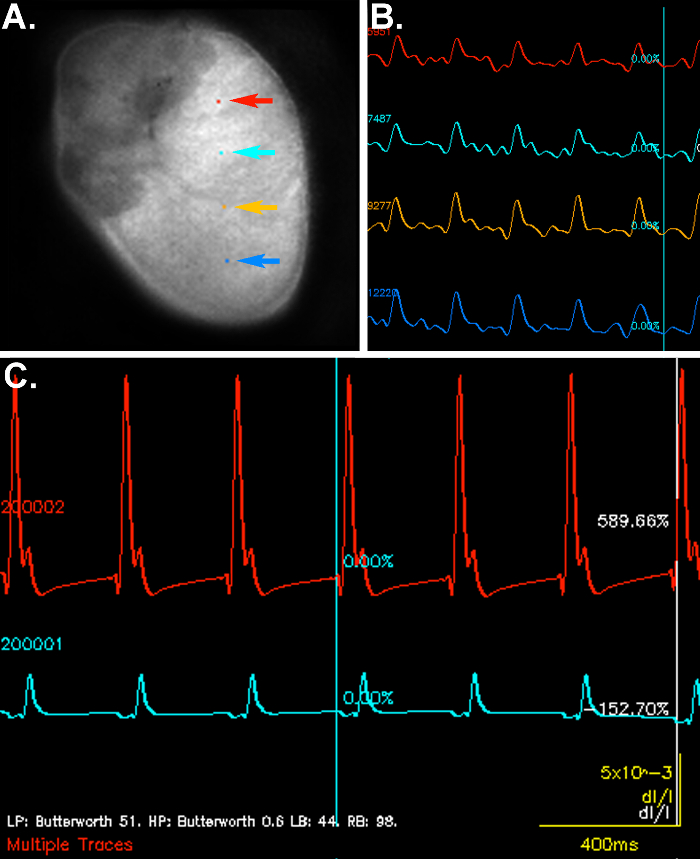
Figure 2. Representative optical signals and electrographic recordings from a perfused Lewis rat heart. Panel A shows an image of the epicardial surface used for optical imaging. The position of the pixels selected to demonstrate the changes in fluorescence emission over time in Panel B are indicated by colored arrows. Electrographic signals are shown in Panel C with the red line indicating atrial activation and the light blue line corresponding to the ventricular signal. Please click here to see a larger version of this figure.
Discussion
Removal of the heart from anesthetized rats must be performed rapidly to avoid myocardial ischemia. If ischemia or insufficient coronary perfusion occurs, the heart will likely develop arrhythmias and may become infarcted. Additionally, these hearts will show insufficient fluorescence emission for informative recordings and subsequent analyses. Before loading the dye, myocardial cells need to be adequately perfused with Krebs-Henseleit solution to establish and maintain a physiologic electrolyte milieu for the stability of electrical impulses. Accurate preparation of the perfusate is also necessary to maintain proper organ viability and function. Differences in the electrolyte concentration or insufficient filtering of the perfusate will probably lead to fatal myocardial dysfunction and heart rhythm disorders. For the optical recordings, the heart must be completely loaded with voltage-sensitive dye. This is especially important for the atrial myocardium as these chambers are not well-perfused by the coronary arteries. We have found that additional intra-cavitary perfusion of the atria will establish a good optical signal. Furthermore, acquisition of top-quality voltage tracings requires the perfused heart to be motionless; otherwise, changes in fluorescence emission cannot be reliably used to track changes in membrane potential with high fidelity due to artifacts caused by signal drift. This will result in multiple peaks for one pixel rather than a single peak. Other methods to eliminate motion artifacts from optical recordings include mechanical immobilization, treatment with other excitation-contraction uncouplers (e.g. cytochalasin D, blebbistatin), signal processing, and by mathematical modeling[1, 8]. Finally, the method described here merely provides information regarding action potential movement on the cardiac epicardial surface. Alternative tissue preparations and infrared potentiometric dyes may resolve the electrical propagation characteristics in other regions of the heart.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work is supported by research grants from the National Institutes of Health (HL068915; HL088206) and contributions to the Cardiac Conduction Fund at Children’s Hospital Boston.
Materials
| Material Name | Type | Company | Catalogue Number | Comment |
|---|---|---|---|---|
| CardioCMOS-SM128f | Equipment | RedShirt Imaging, Decatur, GA 30030 USA | ||
| CardioPlex Software | Equipment | RedShirt Imaging, Decatur, GA 30030 USA | ||
| LUXEON LED Light Source 460-490 nm | Equipment | Lumileds Lighting, US, LLC, San Jose, CA 95131 USA | LXHL-PB02 | |
| ECG Amplifier Type 689 Hugo Sachs Elektronik | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | 730149 | |
| Dichroic Mirror 505 nm | Equipment | Semrock, Rochester, NY 14624 USA | FF505-SDi01-25×36 | |
| Emission Filter 605 nm Long Pass | Equipment | SciMedia, Costa Mesa, CA 92626 USA | ||
| THT Sideways | Equipment | SciMedia, Costa Mesa, CA 92626 USA | 25 BM-8 | |
| Mini Ball Joint Holder | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | BS4 73-0177 | |
| Small Stimulation Electrode Set | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | BS4 73-0160 | |
| BM-6 Benchtop Vibration Isolation Platform | Equipment | Technology Inc., Inglewood, CA 90301 | 25 BM-6 | |
| Monopolar ECG Electrode | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | BS4 73-0200 | |
| Roller Pump SCI 400 | Equipment | Watson-Marlow Bredel Inc., Wilmington, MA 01887 USA | 401U/D1 | |
| Roller Pump MasterFlex Easy Load II | Equipment | Cole Parmer, Vernon Hills, Illinois 60061 USA | Model 77201-60 | |
| Tubing Marprene #14 | Equipment | Watson-Marlow Bredel Inc., Wilmington, MA 01887 USA | 902.0016.016 | |
| MasterFlex Tubing | Equipment | PharMed, Westlake, OH 44145 USA | 06485-25 | |
| S48 Square Pulse Stimulator | Equipment | Grass Technologies, West Warwick, RI 02893 USA | Model S48 | |
| SIU5 RF TRANSFORMER ISOLATION UNIT | Equipment | Grass Technologies, West Warwick, RI 02893 USA | Model SIU5 | |
| 5 Liter Water Jacketed Reservoir | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 120142-5 | |
| 2 Liter Water Jacketed Reservoir | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 120142-2 | |
| 0.5 Liter Water Jacketed Reservoir | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 120142-0 | |
| 0.25 Liter Water Jacketed Reservoir | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 120142-025 | |
| 10 ml Heating Coil | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 158822 | |
| Compliance Bubble Trap | Equipment | Radnoti Glass Technology Inc., Monrovia CA 91016 USA | 130149 | |
| Luer Disconnect Cannula | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | 72-1444 | |
| 3-Way stopcock, FLL to MLT, No Port Covers | Equipment | Harvard Apparatus, Holliston, MA 01746 USA | BS4 72-2630 | |
| Thermocouple Thermometer | Equipment | Cole Parmer, Vernon Hills, Illinois 60061 USA | WU-91100-40 | |
| Ultra Fine IT-Series Flexible Microprobe | Equipment | PhysiTemp Instruments Inc., Clifton, NJ 07013 USA | IT-24P | |
| Oscilloscope Tektronix TDS 1002 | Equipment | Tektronix Inc., Beaverton, OR 97005 USA | TDS 1002B | |
| 2,3-Butanedione monoxime | Reagent | Sigma, St. Louis, MO 63132 USA | B0753 | |
| Ketamine HCl | Reagent | Hospira Inc., Lake Forest, IL 60045 USA | RL-0065 | |
| Xylazine | Reagent | Lloyd Inc., Iowa 51601 USA | LB15705A | |
| E-TOXA-CLEAN® | Reagent | Sigma, St. Louis, MO 63132 USA | E9029 | |
| Di-8-ANEPPS | Reagent | Invitrogen, Carlsbad, CA 92008 USA | D-3167 |
References
- Efimov, I. R., Nikolski, V. P., Salama, G. Optical imaging of the heart. Circ Res. 95 (1), 21-33 (2004).
- Hucker, W. J. Images in cardiovascular medicine. Optical mapping of the human atrioventricular junction. Circulation. 117 (11), 1474-147 (2008).
- Entcheva, E., Bien, H. Macroscopic optical mapping of excitation in cardiac cell networks with ultra-high spatiotemporal resolution. Prog Biophys Mol Biol. 92 (2), 232-257 (2006).
- Entcheva, E. Fluorescence imaging of electrical activity in cardiac cells using an all-solid-state system. IEEE Trans Biomed Eng. 51 (2), 333-341 (2004).
- Stamm, C. Rapid endotoxin-induced alterations in myocardial calcium handling: obligatory role of cardiac TNF-alpha. Anesthesiology. 95 (6), 1396-1405 (2001).
- Choi, Y. H. Cardiac conduction through engineered tissue. Am J Pathol. 169 (1), 72-85 (2006).
- Skrzypiec-Spring, M. Isolated heart perfusion according to Langendorff—still viable in the new millennium. J Pharmacol Toxicol Methods. 55 (2), 113-126 (2007).
- Fedorov, V. V. Application of blebbistatin as an excitation-contraction uncoupler for electrophysiologic study of rat and rabbit hearts. Heart Rhythm. 4 (5), 619-626 (2007).

