Analyzing the Antigenic Relationship between Viruses using an Image-Based Microneutralization Assay
Abstract
Source: Lin, Y., et al. Optimization of a Quantitative Micro-neutralization Assay. J. Vis. Exp. (2016)
This video demonstrates the imaging-based micro-neutralization assay to analyze the antigenic relations between influenza A and B viruses. It provides a quantitative and visual way to assess the effectiveness of antibodies or other treatments in preventing viral infection.
Protocol
1. Virus Titration
NOTE: Depending on the number of viruses and the number of duplicates, a well plate can be set up with the following flexibilities: (1) The viral dilution can be arranged along either the rows or the columns (Figure 1). Each virus should occupy a separate row/column. (2) The viral dilution increment is flexible, but it should start with the highest viral concentration at the top-left corner. (3) There is no restriction on the number of duplicates.
- Aliquot sufficient MDCK cells or MDCK-SIAT cells8 into 96-well plates (200 µl/well). Incubate the cells at 37 °C with 5% CO2 for 2 or 3 days to reach confluence. Propagate the cells in DMEM supplemented with 10% heat-inactivated fetal calf serum (FCS) and antibiotics (100 U/ml penicillin and 100 µg/ml streptomycin). Incubate the SIAT cells at 37 °C with 5% CO2 and 1 mg/ml G418 sulfate.
NOTE: The dilution factor is dependent upon experience and the cell line used. The cell monolayer must be confluent (i.e., no cell wall or visible outline for MDCK cells and a tightly packed layout for MDCK-SIAT1 cells) at the time of virus inoculation. Examples of dilutions based on the subculture of confluent flasks of MDCK or MDCK-SIAT1 cells are given in Table 1. - Wash the cells 3 times with 200 µl of virus growth medium (VGM) per well. Sterilize the multiwall plate washer with 70% EtOH for 30 min, and then rinse it in sterile phosphate-buffered saline (PBS) or VGM before the wash.
- Aspirate the VGM and immediately add 50 µl of VGM to each well.
- Add 900 µl of VGM to each of 6 sterile tubes (or fewer, depending upon the number of test viruses and duplicates). Pipette 100 µl of virus into the first tube, mix well, and serially dilute, changing the tips between each dilution. Repeat for each virus to be titrated.
NOTE: This will give virus dilutions of 10-1 to 10-6. - Add 50 µl of each virus dilution, starting at the highest dilution (1 x 10-5 de Figure 1), to duplicated wells (Columns 9 and 10 in Figure 1) of a 96-well plate.
- Place the plate at 37 °C for 2-3 hr to allow the virus to infect the cells.
NOTE: A 2 to 3-hour incubation is necessary to ensure that the viruses in the inoculum have enough time to reach the monolayer. - Prepare the overlay (10 ml/plate)
NOTE: The overlay consists of 5 ml of 2x DMEM, Trypsin (2 µg/ml final concentration), 20 µl of 1 mg/ml stock, and 5 ml of cellulose cotton linters (see the Materials List). - Remove the inoculum.
- Add the overlay (200 µl to each well). Incubate overnight at 37 °C, UNDISTURBED.
NOTE: The virus budding takes place after approximately 4 hr, so it is important that the plate is not disturbed after this time. - Aspirate the overlay from the wells.
- Add ice-cold 4% paraformaldehyde in PBS A (200 µl/well). Place them at 4 °C for 30 min or at room temperature for 20 min.
NOTE: PBS A is natural-pH phosphate-buffered saline with 10 g of NaCl, 0.25 g of KCl, 1.437 g of Na2HPO4, 0.25 g of KH2PO4, and 1 L of distilled water (see the Materials List).
NOTE: Paraformaldehyde is harmful when inhaled and can cause burns if swallowed. There is also limited evidence of a carcinogenic effect, and it may cause sensitization after skin contact. - Aspirate the paraformaldehyde and wash the plates twice with PBS A (200 µl/well).
- Store the plates in PBS A at 4 °C for future use, or carry on with the following steps.
- Add permeabilization buffer (100 µl/well). Leave it at room temperature for 30 min.
- Wash the plate twice with PBS A (200 µl/well).
- Add 50 µl of the first antibody, mouse MAb against influenza type A (1:1,000 in ELISA Buffer), per well. Incubate it at room temperature for 1 hr (or 4 °C overnight), with shaking.
- Wash it 3 times in 100 µl of wash buffer (0.05% Tween 80 in PBS A, v/v) per well. Incubate it at room temperature for 5 min between washes. Alternately, wash it 3 times without incubation using 300 µl per well.
- Add 50 µl of the second antibody, goat anti-mouse IgG (H+L) HRP conjugate (1:1,000 in ELISA Buffer), per well. Incubate it at room temperature for 1 hr, with shaking.
- Wash it 3 times as described in step 1.17.
- Add the substrate (50 µl/well). Incubate it at room temperature for 30 min or until the development of a blue color is clearly visible.
- To stop the reaction, wash the plate twice with 200 µl of distilled water per well, incubating at room temperature for 2 -3 min between the washes.
- Air-dry the plate and store it in a dark place (e.g., wrap it in aluminum foil).
2. Virus Neutralization
NOTE: Calculate the number of plates required for the neutralization assay. Each plate can accommodate one virus and a number of antisera.
NOTE: Depending on the number of antisera and the number of duplicates, a well plate can be set up with the following flexibilities: (1) The serum dilution can be arranged along either a row or a column (Figure 2). Each serum should occupy a separate row or column. (2) The increment of serum dilution is flexible, but it should start with the highest serum concentration at the top-left corner of a plate. (3) The number of duplicates balances the number of antisera. More duplicates can help to smooth out experimental variations. (4) The number of the VC and the number of the cell control (CC) duplicates are flexible, but they should also be in separate rows or columns. It is expected that the number of VC duplicates is significantly greater than that of the antisera.
- Prepare the cell monolayer, as in steps 1.1-1.3, 2 or 3 days in advance.
- Add 50 µl of VGM to each well of the plate.
- Use Columns 11 and 12 for VC and CC, respectively. Add 50 µl aliquots of 1:20 receptor-destroying enzyme (RDE)-treated serum to the first row (A) of Columns 1-10.
NOTE: For each virus and serum combination, the assay is performed in duplicate, so one plate may, for example, contain one virus tested against five antisera. - Perform 2-fold serial dilutions by transferring 50 µl from Row A to Row H (Columns 1-10 in Figure 2) and discarding 50 µl from Row H.
- Add 50 µl of diluent to each well of the CC column (Column 12 in Figure 2).
- Add 50 µl of the virus to each well of the plate, except for the CC column (Columns 1-11, Rows A-H in Figure 2).
- Incubate it at 37 °C for 2-3 hr. Remove the inoculum. Add the overlay (200 µl) to each well. Incubate it overnight at 37 °C, UNDISTURBED. Fix and stain the plates as for the virus titration (steps 1.11-1.22).
3. Neutralization Quantification
- Place a well plate in the scanning area of a flatbed scanner, as shown in Figure 2a. Use the L-shape position limit to ensure an optimum and repeatable imaging location.
NOTE: It is possible to image two well plates in one scan. - Scan the plate.
- Run the "Wellplate Reader" software to calculate the required viral titers (Figure 3).
- Click the "Load image" button to load an image. Slide the red bar in "Global Threshold" to adjust the sampling threshold. Click the "Update" button to examine the effect.
- Tick the "Calculate Neutralization/Titration" box. Click the "Sampling" button to quantify the ICP. Click the "Save" button to save the sampling results when prompted.
NOTE: After the sampling process, a new window called "Neutralization & Titration Calculation" appears automatically if the "Calculate Neutralization/Titration" box is ticked. - Load or input the well-plate map. Indicate the threshold (e.g., 50%) in "Neutralization: Infection Deduction (%)."
- Select "Neutralization Process" to calculate the titers. Check the titers and click the "Save & Close" button to save the neutralization results.
NOTE: Neutralization is reported as the reciprocal of the highest dilution of the serum with the predefined ICP reduction (such as 50% or 80%) against the VC (the mean of Column 11 in Figure 4. A 50% ICP reduction was used in the Representative Results.
Representative Results
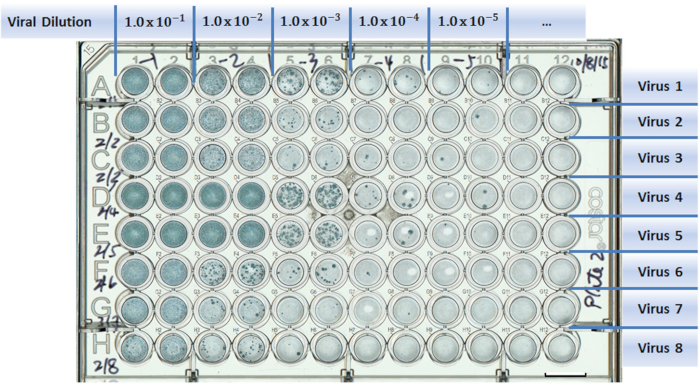
Figure 1: Example of a well-plate setup in a virus titration experiment. Test viruses are assigned to separate rows (A to H). Columns are designed for different viral dilutions (1 to 12). Two columns were used as duplicates for each viral dilution. Scale bar = 10 mm.

Figure 2: Schematics of a sample scanning system. (a) A 96-well plate on the imaging position of a flatbed scanner and (b) dimensions of the L-shape position limit shown in (a).
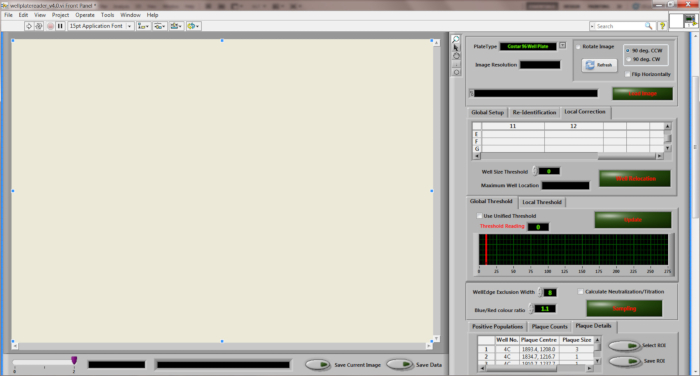
Figure 3: Desktop diagram of the "Wellplate Reader" quantitation software.
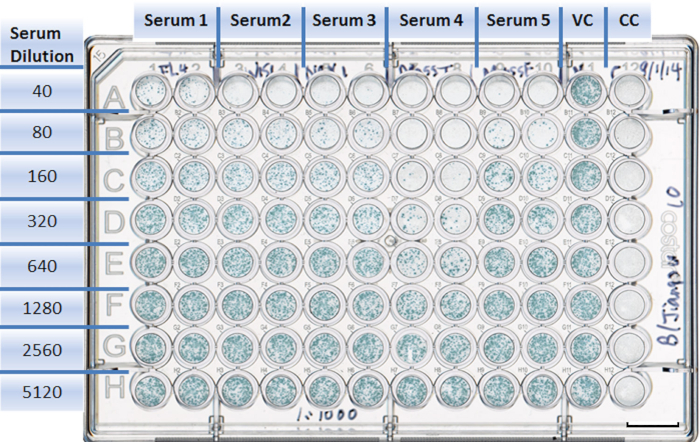
Figure 4: Example of a well-plate setup in a neutralization experiment. Each antiserum is assigned to two columns with duplicates (1 to 10). Rows are designed for different serum dilutions (A to H). The virus control takes Column 11, with eight duplicates (A11 to H11). The cell control is in Column 12, with eight duplicates (A12 to H12). Scale bar = 10 mm.
Divulgations
The authors have nothing to disclose.
Materials
| VGM | Sigma | D6429, P0781 | 500 ml DMEM+5 ml Pen/Strepb |
| PBS A | Nature pH Phosphate-buffered saline: NaCl 10 g, KCl 0.25 g, Na2HPO4 1.437 g, KH2PO4 0.25 g, and Dist. Water 1 L | ||
| Avicell | FMC | RC-581F | 2.4 g in 100 ml distilled water dissolved by agitation on a magnetic stirrer for 1 hr. Sterilize by autoclaving |
| 2x DMEM | Gibco | 21935-028 | |
| Trypsin | Sigma | T1426 | |
| Overlay (10 ml/plate): | 5 ml 2x DMEM, Trypsin 2 μg/ml final concentration, Avicell 5 ml | ||
| Triton X- 100 e | Sigma | T8787 | Permeabilization buffer: 0.2% in PBS A (v/v) |
| Tween 80 | Sigma | P5188 | Wash Buffer: 0.05% Tween 80 in PBS A (v/v) |
| Horse serum | PAA Labs Ltd | B15-021 | ELISA Buffer: 10% in PBS A (v/v) + 0.1% Tween 80 |
| Mouse MAb against influenza type A | Biorad | MCA 400 | 1 st Antibody: 1:1,000 in ELISA Buffer |
| Goat anti-mouse IgG (H+L) HRP conjugate | Biorad | 172-1011 | 2 nd Antibody: 1:1,000 in ELISA Buffer |
| True Blu peroxidase substrate | KPL | 50-78-02 | Substrate: True blue +0.03% H2O2 g (1:1,000 of 30% solution) |
| 96-well flat-bottom microtitre plates | Costar | 3596 | |
| 8 channel multiwall-plate washer and manifold | Sigma | M2656 | |
| Perfection Plate Scanner | Epson | V750 Pro | The imaging software can be downloaded freely from http://www.epson.com/cgi-bin/ Store/support/supDetail.jsp? oid=66134&infoType=Downloads. |
| Software operational environment: LabVIEW | National Instruments Corporation | Window based with NI Vision builder | Version: LabVIEW2012 or above |

