Construction of a Preclinical Multimodality Phantom Using Tissue-mimicking Materials for Quality Assurance in Tumor Size Measurement
Summary
This paper describes in-house procedures of constructing a preclinical multimodality phantom made of tissue-mimicking (TM) materials for quality assurance (QA) of tumor size measurement in animal imaging modalities such as ultrasound (US), computed tomography (CT) and magnetic resonance imaging (MRI).
Abstract
World Health Organization (WHO) and the Response Evaluation Criteria in Solid Tumors (RECIST) working groups advocated standardized criteria for radiologic assessment of solid tumors in response to anti-tumor drug therapy in the 1980s and 1990s, respectively. WHO criteria measure solid tumors in two-dimensions, whereas RECIST measurements use only one-dimension which is considered to be more reproducible 1, 2, 3,4,5. These criteria have been widely used as the only imaging biomarker approved by the United States Food and Drug Administration (FDA) 6. In order to measure tumor response to anti-tumor drugs on images with accuracy, therefore, a robust quality assurance (QA) procedures and corresponding QA phantom are needed.
To address this need, the authors constructed a preclinical multimodality (for ultrasound (US), computed tomography (CT) and magnetic resonance imaging (MRI)) phantom using tissue-mimicking (TM) materials based on the limited number of target lesions required by RECIST by revising a Gammex US commercial phantom 7. The Appendix in Lee et al. demonstrates the procedures of phantom fabrication 7. In this article, all protocols are introduced in a step-by-step fashion beginning with procedures for preparing the silicone molds for casting tumor-simulating test objects in the phantom, followed by preparation of TM materials for multimodality imaging, and finally construction of the preclinical multimodality QA phantom. The primary purpose of this paper is to provide the protocols to allow anyone interested in independently constructing a phantom for their own projects. QA procedures for tumor size measurement, and RECIST, WHO and volume measurement results of test objects made at multiple institutions using this QA phantom are shown in detail in Lee et al. 8.
Introduction
Assessment of the change in tumor size is an important endpoint for evaluating the activity of anti-tumor drugs in both tumor shrinkage and disease progression 9, 10. World Health Organization (WHO) and Response Evaluation Criteria in Solid Tumors (RECIST) are the codified methods for the anatomic assessment of tumor lesions in imaging modalities such as ultrasound (US), computed tomography (CT) or magnetic resonance imaging (MRI). For WHO criteria, the product of tumor maximum diameter and its largest perpendicular diameter in the transverse plane for target regions is calculated 4. In contrast, for RECIST, the sum of longest diameters in the transverse plane for a limited number of target lesions is calculated 4. In spite of continuously growing interest in tumor therapeutic response assessment, there has been no preclinical quality assurance (QA) phantom/QA procedures for the imaging biomarker.
Considering that tumor size measurement based on WHO criteria and/or RECIST is the only imaging biomarker approved by the United States Food and Drug Administration (FDA), as a starting point of QA for any other imaging biomarkers, Lee et al. designed and constructed UTHSCSA/Gammex Mark 1 and Mark 2 phantoms for QA of tumor size measurement in collaboration with Gammex Inc 7. The Mark 1 phantom was a revised version of a Gammex commercial US phantom and thus, the size was too large to fit into animal CT and MR scanners. Also some tools in the Mark 1 phantom were unnecessary for tumor size measurement. The Mark 2 phantom was designed based on RECIST which is the most recent FDA-approved imaging biomarker. However, the size of the Mark 2 phantom was still too large for MR scanners, and CT and MR image quality of the phantom was not acceptable for accurate tumor size measurement 7.
The QA phantom described herein was re-designed to overcome shortcomings of the previous phantoms and constructed using modified tissue-mimicking (TM) materials and protocols developed in our lab. This paper describes the details of the protocols for phantom construction: First, methods are introduced for preparing the silicone molds needed for casting tumor-simulating test objects and for assembling a rotator for rotating a phantom to prevent gravitation sedimentation. Second, protocols for preparing TM materials modified from D’Souza et al.’s for US, CT and MR imaging are described 11. The physical properties of the TM materials were tested in each modality to ensure that the TM materials represented human soft tissues as observed in the images acquired with the various modalities, but the results are not displayed here. Third, the protocol for phantom construction is described. Lastly, US, CT and MR images of the phantom are presented as results.
Protocol
1. Phantom Design
A drawing of the preclinical multimodality phantom is shown in Figure 1 7, 8. The size of the phantom is 38 mm in diameter and 115 mm in length to allow the phantom to be scanned in various animal scanners. The phantom contains five tumor-simulating test objects (diameter: 14, 10, 7, 4 and 2 mm) placed at a depth of 10 mm within the phantom.
2. Silicone Mold Construction
Silicone molds are prepared to cast the tumor-simulating test objects as described in this section 7. All acrylic plates and rods required for preparation of the silicone molds are cut with an accuracy of 25 μm in the machine shop at the University of Texas Health Science Center at San Antonio (UTHSCSA).
- Make five holes (diameter: 14, 10, 7, 4, 2 mm) for test objects and another five holes (diameter: 6 mm) for alignment rods in two acrylic base plates (size: 4.2 cm × 11.5 cm × 0.9 cm) (Figure 2A).
- Cut spacer pairs with height of 7, 5, 3.5, 2 and 1 mm (size: 1.0 cm × 5.5 cm) (Figure 2B).
- Prepare steel balls (diameter: 14, 10, 7, 4 and 2 mm, accuracy: 2.5 μm).
- Place two spacer pairs with height of 7 mm and a base plate onto a thin acrylic plate in sequence and tighten them using C-clamps (Figure 2C).
- Insert the steel ball with 14 mm diameter into 14 mm hole of the base plate and glue it using J-B KWIK (Figure 2C). Repeat the procedures for the rest of the balls (Figure 2D) and for the other base plate. Note that the steel balls in two base plates are glued as mirror images 7.
- Attach four 2.5 cm-high acrylic plates (size: 2.5 cm × 11.5 cm for two plates and 2.5 cm × 4.2 cm for another two plates) onto each base using masking tape like fences (Figure 3A).
- Attach the top plate (size: 4.2 cm × 11.5 cm, five holes with 0.8 cm of diameter, ten holes with 1.2 cm of diameter) in one of the base plate assemblies to insert five acrylic rods (diameter: 0.8 cm and length: 0.5 cm) with 1 mm tips, and to insert five alignment rods (diameter: 0.9 cm and length: 5.0 cm) and to pour silicone (Figure 3A).
- Insert the acrylic rods into the 0.8 cm holes in the top plate all the way to the top of steel balls and glue them using silicone glue. Then insert alignment rods into the holes in the base plate through larger holes in the top plate (Figure 3A).
- Mix part A of silicone rubber compound with part B in the ratio of 10 to 1 by weight.
- Pour the silicone rubber compound into the assembly and dry the assembly at room temperature for about 24 hr (Figure 3B).
3. Rotator Assembly
The rotator is prepared from PVC pipe and a rotisserie motor.
- Grind the end of a bolt to fit the hole of a rotisserie motor.
- Screw the ground bolt to the end of the PVC pipe (length: 270 mm and inner diameter: 75 mm) using a nut and a washer.
- Bend metal plates and glue them onto a plastic plate using J-B KWIK to support the PVC pipe and to adjust the height of PVC pipe 7.
4. TM Material Preparation
The protocols for preparing the TM materials are modified from those developed in Dr. Ernest L. Madsen’s laboratory at the University of Wisconsin Madison and more details are in Lee et al. 8,11.
4.1 Background TM material preparation
- Pass commercial whole milk (200 cc) through 20 μm and then 10 μm mesh filters.
- Dissolve Thimerosal (0.2 g) in the filtered milk (100 cc).
- Using house vacuum, degas this milk solution for 30 sec at room temperature.
- Dissolve dry agarose (2 g) in deionized water (18 MΩ) (100 cc) at room temperature.
- Then add 1-propanol (7.9 cc) and BaSO4 (1 g) to the agarose solution.
- Degas the agarose solution and then heat it in a 95 °C water bath until the agarose solution clears.
- While the agarose solution clears in the 95 °C water bath, heat the condensed milk in a 55 °C water bath.
- Move the molten agarose solution to the 55 °C water bath to cool down.
- Once both solutions are at 55 °C, mix agarose solution (50 cc) with condensed milk (50 cc) to make the ratio of 50 to 50 by volume and slowly stir the mixture followed by air bubble removal from the surface.
- Then add EDTA (0.103 g) and CuCl2·2H2O (0.06 g) to the agarose-milk mixture followed by sufficient stirring to ensure homogeneity.
- Lastly, add glass beads (15 – 60 μm diameter, mean diameter: 35 μm) (0.1 g) and stir the final mixture repeatedly. Prior to use, soak the glass beads in concentrated nitric acid for 24 hr to remove any impurities and then rinse off the acid.
4.2 Test object TM material preparation
The test object TM material is prepared in a similar manner as the background TM material except for the following compositional differences:
- Pass commercial whole milk (20 cc) through 20 μm and then 10 μm mesh filters.
- Dissolve Thimerosal (0.02 g) in the filtered milk (10 cc).
- Dissolve dry agarose (0.60 g) in a room temperature solution of deionized water (10 cc) and 1-propanol (0.79 cc).
- Degas the agarose solution and then heat it in a 95 °C water bath until the agarose solution clears.
- While the agarose solution clears in the 95 °C water bath, heat the condensed milk in a 55 °C water bath.
- Move the molten agarose solution to the 55 °C water bath.
- Once both solutions are at 55 °C, mix the agarose solution (5 cc) with condensed milk (5 cc) and slowly stir the mixture followed by air bubble removal from the surface.
- Then add EDTA (0.0017 g) and CuCl2·2H2O (0.0010 g) to the agarose-milk followed by sufficient stirring.
5. Multimodality Phantom Assembly
Using the silicone molds, the following steps are accomplished to construct the multimodality phantom.
- On the silicone mold without 1 mm holes, attach nylon thread along the center of spheres and glue it at both ends of the mold using silicone glue (Figure 4A).
- Using a soft brush, apply silicone grease on the surface of two molds (Figure 4A) and assemble two molds using alignment rods.
- Prepare test object TM material as described in section 4.2 and pour it through 1 mm holes of the silicone mold using a 22-gauge needle of a syringe.
- To allow the test objects to set, store the molds in a refrigerator (5 °C) for about 30 min.
- In each side of a half-cylindrical container (length: 115 mm and diameter: 38 mm), make two holes of 1 mm at a depth of 10 mm from the phantom surface in order to mount nylon thread with test objects. Make an additional hole of 6 mm to pour background TM material.
- Unload the test objects with nylon thread from the molds (Figure 4B) and then mount them in the half-cylindrical container (Figure 4C).
- Using 3M Scotch-Weld DP-100 and 3M duct tape, adhere thin non-conducting aluminum (thickness: 0.12 mm) onto the acrylic container. Block the 1 mm holes in the acrylic container using the same glue (Figure 4C).
- Prepare background TM material quickly and slowly pour it into the 6 mm hole of the container using a small plastic funnel.
- After removal of any air bubbles, glue the 6 mm hole using 3M Scotch-Weld DP-100.
- Once assembled, rotate the phantom at 2 rpm in the rotator for 4 to 5 hr at room temperature.
- Remove the nylon thread after the TM materials in the phantom completely hardens.
6. Multimodality Imaging
The phantom is scanned in preclinical ultrasound, CT and MRI and images in three modalities are acquired. The imaging protocols are described in detail in Lee et al. 7, 8.
Representative Results
Figure 3B and Figure 5 show two silicone molds for casting test objects, and the multimodality phantom, respectively. The length × width × depth of each mold is 109 mm × 37 mm × 21 mm and two molds are identical mirror-images. One mold has 1 mm holes where TM material can be inserted using a thin needle. Each mold has an additional five holes for alignment rods. The length × width × depth of the phantom is 115 mm × 38 mm × 24 mm and its initial mass was 101.02 g. The size of the phantom is adequate to fit into preclinical scanners.
Images acquired by US, CT and MRI are shown in Figure 6. The contrast between test objects and background is sufficient to distinguish test objects and measure their sizes. No severe artifacts are observed in any images except for small reverberation in the US images.
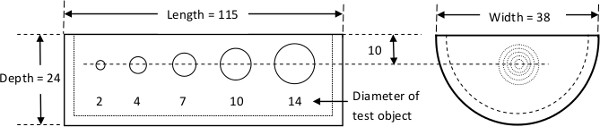
Figure 1. Design of a preclinical multimodality phantom. The phantom has five tumor-simulating test objects with diameter of 2, 4, 7, 10 and 14 mm placed at 10 mm from the phantom surface.

Figure 2. Preparation for casting silicone molds. A. A base plate with five holes for test objects and another five holes for alignment rods. B. Spacer pairs with height of 7, 5, 3.5, 2 and 1 mm.C. Gluing steel balls using thin acrylic plate, spacers, base plate and C-clamps, D. A base plate with five steel balls glued.

Figure 3. Procedures for casting silicone molds. A. Construction of base plate assemblies before pouring silicone compound. B. Silicone molds.

Figure 4. Procedures for casting test objects using silicone molds. A. Preparation before casting test objects in silicone molds using nylon thread, silicone grease and alignment rods. B. Test objects in the silicone mold before unloading. C. Mounting of test objects in an acrylic container.

Figure 5. A multimodality phantom made of tissue-mimicking materials. The phantom fit into various animal scanners in multiple institutions.
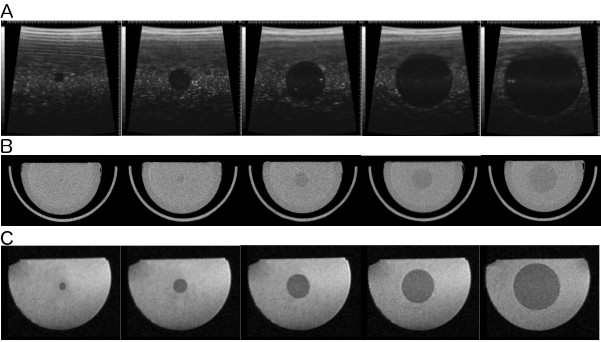
Figure 6. A. US, B. CT and C. T2 weighted MR images of the phantom. Images do not show severe artifacts and air bubbles. Contrast between test objects and background was adequate for size measurement.
Discussion
The goal of this article was to provide the methods for making TM materials for multimodality imaging and constructing a preclinical multimodality phantom as a QA tool for accurate tumor size measurement using different modalities in multiple institutions. As previously mentioned, TM materials were originally developed by Dr. Ernest L. Madsen’s laboratory at the University of Wisconsin Madison for a multi-imaging modality prostate phantom. We modified Dr. Madsen’s TM material protocols for our own purpose in order to have adequate contrast between test objects and background and to represent the physical properties of soft tissue in US, CT and MR images. The methods for phantom construction using our own TM material protocols were briefly introduced by Lee et al. for the first time 7, 8. In this paper, protocols of the TM materials and phantom construction were explained in detail.
Prior to TM material preparations, silicone molds and a rotator were customized in our lab. Since silicone molds can shrink in the process of drying, it is important to choose the right silicone compound for mold preparation. We measured the diameter of each object in the molds using a NIST-traceable caliper after they hardened to ensure that there was minimal shrinkage. The rotator was necessary to prevent gravitational sedimentation of glass beads in the background material.
TM materials were made of several chemicals for the following reasons 7, 11, 12: Milk has the same properties as human tissue; Thimerosal prevents bacterial invasion in milk; Mesh filters remove any impurities that may have been introduced during the prior concentrating and commercial packaging of milk; Agarose is a bonding material and MR T2 relaxation time modifier; Deionized water doesn’t include metal ions which lower the relaxation times unlike tap water; Propanol increases the speed of sound for water (1,484 m/sec) to that for soft tissues (1,540 m/sec); BaSO4 is for CT contrast enhancement; Cu2+/EDTA decreases MR T1 relaxation time; Glass beads are for US contrast enhancement. The contrast in images and physical properties are discussed in Lee et al. 8.
TM material of test objects should be degassed and slowly injected through 1 mm-hole in silicone mold using a syringe to avoid air bubbles in test objects. Once test objects are cast in silicone molds, they should be loaded into an acrylic phantom immediately and the top of the phantom should be covered and glued immediately as well to prevent dehydration of test objects.
Periodic weighing of the phantom is necessary to check dehydration. Our findings showed that there was a maximum 1.68% weight loss in a year in our phantoms 8, which is acceptable for the phantom application. This loss can be corrected by periodically injecting neat replacement water. However, the effect of weight loss on changes in the images needs to be investigated by scanning the phantom and measuring the size of test objects periodically. It is also important to keep the phantom at room temperature and away from humidity to prevent dehydration.
The current QA phantom does not take into account the variability in shape observed in typical animal or human tumors. Thus, a phantom with test objects of irregular shape will need to be constructed and tested as our future study 8. Nonetheless, the current phantom is still usable for other purposes, e.g. accurate imaging system calibration, testing the accuracy of a measurement tool in US, CT or MR systems, and so on. It also can be used clinically with revision of the phantom size.
For tumor size measurement QA using the phantom, small animal imaging systems that have capability of providing three dimensional images (width, length and depth in Figure 5) are required. QA procedures for accurate tumor size measurement including scanning phantom and imaging protocols have been developed 8. For reproducibility of the image quality, the same imaging protocols including the same MR coil used in this study are recommended since imaging contrast depends on imaging parameters. The details of imaging protocols are referred to in our previous articles 7,8 and they are based on small animal imaging protocols that have been used at UTHSCSA. US, CT and MR images obtained in this study had adequate contrast to measure the size of test objects (Figure 6). However, the quality of US and CT images is not as good as that of MR images. In US, more gel should be used to have better contact between phantom membrane and transducer on the surface. For better contrast in US images, a slightly increase in the amount of glass beads in the background TM preparation could be used as long as the US properties are within the range for soft tissues. Similarly, more BaSO4 can be added to the background TM material to improve CT contrast. Another way to improve CT contrast would be to decrease the x-ray tube voltage or to increase tube current, but small animal CT scanners have limited options for modifying these tube parameters.
RECIST, WHO and volume measurement results of test objects are not displayed here since they are out of the scope of this paper. Lee et al. 8 briefly presented experimental data analyzed from three independent measurements on US, CT and MRI in two institutions. In UTHSCSA, standard deviations (SD) of three measurements in diameter of test objects ranged from 0 to 0.06 mm, from 0.01 to 0.26 mm and from 0.01 to 0.09 mm for US, CT and MRI, respectively in three perpendicular directions and in five different diameters. In UC Denver, SDs ranged from 0.02 to 0.21 mm, from 0.01 to 0.31 mm, from 0.06 to 0.29 mm for US, CT and MRI, respectively. Further information is presented in Lee et al. 7, 8. Another future study will include more observers to investigate inter-observer variability.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors are thankful to Dr. Madsen at the University of Wisconsin-Madison and Cristel Baiu at Gammex Inc. for providing advice on TM materials. The authors are also grateful to Dr. Malcolm David Murray for providing the methods to construct the phantom.
Materials
| Reagent/Material | |||
| PVC pipe | N/A | N/A | Home Depot |
| Bolt, nut, washer and metal plates | N/A | N/A | Home Depot |
| Acrylic plates and rods | N/A | N/A | Plastic supply in San Antonio, TX |
| Steel balls | Nordex, Inc. | AEC-M2-2, -4, -7, -10 and -14 | 2, 4, 7, 10 and 14 mm diameter |
| C-clamps | Adjustable Clamp | 1420-C | 2 inch length |
| Masking tape | 3M Industrial Adhesives and Tapes | 2600 | |
| Duct tape | 3M Industrial Adhesives and Tapes | S-3763SIL | |
| J-B KWIK | J-B WELD Co. | 380238 | |
| 3M Scotch-Weld Epoxy Adhesive | 3M Industrial Adhesives and Tapes | DP-100 | |
| Silicone grease | Permatex, Inc. | 22058 | |
| Silicone glue | DAP, Inc. | 688 | |
| Silicone rubber compound | Smooth-ON, Inc. | Smooth-SilTM950 Part A and B | A:B mix ratio = 10:1 by weight |
| Brush | N/A | N/A | Hobby Lobby |
| Syringe | Becton Dickinson | 309604 | 10 ml |
| Needle | Becton Dickinson | 305156 | 22-gauge 1.5 inch length |
| Funnel | N/A | N/A | |
| Mesh filters | Small parts, Inc. | CMN-0010-C and CMN-0020-C | 10 and 20 μm |
| Whole milk | N/A | N/A | HEB in San Antonio, TX |
| Thimerosal | Sigma-Aldrich Co. | T5125 | |
| Propanol | Sigma-Aldrich Co. | 33538 | |
| EDTA | Sigma-Aldrich Co. | 431788 | |
| CuCl2 | Sigma-Aldrich Co. | 459097 | |
| Agarose | Sigma-Aldrich Co. | A0169 | |
| BaSO4 | Sigma-Aldrich Co. | B8675 | |
| Glass beads | Potters Industries, Inc. | 3000E | |
| PET/AL/LLDPE* | Pechiney Plastic Packaging, Inc. | Pechiney Spec 151 | Phantom cover material |
| *Polyethylene terephthalate/aluminum/linear low density polyethylene | |||
| Equipment | |||
| Rotisserie motor | Brinkmann | 812-7103-S | Home Depot |
| Water bath 1 | Precision, Inc. | Model: 282, Serial #: 601091552 | |
| Water bath 2 | VWR, Inc. | Model: 1212, Serial #: 08119606 | |
| Ultrasound | Visualsonics | Serial #: 770/120-259 | |
| CT | Gamma Medica-Ideas | Serial #: GR 0050 | |
| MRI | Bruker | Part #: W3301390, Serial #: 0030 |
References
- Prasad, S. R., et al. CT tumor measurement for therapeutic response assessment: Comparison of unidimensional, bidimensional, and volumetric techniques-Initial observations. Radiology. 225 (2), 416-419 (2002).
- Cortes, J., et al. Comparison of unidimensional and bidimensional measurement in metastatic non-small cell lung cancer. Br. J. Cancer. 87 (2), 158-160 (2002).
- Saini, S. Radiologic measurement of tumor size in clinical trials: past, present, and future. AJR Am. J. Roentgenol. 176 (2), 333-334 (2001).
- Suzuki, C., et al. Radiologic measurements of tumor response to treatment: practical approaches and limitations. Radiographics. 28 (2), 329-344 (2008).
- Therasse, P., et al. RECIST revisited: A review of validation studies on tumour assessment. Eur. J. Cancer. 42 (8), 1031-1039 (2006).
- O’Connor, P. B., et al. Quantitative imaging biomarkers in the clinical development of targeted therapeutics: current and future perspectives. Lancet Oncol. 9 (8), 766-776 (2008).
- Lee, Y. C., et al. Preclinical multimodality phantom design for quality assurance of tumor size measurement. BMC Med. Phys. 11 (1), (2011).
- Lee, Y. C., et al. QA procedures for multimodality preclinical tumor drug response testing. Med. Phys. 37 (9), 4806-4816 (2010).
- Park, J., et al. Measuring response in solid tumors: comparison of RECIST and WHO response criteria. Jpn. J. Clin. Oncol. 33 (10), 533-537 (2003).
- Eisenhauer, E. A., et al. New response evaluation criteria in solid tumors: revised RECIST guideline (version 1.1). Eur. J. Cancer. 45 (2), 228-247 (2009).
- D’Souza, W. D., et al. Tissue mimicking materials for a multi-imaging modality prostate phantom. Med. Phys. 28 (4), 688-700 (2001).
- Mitchell, M. D., et al. Agarose as a tissue equivalent phantom material for NMR images. Magn. Reson. Imaging. 4 (3), 263-266 (1986).

