Imaging Through the Pupal Case of Drosophila melanogaster
Summary
This paper demonstrates the use of a fast scanning confocal microscope to image cell behavior directly through the puparium. By leaving the pupal case intact, this method allows observation and measurement of dynamic cell processes at a stage of Drosophila development that is difficult to study directly.
Abstract
The longstanding use of Drosophila as a model for cell and developmental biology has yielded an array of tools. Together, these techniques have enabled analysis of cell and developmental biology from a variety of methodological angles. Live imaging is an emerging method for observing dynamic cell processes, such as cell division or cell motility. Having isolated mutations in uncharacterized putative cell cycle proteins it became essential to observe mitosis in situ using live imaging. Most live imaging studies in Drosophila have focused on the embryonic stages that are accessible to manipulation and observation because of their small size and optical clarity. However, in these stages the cell cycle is unusual in that it lacks one or both of the gap phases. By contrast, cells of the pupal wing of Drosophila have a typical cell cycle and undergo a period of rapid mitosis spanning about 20 hr of pupal development. It is easy to identify and isolate pupae of the appropriate stage to catch mitosis in situ. Mounting intact pupae provided the best combination of tractability and durability during imaging, allowing experiments to run for several hours with minimal impact on cell and animal viability. The method allows observation of features as small as, or smaller than, fly chromosomes. Adjustment of microscope settings and the details of mounting, allowed extension of the preparation to visualize membrane dynamics of adjacent cells and fluorescently labeled proteins such as tubulin. This method works for all tested fluorescent proteins and can capture submicron scale features over a variety of time scales. While limited to the outer 20 µm of the pupa with a conventional confocal microscope, this approach to observing protein and cellular dynamics in pupal tissues in vivo may be generally useful in the study of cell and developmental biology in these tissues.
Introduction
The vinegar fly, Drosophila melanogaster, is a well-established model for studying many aspects of biology. Drosophila research has a rich history of genetic experimentation that allows sophisticated forms of gene manipulation including expression, knockdown and mutation. With the advent of fluorescent protein labels, this repertoire has expanded to include studies of cells and proteins in living animals. The fly embryo is an excellent system for such studies as it is small and optically clear allowing deep, high-resolution imaging in vivo1-3. Other stages of fly development have proven to be less tractable, requiring anaesthetization4, dissection and short term culture5,6, or the creation of windows in the cuticle for imaging7,8. These manipulations usually compromise animal development in the long term or affect the animal in ways that limit imaging to short periods.
To study novel mutations in genes that resemble cell cycle regulators, it was essential to find an appropriate preparation to study the timing and fidelity of the cell cycle. Since most embryonic cell cycles are truncated (S-M or S-G2-M) and the mutants under study do not show defects until later stages, it was important to observe the cell cycle in pupal stage tissues. Epithelial cells in the pupa have a more typical G1-S-G2-M cell cycle and pupae of this stage are not capable of muscle movements9. The initial starting point for manipulations included intact whole pupae expressing Histone2AV-GFP. Despite the apparent opacity of the pupal case, this intact preparation proved to be excellent for long-term in vivo imaging. This technique is simple enough that undergraduate researchers routinely use it to study aspects of cell and developmental biology in Drosophila and yet the resolution is fine enough to allow discrimination of micrometer scale features. With this method, observations of events over hours, minutes, or seconds are possible simply by adjusting time series parameters. Videos using blue, green, yellow, orange, and red fluorescent proteins, or combinations of these, have been made. Importantly, if care is taken to minimize the laser intensity, even long-term imaging has no effect on development or viability of the animals.
Protocol
1. Fly Work
- Maintain flies on standard cornmeal-agar-molasses-yeast medium at room temperature10.
- For crosses, isolate virgins within 6 hr of eclosion. After crossing to males of the desired genotype, change flies to new vials every 3-4 days.
Note: For these experiments, Gal4 line A9 was used to drive expression of transgenes in the wing. Fly stocks can be obtained from the stock center in Bloomington. Stocks used in these experiments include A9-Gal4 (Bl#8761), His2Av-GFP (Bl#5941), Sco/CyO HsCre (Bl#1092), UAS-ChRFP-Tub (Bl#25773), lollibow11.
2. Selection and Mounting of Pupae
- Stage pupae either by collecting them as white prepupae (WPP) and keeping them at 25 °C until they reach the appropriate age or by using morphological criteria12.
- To observe cell divisions, select pupa that have recently undergone head eversion. From this time until just before eclosion (at >96 hr), pupae are immobile allowing extended time-lapse observation.
- Remove pupae from the vials by first touching them with a paintbrush moistened with water, waiting for a minute to allow the water to loosen the adhesive and then gently prodding them onto the paintbrush.
- Gently wash selected pupae by prodding them with a paintbrush in water to remove the salivary gland adhesive and food particles.
- Transfer clean pupae to a 25 mm Petri dish with a coverslip bottom (number 1 ½ coverslips).
- Using thin strips of either modeling clay or dental wax as a support, mount pupae so that the tissue of interest is closest to the coverslip.
Note: In the studies described here, pupal wings, legs, abdominal histoblasts or dorsal notum have been observed. In practice, any tissue within 20 µm of the surface of the pupal case is observable. - Orient pupae carefully so that the tissue of interest is parallel to the glass coverslip surface.
- Once pupae are mounted, use a paintbrush to transfer a thin layer of thiodiethylene glycol (TDG) to the space between the pupa and the coverslip.
Note: TDG reduces surface scattering, matches the refractive index of oil, and allows oxygen to permeate the tissue13. Use of oils is not recommended, as they tend to deprive the tissues of oxygen, causing rapid cessation of cell behaviors.
3. Imaging
Note: For imaging, a confocal microscope is likely to be essential as the confocality removes most of the obscuring background caused by intense illumination of the pupal case.
- Adjust settings on the confocal to minimize the impact of illumination on pupal development and viability. To do so, strike a balance between the intensity of excitation and the sensitivity of collection. A typical configuration for imaging with the Leica SP5 used the resonant scanner (8,000 Hz), laser power set to 2% (10% transmittance of 20% power), pinhole set to 120 µm, and line averaging set to 8. Settings will vary depending on experimental conditions.
Note: To resolve fine scale features 40X and 63X Oil immersion lenses (0.1 mm working distance) have also been successfully used in this method, though they limit the depth of focus.
4. Analysis
To analyze frames, z-stacks or movies import the data files to FIJI, which has effective tools for viewing, measuring, and modifying files for presentation14. For example time to complete mitosis was measured using the sampling interval and multidimensional image browser to step through frames while watching a cell from prophase to telophase. x-, y-, and z-dimensions can be measured using the measuring tool. For detailed instructions on the software and its use see: http://fiji.sc/Fiji.
Representative Results
Cells in pseudostratified epithelia, such as the developing Drosophila eye, or the ventricular layer of the developing vertebrate central nervous system, undergo nuclear movements, termed interkinetic nuclear migration, in time with the cell cycle. DNA replication occurs when nuclei are at or near the basal surface and cells enter mitosis when the nuclei reach the apical surface15,16. The pupal wing cells form a rapidly dividing monolayer epithelium during the first several hours after head eversion. Data were collected in xyzt mode from an early pupal wing, taking sections every 2 µm at 1 min intervals. In the resulting 3D movies mitosis was only seen at the apical surface (Figure 1A). Sections taken at more basal locations showed no signs of mitosis (Figure 1B) but nuclear movements occurred as the newly divided nuclei returned from the apical surface.
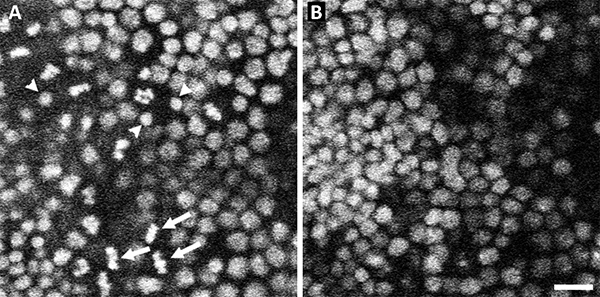
Figure 1. Nuclei move to the top of the epithelium to divide. Representative sections from a xyzt confocal image of wild-type Drosophila pupal wing expressing His2AvGFP. At the top-most sections (A) nuclei can be seen in different phases of the cell cycle. Nuclei in metaphase (arrows) are abundant in this micrograph as are nuclei in telophase (arrowheads). At lower planes of section (B) no evidence of mitosis is apparent though there is variation in nuclear diameter, which is indicative of the state of DNA replication. Scale Bar 10 µm. Click here to view larger image.
Discussion
To visualize, measure, and quantify features of dividing cells, required development of a simple preparation for observing mitosis in the living pupal wing of Drosophila by means of confocal analysis of His2AvGFP expressing cells. This method was used to document that the cell cycle in the pupal wing bears strong similarities to cell cycles in pseudostratified epithelia in that nuclei move to the apical surface of the epithelium where they enter mitosis. Following telophase, nuclei drop back into the epithelial layer. Hence, the cells of this monolayer, apparently unstratified epithelium, show limited interkinetic nuclear migration.
Imaging of publicly available stocks and lines created for brainbow17 type live imaging in Drosophila11, demonstrated that this technique is generalizable to the study of other biological phenomena. The technique described here was used in the production of videos over times ranging from minutes to 10 hr. To some degree, the microscope parameters vary with the experiment. For example, the sampling rate of time-lapse imaging must conform to the nature of the biological phenomenon under observation: for observing cell divisions, which typically take around 12 min from prophase to telophase, collecting a stack of images every minute is appropriate. To observe the behavior of filopodia in the same cells, sections or stacks must be collected at finer (3 sec) intervals.
Under the conditions described, no effect on the survival or morphology of eclosing adults was observed regardless of the period of observation. When the fluorescent marker is of low abundance, laser power was typically increased. In these situations, shortening the duration of the movie or reducing the number of sections or z-stacks, minimized exposure. In controls for other experiments, exposing the tissue to maximum intensity for 5 minutes had no effect on morphology or survival of the adult. Visualization was extended to include red, green, yellow, orange and blue fluorescent proteins tagged to a variety of different proteins, localized to the membrane, and/or filling the cytoplasm. Features in the submicron range were observed over timescales from seconds to hours. This method has been used extensively to observe and measure the timing and fidelity of mitosis in wild type and mutants (in preparation).
The pupal stage of Drosophila development is a period of remarkable plasticity in which the larval form is substantially eliminated while the adult form develops on the scaffold of the larval body. Despite a rich history, the study of metamorphosis has been methodologically hampered. For example, the use of direct observation to study the processes underlying metamorphosis has been limited by the lack of reliable in vivo access to the cells and tissues during remodeling. The surprisingly simple approach presented here allows access to this cryptic period of development in Drosophila. The approach is simple enough that undergraduates can easily learn and use it and robust enough to allow observations of tissue, cell and subcellular organization. The microscope used for these experiments is not optimized for deep tissue imaging, and so our observations are largely constrained to the outer 20 µm of the pupa. It is likely that techniques that permit deeper tissue penetration, such as multi-photon microscopy, would allow a much greater depth of imaging and allow study of the internal structural rearrangements underlying metamorphosis.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors wish to acknowledge Akira Chiba for intellectual support, material support, and stocks. Thanks to Julia Dallman for comments.
Materials
| Fly stuff fly pad | Genesee scientific | 59-114 | for fly anesthetization |
| CO2 gas | Airgas south | CD50 | For fly anesthetization |
| Regulator | Airgas south | CO2 regulator | |
| Fly vials | Genesee scientific | 32-113RLBF | Fly culture |
| Drosophila lines:A9-Gal4 (Bl#8761), His2Av-GFP (Bl#5941), Sco/CyO HsCre (Bl#1092), UAS-ChRFP-Tub (Bl#25773) | Bloomington Stock center | ||
| Glass bottom dishes #1 1/2 | WillCo Wells BV | For microscopy | |
| Thiodiethylene Glycol | Fluka | 88559 | mountant |
| Modeling clay | art supply store | Support to position pupae against | |
| Paintbrushes | art supply store | To manipulate flies | |
| Fine Forceps, Inox #5 | Fine science tools | 11252-20 | Dumont #5 |
| computer | any | 8 Gb RAM for image/movie analysis | |
| Fiji software | Free ware http://fiji.sc/Fiji | Image analysis software | |
| Confocal microscope | Any fast scanning confocal should be sufficient | ||
| 20x dry, and 40x or 63x oil immersion lenses | any | For imaging tissue, cellular and subcellular features | |
| Immersion oil (non fluorescent) | |||
| Stereo microscope | any | For fly manipulation |
References
- Raff, J. W., Jeffers, K., Huang, J. Y. The roles of Fzy/Cdc20 and Fzr/Cdh1 in regulating the destruction of cyclin B in space and time. J. Cell Biol. 157, 1139-1149 (2002).
- Stramer, B., et al. Live imaging of wound inflammation in Drosophila embryos reveals key roles for small GTPases during in vivo cell migration. J. Cell Biol. 168, 567-573 (2005).
- Clark, I. B., Jarman, A. P., Finnegan, D. J. Live imaging of Drosophila gonad formation reveals roles for Six4 in regulating germline and somatic cell migration. BMC Dev. Biol. 7, 52 (2007).
- Fuger, P., Behrends, L. B., Mertel, S., Sigrist, S. J., Rasse, T. M. Live imaging of synapse development and measuring protein dynamics using two-color fluorescence recovery after photo-bleaching at Drosophila synapses. Nat. Protoc. 2, 3285-3298 (2007).
- Siller, K. H., Serr, M., Steward, R., Hays, T. S., Doe, C. Q. Live imaging of Drosophila brain neuroblasts reveals a role for Lis1/dynactin in spindle assembly and mitotic checkpoint control. Mol. Biol. Cell. 16, 5127-5140 (2005).
- Roy, S., Hsiung, F., Kornberg, T. B. Specificity of Drosophila cytonemes for distinct signaling pathways. Science. 332, 354-358 (2011).
- Gho, M., Bellaiche, Y., Schweisguth, F. Revisiting the Drosophila microchaete lineage: a novel intrinsically asymmetric cell division generates a glial cell. Development. 126, 3573-3584 (1999).
- Ninov, N., Martin-Blanco, E. Live imaging of epidermal morphogenesis during the development of the adult abdominal epidermis of Drosophila. Nat. Protoc. 2, 3074-3080 (2007).
- Edgar, B. A., Lehner, C. F. Developmental control of cell cycle regulators: a fly’s perspective. Science. 274, 1646-1652 (1996).
- Wirtz, R. A., Semey, H. G. The Drosophila kitchen: equipment, media preparation, and supplies. Drosophila Information Service. 58, 176-180 (1982).
- Boulina, M., Samarajeewa, H., Baker, J. D., Kim, M. D., Chiba, A. Live imaging of multicolor-labeled cells in Drosophila. Development. 140, 1605-1613 (2013).
- Bainbridge, S. P., Bownes, M. Staging the metamorphosis of Drosophila melanogaster. J. Embryol. Exp. Morphol. 66, 57-80 (1981).
- Staudt, T., Lang, M. C., Medda, R., Engelhardt, J., Hell, S. W. 2,2′-thiodiethanol: a new water soluble mounting medium for high resolution optical microscopy. Microsc. Res. Tech. 70, 1-9 (2007).
- Schindelin, J., et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods. 9, 676-682 (2012).
- Meyer, E. J., Ikmi, A., Gibson, M. C. Interkinetic nuclear migration is a broadly conserved feature of cell division in pseudostratified epithelia. Curr. Biol. 21, 485-491 (2011).
- Sauer, F. C. Mitosis in the neural tube. J. Comp. Neurol. 62, 377-405 (1935).
- Livet, J., et al. Transgenic strategies for combinatorial expression of fluorescent proteins in the nervous system. Nature. 450, 56-62 (2007).

