Assessing Phagocytic Clearance of Cell Death in Experimental Stroke by Ligatable Fluorescent Probes
Summary
We present a new fluorescence technique for selective in situ labeling of active phagocytic cells, which clear off cell corpses in stroke. The approach is important for assessing brain reaction to ischemia because only a small proportion of phagocytes present in ischemic brain participate in clearance of cell death.
Abstract
We describe a new histochemical approach for visualization of phagocytic clearance in focal brain ischemia. The approach permits the study of elimination of dead cells in stroke by waste-management phagocytes of any cellular lineage. Although numerous cells of different origins that are capable of phagocytosis are present in ischemic brain, only part of them actively engulf and digest cell corpses. The selective visualization, quantification and analysis of such active phagocytic waste-management are helpful in assessing brain response to ischemia. Efficient cell death clearance is important for brain recovery from ischemic injury, as it opens the way for the subsequent regenerative processes. The failure to clean the corpses would result in a toxic reaction caused by non-degraded DNA and proteins. The described procedure uses fluorescent probes selectively ligated by a viral topoisomerase to characteristic DNA breaks produced in all phagocytes during engulfment and digestion of cells irreversibly damaged by ischemia. The method is a new tool for the investigation of brain reaction to ischemic injury.
Introduction
Ischemic stroke induces profound changes in the affected brain tissue. It initiates massive cell death, which leads to the rapid activation of resident phagocytic cells of microglial origin. It also sets off infiltration of ischemic brain by various types of blood-derived professional phagocytes including neutrophils, macrophages, dendritic, and mast cells1,2.
It is still debated whether this response to ischemic injury plays a positive or a negative role. Although phagocytosis following stroke can be beneficial because it clears dead cells and suppresses inflammation, it also generates toxic reactive oxygen species affecting neuronal survival and exacerbating tissue damage1-5.
While several types of phagocytic cells infiltrate ischemic brain, not all of them participate in waste-management by engulfing cell corpses and clearing the way for regenerative processes1-3. This creates a requirement for selective identification of waste-management cells that carry out phagocytic clearance of cell death in stroke. When imaging such active waste-management cells, it is also important to answer the question of how efficiently they degrade the engulfed cell corpses. The effective and complete degradation of the dying cell's DNA in phagocytosis is essential because it prevents self-immunization and the release of pathological nuclear material6.
Here we present new probes that use specific DNA breaks as markers of active phagocytic cells. These signature breaks are exclusively produced during breakdown of engulfed nuclei inside functional waste-management cells. Therefore the probes selectively label only those phagocytes that engulf and actively digest cellular corpses. They also permit observing the intensity and completion of DNA breakdown after the engulfment. The probes are helpful in evaluations of intensity and efficiency of phagocytic clearance.
The new probes rely on a non-protein-based marker and therefore can be particularly advantageous in studies of stroke, where extensive ischemic damage can disrupt cellular morphology or deplete protein-based markers, especially inside the core ischemic zone.
The principle of the technique is presented in Figure 1. The figure shows hairpin-shaped oligonucleotide probes ligated by the enzyme vaccinia topoisomerase (VACC TOPO) to 5'OH DNA ends generated by lysosomal DNase II during DNA digestion7.
Protocol
The method is suitable for formaldehyde-fixed, paraffin embedded tissue sections. The technique is demonstrated here in its application to experimental stroke in rat.
1. Preparing Components for Tyramide Signal Amplification (TSA) System
Although the TSA-based enhancement is used in the final stage of labeling, preparing for this reaction can take more than 1 hr. Therefore it is convenient to make the TSA reagents before starting the labeling.
- Make fluorophore tyramide stock solution. Use the fluorophore tyramide reagent supplied in individual vials with the TSA labeling kit. Add 300 µl DMSO to the tyramide reagent vial. Fluorophore tyramide stock solution is stable for at least 3 months when stored at 4 °C.
- Make TNT wash buffer containing: 0.1 M Tris-HCl, pH 7.5; 0.15 M NaCl; 0.05% Tween-20. If needed 0.3% Triton X-100 can be substituted for the 0.05% Tween-20. Alternatively PBS may be used as wash buffer.
- Make TNB blocking buffer containing: 0.1 M Tris-HCl, pH 7.5; 0.15 M NaCl; 0.5% Blocking Reagent (supplied with kit). To completely dissolve the blocking reagent, slowly add it in small increments to the buffer while stirring. Heat this solution gradually to 55 °C with continuous stirring. This can take 30-60 min. The solution will appear milky. Bring to room temperature before using. Aliquot and store at -20 °C for long-term use.
2. Preparation of Sections
Use slides with mounted 5 μm-thick sections cut from formalin-fixed tissues.
- Treat sections with xylene for 15 min.
- Rehydrate sections as follows: soak in 96% EtOH 2x for 5 min each; soak in 80% EtOH for 5 min; wash with water 2x for 5 min each.
- Make Proteinase K working solution by diluting Proteinase K stock solution to 50 µg/ml in PBS.
- Add Proteinase K working solution to sections. Incubate 15 min at 23 °C. The volume of the solution and the time of digestion may need to be adjusted depending on the size of sections and tissue types. For a small (below 5 mm in diameter) section 50 µl of Proteinase solution would be sufficient. For an average size (~10 mm in diameter) section, add 100 µl of Proteinase K solution. For very large sections the volume of the solution can be scaled up. In all cases take care to prevent overdigestion of sections which leads to complete loss of signal and destroys tissue morphology. This often happens if incubation time exceeds 25 min.
- Wash sections in distilled water 2x for 5 min each. Prepare the labeling mix containing active probe while sections are in water.
3. VACC TOPO-based Staining
- Prepare the active probe solution by combining the following:
Water - 11.75 µl
1 M Tris-HCl, pH 7.4 - 1.25 µl
100 pmol of 12-base overhang suicide oligonucleotide - 1 µl
100 pmol of adapter oligonucleotide - 1 µl
50 pmol VACC TOPO - 10 µl - Mix gently by pipetting. Incubate at room temperature (23 °C) for 15 min to allow for probe activation.
- Take sections from wash jar and aspirate the remaining water. Then apply the active probe solution prepared in step 3.1 (25 µl per ~10 x 10 mm section).
- Coverslip the sections and incubate for 15 min at 23 °C. Dip slides in water to gently remove coverslips. Wash in water 3x for 5 min each.
4. TSA Signal Enhancement
- Cover tissue sections with TNB blocking buffer prepared earlier and incubate slides in a humidified chamber for 30 min at room temperature (23 °C).
- Prepare a 1:100 dilution of streptavidin-horseradish peroxidase conjugates (SA-HRP) in TNB blocking buffer. Aspirate TNB blocking buffer and apply SA-HRP solution. Incubate slides with SA-HRP for 30 min at room temperature. Use adequate reagent volume to cover the tissue section, generally 100 µl per section.
- Wash slides 3x for 5 min each in TNT wash buffer (or PBS) at room temperature.
- Prepare a 1:50 dilution of fluorophore tyramide stock in 1x Amplification Diluent from the kit. Pipette the fluorophore tyramide working solution onto each slide. Use enough working solution to completely cover the tissue section, generally 100 µl per section. Make fluorophore tyramide working solution immediately before each labeling. The solution cannot be reused, so discard any unused portion of it after labeling.
- Incubate the slides at room temperature for 3 to 10 min. Monitor fluorescence increase under microscope until the first hints of structures can be seen. Quickly proceed to wash step.
- Wash slides 3x for 5 min each in TNT wash buffer (or PBS) at room temperature with agitation.
- Add the antifading solution with DAPI and coverslip. Observe under the fluorescence microscope. To visualize DAPI and FITC fluorescence use a band-pass filter set:
FITC excitation D490/40, emission 520/10; DAPI excitation D360/40, emission 460/20, or similar.
Representative Results
The principle of the technique for labeling active waste-management phagocytes is presented in Figure 1. The schematic demonstrates that the detection proceeds in three steps. The first step encompasses probe activation (Figure 1A); in the second step the activated probe is ligated to specific DNA breaks in tissue sections (Figure 1C); the third step includes fluorescent signal amplification (Figure 1D). In a more detailed explanation, the detection goes as follows:
1. Probe activation. Prior to the ligation reaction VACC TOPO needs to be covalently linked to the 3' end of the oligoprobe. During probe activation VACC TOPO attaches to the hairpin-adapter duplex and cleaves the upper strand. The 12-base-long part then permanently separates, leaving VACC TOPO attached to the 3' end of the hairpin. This oligonucleotide with the enzyme linked to its 3' end can label DNase II type breaks (Figure 1B). The 12-base overhang and an adapter oligo are required because the enzyme will not cut a shorter strand8 and will therefore be unable to attach to the probe and activate its 3' end.
2. Probe ligation. Biotinylated hairpins are ligated by VACC TOPO to 5'OH blunt ended DNA breaks generated by the phagocytes digesting nuclei of dead cells. The attached probes are visualized by fluorescence using tyramide enhancement9.
3. Florescent signaling is achieved by using Tyramide Signal Amplification (TSA) system9. During this process streptavidin-horseradish peroxidase conjugates attach to biotinylated hairpins and activate binding of fluorescent tyramides in the close vicinity, creating strongly florescent sites.
Application of the technique to experimental stroke is shown in Figure 2. Figure 2 presents fluorescence images of active phagocytic cells clearing cell death in rat brain 48 hr after the start of permanent focal brain ischemia. The cells are labeled by the VACC TOPO probes as described. The probes visualize gigantic phagolysosomes10 with DNase II breaks (green fluorescence), marking phagocytes which actively engulf and digest cell corpses7. Simultaneous co-staining with DAPI labels chromatin of the phagocytes and of the engulfed cells inside phagocytes. The overlays of VACC TOPO and DAPI signals permit easy identification and morphological analysis of active phagocytic cells. For clearer presentation, the images are complemented by diagrams of the events shown.
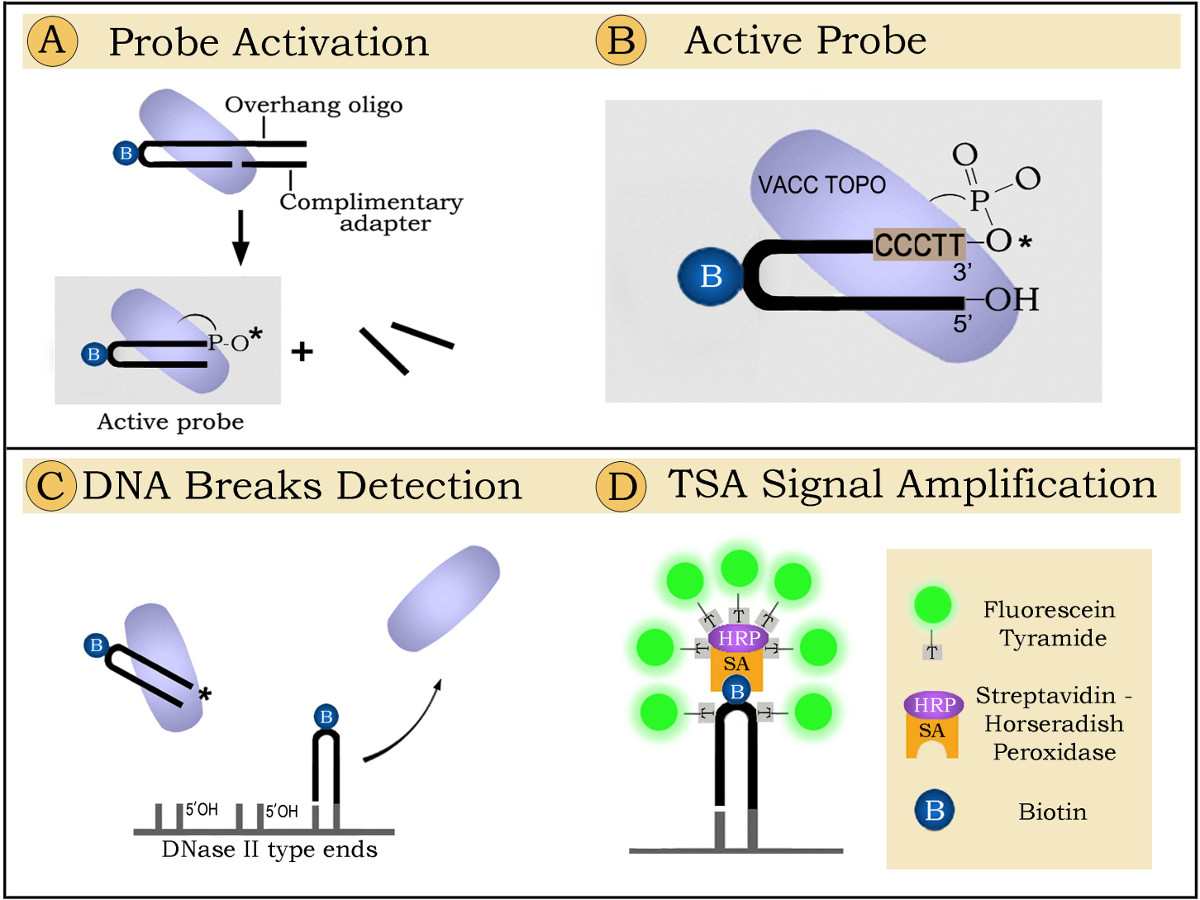
Figure 1. Detection of phagocytic clearance in brain sections by ligatable probes: principle of the method.
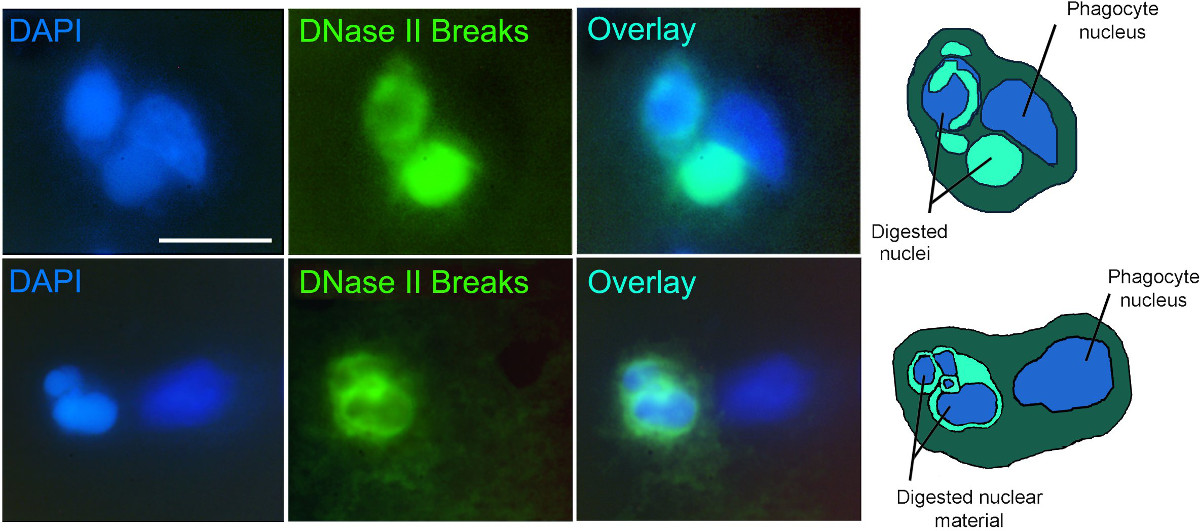
Figure 2. Experimental stroke 48 hr after ischemia onset: phagocytic clearance of cell death. Two examples of phagocytic clearance. Ligatable VACC TOPO probe – green fluorescence. Nuclear stain DAPI – blue fluorescence. Diagrams of the events presented in the images (right column) show nuclei of phagocytes and pycnotic engulfed extra nuclei in various stages of digestion. Scale bar – 15 µm.
Discussion
In this video we demonstrate, in the tissue section format, how to label active phagocytes which clear cell death in focal brain ischemia. The presented technique is the first approach which specifically labels only those waste-management cells which have engulfed and actively digest cellular corpses. This makes it advantageous with respect to other existing methods for labeling of phagocytic cells, which do not have this capability.
The method uses a general and selective DNA-based marker of phagocytic activity – double-stranded, blunt ended 5’OH DNA breaks produced in phagolysosomes of waste management cells during digestion of engulfed nuclei. This specific labeling of only active phagocytic cells permits accurate assessment of the intensity and efficiency of the phagocytic reaction in ischemic brain.
The technique is robust and works well if all steps are followed as described. The proteinase K digestion step is particularly important for strong and reproducible staining. Overdigestion at this step leads to the loss of DNA from the section and might decrease or even eliminate the signal. The other critical point is to use a highly active preparation of the vaccinia topoisomerase enzyme. A limitation of the technique is that it was not optimized for use with fresh frozen sections.
The approach has broad applicability to studies of apoptosis and is expandable to other tissues and conditions beyond ischemic brain. It is particularly convenient when used for visualization of phagocytic reactions in tissues and organs composed of multiple cell types with diverse participating waste-management cells.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This research was supported by grant R01NS062842 from the National Institute of Neurological Disorders and Stroke, National Institutes of Health (V.V.D.) and by grants R21 NS064403 from the National Institute of Neurological Disorders and Stroke, National Institutes of Health through ARRA (V.V.D.) and R21 EB006301 National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health (V.V.D.).
Materials
| Name of the reagent or material | Company | Catalogue number | Comments (optional) |
| Xylene | JT Baker | 9490-22 | |
| Ethanol | |||
| Proteinase K | Roche | 3115879001 | |
| PBS Buffer | Invitrogen | AM9624 | |
| Vaccinia Topoisomerase I | Vivid Technologies | 5 pmol/μL stock | |
| 12-Base Overhang Suicide Oligonucleotide | IDT DNA | 5’-AAG GGA CCT GCB GCA GGT CCC TTG ATA CGA TTC TA -3’ ; B – Biotin-dT; 100 pmol/μL stock in ddH2O | |
| Adapter Oligonucleotide | IDT DNA | 5’-TAG AAT CGT ATC-3’; 100 pmol/μL stock in ddH2O | |
| 1 M Tris-HCl, PH 7.4 | Lonza | 51237 | |
| TSA, Fluorescein System | PerkinElmer | NEL701A001KT | |
| 1 M NaCl | |||
| DMSO | Sigma | ||
| Tween-20 | Sigma | P9416-50ML | |
| Vectashield with DAPI | Vector Laboratories | H-1200 | |
| Coverslips | 22x22mm or 22x40mm glass or plastic coverslips. Plastic coverslips are preferable during the reaction, as they are easier to remove from the section. | ||
| Rat Brain Sections | Rat brain taken 48 hours after reproduction of ischemic stroke as described in 11. 5-6μm-thick sections cut from paraformaldehyde-fixed, paraffin-embedded tissue blocks. Use charged and precleaned slides. | ||
| Equipment | |||
| Inverted Microscope | Olympus | ||
| Digital Video camera and software | Micromax | ||
| Software | Metamorph | ||
References
- Jin, R., Yang, G., Li, G. Inflammatory mechanisms in ischemic stroke: role of inflammatory cells. J Leukoc Biol. 87, 779-789 (2010).
- Schilling, M., Besselmann, M., Müller, M., Strecker, J. K., Ringelstein, E. B., Kiefer, R. Predominant phagocytic activity of resident microglia over hematogenous macrophages following transient focal cerebral ischemia: an investigation using green fluorescent protein transgenic bone marrow chimeric mice. Exp Neurol. 196 (2), 290-297 (2005).
- Faustino, J. V. Microglial cells contribute to endogenous brain defenses after acute neonatal focal stroke. J Neurosci. 31 (36), 12992-13001 (2011).
- Thored, P. Long term accumulation of microglia with proneurogenic phenotype concomitant with persistent neurogenesis in adult subventricular zone after stroke. Glia. 57 (8), 835-849 (2009).
- Sierra, A., Abiega, O., Shahraz, A., Neumann, H. Janus-faced microglia: beneficial and detrimental consequences of microglial phagocytosis. Front Cell Neurosci. 7 (6), (2013).
- Samejima, K., Earnshaw, W. C. Trashing the genome: Role of nucleases during apoptosis. Nat.Rev. Mol. Cell Biol. 6 (9), 677-688 (2005).
- Minchew, C. L., Didenko, V. V. Fluorescent probes detecting the phagocytic phase of apoptosis: enzyme-substrate complexes of topoisomerase and DNA. Molecules. 16 (6), 4599-4614 (2011).
- Shuman, S. Two classes of DNA end-joining reactions catalyzed by vaccinia topoisomerase. Sci. Rep. 267, 16755-16758 (1992).
- Barcia, C., et al. ROCK/Cdc42-mediated microglial motility and gliapse formation lead to phagocytosis of degenerating dopaminergic neurons in vivo. Sci. Rep. . 2, 809 (2012).
- Didenko, V. V., Ngo, H., Minchew, C. L., Boudreaux, D. J., Widmayer, M. A., Baskin, D. S. Visualization of irreparable ischemic damage in brain by selective labeling of double-strand blunt-ended DNA breaks. Mol. Med. 8, 818-823 (2002).

