Testing the Efficacy of Pharmacological Agents in a Pericardial Target Delivery Model in the Swine
Summary
We have developed a swine model for the target delivery of pharmacological agents within the pericardial space/fluid. Using this approach, the relative benefits of administered agents on induced atrial fibrillation, relative refractory periods and/or ischemic protection can be investigated.
Abstract
To date, many pharmacological agents used to treat or prevent arrhythmias in open-heart cases create undesired systemic side effects. For example, antiarrhythmic drugs administered intravenously can produce drops in systemic pressure in the already compromised cardiac patient. While performing open-heart procedures, surgeons will often either create a small port or form a pericardial cradle to create suitable fields for operation. This access yields opportunities for target pharmacological delivery (antiarrhythmic or ischemic preconditioning agents) directly to the myocardial tissue without undesired side effects.
We have developed a swine model for testing pharmacological agents for target delivery within the pericardial fluid. While fully anesthetized, each animal was instrumented with a Swan-Ganz catheter as well as left and right ventricle pressure catheters, and pacing leads were placed in the right atrial appendage and the right ventricle. A medial sternotomy was then performed and a pericardial access cradle was created; a plunge pacing lead was placed in the left atrial appendage and a bipolar pacing lead was placed in the left ventricle. Utilizing a programmer and a cardiac mapping system, the refractory period of the atrioventricular node (AVN), atria and ventricles was determined. In addition, atrial fibrillation (AF) induction was produced utilizing a Grass stimulator and time in AF was observed. These measurements were performed prior to treatment, as well as 30 min and 60 min after pericardial treatment. Additional time points were added for selected studies. The heart was then cardiopleged and reanimated in a four chamber working mode. Pressure measurements and function were recorded for 1 hr after reanimation. This treatment strategy model allowed us to observe the effects of pharmacological agents that may decrease the incidence of cardiac arrhythmias and/or ischemic damage, during and after open-heart surgery.
Introduction
Currently in open-heart procedures, clinicians utilize antiarrhythmic and other treatment agents systemically. Yet, this can be problematic for many patients, especially those who are already clinically compromised. For example, intravenous treatments can result in systemic drops in blood pressure or renal dysfunction; further, they may create anesthesia management issues and/ or other long-term side effects.
Here we have created a model for testing the efficacy of administering pharmacological agents into the pericardial space. For example, this approach can be utilized for testing antiarrhythmic drugs, studying compounds that could increase cardiac function and/or promoting recovery of the myocardium after surgical procedures. There have been observed benefits to the target delivery of treatments into the pericardial space versus intravenous administration: e.g., our laboratory demonstrated that localized delivery of antiarrhythmic drugs, such as metoprolol, is protective against the incidence of arrhythmias while minimizing reductions in blood pressure 1. This target delivery strategy also provides the opportunity for administering higher focal concentrations while minimizing systemic levels. For example, high levels of intravenously delivered concentrations of fatty acids may result in hemolysis, but pericardial delivery minimizes this concern 2.
This study paradigm consists of three major objectives to determine the efficacy of pericardial delivered compounds: 1) in situ determination of refractory periods of the atrial ventricular node, the atria and ventricles, before treatment and 30 and 60 min post treatment; 2) the relative in situ AF burden before treatment and 30 and 60 min post treatment (additional timepoints were often added) 3) functional analysis of the heart after it has been reanimated 3 including hemodynamic monitoring, heart rate, heart metabolism (lactate and glucose) sampled from the coronary sinus, ejection fraction (EF%) and ventricular wall thickness (cm) monitored every 10 min post reanimation This treatment strategy model allowed us to observe the effects of pharmacological agents that may decrease the incidence of cardiac arrhythmias and/or ischemic damage, during and after open-heart surgery or transplantation.
Protocol
This protocol was approved by the University of Minnesota Institutional Animal Care and Use Committee.
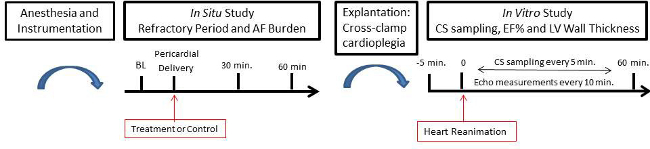
Figure 1. Diagram of Study Paradigm Please click here to view a larger version of this figure.
1. Surgical Preparation of the Swine
- Administer 5-7mg/kg of tiletamine/zilazipam to the swine (70-80 kg) via intramuscular injection. Place an ear vein catheter and administer 5-7 mg/kg methohexital via intravenous access. Intubate the animal with endotracheal tube and ventilate.
- Deliver isoflurane >1.2% MAC in a mixture of house air and oxygen to maintain a surgical plane of anesthesia. Ensure the animal is in a deep plane of anesthesia prior to any manipulations by checking for the absence of jaw tone and keeping the anesthesia above the minimum alveolar concentration for swine. Initial anesthetic depth check is conducted by assuring the absence of jaw tone and the isoflurane levels are monitored continuously throughout the procedure (>1.5 MAC) to ensure a deep plane of anesthesia.
- Carefully access the right external jugular and carotid artery with cautery and blunt dissection. Place an 8.5F Swan-Ganz catheter in the external jugular and inflate the balloon to 1.5 cc. Then traverse through the right atrium, into the ventricle and through the pulmonic valve down the pulmonary artery until a wedge pressure is felt.
- Deflate the balloon and leave the catheter in this position. Record the right atrial pressure and pulmonary artery pressure using the balloon. Additionally, record the wedge pressure or pressure in the lung (or if the lungs are considered normal, then also record the left atrial pressure).
NOTE: This catheter also allows for estimates of the cardiac output. - Place an additional 5.0F balloon pressure catheter in the external jugular and into the right ventricle to continuously record pressure.
- Place a 5.0F balloon pressure catheter in the carotid artery, prolapse through the aortic valve and maintain in the left ventricle. Flush all pressure catheters with 20 units/ml heparanized saline connected to a pressure transducer, and record all pressure data from the left ventricle, right ventricle (and estimated cardiac output), pulmonary artery pressure and right atrial pressure.
- For peripheral monitoring, access a branch of the femoral artery by placing an 18F catheter for real-time blood pressure or blood gas monitoring. Record heart rate and temperature during the procedure.
- Carefully access the left external jugular with cautery and blunt dissection. Place two 11.0F hemostasis introducers in the jugular and secure with 0 silk sutures. Place steerable catheters in the introducers.
- Under fluoroscopic guidance, place active fixation leads in the right atrial appendage and the apex of the right ventricle.
NOTE: A fixation lead is a surgically implanted wire that connects the heart to an implanted pacemaker. - Connectanalyzer cables to the leads and test for capture utilizing a programmer. Set the parameters to 8V, 0.25 msec duration, and set the pacing rate at a minimum of 10 bpm higher than the intrinsic rate at that time. Record the relative impedance for each lead.
- Make a medial surgical incision from the xiphoid process to near the insertion of the sternocleidomastoid muscles. Use a sternal saw to remove the anterior protrusion from the sternum (keel bone) to enable the partial medial dissection of the sternum. Use a shear cut through the remaining portions of sternal bone structure. Dissect the sternal-pericardial ligament and retract the sternum.
- Use blunt dissection to separate the pericardium from the pleural linings. Make a medial sagittal incision (3-5 cm) in the pericardium and create a pericardial cradle with four square knot sutures at each corner (right, left, superior and inferior sites).
- Place a temporary bipolar lead in the apical region of the left ventricle and place a unipolar plunge temporary pacing lead into the left atrial appendage.
- Euthanasia is accomplished by removal the heart under deep anesthesia.
2. Preparation for Electrophysiological Monitoring
- Cardiac Mapping System.
- Select an AF study in a cardiac mapping system Connect the ventricular epicardial bipolar pacing leads and the epicardial leads in the atrial appendage with alligator clips to the appropriate settings in the breakout box. Connect an EKG and ground pads to the system.
- Oscilloscope and Grass Stimulator.
- Connect the bipolar left ventricular pacing lead and unipolar atrial lead in the breakout box to a Grass stimulator which is connected to an oscilloscope for electrical verification.
- Programmed Electrophysiological Stimulation.
- Secure the right atrial leads and right ventricle leads in a pacemaker Place the programmer telemetry head over the pacemaker. Select the pacing electrode stimulation (PES) study and set the parameters to 8V; remove all patient parameters. Set the pacing duration set to 8 pulses at 400 msec (or 300 msec).
3. In Situ Study
- Refractory Period: Study Paradigm.
- Determine the atrial effective refractory period (AERP), atrial ventricular node refractory period (AVNERP) and the ventricular effective refractory period (VERP) at baseline, and at 30 and 60 min post pericardial delivery of compound/drug/control treatment; additional time points may be added as desired.
- Refractory Period: Programmer Settings.
- Burst induce the PES study parameter to 8 paces (400 msec or 300 msec) followed by a minimum of 300 msec pacing. Reduce the pacing until the ventricles/atrium (respectively) fail to contract. This is noted from the sensing of the pacing leads; thus determining the relative refractory period.

Figure 2. EnSiteSystem: AERP, AVNERP and VERP Determination Please click here to view a larger version of this figure.
- Determination of Relative AF Burden: Parameters.
- Sustain the animal’s heart in AF, after 1 min, allow it to remain in AF for up to 10 min: i.e., before shocking the atria with direct paddles at 5 joules to terminate AF. Elicit AF induction up to 10 times at each time point. Note, in this study. AF is defined as an erratic rhythm with an absence of the P wave.
- AF Burden: Grass Stimulator and Oscilloscope Settings.
- Optimize the parameters for the Grass stimulator to deliver 4 Hz with a 2 sec pulse duration to the left atrial appendage (LAA) via the plunge pacing lead; verify this by a standard oscilloscope. Deliver a single pulse to the LAA to induce AF.
4. Heart Explantation and Reanimation (Transplant Model)
- Aortic Root Cannula.
- Carefully dissect the pericardial tissue around the ascending aorta and remove the pericardium.Suture two points (2-3 cm apart) of 2.0 Ethibond suture in the ascending aorta, and administer 30,000 units of Heparin intravenously.
- Place the aortic root cannula between the suture points and secure the aorta. With the delivery system pressurized, remove the stylet-bevel and place a clamp on the cannula not allowing for flow.
- Cardioplegia Preparation.
- Administer cold St. Thomas cardioplegia solution into the ascending aorta under a pressure of 150 mm/hg via a pressurized bag. Secure an irrigation catheter that is attached to the pressurized cardioplegia with a 3-way stop cock on the distal end. Secure the stop cock to the aortic root cannula, release a steady flush of cardioplegia through the catheter and secure to the aortic root cannula.
- Cross-Clamp.
- Clamp the inferior vena cava with an atraumaticclamp 1-2 cm just superior to the diaphragm. Clamp the ascending aorta with atraumatic angled camp 1-2 cm on the ascending aorta superior to the aortic root cannula.
- Cross-Clamp/Cardioplegia.
- Remove the clamp from the aortic root cannula and flush the cardioplegia at 150 mm/hg toward the aortic valve, closing the aortic valve and perfusing the coronaries.
- Clamp the superior vena cava and make a small incision in the pulmonary artery, so as not to over pressurize the heart. Once the heart is stopped (asystolic), excise and remove heart from the chest and place in a bath of cold Krebs-Henseleit buffer (~3-8 °C).
- Reanimation.
- Cannulate the great vessels and prepare the heart utilizing Visible Heart methodologies (3). Once the perfused heart reaches 37±5 °C, restore a native sinus rhythm by delivering 34 J shocks to the ventricles via epicardial patch electrodes.
5. In Vitro Study Paradigm
- Continuously monitor cardiac function for 60 min post reanimation. Continuously monitor echocardiography in 2D mode with a short-axis view: an LV view just below the mitral valve.
- Calculate ventricular wall thickness and ejection fractions every 10 min post reanimation (see below). Take samples from the coronary sinus (1 cc) at -5 min and every 5 min during the evaluation period.
- Monitoring.
- Monitor the hemodynamics for both the left and right ventricles, including pressures, flows and volume changes.
- Echocardiography: Ejection Fraction.
- Place a transthoracic echo probe directly on the heart, capturing a mid-papillary short-axis view. Calculate the ejection fraction (EF %) in 4-chamber working mode every 10 min using the equation: LV End Diastolic Volume − LV End Systolic Volume X 100/LV End Diastolic volume
- Echocardiography: Left Ventricular Wall Thickness.
- Measure ventricular wall thickness (cm) from a mid-papillary short-axis window at 10 min intervals for an hour post reanimation. Measure four corners of the left ventricle using a screen calipers and take the average.
- Myocardial Metabolism.
- Place a steerable catheter in the coronary sinus (CS). Sample 1 cc from the CS 5 min before reanimation and every 5 min post reanimation for 1 hr. Test the samples for lactate and glucose levels; other blood gases can also be assessed.
Representative Results
These results are characteristic of the data that can be collected utilizing this target delivery model of pharmacological agents in the swine. This data demonstrated notable increases in these ventricle effective refractory periods (VERP) following a DHA infusion in situ. In addition, the data establishes an increase in left ventricular pressure of DHA compared to control in vitro. The LV pressure in the DHA treated hearts were significantly higher compared to control at several time-points. This data validates a pharmacological window for testing treatment strategies in situ and in vitro.
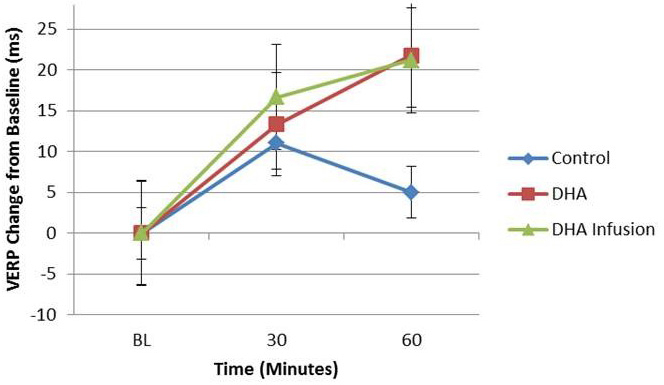
Figure 3. Change in the Ventricle Effective Refractory Period (VERP). The Ventricle Effective Refractory Period (VERP) was determined 5 min before pericardial delivery of control agent (saline) or docosahexaenoic acid (DHA), or DHA infusion, in addition to 30 and 60 min post-pericardial delivery of either the treatments or control. The VERP of the DHA group trended toward increasing compared to controls. Please click here to view a larger version of this figure.
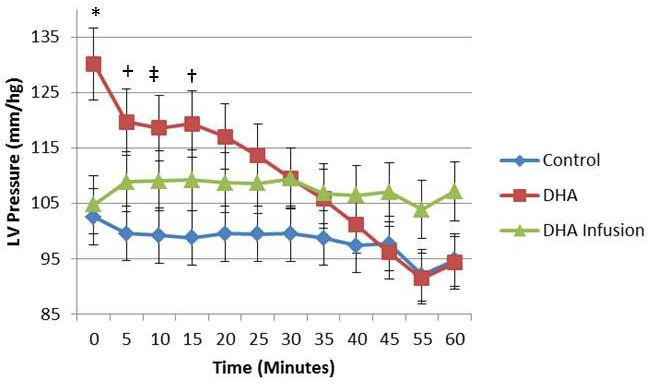
Figure 4. Pressure of Left Ventricle in 4-Chamber Working Model The maximum pressure (mmHg) was determined every 5 min for 60 min after the heart was reanimated using the visible heart methodologies. DHA and DHA infusion (treated for over 10 min) treated hearts trended to elicit initial higher pressure than control for the majority of the testing period. An un-paired T-test was completed for each time-point. (*, P=0.065, +, P=0.056, †, P=0.059 ‡.P=0.058) Please click here to view a larger version of this figure.
The Ventricle Effective Refractory Period (VERP) was determined 5 min before pericardial delivery of control agent (saline) or docosahexaenoic acid (DHA), or DHA infusion (over 10 min), in addition to 30 and 60 min post-pericardial delivery of either the treatments or control. The VERP of the DHA group trended toward increasing compared to controls.
The maximum pressure (mmHg) was determined every 5 min for 60 min after the heart was reanimated using the visible heart methodologies. DHA and DHA infusion treated hearts trended to elicit initial higher pressure than control for the majority of the testing period. An un-paired T-test was completed for each time-point. (*, P=0.065, +, P=0.056, †, P=0.059 ‡.P=0.058)
Discussion
Here, we have demonstrated a unique approach for testing the potential efficacy of target delivered compounds into the pericardial space. This study paradigm can be utilized to test current market released pharmaceutical products or experimental compounds, thus providing direct translational applications to various clinical settings. Within this treatment strategy there are two major clinical applications for delivering pharmaceuticals to the pericardial space: 1) open-heart or minimally invasive cardiac surgical procedures; and 2) heart transplantation and organ preservation. In addition, this study paradigm has multiple parameters that can be analyzed to better understand the predictive efficacy of the pharmaceutical agents themselves within these described treatment strategies. In situ parameters include: 1) the relative refractory periods of the AVN, atria and/or ventricles; 2) the determination of relative AF susceptibility/burden; and 3) hemodynamic and heart rate responses. In vitro parameters include: 1) relative ejection fractions; 2) LV wall thickness responses; 3) cardiac metabolic alterations; and 4) hemodynamics and heart rate changes. The historical data we present here demonstrate a selection of these described parameters. The most notable translational aspects of these studies are for open- or minimally invasive heart procedures, as well as organ procurement prior to transplantation.
More specifically, a formed pericardial cradle placed during an open-heart surgery, either minimally invasive or following a full-sternotomy, provides an important opportunity to administer localized compounds: i.e, at higher concentrations than may be administered intravenously and/or without systemic side effects. For even less invasive cardiac procedures (e.g., subxyphoid access), a small incision is made in the pericardium that could also be used as a conduit for target drug delivery. Myocardial irritability/insult is greatly increased while the heart is manipulated during various cardiac procedures. Treating for arrhythmias and/or functional deficits directly may be a beneficial and easy means for improving patient outcomes.
Similar applications could be easily performed during procedures for organ recovery, for example, preconditioning the heart prior to procurement. It is crucial that the myocardium is optimally conditioned for a complicated procedure that includes defibrillating the heart back into a normal sinus rhythm after transplantation. In other words, there are increased risks of atrial and ventricular tachycardias and/or fibrillation during heart procurement and after transplantation. Also, it is important to preserve the other organs in the donor during the procurement process, where peripherally administered drugs can compromise these organs. For example, intravenously administered metoprolol can result in acute kidney dysfunction when given to treat arrhythmias during surgical procedures/procurements 1. Currently, the heart must be transplanted to the recipient within approximately 4-6 hr. This time constraint remains as one of the limiting factors in performing heart transplantations today. Thus, employing the experimental paradigm we describe here could be an important means for evaluating compounds that may be beneficial in prolonging the acceptable range of ischemic time after organ recovery.
Previously collected data from our laboratory utilizing this study paradigm have demonstrated that it can be highly useful for obtaining multiple parameters that can be analyzed to predict the efficacy of the pharmaceutical agents themselves within the described treatment strategy. More specifically, studying the effective refractory periods following pericardial delivered DHA, DHA infusion, metoprolol and other drugs in situ has been important so to understand their antiarrhythmic potential for this target delivery method. Prolonging the effective refractory periods and/or reducing the conductance velocities within the AVN can often terminate certain types of arrhythmias 4. Here we have demonstrated notable increases in these effective refractory periods following a DHA infusion, as well as after delivering metoprolol as compared to control (previously published, 1. In addition, it is of interest to determine the relative AF burden because of its relationship to the probability for arrhythmic potentiation in a given patient; here we also noted alterations in this response following pharmacological preconditioning.
The investigation of clinically relevant clinical parameters after the reanimation of a preconditioned heart also gives translational insights as to the potential benefits of this pharmacological administration on a transplant recipient. To date using this overall target delivery/preconditioning research approach, our laboratory has investigated a variety of clinically administered agents as well as novel pharmacological compounds that may minimize arrhythmias or ischemic damage to the heart (metoprolol, amiodarone, lidocaine, delta opioids, omega-3 fatty acids, ursodyoxycholic acid, lipovenous, docosahexaenoic acid and/or combinations of these). It should also be noted that these agents could also be administered as post-conditioning agents in the in vitro aspect of the protocols described within. Observing the in vitro hemodynamics, the representative data for the LV pressures are notably higher in hearts that have been treated with DHA or a DHA infusion compared to a vehicle control. In addition, our laboratory has observed changes in ejection fraction and LV wall thickness in treated hearts compared to controls. The clinical parameter/factors that can be studied are important so to evaluate effective function and to observe the onset of edema. Further, samples from the coronary sinus can also be obtained for analysis of various metabolic factors: e.g., lactate and glucose levels. These parameters are imperative to assess the relative cardiac metabolism and/or overall heart function. For example, increased lactate levels are often indicative of acidosis, in turn resulting in poor cardiac function.
The data we have obtained from such studies have demonstrated a viable experimental model (with noted limitations because of the acute nature of the study) to determine the practicality of utilizing either clinically available drugs or experimental agents for a pericardial target delivery strategy. We consider that the investigative approach we describe here is highly reproducible and provides novel insights relative to the target and systemic benefits of the delivery of various pharmacological agents within the pericardial space. The results one may obtain in such designed protocols could have important translational implications for both cardiac surgery and heart transplantation.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We would like to give a big thanks to the Visible Heart Laboratory staff and students that have helped with this project: Nate Menninga, Lars Mattison and Megan Schmidt.
Materials
| SelectSecure® 3830 lead | Medtronic | N/A | Pacing Lead |
| C304 Deflectable Catheter | Medtronic | N/A | Steerable catheter for placing leads |
| SelectSecure® 3830 lead | Medtronic | N/A | active fixation pacing leads |
| Grass S48 Stimulator | N/A | N/A | Electrical Stimulator |
| Premium 6500 Unipolar Pacing | N/A | Plunge pacing lead for LAA | |
| EnSite™ Cardiac Mapping | N/A | Electrophysiology mapping system | |
| CareLink Programer 2092 | Medtronic | N/A | programmer for pacing leads |
| GEM II ® pacemaker | Medtronic | N/A | pacemaker can |
| DLP ® Aortic Root Cannula | Medtronic | N/A | aortic root cannula for transplant |
| C-Arm Fluoroscopy | Ziehm | N/A | fluoroscopic imaging |
| Oscilliscope | Tektronix | N/A | |
| 11F Hemostasis introducer | SafeSheath | N/A | Hemostasis introducers |
| Swan-Ganz Catheter 8.0F | ICU Medical | N/A | thermal dilution catheter |
| Venogram balloon | Oscor | N/A | pressure monitoring |
| Ultraview SL | Spacelabs | N/A | EKG and blood pressure |
| s/5 Avance | General Electric | N/A | Anesthesia machine |
| Atrial 6492 – Unipolar Temporary Atrial Pacing Lead | Medtronic | N/A | temporary pacing lead |
| VIVID i | General Electric | 2D electrocardiography unit |
References
- Richardson, E. S., Rolfes, C., Woo, S. O., Elmquist, W. F., Benditt, D. G., Iaizzo, P. A. Cardiac Responses to the Intrapericardial Delivery of Metoprolol: Target Delivery Compared to Intravenous Administration. J. Cardiovasc. Translational Research. 5 (4), 535-540 (2012).
- Xiao, Y. F., Sigg, D. C., Ujhelyi, M. R., Wilhelm, J. J., Richardson, E. S., Iaizzo, P. A. Pericardial delivery of omega-3 fatty acid: a novel approach to reducing myocardial infarct sizes and arrhythmias. Am J Physiol Heart Circ Physiol. 294 (5), 2212-2218 (2008).
- Chinchoy, E., et al. Isolated four-chamber working swine heart model. Ann Thorac Surg. 70 (5), 1607-1614 (2000).
- Moreno, R., Waxman, S., Rowe, K., Verrier, R. L. Intrapericardial beta-adrenergic blockade with esmolol exerts a potent antitachycardic effect without depressing contractility. J Cardiovasc Pharmacol. 36 (6), 722-727 (2000).
- Jansen, B. J., et al. Intrapericardial delivery enhances cardiac effects of sotalol and atenolol. J Cardiovasc Pharmacol. 44 (1), 50-56 (2004).
- Vereckei, A., Gorski, J. C., Ujhelyi, M., Mehra, R., Zipes, D. P. Intrapericardial ibutilide administration fails to terminate pacing-induced sustained atrial fibrillation in dogs. Cardiovas Drugs Ther. 18 (4), 269-277 (2004).
- Kumar, K., Nguyen, K., Waxman, S., Nearing, B. D., Wellenius, G. A., Zhao, S. X., Verrier, R. L. Potent antifibrillatory effects of intrapericardial nitroglycerin in the ischemic porcine heart. J Am Coll Cardiol. 41 (10), 1831-1837 (2003).
- Fei, L., Baron, A. D., Henry, D. P., Zipes, D. P. Intrapericardial delivery of L-arginine reduces the increased severity of ventricular arrhythmias during sympathetic stimulation in dogs with acute coronary occlusion: nitric oxide modulates sympathetic effects on ventricular electrophysiological properties. Circulation. 96 (11), 4044-4049 (1997).
- Ayers, G. M., Rho, T. H., Ben-David, J., Besch, H. R., Zipes, D. P. Amiodarone instilled into the canine pericardial sac migrates transmurally to produce electrophysiologic effects and suppress atrial fibrillation. J Cardiovasc Electrophysiol. 7 (8), 713-721 (1996).
- Darsinos, J. T., Karli, J. N., Samouilidou, E. C., Krumbholz, B., Pistevos, A. C., Levis, G. M. Distribution of amiodarone in heart tissues following intrapericardial administration. Int. J. Clin. Pharacol Ther. 37 (6), 301-306 (1999).
- Ujhelyi, M. R., Hadsall, K. Z., Euler, D. E., Mehra, R. Intrapericardial therapeutics: a pharmacodynamic and pharmacokinetic comparison between pericardial and intravenous procainamide delivery. J Cardiovasc Electrophysiol. 13 (6), 605-611 (2002).

