Isolation and Propagation of Circulating Tumor Cells from a Mouse Cancer Model
Summary
Circulating tumor cells (CTCs) have been shown to play an important role in tumor metastasis. Here, a method for the isolation and propagation of CTCs from the whole blood of a syngeneic mouse tumor model of hepatocellular carcinoma (HCC) metastasis is described.
Abstract
Cancer metastasis is the foremost cause of cancer-associated deaths. Recent studies have shown that circulating tumor cells (CTCs) are important in cancer metastasis. Indeed, the number of CTCs correlates with tumor size. Here, a detailed description is provided of a methodology for isolation and propagation of CTCs from a syngeneic mouse model of hepatocellular carcinoma (HCC) which allows for downstream analysis of potentially important molecular mechanisms of solid organ tumor metastasis. This method is efficient and reproducible. It is a non-invasive technique and, therefore, has potential to replace the invasive biopsy of tissues from humans which may be associated with complications. Therefore, the method discussed here allows for the isolation and propagation of CTCs from whole blood samples such that they can be examined and characterized. This has potential for future adaptation for clinical applications such as diagnosis, and personalized targeted therapy.
Introduction
The cancer research community has known of the existence of circulating tumor cells (CTCs) since first being observed by Thomas Ashworth in 18691. Since then, CTCs have been shown to be important in tumor metastasis and disease progression2-5. Today, solid tumors are a major cause of morbidity and mortality worldwide. CTCs are rare cells that originate from primary tumors and travel through the blood stream to different organs of which only a small fraction ultimately develop into metastasis2-5. Notably, there is positive correlation between tumor size and CTC number3,4.
An understanding of CTC biology can contribute to the search for targeted therapy. Furthermore, CTCs may have diagnostic applications. To achieve these potential clinical applications, one needs to overcome some current challenges to studying CTCs. One challenge is related to the fact that CTCs may be present as single cells or as clusters and they may even be able to change their phenotype in response to the blood microenvironment2. Moreover, detection can be very challenging, in part, due to the low count of CTCs (a few to hundreds per milliliter) among one billionhematologic cells per milliliterin the blood6. Nevertheless, in recent years, research into the potential clinical applications of CTCs from solid organ cancers has intensified.
Despite these efforts, the challenges of studying and understanding the role of CTCs persist due to the rarity of CTCs and the inadequacy of the technological tools currently available. Despite these challenges, the tremendous potential for clinical applications continues to be an incentive to pursue research into the role of CTCs in cancer metastasis.
We were recently successful in isolating and propagating in cell culture CTCs from an orthotopic syngeneic mouse model of hepatocellular carcinoma (HCC) metastasis5. The purpose of the current paper is to describe in detail all aspects of the successful methodology. The significance of this methodology lies in the fact that this approach may be modified in order to successfully isolate and propagate in culture human CTCs, thus enhancing the possibility of in vitro studies of CTC biology.
There are multiple potential clinical applications for the use of CTCs. CTCs may be useful for prognosis, response monitoring, screening, dynamic monitoring of tumor molecular alterations, and personalized therapy4. Therefore, a better understanding of the biology of CTCs has high potential for clinical impact.
Protocol
Ethics Statement: All animal studies were approved by the Institutional Animal Care and Use Committee (IACUC) of Hunter College of The City University of New York.
1. Pre-experiment Procedures
- Upon arrival to the animal facility, initially house 3 BALB/c mice to a cage with a filtered top and wood chip bedding. Ensure that the mice have free access to food, purina rodent chow, and water. Bedding and food should be changed twice a week while cages should be cleaned once a week.
- Induce general anesthesia in mice using an isoflurane vaporizer along with oxygen at a constant flow rate of 1 L per min. The system is self-reliant and includes a precision vaporizer.
- Position animal in a plastic box that has the precision vaporizer and gas/anesthetic system connected to it. Initially set the vaporizer to 2.5 until animal is unconscious, at which point lower the vaporizer to 2.
- At that time, switch the valve line, sealing it to the box and opening it to the nose cone.
- Move the animal to the preparation area, with the nose cone placed properly and connected (for maintenance anesthesia).
- Prepare 5 × 106 BNL 1ME A.7R.1 mouse HCC cells for implantation per mouse. BNL 1ME A.7R.1 should be growing in medium (10% heat inactivated FBS, 1% penicillin/streptomycin and 2 mM L-Glutamine) incubating in a cell culture dish at 37 °C in a humidified incubator with air and 5 % CO2.
- With a syringe having a 20 G needle, implant 100 µl of PBS containing 5 x 106 BNL 1ME A.7R.1 mouse hepatocellular carcinoma cells into a lobe of the livers of BALB/c mice to produce the primary tumors. Post-implantation, house the BALB/c mice in a cage with the same house requirements as pre-implantation.
- At the experimental end point, clinical evidence of hepatocellular carcinoma is observable as significant reduction in body condition score (usually detected after 4 weeks). At this point, there is a likelihood that primary tumors have developed in the liver while metastatic deposits have formed in the lungs. Thereafter, commence the process of the isolation and propagation of circulating tumor cells.
2. Collection of Whole Blood for Circulating Tumor Cell Isolation
NOTE: Properly sanitized the surgery area and make sure it is clutter-free. Autoclave all surgical instruments or soak in a disinfectant according to manufacturer’s recommendation
- Sacrifice mice by humane euthanasia at experimental end-point (clinical evidence of tumor development). Euthanization of mice should involve CO2 asphyxiation. Observe the mice properly to confirm anesthesia is correctly distributed. Carbon dioxide should be released slowly into the chamber over a period of 5 – 10 min, causing respiratory arrest. Follow by intracardiac exsanguination and cervical dislocation.5
- Use a 25 G needle attached to a 1 ml syringe for collection of whole blood. Heparanize the syringe with 0.01 ml heparin. Puncture skin immediately with the needle inferior to the xyphisternum at approximately 45° angle. Advance needle slowly to puncture the heart, and then lower the angle of the needle. Steadily hold the needle and syringe in place, and draw out 500 µl – 1,000 µl of whole blood from the heart.
- Remove the syringe and needle and transfer the blood into a pre-labelled 1.5 ml pyrogen-free tube. Centrifuge the whole blood collected into 1.5 ml pyrogen-free tube at 1,167 x g for 5 min.
NOTE: The centrifugation separates the whole blood sample into three layers: the bottom or hematocrit layer consisting of red blood cells, an intermediate thin layer of buffy coat, and a top plasma layer. - Remove the plasma carefully by pipetting and collecting it into a separate 1.5 ml pyrogen-free tube for further experiments such as detection of cell-free nucleic acids7.
- For isolation of circulating tumor cells (CTCs), collect the buffy coat into a separate 1.5 ml pyrogen-free tube in preparation for red blood cell (RBC) lysis. This is necessary to remove residual RBCs in the buffy coat layer.
3. Recipe for Red Blood Cell (RBC) Lysis Buffer
- Make a 1 L solution of RBC lysis buffer.
- Make a 500 ml of Ammonium Chloride and 500 ml of Tris. Then mix to create a 1 L solution of RBC lysis buffer with a final concentration of 155 mM of Ammonium Chloride and 10 mM of Tris.
- Buffer the solution to a pH of 7.5. Store the RBC lysis buffer at +2 to +8 °C, and bring to RT immediately prior to use.
4. RBC Lysis of Buffy Coat
- Add 1 ml of RBC lysis buffer to the sterile pyrogen-free 1.5 ml tube containing the collected buffy coat.
- Mix the contents of the tube by inverting multiple times. Incubate the mixed contents of the tube for 5 min at RT on the bench. Centrifuge the mixed contents for 5 min at 1,167 x g.
- After centrifugation, a whitish pellet will be obtained. Discard the reddish supernatant slowly and carefully into 10% bleach without any disruption to the pellet. If the pellet is not whitish, re-do steps 3.1 – 3.5. Increasing the incubation time in RBC lysis buffer to 10 – 15 min may also be helpful.
- Safely dispose the biological waste once decontamination of biological waste in 10% bleach is complete after 24 hr.
5. Propagation in Cell Culture
NOTE: Tumor cells are typically rapidly growing cells white blood cells that are abundant in the original mix of cells seeded into the dish. Following repeated change of medium, and subsequent passages of CTCs, all the white blood cells are removed and a relatively pure population of CTCs remains.
- After discarding the supernatant into 10% bleach, wash the whitish pellet in 1 ml phosphate-buffered saline (PBS) at least once, and resuspend in complete culture medium for propagation in cell culture.
- Once wash is completed, add 1 ml of complete BNL 1ME A.7R.1 culture medium (10% heat inactivated FBS, 1% penicillin/streptomycin and 2mM L-Glutamine) to the pellet and resuspend the pellet in the culture medium.
- Seed the resuspended mix of cells into a cell culture dish. Incubate the cell culture dish at 37 °C in a humidified incubator with air and 5% CO2.
- Change the cell culture medium every two to three days. After five to seven days, CTCs will have adhered to the cell culture dish and started proliferating. These cells will remain viable beyond 25 passages and after repeated freezing and thawing cycles5.
6. Verification of Hepatocellular Carcinoma Circulating Tumor Cell Line
- Perform a polymerase chain reaction (PCR)-based method, to amplify only one specific DNA segment of the mouse β-globin gene to confirm that the novel established CTC cell lines are mouse cells like the original implanted BNL 1ME A.7R.1 HCC line. Method was originally described and validated by Steube et al.5,8
- Perform immunostaining for the hepatocyte-specific marker cAMP responsive element binding protein 3-like 3 (CREB3L3) by using a specific antibody. This will confirm that novel established CTCs are indeed hepatocytes like the original implanted BNL 1ME A.7R.1 cell line.5
Representative Results
The methodology described here has shown that CTCs can be isolated. Mice were humanely euthanized at experimental end-point. The process involved carbon dioxide asphyxiation, intra-cardiac exsanguination, and cervical dislocation. A schematic of key steps of the procedure are illustrated in Figure 2. Whole blood samples collected by intra-cardiac exsanguination were processed using the protocol described above. After which, centrifugation was used to determine the buffy coat layer that was subjected to the RBC lysis. This was then followed by another centrifugation step to collect a whitish pellet that was washed in PBS, and resuspended in complete cell culture medium for propagation in cell culture. An issue that may come up is that the pellet obtained may not be clear in color. This problem can be easily resolved by modifying the incubation time in RBC lysis buffer by increasing it to 10 – 15 min, or possibly by performing it additional times. Representative images of CTCs being propagated in culture from three different mice are shown in Figure 1 as compared to BNL 1ME A.7R.1 cells. Rapid proliferation and morphology will help identify if cells seeded are CTCs. However, verification techniques such as PCR and immunostaining described in steps 5.1 – 5.2 are needed to confirm the establishment of a viable CTC line.
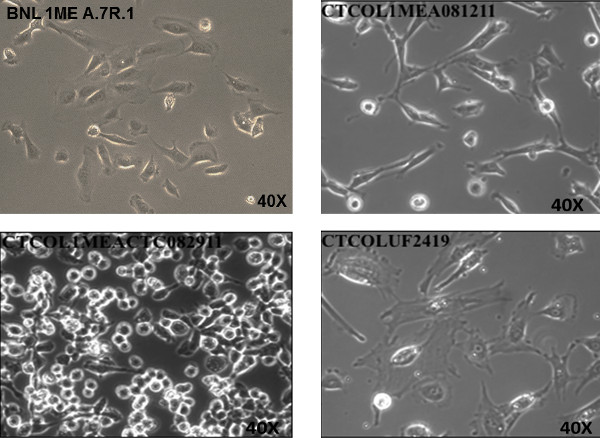
Figure 1. Propagation of Circulating Tumor Cells (CTCs) in Cell Culture from an Orthotopic Syngeneic Mouse Model of Hepatocellular Carcinoma Metastasis. Cells were propagated in 15 mm x 60 mm cell culture dishes in BNL 1ME A.7R.1 culture medium. Proliferating tumor cells adhered to dish and are shown from three separate mice. Tumor cells have remained viable through continuous passages. Phase contrast image of CTCOL1MEA081211, CTCOL1MEACTC082911 and CTCOLUF2419 were taken at 40X magnification (objective lens) in comparison to BNL 1ME A.7R.1 cells (40X magnification). Please click here to view a larger version of this figure.

Figure 2. Schematic of the Procedure of Isolating and Propagating Circulating Tumor Cells from a Mouse Cancer Model System. When clinical evidence of hepatocellular carcinoma was observed as significant reduction in body condition score, whole blood was collected from mice. After several centrifugations, RBC lysis, and several wash steps, the CTCs were seeded into cell culture dishes for propagation. Tumor cell proliferation was observed after several days. Please click here to view a larger version of this figure.
Discussion
In this study, how to isolate and propagate CTCs from the whole blood of a syngeneic mouse with HCC were described. The objective of this work is to enhance the ongoing studies of the mechanisms of cancer metastasis.
A factor that contributes to the poor prognosis of many cancers is the lack of timely detection and consequent widespread dissemination of the malignancy3. CTCs originate from primary cancers and spread via the blood stream to distant organs. As such, CTCs are important in cancer metastasis and have potential for multiple clinical applications, such as for diagnosis3,9,10. This is supported by data from several clinical studies that have showed that CTCs have a strong correlation with the progression of the disease in various cancer types such as pancreatic, prostate and breast, among others3,11,12. Despite the fact that CTCs may be very useful for cancer diagnosis, isolating and characterizing CTCs are challenging because very few are present among billions of blood cells3,7.
A significance of isolating CTCs using our technique is that it provides a reproducible model which can be optimized for potential utility in a clinical setting. This may ultimately lead to the possibility of using this method to replace current biopsy procedures that are more invasive and associated with side effects3,13. Modifications of this method, such as peripheral intravenous blood collection, would be expected to utilize it in a clinical setting. Of course, it is acknowledged that isolating CTCs from humans is a much more challenging endeavor due to general low yield4,14. But, in many cases such as in pancreatic cancer, histo-pathologic or cytological validation using tissue biopsies is fruitless in spite of multiple invasive attempts4. Isolating CTCs has been described as “liquid biopsy” or “live biopsy” of a tumor which enables noninvasive cancer diagnosis and real-time monitoring of therapeutic response3. This methodology models the isolation of CTCs in a mouse model. Thus far, only one FDA- cleared assay, such as Veridex CellSearch System, has been approved to detect CTCs. In one study, this technology identified CTCs in only 11 of 26 patients. This system is associated with some inefficiency, limited purity and high cost4. Moreover, it lacks the important feature of being able to collect viable CTCs whether they are EpCAM-expressing or not for extensive analyses using multiple in vitro and in vivo methods.
An important feature of the described method is that it preserves the viability of the isolated CTCs3. Therefore, using this method allows for the subsequent extensive downstream analysis of both the molecular and functional characteristics of CTCs using a variety of techniques.
In conclusion, a methodology for isolating and propagating CTCs that are viable and can be characterized molecularly and functionally is presented. There is potential that this method can be optimized for clinical applications.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Work in Dr. Ogunwobi’s laboratory is supported by a Research Centers in Minority Institutions Program grant from the National Institute on Minority Health and Health Disparities (MD007599) of the National Institutes of Health. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of the NIMHD or the NIH.
Materials
| Heparin Sodium Salt 1G | VWR | 89508-852 | |
| BTX Tube Micro 1.5mL Clear NS | VWR | 89511-254 | 1.5mL pyrogen-free eppendorf Tubes |
| Needle Sterile Disp BD 25GX1IN | VWR | BD305125 | 25G Needle |
| Slp Tip SRNG 1ml 200 each per pack | VWR | BD309659 | 1mL syringe tip |
| Syringe 1ml leur lok Pk 100 | VWR | BD309628 | 1 mL syringe |
| VWR Forceps Tissue 6 | VWR | 82027-446 | Forceps |
| Cyromold intrm 15X15X5MM PK 100 | VWR | 25608-924 | Cyromold |
| Cryo-oct compund 4oz | VWR | 25608-930 | Oct compound |
| VWR Slide sprfrst 25X75MM PK72 | VWR | 48311-703 | Slides |
| VWR Cover Glass #2 22X5oMM OZ | VWR | 48-382-128 | Cover Glass |
| VWR Slide Box True North Fm Pu | VWR | 89140-278 | Slide Box |
| Super HT PAP Pen | VWR | 89427-058 | PAP pen |
| Water RNASe and DNAse free 2L | VWR | 101454-204 | Nuclease Free Water |
| Buffer Tris Ultra Pure Grade 500G | VWR | 97061-796 | Tris Buffer |
| Ammonium Chloride ACS Grade 2 5KG | VWR | 97062-048 | Ammonium Chloride Buffer |
| Falcon Tissue Culture Dish 60 x 15mm Style polystyrene | VWR | 353002 | Tissue Culture Dish |
| Clorox® Germicidal Bleach, Regular | VWR | 89501-620 | Clorox Bleach |
| PBS, 1X (Phosphate-Buffered Saline) without calcium & magnesium (500mL) | Thermo Fischer Scientific | 21-040-CV | PBS, 1X |
| DMEM, 1X with 4.5 g/L glucose & L-glutamine without sodium pyruvate | Thermo Fischer Scientific | 10-017-CV | DMEM 1X media for BNL 1ME A.7R.1 cells |
| Fetal Bovine Seru8m | Thermo Fischer Scientific | 35-010-CV | FBS |
| Penicillin Streptomycin Solution, 100X | Thermo Fischer Scientific | 30-002-Cl | Penicillin Streptomycin |
| Sorvall Biofuge pico | Thermo Fischer Scientific | 75002411 | 13000rpm Centrifuge |
References
- Ashworth, T. R. A case of cancer in which cells similar to those in the tumors were seen in the blood after death. Australian Medical Journal. 1869, 146-147 (14).
- van de Stolpe, A., den Toonder, J. Circulating Tumor Cells: What Is in It for the Patient? A Vision towards the Future. Cancers. 6, 1195-1207 (2014).
- Sheng, W., et al. Capture, release and culture of circulating tumor cells from pancreatic cancer patients using an enhanced mixing chip. Lab on a chip. 14, 89-98 (2014).
- George, T. J., Ogunwobi, O. O., Sheng, W., Fan, Z. H., Liu, C. "Tissue is the issue": circulating tumor cells in pancreatic cancer. Journal of gastrointestinal cancer. , (2014).
- Ogunwobi, O. O., Puszyk, W., Dong, H. J., Liu, C. Epigenetic upregulation of HGF and c-Met drives metastasis in hepatocellular carcinoma. PloS one. 8, e63765 (2013).
- Cheng, B., et al. Transparent, biocompatible nanostructured surfaces for cancer cell capture and culture. International journal of nanomedicine. 9, 2569-2580 (2014).
- Yu, M., Stott, S., Toner, M., Maheswaran, S., Haber, D. A. Circulating tumor cells: approaches to isolation and characterization. The Journal of cell biology. 192, 373-382 (2011).
- Steube, K. G., Koelz, A. L., Drexler, H. G. Identification and verification of rodent cell lines by polymerase chain reaction. Cytotechnology. 56, 49-56 (2008).
- Pantel, K., Alix-Panabieres, C., Riethdorf, S. Cancer micrometastases. Nature reviews. Clinical oncology. 6, 339-351 (2009).
- Pantel, K., Brakenhoff, R. H., Brandt, B. Detection, clinical relevance and specific biological properties of disseminating tumour cells. Nature reviews. Cancer. 8, 329-340 (2008).
- Cristofanilli, M., et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. The New England journal of medicine. 351, 781-791 (2004).
- De Giorgi, U., et al. Circulating tumor cells and bone metastases as detected by FDG-PET/CT in patients with metastatic breast cancer. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. 21, 33-39 (2010).
- Eloubeidi, M. A., et al. Endoscopic ultrasound-guided fine needle aspiration biopsy of patients with suspected pancreatic cancer: diagnostic accuracy and acute and 30-day complications. The American journal of gastroenterology. 98, 2663-2668 (2003).
- O’Toole, D., et al. Assessment of complications of EUS-guided fine-needle aspiration. Gastrointestinal endoscopy. 53, 470-474 (2001).

