Fabrication of a Functionalized Magnetic Bacterial Nanocellulose with Iron Oxide Nanoparticles
Summary
Here, we present a protocol to make a bacterial nanocellulose (BNC) magnetic for applications in damaged blood vessel reconstruction. The BNC was synthesized by G. xylinus strain. On the other hand, magnetization of the BNC was realized through in situ precipitation of Fe2+ and Fe3+ ferrous ions inside the BNC mesh.
Abstract
In this study, bacterial nanocellulose (BNC) produced by the bacteria Gluconacetobacter xylinus is synthesized and impregnated in situ with iron oxide nanoparticles (IONP) (Fe3O4) to yield a magnetic bacterial nanocellulose (MBNC). The synthesis of MBNC is a precise and specifically designed multi-step process. Briefly, bacterial nanocellulose (BNC) pellicles are formed from preserved G. xylinus strain according to our experimental requirements of size and morphology. A solution of iron(III) chloride hexahydrate (FeCl3·6H2O) and iron(II) chloride tetrahydrate (FeCl2·4H2O) with a 2:1 molar ratio is prepared and diluted in deoxygenated high purity water. A BNC pellicle is then introduced in the vessel with the reactants. This mixture is stirred and heated at 80 °C in a silicon oil bath and ammonium hydroxide (14%) is then added by dropping to precipitate the ferrous ions into the BNC mesh. This last step allows forming in situ magnetite nanoparticles (Fe3O4) inside the bacterial nanocellulose mesh to confer magnetic properties to BNC pellicle. A toxicological assay was used to evaluate the biocompatibility of the BNC-IONP pellicle. Polyethylene glycol (PEG) was used to cover the IONPs in order to improve their biocompatibility. Scanning electron microscopy (SEM) images showed that the IONP were located preferentially in the fibril interlacing spaces of the BNC matrix, but some of them were also found along the BNC ribbons. Magnetic force microscope measurements performed on the MBNC detected the presence magnetic domains with high and weak intensity magnetic field, confirming the magnetic nature of the MBNC pellicle. Young's modulus values obtained in this work are also in a reasonable agreement with those reported for several blood vessels in previous studies.
Introduction
The bacterian nanocellulose (BNC) is synthesized by Acetobacter xylinum strain, also known as Gluconacetobacter xylinus, and deposited in the form of films or pellicles on the air-liquid interface during stationary culture. These BNC pellicles adopt the form of the container where they are grown, and their thickness depends on the number of days in culture. A. xylinus uses the glucose in the medium for the synthesis of the cellulose microfibrils through a process of polymerization and subsequent crystallization. The polymerization of the glucose residues is carried out at the bacterial extracellular membrane where glucan chains are extruded from single pores distributed over the cell envelope. The crystallization of the cellulose microfibrils occurs in the extracellular space with the formation of glucan chain sheets by van der Waals bonding followed by stacking of the sheets by H-bonding1.
Magnetic nanoparticles integrated to a BNC matrix can be manipulated easily by an external magnetic field in order to increase the force necessary to direct and confine smooth muscle cells (SMCs) containing magnetic nanoparticles, at the damaged site of the arterial wall. This strategy keeps the SMCs away from other tissues, and holds the cells in place against the force exerted by the blood flow. It has been shown that SMCs play an important role in the vasoelasticity of the blood vessel, where they form abundant layers located mainly in the tunica media2.
The method used for the synthesis of MBNC involves BNC pellicle immersed and stirred in a solution of iron(III) chloride hexahydrate and iron(II) chloride tetrahydrate at 80 °C. Ammonium hydroxide is added to form iron oxide nanoparticles inside the BNC mesh. The addition of ammonium hydroxide changes the color of the solution from orange to black. The IONPs compact together along the BNC fibrils with a non-uniform distribution.
This protocol focuses on the design of a bacterial nanocellulose-magnetic nanoparticle pellicle, which we have named magnetic bacterial nanocellulose (MBNC), which is intended to use as a substitute for missing, damaged or injured small-diameter blood vessels. H. S. Barud and coworkers have recently published a similar work to produce a BNC-based flexible magnetic paper by mixing BNC pellicles in a stable aqueous dispersion of PEG and superparamagnetic iron oxide nanoparticles3. Here, we describe the production of bacterial cellulose and its impregnation in situ with magnetic nanoparticles. A cytotoxicity assay based on detection of single DNA strand breaks was used to test the biocompatibility of the BNC and MBNC pellicles.
Protocol
1. Preparation of Bacterial Nanocellulose (BNC)
Note: All of the steps are performed under aseptic conditions, unless otherwise indicated.
- Prepare culture medium.
- Prepare 500 ml of liquid culture medium by combining 25 g of yeast extract, 15 g of peptone, 125.0 g of mannitol, and 500 ml of high purity water. Autoclave this mixture at 120 °C for 20 min and store at 4 °C.
- Prepare 100 ml of semisolid media by adding 15 g of agar to 5.0 g of yeast extract, 3.0 g of peptone, 25.0 g of mannitol, and 100 ml of high purity water. Autoclave this mixture at 120 °C for 20 min. Once autoclaved, deposit 5 ml of the mixture in a 90 mm x 16 mm plastic Petri dish. Allow the solution to gel at 4 °C and store at this temperature until further use.
- Rehydrate G. xylinus strain preserved in freeze-dried vials by adding 1 ml of liquid culture medium and pipetting up and down, as indicated by the manufacturer's instructions.
- Inoculate the Petri dishes containing semisolid media with small droplets of bacterial suspension using an inoculating loop. Make sure that the inoculum covers the entire Petri dish by moving the loop in a zig zag direction from the edge to the center of the dish.
- Incubate the Petri dishes at 26 °C for 72 hr in an incubator without CO2. Once the incubation period is complete, small white colonies are visible. If the colonies are not immediately used, store the Petri dishes at 4 °C by sealing the lid with Parafilm and placing the dishes upside down. The colonies can be stored in that way for up to 6 months.
- Transfer 2 ml of the liquid culture medium prepared in step (1.1.1) into each well of a 24-well tissue culture plate. Take two colonies with an inoculating needle from the inoculated Petri dishes in step (1.3) and place them into the first well of the tissue culture plate. Repeat the same procedure for the remaining 23 wells.
- Incubate the tissue culture plate at 30 °C for 7 days. This will yield a total of 24 BNC pellicles with diameter of 16 mm and a thickness of approximately 2-3 mm diameter as depicted in Figure 1.
Note: Do not disturb the bacterial culture at any point during incubation period, for example by shaking the plates. During the incubation period, G. xylinus extrudes glucopyranose sugar molecules to form a polymeric crystalline mesh in the air-liquid interface, which adopts the shape and size of the flask under static cultivation conditions. This polymeric matrix, known as bacterial nanocellulose (BNC), is conspicuous at end of the incubation period. - Collect the BNC pellicles from the growth media and sterilize them in 200 ml of 1% NaOH solution for 1 hr at 50 °C, in order to remove all traces of G. xylinus. Optionally, stir this solution at 300 rpm using a magnetic bar and a stirring plate. Discard the NaOH solution and add 200 ml of freshly prepared 1% NaOH solution. Repeat the same process once more or until the BNC pellicles in solution acquires a translucent appearance.
- Rinse the BNC pellicles with water three times and store them in high purity water at RT. Make sure the BNC pellicles are completely submerged in the water and are not allowed to dry at any time.
- Autoclave the BNC pellicles at 121 °C for 20 min.
Note: An in vivo subcutaneous study in the rat performed by Märtson and coworkers showed non-degradation signs of the BNC after 60 weeks implantation. Indeed, BNC is degradable in nature by microbial and fungal enzymes, which are absent in mammals. On the other hand, the biodegradability of the BNC can be the result of mechanical, chemical, and biological processes that weaken the microfibril network in vivo4.
2. Synthesis of Polymer-coated Iron Oxide Nanoparticles and Its Deposition in a Bacterial Nanocellulose Membrane
- Bubble 1,000 ml of high purity water with nitrogen gas in order to remove any dissolved oxygen in the water and replace it with nitrogen.
- Use a three-neck round bottom flask to prepare a solution in a 2:1 molar ratio of iron(III) chloride hexahydrate (FeCl3·6H2O) and iron(II) chloride tetrahydrate (FeCl2·4H2O) diluted with the deoxygenated high purity water. For example, use 5.4 g of FeCl3·6H2O and 1.98 g of FeCl2·4H2O in 10 ml of deoxygenated high purity water. If this preparation turns too viscous and difficult to stir, use 0.54 g of FeCl3·6H2O and 0.198 g of FeCl2·4H2O in 20 ml of deoxygenated high purity water.
Note: Reduce the exposure time of the FeCl2·4H2O to air by weighing this chemical compound as fast as possible. Once introduced in the three-neck round bottom flask, close the three-neck round bottom flask with septum stoppers until it is connected to the nitrogen gas supply and the condenser tube. - Use two necks of the vessel to provide a constant entrance and output of nitrogen gas by connecting the nitrogen gas supply to a needle punched in a septum stopper and fixed to the vessel's necks.
- Place 1 BNC pellicle which was prepared previously in step 1.5 (15.6 mm of diameter and 2-3 mm of thickness) in the vessel with the reactants. Make sure the sample is completely submerged in the liquid.
- Connect the remaining neck of the vessel to a condenser tube. Additionally, use a drying tube filled with anhydrous calcium sulfate on top of the condenser tube. Run water through the condenser tube.
- Seal all of the glass joints with vacuum grease.
- Heat the solution in a silicone oil bath to 80 °C using a stirring hotplate and hold this temperature until step 2.10. Use a small magnetic stir bar to mix the reactants at 350 rpm for 5 min. Make sure the BNC is appropriately impregnated with the ferrous solution and the reactants are completely dissolved. Keep stirring the mixture until the end of the experiment.
Note: Utilize a thermometer to verify the temperature of the silicone oil. It should be stable to 80 °C. - Increase the stirring velocity to 700 rpm and add (by dropping), in a time interval of 5 min, 5 ml of ammonium hydroxide (NH4OH, 14%) to the 10 ml of ferrous solution using a pipetting needle, which has been also punched in a septum stopper. After addition of the ammonium hydroxide, the color of the solution changes from yellow/orange to black.
- Continue stirring the solution at 80 °C for another 5 min. Avoid high-speed stirrings in order to maintain the integrity of the sample. High speeds, i.e., higher than 1,000 rpm, can destroy the sample.
- Lower the temperature of the solution to 30 °C using the temperature control bottom of the stirring hotplate and keep stirring for another 5 min. Then, turn off the hot plate. At this point, the IONP have been incorporated into the BNC mesh.
- Cool the mixture down to RT and separate the magnetic nanoparticles (MNP) and BNC using a strong permanent magnet (e.g., 1 Tesla). To do this, transfer the mixture to a vessel flask and then, while keeping the magnet close to the vessel, hold the MNPs and the BNC in place while decanting the supernatant.
Note: Be careful when handling strong magnets since they can be harmful when used incorrectly. For steps (2.12)-(2.14) and (2.16) use the deoxygenated high purity water prepared previously in (2.1) to prevent particles from oxidation. - Resuspend the MNPs and BNC in 100 ml water. Gently shake the solution to remove all the MNPs that are not strongly incorporated into the BNC. Decant the supernatant again by holding the MNPs and the BNC in place using the magnet.
- Wash the MNPs and the BNC several times with water until the supernatant reaches neutral pH (pH~7), as measured using a colorimetric strip.
- Separate the magnetic-functionalized BNC or magnetic bacterial nanocellulose (MBNC) from the MNPs using tweezers and rinse the MBNC several times with water until the water runs clear.
- Sterilize the MBNC by exposing the MBNC O/N to UV (110-280 nm).
- Autoclave 500 ml of deoxygenated high purity water at 120 °C for 20 min and store the MBNC in 20 ml of this water.
- Aseptically, immerse the sample in 1% of PEG and stir for 2 hr at RT (37 °C). This procedure improves the biocompatibility and stability of the iron oxide nanoparticles deposited in the BNC, specifically those exposed at the surface5-7. The PEG coating will be distributed over the MBNC 3D network.
Note: Naked IONP are easily oxidized in air because of their high chemical activity8. Even though PEG is considered a non-biodegradable material, its chemical stability depends on the applied biological conditions such as water content, pH, temperature, presence of enzymes, reactive oxygen species, reactive nitrogen species, and others9.
3. Characterization of the BNC and MBNC Pellicles
- Mechanical properties
- Perform normal loading and unloading nanoindentation test using a Berkovich indenter. The radius of Berkovich diamond indenter is 20 nm.
- Use fused silica and tungsten to calibrate contact area as a function of indentation depth at RT. During the test, mount the samples on the indentation using glue. The indenter approached the samples in its thickness direction.
- Randomly select indentation locations on samples surfaces. Keep the spacing between 2 indents between 200-300 mm.
- Apply the load to the samples in steps and record the corresponding displacement of the indenter. Analyze the plot of load vs depth to find the Young's modulus.
- Carry out the nanoindentation test of the samples in the presence of deionized water (DI water), and test by applying load rates between 0.0001 mN/sec and 0.005 mN/sec, with peak load between 0.01 mN and 0.60 mN.
- Use a liquid cell and keep samples under the liquids environment. This unique setup for nanomechanical characterization immersed in a fluid environment is ideal to effectively simulate the reach biomechanical functionality of BNC and MBNC membranes.
- Structural Characterization by SEM
- Characterize the nanocellulose fiber structure by scanning electron microscopy (SEM).
- Lyophilize the samples for 24 hr at -80 °C. Then mount on SEM studs, sputter with Au-Pd film for 10 sec and analyze using SEM.
- Takes images at a magnification of 22,000X and 60,000X, with an acceleration voltage of 5 kV.
- Magnetic domains
- Allow the MBNC pellicles to completely dry at RT, and subsequently expose for 5 min to a permanent magnet (1 Tesla).
- Immediately, carry out the magnetic force measurements using a bio-AFM according to manufacturer's protocol.
- For each measurement, capture first the topography features and acquire the magnetic domains during a second pass. Obtain both measurements with the bio-AFM in non-contact mode.
- Magnetic characterization of the nanoparticles is conducted using vibrating sample magnetometer (VSM) in the physical property measurements system (PPMS) of Quantum Design, at RT (300 K), with a magnetic field in the range of -10,000 to 10,000 Oe.
- Cytocompatibility
- Seed human aortic smooth muscle cells (HASMC) in a 6-well tissue culture plate at a density of 1.0×103 cells/cm2 and incubate for 24 hr in the presence of the test samples: BNC and MBNC pellicles (each with a 15.6 mm of diameter).
- Use populations of untreated and hydrogen peroxide treated cells as negative and positive controls, respectively.
- Perform the Comet assay according to manufacturer's protocols and the guidelines suggested by A. Azqueta & A.R. Collins10.
- Use the nucleic acid dye SYBR Gold in this assay to intercalate and fluorescently label the DNA contained in the electrophoresed samples according to manufacturer's protocol.
Note: Cells that do not undergo any DNA damage in the presence of BNC and MBNC samples, will show a fluorescent round green nucleoid, whereas DNA damaged cells will have long comets – positive samples will have nucleoids (the head of the comet) followed by tails which contain fragmented DNA material (percentage of DNA in the tail).
Representative Results
The incubation period of G. xylinus was a total of 9 days, but the pellicles began to form earlier and were evident after about 2 days. The macroscopic appearance of the BNC is displayed in Figure 1, the shape of which mimics that of the dish-grown culture. Figure 2 describes the process for producing BNC-IONP pellicles, which summaries the main steps involved in the protocol above as well as the configuration of the main components.
SEM images were used to resolve the microstructure, morphology, and spatial distribution of the fibers of BNC (Figure 3) and IONP distribution in functionalized BNC (Figure 4). The BNC is formed by fine ribbons (approximately 50 nm in diameter) that form open pores across the entire network without a defined pattern. The IONP are preferentially located between the pores formed by fibril interlacing, forming clusters of 100 nm or more in size. Individual IONP are also bound along the ribbons. The MBNC exhibits a less compacted fibril structure compared to the BNC, probably because IONP bring together the BNC's ribbons. Magnetic force microscope was used to reconstruct the magnetic profile at the topography of the MBNC (Figure 5A, B). Large pores of 500 nm diameter or larger are formed in the MBNC, which were not observed in untreated BNC (Figure 5A). This is in agreement with the observations in the SEM microphotographs, where the MBNC displays a more porous structure than the unmodified BNC. A magnetic force gradient with two domains of different magnetization was detected across the MBNC surface (Figure 5B), whose contrast does not correlate with the hills and valleys formed by IONP-rich regions in the MBNC topographic images (Figure 5A). High and weak intensity magnetic fields are denoted as yellow and green in Figure 5B respectively. The hysteresis loop of the nanoparticles, which is measured embedded in the bacterial nanocellulose, is shown in Figure 5 providing evidence that all the IONPs were superparamagnetic at RT, with no hysteresis.
HASMC were cultured in the presence of BNC and of MBNC to test for any detrimental effect on the viability of individual cells as a result of exposure to these foreign materials. The extent of damage in individual cells was quantified by the detection of DNA strand breaks (Figure 6). The results were compared to HASMC growing under normal culturing conditions of 37 °C, 95% air, and 5% CO2 (negative control) and to HASMC with hydrogen peroxide-induced genotoxicity (100 µM H2O2) for 30 min (positive control). Paired comparisons using t-test showed that the effects of the MBNC on cell viability were significantly different from those induced with hydrogen peroxide treatment on HASMC (p-value < 0.001, ***).

Figure 1. Macroscopic aspects of bacterial nanocellulose. BNC pellicles have been obtained after an 11-day incubation period, which are approx. 3 mm in thickness. The incubation period depends on the requirements for the intended use. Please click here to view a larger version of this figure.
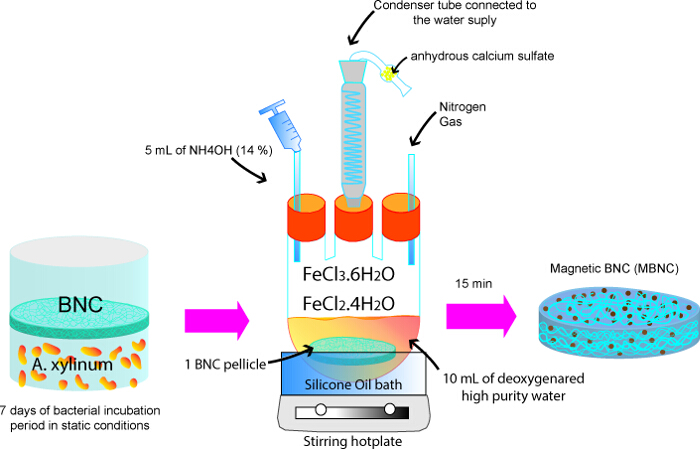
Figure 2. Fabrication of magnetically functionalized bacterial nanocellulose. Iron oxide nanoparticles are assembled and incorporated in situ within the BNC, yielding a MBNC. Please click here to view a larger version of this figure.

Figure 3. SEM image of BNC. The BNC displays a fine network and non-aggregated ribbons with sizes of 50 nm or less. Please click here to view a larger version of this figure.

Figure 4. SEM image of BNC-IONP pellicle. Iron oxide nanoparticles (IONP) are preferentially positioned between the interlacing ribbons. Please click here to view a larger version of this figure.
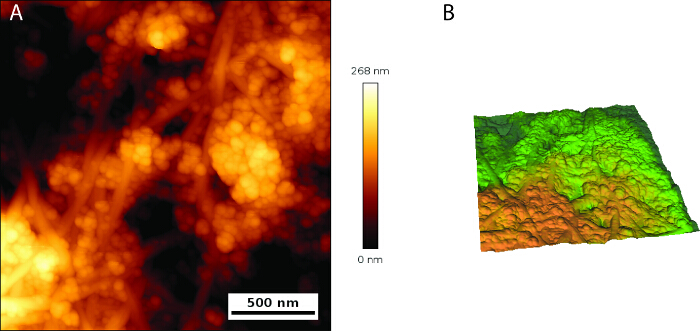
Figure 5. AFM topography of MBNC and magnetic domain structures. (A) Surface topography of MBNC showing spots of highly packaged nanoparticles, which stand above the nanofibril structure. (B) Yellow and green domains denote two regions of different magnetization of high and weak intensity magnetic field respectively. Please click here to view a larger version of this figure.
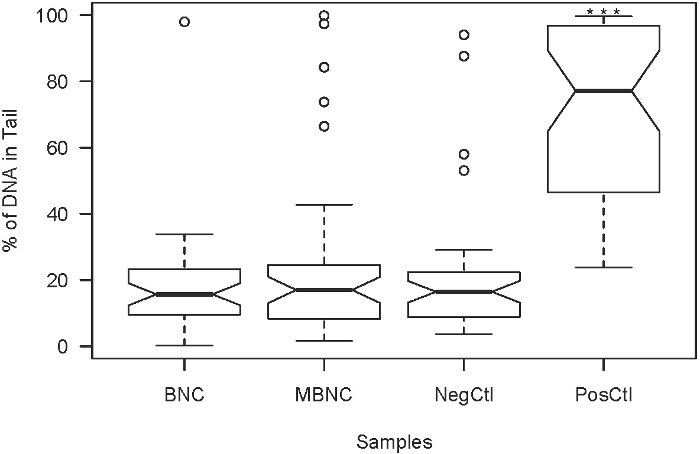
Figure 6. Extent of DNA damage in HASMC after being exposure to BNC, and MBNC respectively. PosCtl denotes HASMC that underwent hydrogen peroxide treatment for comparative purposes. NegCtl denotes HASMC growing under normal culturing conditions. The detrimental effects of the MBNC on HASMC viability were significantly different from those observed in the PosCtl (p-value<0.001, ***). Please click here to view a larger version of this figure.
Discussion
The thickness and size of the BNC pellicle can be easily manipulated by changing the incubation time and the size of the flask in which it is grown during static cultivation. The microproperties of the BNC, such as porosity, can be modified by changing the oxygen ratio in the static culture. Higher oxygen concentrations yield tougher BNC11. A. Bodin and coworkers produced tubes of BNC with a burst pressure up to 880 mm Hg by changing the oxygen ratio from atmospheric oxygen to 100% oxygen during the fermentation process of G. xylinus12. Similarly, the porosity of the BNC can also be introduced by incorporating porogens such as paraffin wax microspheres into the fermentation process. The resulting porosity and pore interconnectivity in this case will depend on the porogen size13.
The porous network of the BNC allows them to be functionalized with nanoparticles, e.g., for drug delivery agents. In our study, we have functionalized BNC with IONP by synthesizing and growing in situ the nanoparticles into the BNC membrane, in order to implement a magnetic protocol for rapid cell recruitment and attachment in BNC-based scaffolds. Nanomechanical tests reveal that nanoscale response of BNC behaves similarly with blood vessels14 with a very low Young's modulus, EBNC = 0.0025 GPa inside the samples to 0.04 GPa at the surface. The values obtained are in the range with those observed by Fu et al.15.
The excess of IONP could easily be removed from the BNC due to the high porosity of the material. SEM photographs showed that the nanoparticles are distributed mainly in the spaces formed by fibril interlacing and dispersed along the ribbons. The concentration of the iron species used in this protocol yielded high densely packaged IONP, which brought together the BNC's ribbons. This resulted in a MBNC with larger pores than those of the unmodified BNC. Olsson et al., who used different concentrations of FeSO4/CoCl2 salts with the same volume fraction of BNC in the synthesis of cellulose nanofibril aerogels, reported a similar increase in the BNC porosity when they changed the volume fraction of the ferromagnetic cobalt ferrite nanoparticles from 0.7% to 5.7%16. This high porosity in the MBNC can be advantageous for the deposition of drugs that increase the recovery time and avoid restenosis at damaged arterial walls.
The lack of correlation between the topographical features and magnetic phase images have also been described by B. Torre et al.17, who pointed out the independence between the topography and the magnetic signals of sparse nanoparticle films. Further characterization studies need to be conducted to determine the magnetization hysteresis (M-H) loops of the MBNC via SQUID-VSM systems.
The MBNC showed low potential for toxic effects, according to the results observed in the Comet Assay, indicating that this material is biocompatible for using in contact with cells.
The most critical steps in the procedure are related to the amount of ammonium hydroxide and the speed at which it is added, as well as ensuring the complete immersion and stirring of the BNC in the solution during the reaction. The first aspect determines the size of the resulting iron oxide nanoparticles, while the second one influence how the nanoparticles are distributed across of the BNC matrix. In order to better control the size of the MNPs, a burette with a stopcock can be used to regulate the addition by dropping ammonium hydroxide into the reaction. Small pieces of BNC that can be completely submerged in the solution are advised, e.g., sizes of approximately 1.9 cm2 for a total volume of 10 ml of solution. One limitation of this technique is the inhomogeneous distribution of the IONP inside the BNC mesh.
This protocol describes a method for incorporating iron oxide nanoparticles in BNC to form a composite. Because of the biocompatibility and the physical and mechanical properties of both the BNC and the iron oxide nanoparticles, the MBNC can be used in a variety of biomedical applications such as drug delivery systems and scaffolds for cellular growth.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was funded by Department of Defense under contract No. W81XWH-11-2-0067
Materials
| Glucoacetobacter Xylinus | ATCC | 700178 | |
| Agar | Sigma Aldrich | A1296-500G | |
| D-Mannitol Bioxtra | Sigma Aldrich | M9546-250G | |
| Yeast Extract | BD Biosciences | 212750 | |
| Bacteriological Peptone | Sigma Aldrich | P0556 | |
| Sodium Hydroxide, 50% Solution In Water | Sigma Aldrich | 158127-100G | |
| Iron(III) Chloride Hexahydrate | Sigma Aldrich | 236489-100G | |
| Ammonium Hydroxide | Macron Fine Chemicals | 6665-46 | |
| Poly(Ethylene Glycol), Average Mn 400 | Sigma Aldrich | 202398-250G | |
| Iron (II) chloride tetrahydrate | Sigma Aldrich | 44939-250G | |
| Disposable petri dish | Sigma Aldrich | BR452000 | |
| Disposable Inoculating Loop | Fisher Scientific | 22-363-604 | |
| Anhydrous Calcium Sulfate | W.A. Hammond Drierite | 13001 | |
| High vacuum grease | Sigma Aldrich | Z273554-1EA | |
| Laboratory pipetting needle with 90° blunt ends | Sigma Aldrich | CAD7937-12EA | |
| pH test strips | Sigma Aldrich | P4786-100EA | |
| Round-bottom three neck angle type distilling flask | Sigma-Aldrich | CLS4965250 | |
| Silicone oil for oil baths | Sigma-Aldrich | 85409-250ML | |
| Drying Tube | Chemglass | CG-1295-01 | |
| Septum Stopper, Sleeve Type | Chemglass | CG-3022-98 | |
| Magnetic stir bar | Chemglass | CG-2001-05 | |
| Condenser | Chemglass | CG-1218-01 | |
| Temperature Controller | BriskHeat | SDC120JC-A | |
| Stirring Hotplate | Fisher Scientific | 11-100-49SH | |
| Comet Assay Kit | Trevigen | 4250-050-K | |
| SYBR Gold Nucleic Acid Gel Stain | Life Technologies | S-11494 | |
| bio-AFM | JPK Instruments | NanoWizard 4a BioScience AFM | |
| Nanoindenter | Micro Materials Ltd | Multi-module mechanical tester | |
| Scanning electron microscopy (SEM) | Hitachi High Technologies America | Hitachi S-4800 | |
References
- Saxena, I. M., Brown, R. M. Biosynthesis of bacterial cellulose. Bacterial Nanocellulose: A Sophisticated Multifunctional Material. , 1-18 (2012).
- Chan-Park, M. B., Shen, J. Y. Biomimetic control of vascular smooth muscle cell morphology and phenotype for functional tissue-engineered small-diameter blood vessels. J.Biomed.Mater.Res.A. 88, 1104-1121 (2009).
- Barud, H. S., et al. Biocellulose-based flexible magnetic paper. J. Appl. Phys. 117, (2015).
- Märtson, M., Viljanto, J., Hurme, T., Laippala, P., Saukko, P. Is cellulose sponge degradable or stable as implantation material? An in vivo subcutaneous study in the rat. Biomaterials. 20, 1989 (1999).
- Illésa, E., Tombácza, E., Szekeresa, M., Tótha, I., Szabób, &. #. 1. 9. 3. ;., Iván, B. Novel carboxylated PEG-coating on magnetite nanoparticles designed for biomedical applications. J. Magn. Magn. Mater. 380, 132 (2015).
- Torrisi, V., et al. Preventing corona effects: multiphosphonic acid poly(ethylene glycol) copolymers for stable stealth iron oxide nanoparticles. Biomacromolecules. 15, 3171 (2014).
- Cai, Z., Kim, J. Bacterial cellulose/poly(ethylene glycol) composite: characterization and first evaluation of biocompatibility. Cellulose. 17, 83 (2010).
- Wu, W., He, Q., Jiang, C. Magnetic iron oxide nanoparticles: synthesis and surface functionalization strategies. Nanoscale Res. Lett. 3, 397-415 (2009).
- Ulbricht, J., Jordan, R., Luxenhofer, R. On the biodegradability of polyethylene glycol, polypeptoids and poly (2-oxazoline)s. Biomaterials. 35, 4848 (2014).
- Azqueta, A., Collins, A. R. The essential comet assay: a comprehensive guide to measuring DNA damage and repair. Arch. Toxicol. 87 (6), 949-968 (2013).
- Scherner, M., et al. In vivo application of tissue-engineered blood vessels of bacterial cellulose as small arterial substitutes: proof of concept. J. Surg. Res. 189, 340 (2014).
- Bodin, A., et al. Influence of cultivation conditions on mechanical and morphological properties of bacterial cellulose tubes. Biotechnol Bioeng. 97, 425 (2007).
- Zaborowska, M., et al. Microporous bacterial cellulose as a potential scaffold for bone regeneration. Acta Biomaterialia. 6, 2540 (2010).
- Karimi, A., et al. A comparative study on the mechanical properties of the umbilical vein and umbilical artery under uniaxial loading. Artery Res. 8, 51 (2014).
- Lina, F., Ping, Z., Shengmin, Z., Guang, Y. Evaluation of bacterial nanocellulose-based uniform wound dressing for large area skin transplantation. Mater. Sci. Eng. C. 33, 2995 (2013).
- Olsson, R. T., et al. Making flexible magnetic aerogels and stiff magnetic nanopaper using cellulose nanofibrils as templates. Nature Nanotech. 5 (8), 584-588 (2010).
- Torre, B., et al. Magnetic force microscopy and energy loss imaging of superparamagnetic iron oxide nanoparticles. Sci. Rep. 1 (202), 1-8 (2011).

